Abstract
Background
Persistent post-traumatic headache (PTH) is a prevalent and disabling neurological disorder, often attributed to mild traumatic brain injury and resembling migraine in clinical features. The underlying cortical morphometric changes and their relevance to persistent PTH remain unclear.
Methods
This cross-sectional magnetic resonance imaging (MRI) investigation enrolled 103 adults with persistent PTH, 296 with migraine and 155 healthy controls (HC), to undergo structural MRI at 3T. Cortical surface area, thickness and volume were evaluated in FreeSurfer. The analyses applied cluster-determining thresholds of p < 0.001 and cluster-wise thresholds of p < 0.05, adjusted for age, sex and total intracranial volume.
Results
Participants with persistent PTH exhibited larger surface area in the right anterior and posterior cingulate cortex (pcluster = 0.003), as well as the right superior parietal cortex/postcentral gyrus, compared to HC (pcluster < 0.035). No morphometric differences were observed between participants with persistent PTH and migraine (including subgroups: episodic, chronic, with aura without aura).
Conclusions
These findings reveal morphometric alterations in persistent PTH, specifically within pain processing regions of the mid-cingulate and somatosensory cortex. Similar changes have been reported in migraine, suggesting a shared neurobiological substrate. These enlargements might reflect adaptations to recurrent nociceptive stimuli that sustain persistent PTH.
Introduction
Post-traumatic headache (PTH) is a prevalent neurological disorder that impairs quality of life and imposes an enormous burden on those affected (1,2). The most common cause is mild traumatic brain injury (mTBI), often referred to as concussion (3). According to the International Classification of Headache Disorders, 3rd edition (ICHD-3), PTH attributed to mTBI can be classified as either acute or persistent (4). Acute PTH develops within seven days of injury and resolves by three months (4). However, a considerable proportion of patients experience headache that persists beyond three months (5), resulting in a diagnosis of persistent PTH (4). The clinical presentation of persistent PTH resembles chronic migraine and includes near-daily to daily headache of moderate-to-severe intensity, often accompanied by photophobia, phonophobia, nausea and vomiting (6,7). Despite its disabling nature, the neurobiological underpinnings of persistent PTH and its potential overlap with migraine remain poorly understood (2).
One proposed mechanism involves long-term adaptations within supraspinal pain-processing areas, triggered by recurrent nociceptive signaling post-mTBI (2,8). Evidence from magnetic resonance imaging (MRI) studies of migraine has revealed morphometric alterations in pain-processing areas such as the anterior cingulate cortex and the somatosensory cortex (9,–11). More pronounced changes have been noted in chronic migraine, possibly reflecting sustained nociceptive input (12). By contrast, data on cortical morphometric changes in persistent PTH are scarce and often limited by methodological issues (13,–17). A recent systematic review underscored these limitations, citing small samples, the absence of multiple comparison corrections and use of suboptimal statistical thresholds (12). Therefore, further investigations are needed to determine whether cortical morphometry differs among people with persistent PTH, individuals with migraine, and healthy controls (HC). Such insights will advance our understanding of persistent PTH and reveal whether identified morphometric alterations mirror those observed in migraine (12), given the striking clinical parallels (6,7).
The present cross-sectional study examined cortical morphometry in a large sample of adults with persistent PTH after mTBI in comparison with those with migraine and HC. Brain MRI at 3T was used to quantify cortical surface area, thickness and volume. Our analyses also encompassed distinct migraine subtypes (episodic, chronic, with aura, without aura) and explored correlations between identified morphometric differences and relevant clinical features.
Methods
The study protocol was approved by the Scientific Ethics Committee for the Capital Region of Denmark (Identifier: H-20033264). All study-related assessments or procedures were conducted in accordance with the Declaration of Helsinki, with later revisions (18). All participants provided written informed consent before enrollment. The study was conducted from November 2020 to October 2023. All of the investigators have reviewed and approved the final version of the manuscript. The presented data on adults with migraine and HC derive from the MRI Core of the prospective Registry for Migraine (REFORM) study (19). A previous report described cortical morphometric changes in migraine and its subtypes compared to age- and sex-matched HC (12). The present work builds on these findings by analyzing cortical morphometry in participants with persistent PTH compared to both participants with migraine and HC in REFORM.
Design and participants
This study applied a cross-sectional design involving adults with persistent PTH or migraine, as well as HC. Participants with persistent PTH and those with migraine were identified predominantly through referrals from neurologists, whereas HC were recruited using advertisements posted on a national research volunteer website (https://forsoegsperson.dk). A complete account of inclusion and exclusion criteria for all groups is provided in the Supplementary material (Tables S1–S3).
