Abstract
Background
Integrating brain structure and function may help characterize neurobiological heterogeneity in migraine alongside symptom presentation.
Aim
To apply a multimodal, exploratory, data-driven approach to identify migraine subgroups using structural and functional MRI, and to describe the clinical characteristics of the resulting subgroups.
Methods
Resting-state functional connectivity (FC) across cortical and subcortical regions, along with structural measures including cortical thickness, cortical volume, and subcortical volumes, were extracted from 111 individuals with migraine (75 chronic, 36 episodic) classified according to ICHD-3 criteria. After dimensionality reduction using principal component analysis, hierarchical agglomerative clustering was applied to identify multimodal imaging-derived subgroups. For comparison, secondary unimodal clustering models were constructed using functional-only and structural-only feature sets. The optimal number of clusters was determined using silhouette coefficients, and clustering concordance across models was quantified using the Adjusted Rand Index (ARI). Group differences in clinical characteristics, FC, and cortical and subcortical structure were assessed using covariate-adjusted statistical models with false discovery rate (FDR) correction.
Results
Multimodal clustering identified two subgroups with distinct clinical and imaging profiles, Migraine Cluster 1 (M1 f + s ) and Migraine Cluster 2 (M2 f + s ). M2 f + s showed older age, longer disease duration, greater migraine disability, widespread increases in cortical-subcortical FC (including Dorsal Attention, Somatomotor, and Visual networks), and reduced cortical volumes across frontal, parietal, temporal, and insular regions compared with M1 f + s . This subgroup also exhibited increased connectivity relative to controls. In contrast, M1 f + s showed preserved cortical structure and stronger Control-network–subcortical connectivity compared to M2 f + s , and no significant functional or structural deviations from controls. Unimodal analyses revealed that Functional-only clustering aligned moderately with the multimodal cluster solution (ARI = 0.427), showing that FC was a primary determinant of the multimodal cluster structure, whereas structural-only clustering showed negligible overlap (ARI = 0.001), reflecting an orthogonal dimension of heterogeneity captured by structural variation.
Conclusion
Data-driven multimodal neuroimaging-based clustering in migraine identified two subgroups with distinct clinical and imaging patterns, highlighting heterogeneity and providing a framework for further investigation of imaging-informed characterization.
This is a visual representation of the abstract.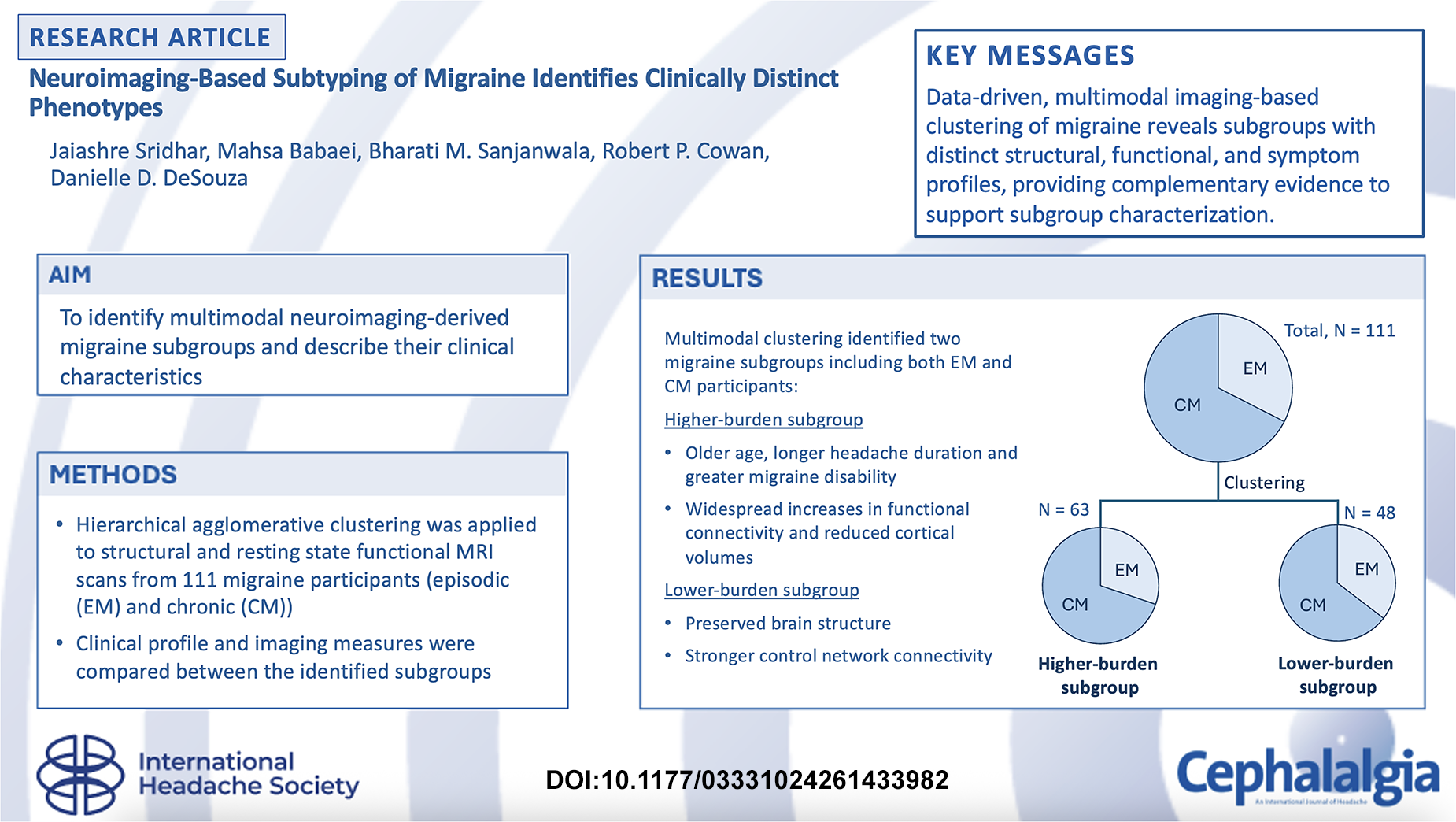
Keywords
Introduction
Migraine is a heterogeneous neurological disorder characterized by significant variability in clinical profile, disease progression, and treatment response. Current classification using International Classification of Headache Disorders (ICHD-3), 1 categorizes migraine based on clinical criteria, including attack frequency, aura presence, and associated symptoms. However, current diagnostic approaches often do not guide effective treatment selection, in part due to underlying neurobiological differences between individuals.2,3 Improving diagnosis and treatment strategies may benefit from approaches that integrate neuroimaging and other biological markers with clinical profiles to support a more comprehensive understanding of migraine subgroups.4,5
Previous studies have explored neuroimaging-based approaches to identify migraine subgroups. In one study, Krimmel and colleagues 6 examined resting-state functional connectivity in a two-site study that included a subset of the current cohort and identified four migraine biotypes derived from three connectivity-based dimensions linked to clinical symptoms. In another study, Schwedt and team 7 used structural MRI metrics such as cortical thickness, surface area, and volume, and identified two subgroups that differed in allodynia, years with migraine and migraine-related disability.
