Abstract
Background
Glutamate has been implicated in migraine pathogenesis, and is elevated in the plasma, cerebrospinal fluid, and saliva in migraineurs. However, no comparison of glutamate levels among chronic migraine, episodic migraine and controls has been reported. The aim is to compare salivary glutamate levels of individuals with chronic migraine with those of individuals with episodic migraine and healthy controls.
Methods
We investigated salivary glutamate level of 46 women with chronic migraine, 50 women with episodic migraine, and 19 healthy controls via enzyme linked immunosorbent assay.
Results
The salivary glutamate level of the chronic migraine group (median and interquartile range, 20.47 [15.27–30.15] pmol/mg total protein) was significantly higher than those of the episodic migraine (16.17 [12.81–20.15] pmol/mg total protein,
Conclusions
Salivary glutamate level was elevated in chronic migraine participants. These data suggest that salivary glutamate level could be an indicator of CM
Introduction
Migraine is subdivided into both chronic migraine (CM) and episodic migraine (EM) subtypes, of which CM affects 1–5% of the general population (1,2). Recent studies suggest central sensitization plays a role in the pathogenesis of CM (3). Central sensitization refers to enhanced central nociception caused by an increase in spontaneous activity, a decrease in the response threshold, and an enhanced responsiveness to nociceptive stimuli that manifests as increased pain sensitivity (hyperalgesia) and pain response to non-nociceptive stimuli (allodynia) (4). Individuals with CM exhibit more hyperalgesia during the interictal period and more allodynia symptoms during the ictal period compared to individuals with EM (5).
Glutamate is the most abundant excitatory neurotransmitter in the central nervous system of vertebrates (6), and as such has been implicated in aspects of migraine pathogenesis including cortical spreading depression, trigeminal neuron activation, and central sensitization (7). Moreover, elevation of the glutamate level has been described in plasma, cerebrospinal fluid (CSF) and saliva samples from migraine patients during the ictal and interictal periods (8–11). Most of these previous studies did not differentiate between EM and CM, or alternatively diagnosed CM according to previous criteria. Of note, the criteria for CM were recently revised in the International Classification of Headache Disorders third edition beta version (ICHD-3 beta) (2).
Saliva has been used as a diagnostic tool to study activation of trigeminal nerves in disease conditions (10,12). Saliva contains a wide variety of neuropeptides due to salivary gland innervation by the nerve terminals of the trigeminovascular system, and could therefore provide certain clues about nervous system pathophysiology (13). In practical clinical terms, saliva is readily accessible, safer to handle, and collected noninvasively (14).
We hypothesized that salivary glutamate would be elevated in CM participants compared to EM participants or control subjects, ultimately demonstrating its potential as a clinical indicator of CM. The aim of this study is to compare salivary glutamate level of participants with CM with those of individuals with EM and healthy controls.
Methods
Participants
Participants were recruited from the neurology outpatient clinic of a university hospital between August 2015 and February 2016. Only women with EM or CM were included in the study. The participants a) were aged between 19 and 65 years; b) fulfilled the ICHD-3 beta criteria for EM (code 1.1 or 1.2) or CM (code 1.3); and c) for EM, provided a salivary sample at least 48 hours after the termination of a headache attack, and for CM, indicated a headache intensity of no more than mild. The exclusion criteria were as follows: a) secondary headache other than medication-overuse headache (MOH); b) chronic pain condition other than fibromyalgia (FM) which involves 30–60% of CM patients (15); and c) current medical or psychiatric treatment except for migraine. Fibromyalgia was diagnosed according to the 2010 American College of Rheumatology criteria (16). Medication-overuse headache was diagnosed according to the ICHD-3 beta (2).
Control subjects were recruited from women who visited for a routine check-up or who were accompanying patients. Subjects were eligible if they did not experience any form of headache during the previous year and did not report migraine or a probable migraine attack during their lifetime.
The study protocol was approved by the institutional review board/ethics committee of Hallym University Sacred Heart Hospital in Korea (IRB No. 2015-I083). All participants provided written informed consent.
Measurement of salivary glutamate
Saliva collection was performed as previously described (17). In brief, participants prepared the mouth by rinsing with tap water and expectorating. Sterile 2% citric acid (Sigma Aldrich, St. Louis, MO, USA) was applied to the sides and tip of the tongue with a cotton-tipped applicator to stimulate the sublingual glands to produce saliva. Citric acid was continually applied every 30 seconds until 5 mL of saliva was collected. The saliva collected in the first 2 minutes was discarded to prevent mixing of stimulated and unstimulated saliva. Following collection, samples were stored at −20℃ until glutamate measurement. To minimize the interference of dietary glutamate, samples were collected after at least 2 hours of fasting (18). Saliva collection was performed between 9:00 and 12:00 a.m. We set each saliva sample as 0.92 µg of total protein and assayed using a glutamate enzyme-linked immunoassay kit (Abnova, Taiwan) according to manufacturer specifications. The detection limit of the ELISA kit was 0.3 µg/ ml. The inter-assay and intra-assay coefficient variance were 13.2% and 8.0%, respectively.