Persistent PTH group
Eligible participants were required to have a diagnosis of persistent PTH attributed to mTBI for at least one year, according to ICHD-3 criteria (4). In addition, headache had to occur on at least four days per month during the preceding three months. Individuals were excluded if they had a history of multiple TBIs, whiplash injury, clinically significant neurological or somatic diseases, or any personal history of primary headache disorders, except for infrequent episodic tension-type headache.
Migraine group
Participants eligible for inclusion were required to have an ICHD-3 diagnosis of migraine without aura, migraine with aura, or chronic migraine for at least one year (4). Furthermore, participants needed to report at least 4 monthly migraine days during the past three months. The main exclusion criteria were any significant neurological or somatic disease.
HC group
Eligible HC were adults reporting no personal history of primary or secondary headache disorders, except for infrequent episodic tension-type headache. In addition, HC were excluded if they reported having first-degree relatives diagnosed with a primary headache disorder, clinically significant medical conditions, or regular use of any medication.
Procedures
Both participants and HC underwent standardized physical and neurological examinations. Furthermore, site investigators performed a semi-structured interview on participants with persistent PTH or migraine. This interview captured detailed clinical features, medication use, medical history and psychiatric comorbidities. Participants were also asked about the timing of their most recent headache episode or migraine attack, aura symptoms and menstrual status, as appropriate.
Moreover, both participants and HC were required to abstain from specific substances before their MRI session. Treatment with analgesics, anti-inflammatory drugs, sedatives and antihistamines was not allowed for at least 48 hours prior to the scan session. In addition, caffeine or caffeinated products were not permitted within 12 hours before the MRI scan.
MRI procedures
All MRI data were acquired on a single 3.0 Tesla Siemens MAGNETOM Prisma scanner (Siemens Healthineers, Erlangen, Germany) equipped with a 32-channel head coil. Head motion artifacts were minimized by instructing participants and HC to remain as still as possible and by placing foam pads at both temple regions. The imaging protocol included magnetization prepared rapid acquisition gradient echo (MPRAGE) and fluid-attenuated inversion recovery (FLAIR) sequences. The MPRAGE sequence was acquired with voxel size 1.0 × 1.0 × 1.0 mm, repetition time 2300 ms, echo time 2.0 ms, inversion time 900 ms, as 208 interleaved slices, with 1 min and 57 s acquisition time. The FLAIR sequence was acquired with 0.5 × 0.5 × 1.0 mm resolution, repetition time 5000 ms, echo time 394 ms, inversion time 1600 ms, as 160 interleaved slices, with 5 min and 27 s acquisition time. Additional acquisitions were also made using other sequences to capture imaging data on brain structure and function (see Supplementary materials, Table S4). The entire scan session lasted about 55 min.
Preprocessing
All scans were preprocessed and analyzed in FreeSurfer, version 7.2.0 (https://surfer.nmr.mgh.harvard.edu) on a single MacOS workstation (macOS Big Sur, version 11.6; Apple Inc., Cupertino, CA, USA). The MPRAGE and FLAIR sequences were used to optimize grey matter segmentation (20,21). The FreeSurfer pipeline encompassed motion correction, intensity normalization, skull-stripping, segmentation and parcellation. These steps produced three-dimensional reconstructions of the pial and white matter surfaces, which were used to delineate the cerebral cortex at subvoxel resolution (21). The surfaces comprised triangular faces adjoining at defined vertices. Three independent assessors reviewed the segmentation and performed manual corrections when necessary. Following surface reconstruction, outcome measures were mapped to the standard FreeSurfer template (fsaverage) for interindividual comparisons (21). A 10-mm Gaussian smoothing kennel was then applied to all imaging data to facilitate robust statistical analyses.
Participant classification: categories and subgroups
For comparison with the participants with persistent PTH, we subgrouped the participants with migraine according to the ICHD-3 (4). We used a non-mutually exclusive classification system wherein participants with migraine were classified as having either episodic or chronic migraine, as well as either migraine with or without aura. If a participant had both migraine without and with aura, they were described in the cohort of participants with migraine with aura.