These foundational studies demonstrated the potential of neuroimaging to identify migraine subtypes by focusing on either structural or functional brain measures but, in some cases, restricted analyses to cortical regions. Building on this work, we applied an exploratory, data-driven clustering approach using multimodal neuroimaging that integrated functional and structural measures from both cortical and subcortical regions to identify migraine subgroups. To explore the robustness and specificity of these subgroup patterns, we additionally evaluated unimodal clustering models based solely on functional or structural features. Our overall aim was to define neurobiologically informed migraine subgroups, characterize their functional, structural and clinical profiles, and clarify the contributions of different imaging modalities to subgroup identification.
Methods
Participants
Patients with migraine and controls without migraine were recruited through the Stanford Headache Clinic as well as from the surrounding community. The migraine group comprised individuals with episodic (EM) or chronic migraine (CM), with or without aura, classified according to the criteria established in the ICHD-3. 1 The control group included participants who reported no or only very infrequent non-migraine headache days over the course of a year (<3 days). Participants were enrolled between January 2015 and March 2024. Exclusion criteria included the presence of any neurological disorder other than headache or contraindications for MRI. Only participants who had both structural and resting-state functional MRI scans were included in the study. All participants completed comprehensive questionnaires detailing their current clinical status and past medical history.
The study was approved by Stanford University's institutional review board (IRB). Written informed consent was obtained from all participants prior to data acquisition.
Clinical measures
Participants completed self-administered online questionnaires assessing demographics, headache characteristics (frequency, duration, years with headache, severity, triggers, and associated symptoms), migraine-related disability, lifestyle habits, overall health, and medication effectiveness. Monthly migraine frequency was recorded as the average number of headache days over the past three months, and headache severity was rated on a scale of 1–10. Migraine-related disability was measured using the Migraine Disability Assessment (MIDAS). To evaluate comorbid psychological and behavioral conditions, participants completed validated psychometric scales, including the Patient Health Questionnaire-9 (PHQ-9) for depression, Generalized Anxiety Disorder-7 (GAD-7) for anxiety, Pain Catastrophizing Scale (PCS) for negative pain-related thoughts, Pittsburgh Sleep Quality Index (PSQI) for sleep disturbances, Patient Health Questionnaire-15 (PHQ-15) for physical symptom burden and Pain Self-Efficacy Questionnaire (PSEQ) for confidence in managing pain. Acute treatments included triptans, over-the-counter medications, and nonsteroidal anti-inflammatory drugs (NSAIDs), while preventive treatments included botulinum toxin injections, supplements or non-traditional (riboflavin, magnesium, melatonin, cannabis/THC, etc.), and alternative therapy (physical therapy, acupuncture, massage, ayurvedic, etc.). Participants reported the medications used and those providing symptom improvement.
MRI acquisition
MRI data were acquired using a GE 3T MRI scanner (GE Healthcare, Milwaukee, WI) with an 8-channel phased-array head coil. Structural MRI (sMRI) was performed using high-resolution T1-weighted 3D axial FSPGR IR-prepared sequences with the following parameters: repetition time (TR) = 5.9 ms, echo time (TE) = minimum (i.e., minimum achievable TE for this sequence), flip angle = 15°, voxel size = 0.9 × 0.9 mm, matrix size = 256 × 256, and slice thickness = 1 mm (no gap). For resting-state functional MRI (rsfMRI), T2*-sensitive spiral-pulse sequences were used with parameters: TR = 2000 ms, TE = 30 ms, flip angle = 80°, voxel size = 3.4 × 3.4 mm, slice thickness = 4 mm, and interslice spacing = 0.5 mm.
sMRI processing
sMRI scans were processed using the FreeSurfer image analysis suite (version 7.4.1). 8 The resulting reconstructions were manually inspected to ensure accuracy. FreeSurfer's automated segmentation was employed to parcellate the brain into cortical 9 and subcortical 10 regions of interest (ROIs). For each ROI, cortical and subcortical volumes were extracted, averaged across hemispheres, and normalized to each participant's estimated total intracranial volume. Regional cortical thickness measures were also extracted and averaged bilaterally. This resulted in 34 cortical 9 ROIs and 9 subcortical ROIs, including the thalamus, caudate, putamen, pallidum, hippocampus, amygdala, accumbens, ventral diencephalon and brainstem.
rsfMRI processing
rsfMRI scans underwent preprocessing using fMRIPrep (version 23.2.0), 11 which included motion correction, slice-timing correction, co-registration to sMRI, and frame-wise displacement (FD) estimation. The quality of preprocessing steps was verified by inspecting the output reports generated by fMRIPrep. Subsequent post-processing with XCP-D (version 1.8.6) 12 included removing initial non-steady-state volumes, filtering outliers at FD threshold of 0.5 mm, 13 despiking, and bandpass filtering (0.01–0.1 Hz). Nuisance regressors included the top five principal components from white matter and cerebrospinal fluid, six motion parameters, and the mean global signal.