Statistical analysis
The Kolmogorov–Smirnov test was used to test the normality of each distribution; for continuous variables with normal distributions, we used Student’s t-tests or an analysis of variance. For variables with non-normal distributions, we used Mann–Whitney
Results
Participant demographic and headache characteristics
Participant demographics and clinical characteristics.

Compared among controls, episodic migraine participants and chronic migraine participants.
Compared between episodic migraine participants and chronic migraine participants.
CM: chronic migraine, EM: episodic migraine, MOH: medication-overuse headache, SSRI: selective serotonin reuptake inhibitor.
Salivary glutamate levels
Salivary glutamate was significantly higher in the CM group (median and interquartile range, 20.47 [15.27–30.15] pmol/mg total protein) compared to the control group (12.18 [9.40–16.24] pmol/mg total protein, Box plots of salivary glutamate concentrations in the control, episodic migraine, and chronic migraine groups.
To evaluate the utility of salivary glutamate level as a marker for CM, ROCs were generated and the AUC for each ROC was calculated. The maximal Youden index for distinguishing CM participants from control subjects was achieved at a glutamate level of 16.58 pmol/mg total protein with an AUC of 0.769 (95% bootstrap confidence interval [CI], 0.647–0.890) (Figure 2(a)). Using this threshold, 69.6% of CM participants and 84.2% of control subjects were correctly assigned. The maximal Youden index for distinguishing CM participants from EM participants was achieved at a glutamate level of 17.94 pmol/mg total protein with an AUC of 0.657 (95% bootstrap CI, 0.544–0.796) (Figure 2(b)). Using this threshold, 67.4% of CM participants and 68.0% of EM participants were correctly assigned.
Receiver operating characteristic (ROC) curves for the chronic migraine and control groups (a) and for the chronic migraine and episodic migraine groups (b). The points for maximal area under the curve are highlighted with specificity and sensitivity values.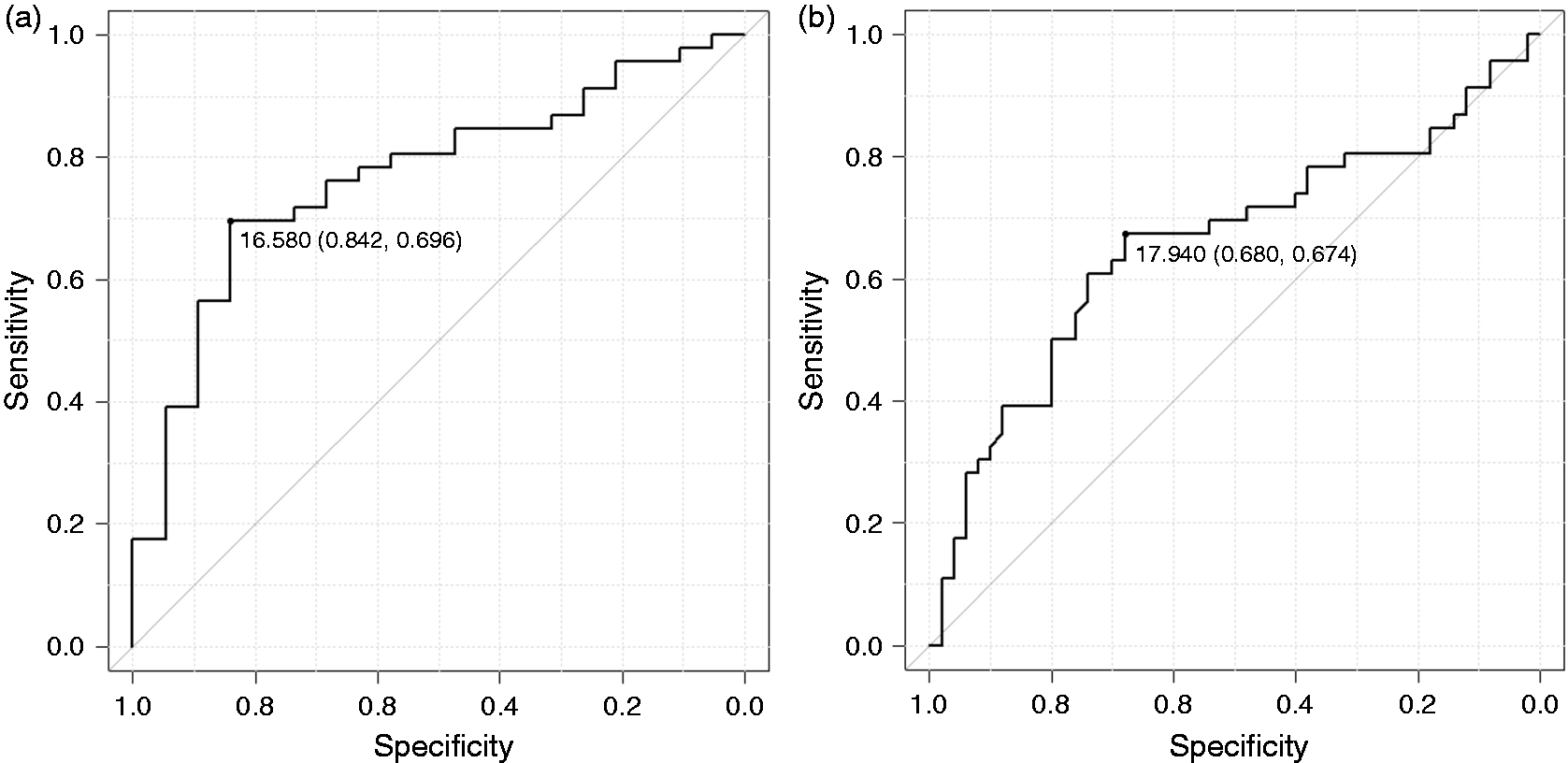
Salivary glutamate level in CM participants according to disease comorbidity or preventive treatment
Salivary glutamate was not significantly different between five CM participants with MOH (16.58 [12.73–44.82] pmol/mg total protein) and 41 CM participants without MOH (20.72 [15.36–19.63] pmol/mg total protein,
Discussion
The main findings of the present study were as follows: a) the salivary glutamate level of participants with CM was significantly higher than that of participants with EM or control subjects; b) the salivary glutamate level of participants with EM was marginally elevated from that of control subjects; and c) the salivary glutamate level of participants with CM did not differ significantly according to the presence of MOH or FM, or current preventive treatment.