Participants with persistent PTH were also grouped descriptively as having a migraine-like phenotype or a tension-type headache-like phenotype based on ICHD-3 criteria (4). Some individuals with PTH can experience post-concussive symptoms of photophobia or phonophobia irrespective of the presence of headache. For these individuals, it was decided that they would only fulfill the criteria of photophobia and phonophobia during headache, if they experienced an exacerbation during their headache attacks, in agreement with prior studies (22,–24). Furthermore, the participants were classified as having either episodic or chronic phenotypes depending on the frequency of the migraine-like and tension-type headache-like attacks. This classification likewise followed ICHD-3 criteria (4).
Outcomes
Participants with persistent PTH were separately compared to those with migraine (including defined subgroups) and HC across each outcome measure. The migraine group was classified according to ICHD-3 criteria into two non-mutually exclusive categories: episodic migraine versus chronic migraine, and migraine with aura versus migraine without aura (4). Each of these categories comprised two distinct subgroups to allow detailed comparisons. Moreover, in participants with persistent PTH, post-hoc analyses were performed to explore potential correlations between significant morphometric clusters and both the number of monthly headache days and the disease duration measured in years. Additional whole-brain post-hoc analyses assessed potential confounding effects of preventive medication use, comorbid anxiety, or comorbid depression.
Statistical analysis
All demographic and clinical continuous variables were tested for normality using the Kolmogorov–Smirnov test. Depending on the results, continuous variables were reported either as the mean ± SD or as medians with interquartile ranges. Categorical variables were summarized as frequencies (counts) and percentages. For group balancing, age was compared using Student's t-tests, age distribution using two-sample Kolmogorov-Smirnov's tests, and sex using chi-squared tests. p < 0.05 was considered statistically significant for these tests. All statistical analyses were conducted in R, version 4.1.0 (R Foundation, Vienna, Austria).
Group comparisons of cortical morphometric measures were performed in FreeSurfer using general linear models (GLM) for each hemisphere (20,21). We corrected for multiple comparisons across the entire cortex using a Monte Carlo simulation with a cluster-determining (i.e. vertex-wise) threshold of p < 0.001 and a cluster-wise threshold of p < 0.05. This approach has been shown to control false-positive rates at ≤5% for all measures (25). Age, sex and total intracranial volume were included as covariates in all interindividual analyses. For morphometric outcomes showing significant differences, whole-brain post-hoc analyses were conducted using the same statistical thresholds. The cortex was parcellated according to the Desikan-Killiany Atlas (26).