Cortical timeseries from the 100 ROI Schaefer parcellation 14 were averaged within the seven functional networks defined by Yeo et al. 15 – Control network, Default Mode network, Dorsal Attention network, Limbic network, Salient/Ventral Attention network, Somatomotor network, and Visual network. Subcortical timeseries were extracted from eight ROIs defined in Scale-I of Tian atlas, 16 including the anterior and posterior thalamus, caudate, putamen, pallidum, hippocampus, amygdala and accumbens. This yielded a total of seven cortical network-level timeseries and eight subcortical ROI timeseries. Only participants and ROIs with complete and valid time-series data were included in all analyses. Functional connectivity (FC) was computed using Pearson's correlation (r) and Fisher r to z transformed.
Cluster analysis
Cluster analysis was performed using a multimodal framework integrating functional and structural neuroimaging features from the migraine participants as an exploratory, data-driven approach. The Fisher z-transformed FC matrices were vectorized by extracting the upper-triangular elements and flattening them into feature vectors, which were then grouped into cortical–cortical (CC), cortical–subcortical (CS), and subcortical–subcortical (SS) blocks. Structural features were organized into cortical volume, cortical thickness, and subcortical volume blocks. All functional and structural blocks were z-score standardized and weighted by the inverse of their first singular value to balance the contribution of feature sets with differing dimensionalities.17,18 Weighted blocks were concatenated and reduced using principal component analysis (PCA), retaining components explaining 80% of total variance.19,20 Hierarchical agglomerative clustering was applied to the PCA scores using cosine distance and average linkage to emphasize similarity in feature patterns, and silhouette scores were computed for cluster size, k = 2–6 to identify the optimal cluster structure within this exploratory framework.
For unimodal analyses, the functional-only model included only FC blocks (CC, CS, SS), and the structural-only model included cortical thickness, cortical volume, and subcortical volume blocks, each processed using the same weighting, PCA, and clustering pipeline. As a sensitivity analysis, the multimodal clustering was repeated using a finer functional parcellation, computing FC across all 100 cortical 14 and 44 subcortical ROIs described in Scale-IV of Tian atlas, 16 while structural features retained at their original parcellation.9,10 PCA plots were generated for the optimal cluster solution based on silhouette criteria, and cluster labels were assigned to participants for post hoc characterization of subgroup-specific functional, structural, and clinical profiles.
Statistical analysis
Group differences in demographics, clinical characteristics, FC and brain structure were examined between migraine clusters and between each cluster and the control group. Age and sex distributions were compared using t-tests and chi-squared tests, respectively. Continuous clinical variables were compared using linear regression models with age and sex as covariates. Normality of residuals was assessed using the Shapiro–Wilk test; for variables showing clear deviations from residual normality (p < 0.01), p-values were obtained from models refit with HC3 robust standard errors. Categorical clinical variables were compared using logistic regression, with age and sex as covariates. Multiple comparisons were controlled using the false discovery rate (FDR) procedure.
For FC analyses, group differences were evaluated for each FC feature (CC, CS and SS) using an ANCOVA model, adjusting for age, sex, and headache during scan. Resulting p-values were FDR-adjusted across FC features, and group-averaged FC matrices and significant differences were visualized.
Differences in cortical thickness, cortical volume, and subcortical volume were evaluated using the same ANCOVA framework, adjusting for age, sex, and headache during scan, with FDR correction applied across regions.
The similarity between clustering solutions from the unimodal functional-only and structural-only and the finer ROI parcellation was quantified using the Adjusted Rand Index (ARI), comparing each alternative solution to the multimodal clustering result.
All statistical analyses were performed in Python (v3.9) using the statsmodels, SciPy, and scikit-learn libraries.
Results
Participant demographics
After excluding participants with MRI issues and incomplete data, the final analysis included 111 participants with migraine (75 CM and 36 EM), and 51 controls. There were no significant differences between age and sex between migraine participants (Age: 40 ± 13 (Mean ± SD), Sex: 83% Females) and healthy controls (Age: 38 ± 14, Sex: 53% Females).
Multimodal clustering
The multimodal clustering model integrated functional and structural MRI features, including 21 CC, 56 CS, and 28 SS connectivity features, together with 68 cortical structural measures (34 cortical thickness and 34 cortical volume) and nine subcortical volumetric measures. PCA reduced this feature set to 31 components explaining 80% of the total variance. Hierarchical clustering of these components yielded an optimal two-cluster solution (silhouette = 0.107), resulting in Migraine Cluster 1, M1 f + s with 48 participants and Migraine Cluster 2, M2 f + s with 63 participants. A PCA-based visualization of the cluster separation is shown in Figure 1.

Overview of the clustering framework and results across multimodal and unimodal models. (a) Lists the input feature sets used for multimodal, functional-only, and structural-only clustering models. (b) Shows silhouette values for k = 2–6 where higher scores indicate greater within-cluster cohesion and between-cluster separation. The optimal number of clusters (highest silhouette score) for each model is highlighted in red. (c) Displays PCA-based visualizations of cluster separation for the selected k solutions. (CC: Cortical-Cortical, CS: Cortical-Subcortical, SS-Subcortical-Subcortical functional connectivity features)
Demographics and clinical profile:
Cluster M2
f
+
s
was older than M1
f
+
s
(p = 0.01) while BMI and sex distribution were similar across clusters, with both groups predominantly female (Table 1). The proportions of CM and EM were comparable, as were headache frequency and intensity. However, M2
f
+
s
reported longer headache duration (p = 0.04), more years with migraine, and greater migraine-related disability (p = 0.05), along with lower pain self-efficacy. Rates of medication overuse headache, prodrome, allodynia, and aura were higher in M2
f
+
s.
Pain catastrophizing did not differ between clusters, and both groups exhibited mild depressive symptoms, mild anxiety, medium somatic symptom severity, and poor sleep quality. Regarding treatment response, M1
f
+
s
reported a significantly better response to NSAIDs than M2
f
+
s
(p = 0.03) Functional connectivity profile:
Demographic and clinical characteristics across multimodal and unimodal clustering solutions.