Increased salivary glutamate level among CM participants indicates a dysfunction of the glutamatergic system may exist in CM. An excess of an excitatory neurotransmitter, glutamate, may reflect a neuronal hyperexcitability in CM. Neuronal hyperexcitability has been proposed as an underlying mechanism of CM (19). Accordingly, the present study data may add an evidence of neuronal hyperexcitability in CM. Future studies should examine the glutamatergic system as the diagnostic and therapeutic target of CM.
Several studies have evaluated glutamate concentrations in the bodily fluids of individuals with migraine (8–11); however, these studies did not separately investigate glutamate level in EM patients. Here, we firstly found that salivary glutamate was higher in CM patients compared to EM patients and control subjects. Our analyses demonstrated that salivary glutamate level could be utilized as a biomarker to differentiate patients with CM from control subjects with moderately accurate sensitivity and specificity (Figure 2(a)).
Glutamate level of EM participants was marginally elevated from that of controls but significantly lower than that of CM participants in the present study. The sensitivity and specificity of differentiating between the CM and EM groups based on ROC were lower compared to the corresponding values for differentiating between the CM and control group. These findings suggest that the glutamate level was elevated to some degree in participants with EM, but was significantly lower than that in participants with CM (Figure 2(b)) Our observations were in agreement with the previous report that central sensitization occurred in some individuals with EM but the degree was less than that in the individuals with CM (20).
The saliva collection method must be standardized throughout the collection period. We used stimulated saliva for better control of saliva. Unstimulated saliva is more variable than stimulated saliva, since a wide range of stimuli can stimulate salivary gland secretion to a level above the normal basal state (21). Furthermore, sufficient and consistent quantities can be collected in a shorter period than the amount obtained using unstimulated saliva (22). We used gustatory stimuli using 2% citric acid. This procedure primarily stimulates submandibular and sublingual glands, which are innervated by the trigeminal nerve. Elevation of calcitonin-gene related peptide and vasoactive intestinal peptide levels was successfully investigated in migraineurs using 2% citric acid-stimulated saliva (12).