For post-hoc analyses, we extracted averaged cortical morphometric values from significant clusters. We then correlated these values with both the average number of monthly headache days in the past three months and disease duration within the persistent PTH group. These analyses were performed in R, version 4.1.0, using GLMs adjusted for age, sex, and total intracranial volume. p < 0.05 was considered statistically significant for these associations.
Results
From November 2020 to October 2023, MRI scans were conducted on 105 participants with persistent PTH, 306 participants with migraine and 160 HC. Incidental findings led to the exclusion of two participants with persistent PTH, 10 participants with migraine and five HC. The final sample comprised 103 participants with persistent PTH, 296 participants with migraine and 155 HC. Table 1 provides the demographic and clinical characteristics of all study population groups.
Demographic and clinical characteristics of study populations.
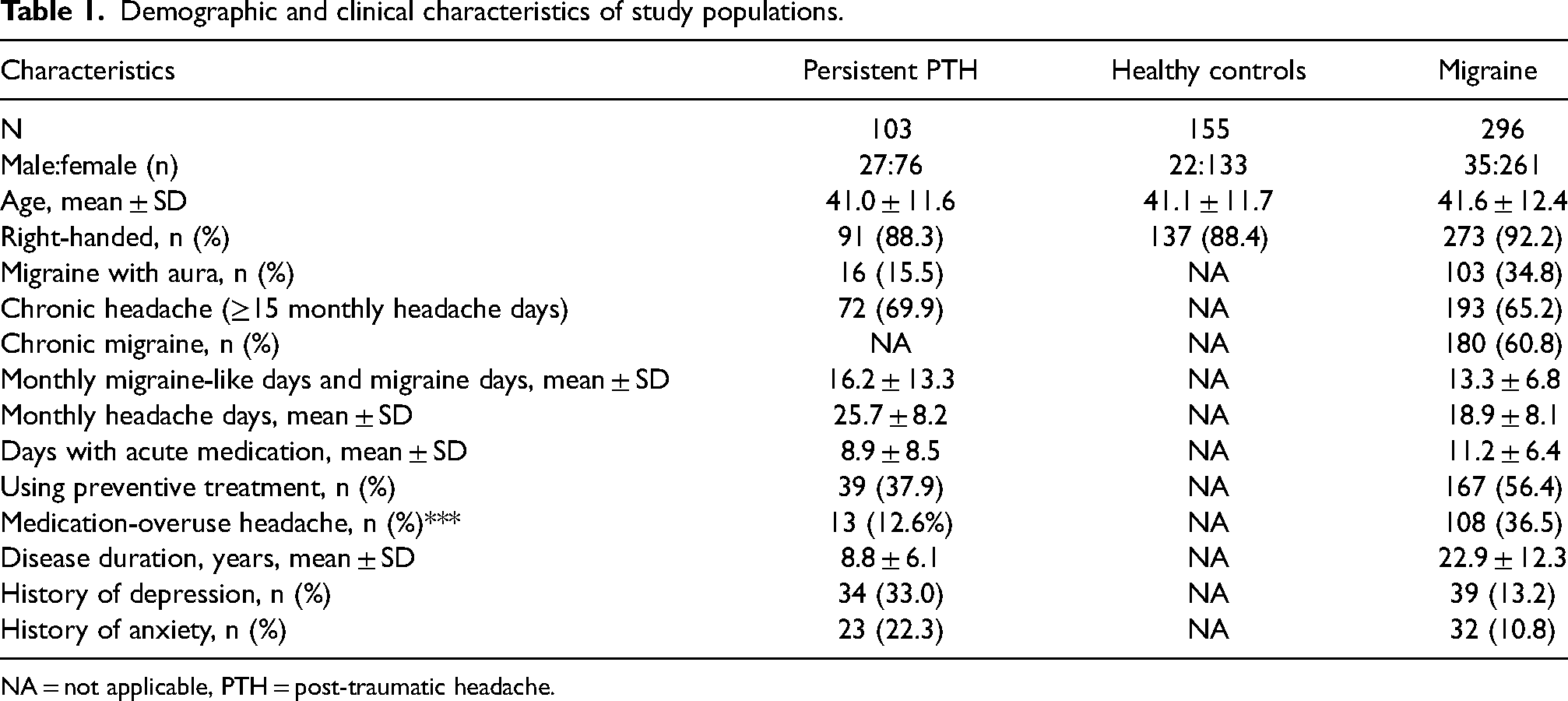
NA = not applicable, PTH = post-traumatic headache.
The participants with persistent PTH had a mean ± SD age of 41.0 ± 11.6 years, with 76 (73.8%) being female. These participants experienced an average of 25.7 ± 8.2 monthly headache days, and 95 (92.2%) exhibited a migraine-like phenotype. Acute headache medication was used on 8.9 ± 8.5 days per month, and 39 (37.9%) participants reported current use of preventive headache medication. During the MRI scan, 96 (91.3%) experienced headache of any intensity.
The age distribution did not differ between participants with persistent PTH and those with migraine (age, p = 0.343; age distribution, p = 0.417). Likewise, no differences in age were observed between participants with persistent PTH and HC (age; p = 0.671, age distribution; p = 0.934). However, a significantly higher proportion of participants with persistent PTH were male compared to those with migraine (p < 0.001) and HC (p = 0.016).
Persistent PTH versus healthy controls
The participants with persistent PTH exhibited a larger surface area in a cluster spanning the right caudal anterior and posterior cingulate cortex compared to HC (pcluster = 0.003) (Figure 1 and Table 2). The majority of the cluster was located in the caudal anterior cingulate cortex (Figure 1). Furthermore, participants with persistent PTH also had a greater surface area in a cluster encompassing the right superior parietal cortex/postcentral gyrus compared to HC (pcluster = 0.035) (Figure 2 and Table 2). The majority of the cluster was located in the postcentral gyrus (Figure 2). No differences in cortical thickness or volume were detected between these groups.

Medial view of the right hemisphere. Left: participants with persistent post-traumatic headache exhibited a significantly larger surface area (red) in the caudal anterior and posterior cingulate cortex compared to healthy controls. These findings were significant at a cluster-determining threshold of p < 0.001 and cluster-wise threshold of p < 0.05. Cluster size of the right anterior and posterior cingulate cortex: 448.0 mm² (MNI coordinates: x = 5.8, y = 2.0, z = 32.6). Right: uncorrected statistical significance map (–log(p)). Warm colors indicate larger surface area in persistent post-traumatic headache, whereas cold colors indicate smaller surface area compared to healthy controls. Parcellated according to the Desikan-Kiliany atlas (26,). MNI = Montreal Neurological Institute.