This table summarizes demographic, clinical, and headache-related characteristics for the subgroups derived from the multimodal, functional-only, and structural-only clustering models. Continuous variables are presented as mean ± standard deviation or median [interquartile range], depending on distribution. Categorical variables are reported as percentages. Variables showing significant between-cluster differences are denoted as ** (FDR-corrected p < 0.05) and * (uncorrected p < 0.05). Abbreviations: BMI - body mass index; CM - chronic migraine; MIDAS - Migraine Disability Assessment; PSEQ - Pain Self-Efficacy Questionnaire; PCS - Pain Catastrophizing Scale; PHQ9 - Patient Health Questionnaire-9; GAD7 - Generalized Anxiety Disorder-7; PHQ15 - Somatic Symptom Severity Scale; PSQI - Pittsburgh Sleep Quality Index; OTC - over-the-counter medications; NSAID - non-steroidal anti-inflammatory drugs. Overall, in our cohort, 86% reported use of over-the-counter medications (OTC), 85% NSAIDs, 69% Triptans, 66% alternative therapy, 51% supplement or non-traditional medications and 48% reported use of BOTOX.
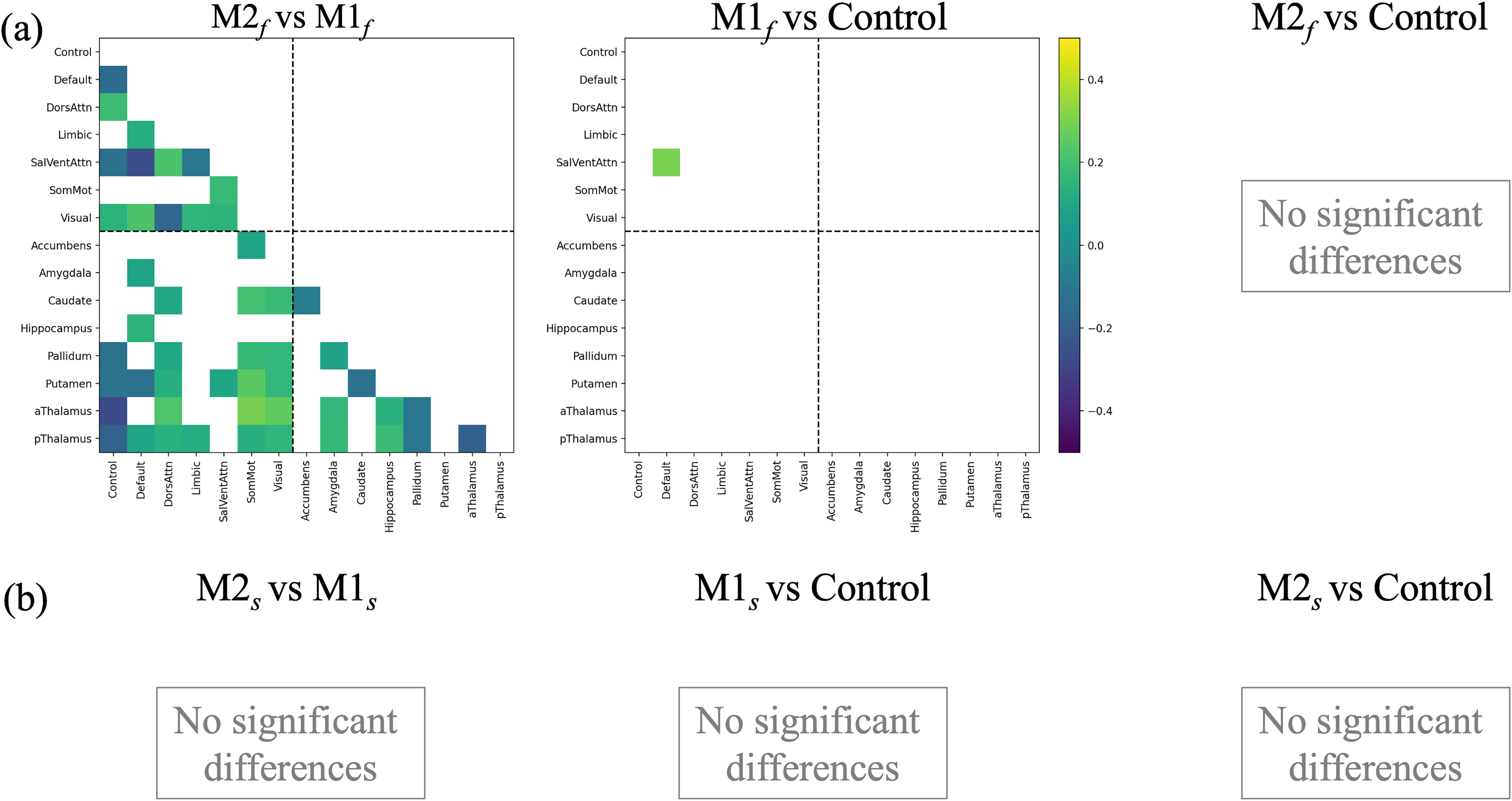
The average FC matrices of M1 f + s, M2 f + s and the control groups are shown in Figure 2(a). M2 f + s exhibited broadly increased FC between the Dorsal Attention, Somatomotor, and Visual networks and multiple subcortical regions, including the thalamus, basal ganglia, and limbic-related structures (Figure 2(b)). The Limbic network also showed stronger connectivity with the Visual and Somatomotor networks, as well as with the posterior thalamus, in M2 f + s . Within the cortical networks, M2 f + s demonstrated greater connectivity among the Dorsal Attention, Salient/Ventral Attention, and Limbic networks, and the Default Mode network showed increased connectivity with both the hippocampus and the Visual network. Subcortically, the amygdala and hippocampus displayed stronger connectivity with the pallidum and thalamus in M2 f + s compared with M1 f + s . In contrast, M1 f + s showed greater connectivity between the Control network and multiple subcortical regions, along with increased connectivity between the Dorsal Attention and Somatomotor networks and between the Default Mode network and both the putamen and Salient/Ventral Attention network.

Multimodal clustering: functional connectivity differences. (a) Shows the average Fisher z-transformed FC matrices for each migraine subgroup (M1f + s, M2f + s) and for the control group. Warmer colors reflect stronger positive connectivity, whereas cooler colors indicate weaker or negative connectivity. (b) Displays FC edges that differed significantly between the two migraine subgroups and between each subgroup and controls after covariate adjustment (age, sex, headache during scan) and FDR correction (q < 0.05). Values correspond to the β coefficients from the group-difference models. Positive coefficients indicate greater connectivity in M2f + s relative to M1f + s (or M1f + s/M2f + s compared to Control), whereas negative coefficients indicate the opposite. Cortical and subcortical boundaries are shown for interpretability.
Compared with controls, M1 f + s did not show any significant FC differences. In contrast, M2 f + s demonstrated increased cortical-subcortical connectivity relative to controls (Figure 2(b)). M2 f + s showed significantly stronger connectivity between the Dorsal Attention and Somatomotor networks and multiple subcortical regions, including the anterior and posterior thalamus, putamen, pallidum, and caudate. Additionally, M2 f + s exhibited higher connectivity between the Visual network and caudate compared to controls.
All FC differences remained significant after FDR correction and adjustment for age, sex, and headache during scan (Figure 2).
Structural profile
M2 f + s showed significantly lower cortical volumes than M1 f + s across several regions after adjusting for age, sex, headache during scan and applying FDR correction (FDR-p < 0.05) (Figure 3). These reductions spanned frontal regions (including medial, middle, and superior frontal territories and anterior cingulate), parietal sensorimotor areas (precentral, postcentral, paracentral, superior parietal, supramarginal, and precuneus), temporal cortex (fusiform, middle and superior temporal regions, temporal pole, transverse temporal), and the insula. A small but significant (FDR-p = 0.045) reduction in pars orbitalis thickness was also present in M2 f + s . There were no volumetric differences in any subcortical regions between the clusters, and no structural differences were identified when either cluster was compared with the control group (Figure 3).
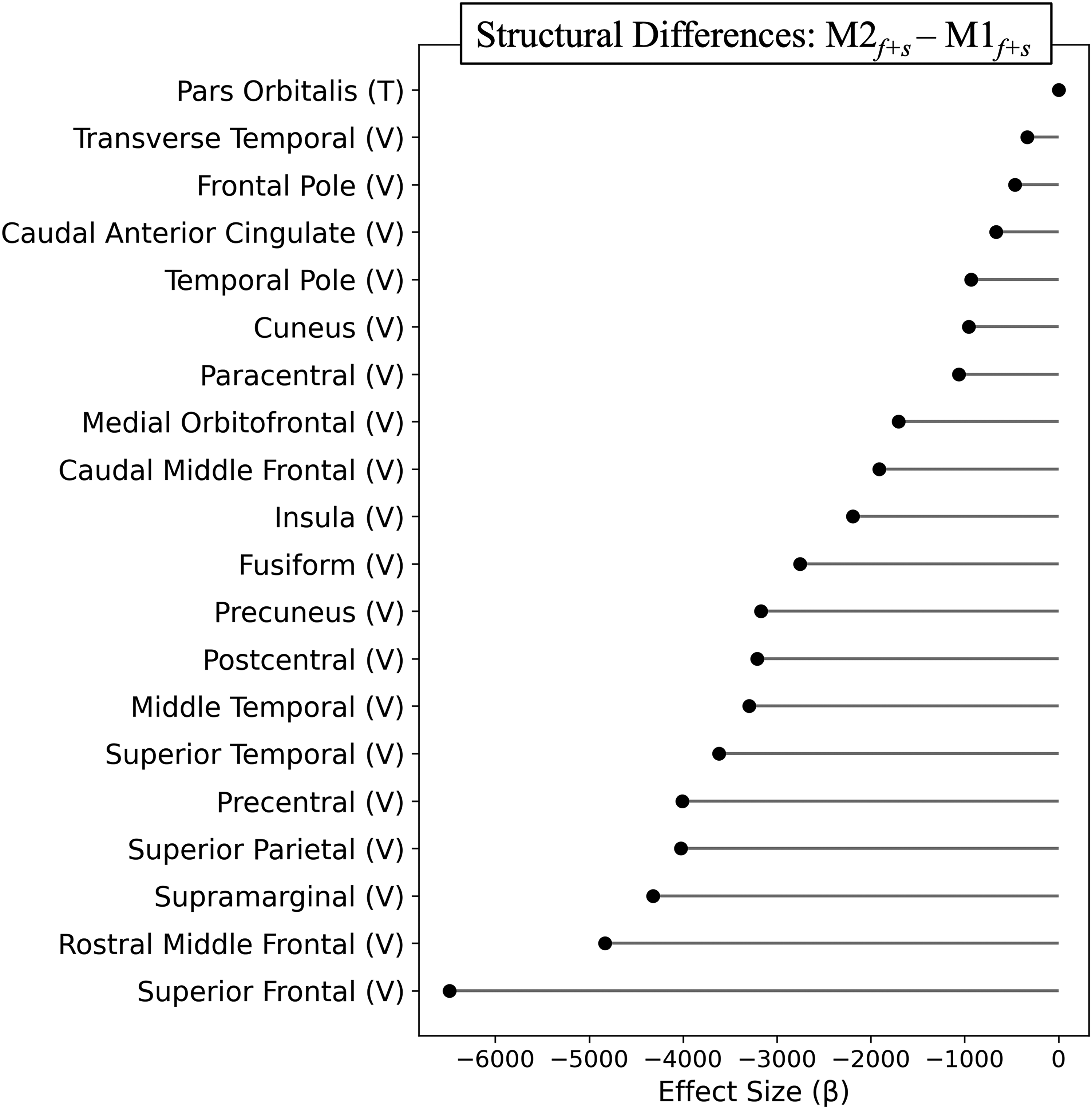
Multimodal clustering: structural differences between migraine subgroups. Shows regions with significant volumetric (V) and thickness (T) differences between the two multimodal migraine subgroups after adjusting for age, sex, and headache during scan. Points represent β estimates (M2f + s vs. M1f + s) with 95% confidence intervals; only FDR-significant regions (q < 0.05) are displayed.