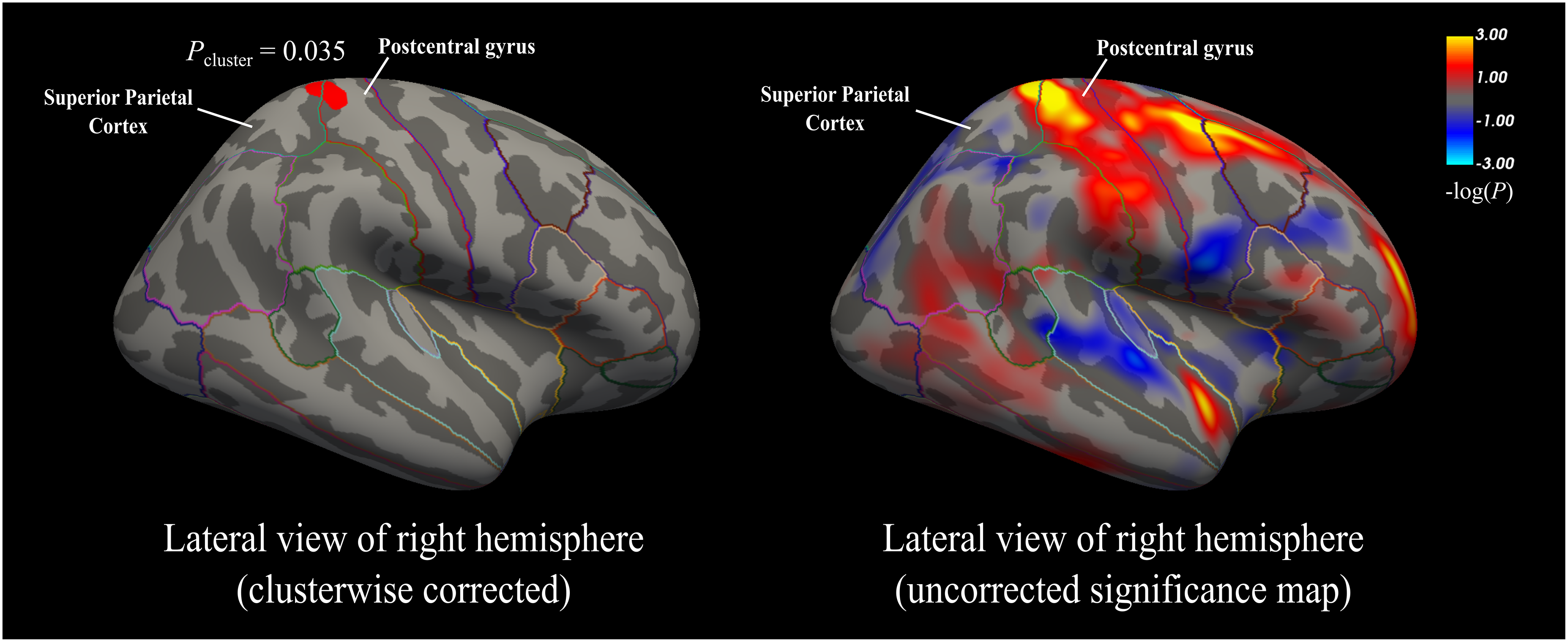
Lateral view of the right hemisphere. Left: participants with persistent post-traumatic headache exhibited a significantly larger surface area (red) in the superior parietal cortex/postcentral gyrus compared to healthy controls. These findings were significant at a cluster-determining threshold of p < 0.001 and cluster-wise threshold of p < 0.05. Cluster size of the right superior parietal cortex/postcentral gyrus: 300.4 mm2 (MNI coordinates x: 19.6, y: −39.1, z: 59.8). Right: uncorrected statistical significance map (–log(p)). Warm colors indicate larger surface area in persistent PTH, whereas cold colors indicate smaller surface area compared to healthy controls. Parcellated according to the Desikan-Kiliany atlas (26). MNI = Montreal Neurological Institute.
Cortical surface area differences in participants with persistent post-traumatic headache versus healthy controls.

MNI = Montreal Neurological Institute.
Persistent PTH versus migraine
The comparisons revealed no significant differences in cortical thickness, surface area, or volume between participants with persistent PTH and those with migraine. Furthermore, no differences were identified between participants with persistent PTH and any migraine subgroup, including episodic migraine, chronic migraine, migraine without aura and migraine with aura.