Unimodal clustering
Functional-only clustering:
The functional-only clustering model, which used FC features reduced via PCA to 27 components explaining 80% of the variance, yielded an optimal two-cluster solution (silhouette = 0.117), resulting in subgroups M1 f (n = 35) and M2 f (n = 76) (Figure 1). The clinical profiles of these clusters broadly mirrored those identified in the multimodal model (Table 1): M2 f demonstrated longer headache duration (p = 0.05), greater migraine-related disability, more years with headache, and lower pain self-efficacy compared with M1 f , however, none of these differences survived FDR correction. The pattern of FC differences between M1 f and M2 f was also consistent with the multimodal clustering solution (Figure 4(a)), however, neither functional cluster showed meaningful FC differences relative to the control group. Similarly, no structural differences were observed either between the functional clusters or between clusters and controls. The correspondence between the functional-only and multimodal clustering solutions was ARI = 0.427 (Figure 4), Figure 5 (a).
Unimodal clustering: functional connectivity differences. (a) and (b) show significant FC differences identified in the functional-only and structural-only clustering models, respectively. In both models, group differences that remained significant after covariate adjustment (age, sex, headache during scan) and FDR correction (q < 0.05) are displayed. Values represent the β coefficients from the group-difference models. Positive coefficients indicate greater connectivity in M2 relative to M1 (or in the migraine subgroup relative to controls), whereas negative coefficients indicate the opposite. Cortical and subcortical network boundaries are overlaid to aid interpretability.
Structural-only clustering:
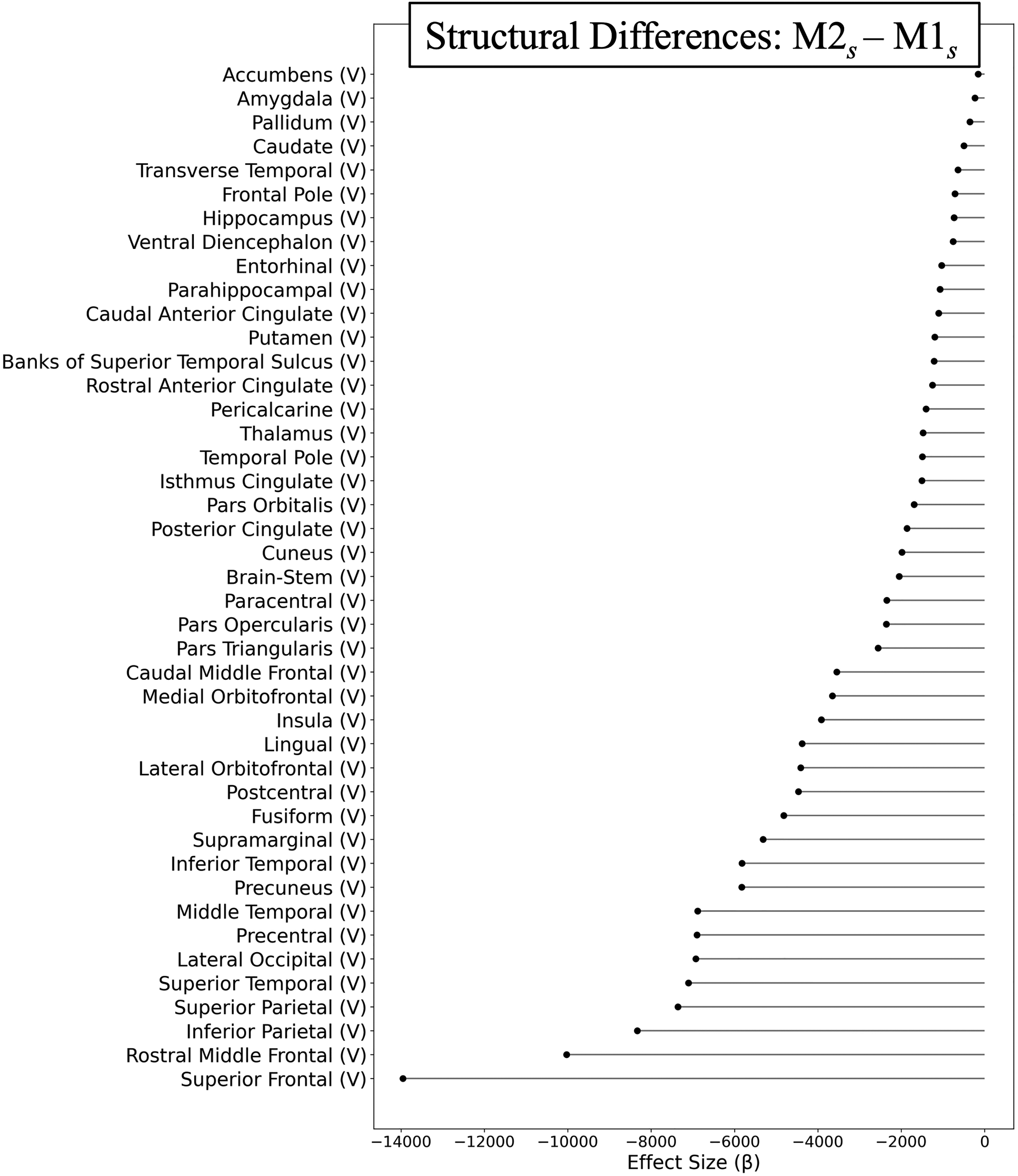
The structural-only clustering model, based on PCA retaining 11 components explaining 80% of the variance, yielded an optimal two-cluster solution (silhouette = 0.243), resulting in subgroups M1 s (n = 52) and M2 s (n = 59) (Figure 1). Clinically, the two clusters were largely similar, although M1 s was older (FDR-p < 0.01) and reported longer headache duration and higher headache frequency while M2 s reported higher rates of aura (Table 1). Structurally, M2 s exhibited widespread reductions in both cortical and subcortical volumes relative to M1 s after covariate adjustment and FDR correction Figure 6. When compared with controls, M1 s showed widespread increases in cortical volume and, subcortically, in the ventral diencephalon whereas the isthmus cingulate exhibited reduced cortical thickness (FDR-p < 0.05, Figure 7). Despite these structural distinctions, the structural-only clusters did not differ in FC either between clusters or relative to controls. The correspondence between the structural-only and multimodal clustering solutions was ARI = 0.001 Figure 5 (b).