Correlations with clinical variables
Correlation analyses were performed among participants with persistent PTH, focusing on cluster of increased cortical surface area identified in comparisons with HC. The surface area in the cluster spanning the right caudal anterior and posterior cingulate cortex did not correlate with monthly headache days (p = 0.14) or disease duration (p = 0.06). Likewise, the surface area in the cluster encompassing the right superior parietal cortex/postcentral gyrus showed no correlation with monthly headache days (p = 0.71) or disease duration (p = 0.84).
Post-hoc analyses
Among participants with persistent PTH, post-hoc analyses were performed to assess whether preventive medication use, comorbid anxiety or comorbid depression impacted the main cortical surface area findings. Neither preventive medication use nor comorbid anxiety showed a significant effect. However, comorbid depression was associated with increased surface area in two small clusters: the right rostral middle frontal cortex (pcluster = 0.028) and the left precentral cortex (pcluster = 0.049) (see Supplementary material, Figure S1 and Table S4). No significant effects were observed in the cingulate or postcentral cortices.
Discussion
This MRI investigation revealed that participants with persistent PTH had a larger surface area in both the cingulate cortex and superior parietal cortex/postcentral gyrus compared to HC. These regions are implicated in supraspinal pain processing (27,–29), indicating that persistent PTH is associated with distinct neuroplastic adaptations. Furthermore, no discernible differences were identified regarding cortical thickness and volume between participants with persistent PTH and HC.
Our results also revealed no differences in cortical morphometry between participants with persistent PTH and those with migraine. Because both individuals with persistent PTH and those with migraine (12) showed similar cingulate changes, this could suggest overlapping neurobiological alterations. Both headache disorders often share clinical features and might arise from convergent pathophysiologic mechanisms (6,7).
Existing evidence and added value
Multiple MRI studies have examined morphometric alterations in persistent PTH, yet the findings remain inconsistent. Among the implicated regions are the anterior cingulate, prefrontal, orbitofrontal, precentral, and caudal middle frontal cortices (13,–15). Additional affected areas include the temporal, parahippocampal and supramarginal gyri, as well as the precuneus (13,–15). A 2025 systematic review identified major methodological limitations in these studies (9). For example, sample sizes were small to moderate, ranging from 28 to 49 participants with persistent PTH (9). Some investigations combined data from participants with and without pre-existing headache (including migraine) disorders or allowed the inclusion of people with multiple TBIs, introducing considerable sample heterogeneity (13,14,30). Moreover, suboptimal statistical methods further complicate interpretation because certain studies reported significant results without specifying whether or how corrections for multiple comparisons were applied (14,15). Others applied liberal statistical thresholds that amplified the risk of false positives (13). Because morphometric analyses can entail thousands of comparisons across the voxels of the cortex, multiple comparison correction becomes critical to control the risk of false discovery (25). These factors hinder efforts to identify cortical morphometric alterations unique to persistent PTH or to place such changes in context with migraine.
The present study addressed these issues by enrolling a large sample of adults with persistent PTH, migraine and HC. Direct comparisons to participants with migraine clarified whether persistent PTH and migraine share similar cortical morphometric profiles. Stringent cluster-determining (p < 0.001) and cluster-wise (p < 0.05) thresholds minimized the risk of type I errors, which, with the current approach, is validated to limit false-positive rates to below 5% for all investigated cortical measures (25). This might explain why we did not replicate the numerous regional alterations reported in earlier studies (9). Some of those findings, though inconsistent, have been proposed as potential ‘biomarkers’ of persistent PTH (16). By contrast, our results revealed morphometric alterations exclusively in cortical regions involved in pain processing. These findings align well with a plausible neurobiological basis of headache and advance our understanding of structural changes in persistent PTH.
Cingulate cortex
Our analyses revealed greater surface area in a cluster overlapping the right anterior cingulate as well as an anterior region of the posterior cingulate cortex, with this specific area often being termed the midcingulate cortex in other brain atlases (31,32). The anterior cingulate cortex is central to the affective dimension of pain, attributing negative or unpleasant valence to noxious stimuli (28,29,33). Evidence from animal and human studies of pain confirms robust activation in this region, and partial ablation can ameliorate intractable cancer pain (34,35). This activation likewise extends to involve mid-cingulate regions consistent with our posterior cingulate coordinates (36,–40).
The anterior and midcingulate cortex hosts neurons that respond specifically to pain (41,42). Such neurons undergo synaptic plasticity during states of chronic pain, characterized by long-term potentiation (29). Interestingly, long-term potentiation is closely associated with structural changes such as dendritic spine enlargement. These alterations might manifest as increased cortical surface area on structural MRI (43,44).