Concordance between clustering solutions. (a–c) show adjusted Rand index (ARI) values and corresponding confusion matrices comparing the multimodal clustering solution with the functional-only model (a), structural-only model (b), and ROI-level sensitivity analysis (c). Each cell represents the percentage of participants in a multimodal cluster (rows) assigned to each alternate cluster (columns). Higher ARI values indicate greater agreement between clustering solutions.

Unimodal structural-only clustering: structural differences between migraine subgroups. This figure shows regions with significant cortical thickness (T) and volumetric (V) differences between the two migraine subgroups (M1s and M2s) identified in the structural-only clustering model. Results are adjusted for age, sex, and headache during scan. Points represent estimates (M2s vs. M1s) with 95% confidence intervals; only regions that remained significant after FDR correction (q < 0.05) are displayed.

Unimodal structural-only clustering: structural differences between M1s and controls. This figure shows regions with significant cortical thickness (T) and volumetric (V) differences between the M1s subgroup and the control group, adjusting for age, sex, and headache during scan. Points represent estimates (M1s vs. Control) with 95% confidence intervals; only regions that survived FDR correction (q < 0.05) are shown.
Sensitivity analysis
As a sensitivity analysis, the multimodal clustering was repeated using a finer functional parcellation. Some regions including the temporal pole, orbitofrontal cortex, mammillary nuclei, hypothalamus, and ventral pallidum were excluded due to insufficient signal coverage across participants. The final dataset included 103 participants, 95 functional cortical ROIs, and 38 functional subcortical ROIs. After concatenation with structural features and PCA reduction, 65 components explaining 80% of the variance were retained. Hierarchical clustering of these components yielded an optimal two-cluster solution (silhouette = 0.069) with cluster sizes of 42 and 61 participants (Online Supplementary Figure 1). The correspondence between this clustering solution and the primary multimodal clustering was ARI = 0.554 Figure 5 (c).
Discussion
This study aimed to identify imaging-based migraine subgroups using a data-driven clustering approach that integrated functional and structural measures across cortical and subcortical regions, and to examine their clinical, functional, and anatomical profiles. This multimodal model yielded two subgroups, M1 f + s and M2 f + s , that differed across clinical and imaging measures. M2 f + s was characterized by older age, longer disease duration, greater migraine-related disability, widespread increases in cortical–subcortical connectivity, prominently involving the Dorsal Attention, Somatomotor, and Visual networks, and reduced cortical volumes across frontal, parietal, temporal, and insular regions compared to M1 f + s . Many of these FC abnormalities were also elevated relative to controls, suggesting a more sensitized and highly integrated pattern of pain-related network organization. In contrast, M1 f + s showed preserved cortical structure, greater Control network connectivity to subcortical regions compared to M2 f + s , and no significant functional or structural differences from controls, consistent with a comparatively less clinically burdened profile.
The unimodal functional-only clustering yielded two subgroups (M1 f and M2 f ) whose clinical and functional profiles closely resembled those identified in the multimodal model, including the presence of a higher-burden group (M2 f ), but no structural differences emerged between these functional subgroups. In contrast, the structural-only model revealed widespread cortical and subcortical volumetric differences between its subgroups (M1 s and M1 s ) yet showed no accompanying functional distinctions. Together, these findings indicate that structural variation captured a different dimension of migraine heterogeneity than functional variation, with each modality highlighting unique aspects of variability in brain organization.
The findings of the present study closely parallel those reported in Krimmel et al., 6 who used canonical correlation analysis followed by k-means clustering to identify FC-based migraine biotypes. Similar to our results, Krimmel showed that network-level functional disruptions produced subgroups that aligned more strongly with dimensions of symptom burden than with conventional diagnostic categories such as episodic versus chronic migraine. By integrating subcortical regions and structural features, the current multimodal approach extends this earlier work and captures broader cortical–subcortical connectivity patterns that were not captured in Krimmel's FC-only framework.
The correspondence across clustering solutions further clarified the contribution of each modality. The functional-only model showed moderate agreement with the multimodal solution (ARI = 0.427), indicating that FC captures a substantial portion of the variance underlying the multimodal clusters. In contrast, the structural-only model showed almost no overlap with the multimodal solution (ARI = 0.001), suggesting that structural variation alone captures an orthogonal dimension of heterogeneity. Notably, the ROI-level multimodal clustering using finer functional parcellations showed the strongest concordance with the primary network-level multimodal solution (ARI = 0.554), with roughly 90–95% of participants assigned to the same cluster. This consistency demonstrates that the multimodal subgroups are robust across parcellation scales, even though parcellation granularity can influence subgroup structure. Fine-grained cortical parcellation, especially in orbitofrontal and anterior temporal regions, showed lower signal-to-noise ratio and missingness, which substantially reduced the usable sample size. For this reason, network-level cortical FC was used in the primary analysis to preserve statistical power, and the ROI-level approach was implemented as a sensitivity analysis.