Sustained nociceptive messaging following mTBI might drive such structural remodeling. These changes could reflect either adaptive or maladaptive responses, consistent with the midcingulate cortex's role in modulating nociceptive hypersensitivity (40). The observed surface area enlargement in the caudal anterior and posterior cingulate cortices likely reflects altered pain processing in people with persistent PTH (45).
This finding aligns well with our recent MRI investigation, which included the same migraine population and HC (12). We identified a larger surface area of the caudal anterior cingulate cortex in specifically people with chronic migraine compared to age- and sex-matched HC (12). This overlap might explain our present results showing no differences between participants with persistent PTH and those with migraine. An enlarged surface area of the right anterior cingulate cortex could be a shared feature of persistent PTH and migraine. Here, it merits emphasis that none of the participants with persistent PTH had a pre-mTBI history of migraine or frequent headache. This detail might suggest that the identified morphometric changes arose de novo post-mTBI rather than reflecting a pre-existing vulnerability.
From a clinical standpoint, both persistent PTH and migraine present with headache exacerbations accompanied by photophobia, phonophobia, nausea and sometimes vomiting (6,7,46). The pathogenesis of persistent PTH remains poorly understood but is considered to involve neuronal damage and dural or CNS inflammation post-mTBI (2). These mechanisms might cause peripheral and/or central sensitization, which, in turn, could explain the hypersensitivity to migraine-related neuropeptides such as calcitonin gene-related peptide and pituitary adenylate cyclase-activating polypeptide (22,47). Together, these observations suggest that shared neurobiological mechanisms may underlie both headache disorders.
In addition to neuroplastic alterations, a possible genetic predisposition for both migraine and persistent PTH might underlie our findings. It is conceivable that some people could have inherent susceptibilities, making them more prone to develop persistent PTH after sustaining a mTBI. Alternatively, morphometric changes might manifest soon after mTBI, promoting the transition from acute to persistent PTH. Large-scale longitudinal MRI studies are needed to clarify the neuroplastic nature of these alterations by documenting their evolution over time or their response to early initiation of preventive treatments.
Of interest, our findings align with a previous report that identified morphometric changes in the anterior and posterior cingulate cortex during the transition from acute to persistent PTH (15). Specifically, Niu et al. (15) used voxel-based morphometry in 60 participants with acute PTH post-mTBI, 17 participants with TBI without PTH and 42 HC. Participants with acute PTH demonstrated larger grey matter volume in these cingulate regions compared to participants with migraine and HC. These alterations persisted in the 15 participants who transitioned from acute to persistent PTH. In light of our present data, it is plausible that these grey matter differences might reflect an increased surface area in the anterior and posterior cingulate cortex. Nevertheless, it is important to note that the anterior and midcingulate cortices are involved in pain processing in general, and not exclusively cephalic pain. For example, structural alterations in the cingulate cortex have been reported in chronic low-back pain (48,49). However, these studies have not identified significant changes in cortical surface area.
Somatosensory cortex
The participants with persistent PTH exhibited a larger surface area in a cluster spanning the superior parietal cortex/postcentral gyrus. Interestingly, the postcentral gyrus hosts the primary somatosensory cortex, which localizes and discriminates painful stimuli (27). A larger surface area in this region might reflect aberrant pain processing or compensatory responses to increased peripheral nociceptive input (27). Notably, no previous MRI studies have documented structural alterations in the postcentral gyrus among participants with persistent PTH, possibly because few investigations have focused on the cortical surface area.
Persistent pain signaling after mTBI might drive these changes because nociceptive input travels from meningeal nociceptors to the somatosensory cortex via ascending trigeminal pathways (50). Alternatively, these alterations could reflect adaptive responses within somatosensory circuits in response to mTBI itself. The absence of similar findings in participants with migraine suggests that morphometric alterations in the postcentral gyrus might distinguish persistent PTH from migraine. However, our results showed no significant differences between these groups. Further research is needed to ascertain whether morphometric changes in the somatosensory cortex confer susceptibility to or maintenance of persistent PTH.
Future research should investigate whether the observed structural alterations reflect functional changes in the same regions, as suggested by some MRI studies (45). In addition to resting-state functional MRI, task-based paradigms using headache-provoking stimuli might clarify pain-processing mechanisms in persistent PTH. Another important question is whether similar structural changes are present in non-cephalic pain disorders, such as fibromyalgia or chronic low-back pain. If not, the alterations could be specific to cephalic pain. Even so, it remains essential to examine whether these changes are also found in other headache disorders, including tension-type headache. This distinction should help determine whether the morphometric features reflect a broader migraine-like phenotype common in persistent PTH.
Psychiatric comorbidities
Among our participants with persistent PTH, post-hoc analysis showed that comorbid depression was associated with increased surface area in the right rostral middle frontal and left precentral cortices. The dorsolateral prefrontal cortex, which corresponds to the rostral middle frontal region in our atlas, has been linked to emotional processing in depression ((51). However, these findings should be interpreted with caution because of the small sample sizes and limited statistical power. Furthermore, our study was not designed to investigate depression-related cortical changes in detail. Notably, neither comorbid depression nor comorbid anxiety affected surface area in the cingulate or somatosensory cortices.
Strengths and limitations
This study offers notable strengths. First, we enrolled a large, well-characterized sample of participants with persistent PTH, migraine and HC. Second, we applied conservative neuroimaging statistics that ensured robust examination of cortical morphometry and minimized false-positive findings. Moreover, all participants with persistent PTH had sustained a single mild TBI and reported no history of primary headache disorders, providing group homogeneity.
However, our study also has several limitations that warrant mention. The cross-sectional design precludes causal inferences regarding the temporal relationship between cortical alterations and headache onset. Differences in acute and preventive headache medication use between and within our persistent PTH and migraine samples might confound comparisons. In addition, our migraine population was heterogenous, including episodic and chronic migraine, migraine with and without aura, and participants experiencing headache during the scan. However, it is worth noting that no significant morphometric differences were identified between persistent PTH and any migraine subgroup. Lastly, participants with persistent PTH were not age- and sex-matched to those with migraine and HC; however, all analyses were adjusted for these variables to mitigate potential confounding effects.
Conclusions
Persistent PTH is associated with increased surface area in the cingulate cortex and somatosensory cortex, both of which are regions implicated in pain processing. The identified structural alterations distinguish people with persistent PTH from HC and parallel changes seen in migraine. Thus, our findings suggest that persistent PTH and migraine might share common pathophysiological alterations in neural structures governing pain processing.
Article highlights
Persistent PTH was linked to greater surface area specifically within key pain-processing regions of the cingulate cortex and postcentral gyrus. Similar cingulate regions were previously enlarged in chronic migraine, suggesting shared pathophysiology. No structural cortical differences were observed between individuals with persistent PTH and migraine. These enlargements might reflect plastic adaptations to nociception in persistent post-traumatic headache.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251362830 - Supplemental material for Differences in cortical morphometry between persistent post-traumatic headache, migraine and healthy controls
Supplemental material, sj-docx-1-cep-10.1177_03331024251362830 for Differences in cortical morphometry between persistent post-traumatic headache, migraine and healthy controls by Rune Häckert Christensen, Haidar Muhsen Al-Khazali, Messoud Ashina, Nina Vashchenko and Rogelio Dominguez-Moreno, Daniel Tolnai, Håkan Ashina in Cephalalgia
Footnotes
Author contributions
Study concept and design: MA and HA. Data acquisition and analysis: RHC, NV, RM and DT. Drafting of the manuscript: RHC, HMA and HA. Critical revision of the manuscript: RHC, HMA, NV, RM, MA, DT and HA. Funding acquisition: MA and HA.
Data availability
Upon reasonable request, the corresponding author will provide the necessary data and materials to interested researchers for the purpose of academic scrutiny, reproducibility and further scientific investigation.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: HMA has received personal fees from Pfizer, outside of the submitted work. RHC has received personal fees from Teva and Pfizer, outside of the submitted work. MA has received personal fees from AbbVie, Amgen, AstraZeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer and Teva, outside of the submitted work. MA is also a Primary Investigator for ongoing clinical trials sponsored by AbbVie, Lundbeck and Pfizer. MA also serves as an Associate Editor of Brain and The Journal of Headache and Pain. HA has received personal fees from AbbVie, Lundbeck, Pfizer and Teva, outside of the submitted work. HA also serves as an Editorial Board Member of The Journal of Headache and Pain. The remaining authors declare no potential conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by research grants from the Lundbeck Foundation (R403-2022-1352 to HA and R310-2018-3711 to MA).
Ethical statement
The study protocol was approved by the Scientific Ethics Committee for the Capital Region of Denmark approved the study protocol (Identifier: H-20033264). All study-related assessments or procedures were conducted in accordance with the Declaration of Helsinki. All participants provided their written informed consent before enrollment.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