The FC differences observed between clusters align with known migraine neurobiology. Enhanced Somatomotor–basal ganglia connectivity is consistent with prior studies21–23 linking basal ganglia dysfunction to pain modulation. Altered Default–Visual interactions resemble findings in migraine with aura 24 and without aura, 25 as well as chronic migraine, 26 supporting visual cortex hyperexcitability. Increased amygdala–thalamus connectivity aligns with limbic abnormalities widely reported in episodic and chronic migraine27,28 and supports thalamic dysregulation in central sensitization.29,30 The higher prevalence of allodynia in clinically burdened subgroups is consistent with Schwedt et al. 7 Unlike our previous findings from DeSouza et al. 31 in our cohort that emphasized amygdala-DMN and salience network interaction, our current results suggest more focal cortical-subcortical interactions, possibly due to differing subtyping methodologies.
Structural differences also aligned closely with existing literature. Reductions in frontal, parietal, temporal, and insular volumes are consistent with prior reports of decreased cortical volume in migraineurs5,32,33 and with chronic pain studies highlighting prefrontal cortex involvement, 34 as well as with observations of hypertrophy in selected migraine populations. 35 Cortical thinning in pain-related areas has also been documented. 36 Volume reductions in prefrontal and cerebellar areas have been shown to differentiate migraine from other headache types, 37 and machine-learning models have successfully leveraged frontal and temporal differences to classify migraine. 38 Multiple studies have also linked higher attack frequency to reduced frontal, limbic, and parietal volumes,33,37,39 consistent with the trends observed in our study, although Schwedt et al. 7 reported no frequency differences between their structural subtypes, underscoring the substantial neuroanatomical heterogeneity present across migraine populations.
Integrating these observations, M2 f + s represents a more clinically burdened profile, marked by widespread increases in cortical–subcortical connectivity and diffuse reductions in cortical volume. In contrast, M1 f + s reflects a comparatively compensated profile with network organization that more closely resembles controls. To further support this characterization, FC comparisons between M1 f + s and controls using a more liberal FDR threshold (q = 0.10), still revealed no significant difference, reinforcing that this subgroup's FC patterns remain broadly comparable to healthy individuals. These subgroup distinctions are descriptive and imaging-derived and should be interpreted with caution. Although M1 f + s showed higher rates of NSAID response, this finding is exploratory and requires confirmation in larger, prospective studies. Across all clustering solutions, the subgroups did not differ significantly on diagnostic measures, including episodic versus chronic migraine status, indicating that subgroup structure was not driven by conventional clinical categorization. While some clinical variables clearly differed between subgroups, these features were examined only post hoc and were not used to derive cluster membership. Accordingly, the present findings support the interpretation that multimodal neuroimaging captures patterns of heterogeneity that are associated with, but not reducible to, clinical features. Alternative approaches to subgrouping migraine based on clinical features alone have been explored in prior work. 40 However, the present study was not designed to compare clinical-only and imaging-based subgrouping approaches, and therefore conclusions are limited to describing how imaging-derived subgroups relate to observed clinical characteristics.
Several limitations should be acknowledged. The sample size was modest, and the cross-sectional design limits the ability to determine how these subgroups evolve over time. Although headache during scanning was included as a covariate, migraine phase (e.g., ictal vs interictal) and preventive medication use at the time of imaging were not systematically recorded, which may introduce state- or treatment-related variability. Clinical differences between clusters were subtle, underscoring the need for larger samples to improve statistical power and clarify the potential relevance of these imaging-derived subgroups. Future work should integrate larger cohorts, supervised classification approaches, longitudinal and phase-specific imaging, detailed medication tracking, and external validation to move neurobiologically informed migraine subtyping toward clinical application.
Conclusion
In conclusion, this study applied a data-driven integration of functional and structural MRI features to characterize heterogeneity within migraine. The multimodal clustering approach identified two migraine subgroups that differed in cortical–subcortical connectivity, cortical volume, and clinically relevant measures such as disease duration, and migraine disability. Importantly, functional connectivity emerged as a primary driver of subgroup differentiation, while structural variation contributed complementary information and highlighted anatomical heterogeneity. Together, these findings suggest that migraine encompasses distinguishable patterns of functional and structural brain organization. Further work is needed to evaluate the reproducibility, clinical relevance, and potential translational implications of these imaging-informed subgroups.
Article highlights
Data-driven multimodal neuroimaging-based clustering identified two distinct migraine subgroups with different clinical characteristics.
One subgroup showed widespread increases in cortical–subcortical functional connectivity and diffuse cortical volume reductions and was associated with greater symptom burden.
Functional connectivity contributed most strongly to subgroup differentiation, while structural variation provided complementary, orthogonal information, highlighting heterogeneity in brain organization within migraine.
Supplemental Material
sj-png-1-cep-10.1177_03331024261433982 - Supplemental material for Neuroimaging-based subtyping of migraine identifies clinically distinct phenotypes
Supplemental material, sj-png-1-cep-10.1177_03331024261433982 for Neuroimaging-based subtyping of migraine identifies clinically distinct phenotypes by Jaiashre Sridhar, Mahsa Babaei, Bharati M. Sanjanwala, Robert P. Cowan and Danielle D. DeSouza in Cephalalgia
Footnotes
Acknowledgements
We are grateful to the participants for their time and commitment to this research.
Ethical considerations
The study was approved by Stanford University's institutional review board (IRB).
Consent to participate
Written informed consent was obtained from all participants prior to data acquisition.
Author contributions
J.S.: Conceptualization, Data curation, Methodology, Formal analysis, Writing – original draft.
M.B.: Data Curation, Methodology, Writing – review & editing.
B.M.S.: Project administration, Data curation, Writing – review & editing.
R.P.C.: Conceptualization, Funding acquisition, Writing – review & editing.
D.D.D.: Conceptualization, Supervision, Writing – review & editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the SunStar Foundation to Robert P. Cowan.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: No conflicting interests for J.S., M.B., B.M.S. and D.D.D. R.P.C. is an advisor (unrelated to current study) for Alder, Amgen, Allergan, Biohaven, Curex, Teva, and Xoc.
Data availability statement
De-identified clinical and imaging data may be shared with qualified researchers upon request, following review and approval by Stanford University in accordance with institutional data sharing policies.
Supplemental material
Supplemental material for this article is available online.