What are possible sources for the elevated salivary glutamate level in CM? One possible explanation is that the elevated salivary glutamate is attributable to elevated plasma glutamate. Elevated plasma glutamate has been reported in migraineurs. Constituents of plasma enter saliva through passive diffusion, active transport, or extracellular ultrafiltration (14). Thus, salivary glutamate may originate from plasma glutamate (8,23,24). The main sources of plasma glutamate are neurons and platelets, and elevation of glutamate in the brain and platelets has been noted in migraineurs (8,9,11,25). Migraine attacks may cause a permeability change of the blood-brain barrier (BBB), and glutamate transfer from the brain to plasma may occur. Excessive neuronal glutamate increases the permeability of BBB via N-Methyl-D-aspartic acid (NMDA) receptor activation (26). A disrupted BBB was observed in an episode of familiar hemiplegic migraine, a genetic variant of migraine with aura (27). In contrast, dynamic contrast-enhanced magnetic resonance imaging showed no significant change in the permeability of BBB during spontaneous migraine attacks (28). Therefore, BBB permeability change during migraine attacks is currently uncertain. Nevertheless, plasma glutamate is positively correlated with glutamate in CSF in healthy volunteers, and other mechanisms may play a role in the close association between plasma and CSF glutamate levels (29). The mechanism for elevated platelet glutamate in migraine is still unknown. Another possible explanation is that glutamate originates from the activated trigeminal nerve. Salivary glands are innervated by nerve terminals of the trigeminal nerve. Glutamate is involved in the activation of the trigeminal nerve (30) and activation of the trigeminal nerve in CM was observed. Therefore, glutamate from the activated trigeminal nerve may contribute to increased salivary glutamate in CM.
Accumulating evidence suggests that glutamate and its receptors plays major roles in central sensitization. Calcium entry through NMDA receptors activates intracellular kinase, which ultimately leads to post-translation changes such as phosphorylation of membrane receptors and ion channels (31). These changes lead to increased excitability and to recruitment of neuronal response to normally subthreshold inputs. A NMDA receptor antagonist, memantine, effectively reduces the nociceptive response after experimental pain in mice (32). FM is a well-known state of central sensitization, and elevated CSF glutamate has been noted in FM. Therefore, elevated salivary glutamate level in CM may reflect an elevated neuronal glutamate level and an enhanced central sensitization state in CM is proposed.
Glutamate is widely found in foods, and the plasma glutamate level is affected by dietary glutamate. After ingestion of glutamate, the plasma glutamate level reaches a peak between 30 minutes and 1 hour later, and returns to the basal level after between 1 and 1.5 hours (18). Although no data are available on the effect of dietary glutamate on the salivary glutamate level, we collected saliva samples after at least 2 hours of fasting to avoid any effect of dietary glutamate on the results. Further studies on the effect of dietary glutamate on the salivary glutamate level should be conducted to evaluate the diagnostic utility of salivary glutamate level.
Previous studies have demonstrated that plasma glutamate level is not different between men and women (33,34). Nonetheless, several studies have reported sex-based difference in the plasma glutamate level under some conditions (19). Therefore, we enrolled only women in the present study to avoid sex-related difference in the salivary glutamate level.
We included CM participants with FM as a comorbidity and excluded other chronic pain conditions. Fibromyalgia is a chronic pain syndrome, and affects 30–60% of individuals with CM (15). Peres et al. reported that glutamate level in the CSF were significantly higher in CM participants with FM than in those without FM (9). Accordingly, we assumed that the presence or absence of FM may affect the salivary glutamate level of individuals with CM. However, in the present study, we found that the salivary glutamate level was not significantly different among CM participants based on FM status.
In the present study, the number of healthy controls was relatively small compared to that of CM participants. Considering the mean and standard deviations of glutamate level for healthy controls (13.32 ± 7.02 pmol/mg total protein) and CM (24.05 ± 14.58 pmol/mg total protein) participants, the estimated power for comparing two groups was 84.6%. Therefore, the present study had sufficient sample size for comparing glutamate levels between controls and CM groups.
Even information regarding salivary glutamate in neurological disorders other than migraine is scarce: An increase in plasma glutamate has been documented in ischemic stroke, epilepsy, amyotrophic lateral sclerosis and others (35). Although the frequency of migraine was not reported in these studies, elevation of salivary glutamate may not be specific to CM. Therefore, salivary glutamate could be used for assessing disease status or effect of treatment rather than screening CM. Plasma glutamate decreased after successful prophylactic treatment of migraine, regardless of prophylactic medications (8).
The present study had several limitations. First, we recruited a limited number of control subjects and migraine participants from a single university hospital; therefore, our findings may lack generalizability to larger or more diverse populations. Second, we did not include chronic tension-type headache or new daily persistent headache, which are two common subtypes of chronic headache. Future studies should be conducted on larger populations of patients with various subtypes of chronic headaches. Thirdly, we compared the glutamate level of CM participants according to various conditions such as preventive treatment, use of anti-glutamatergic medications, comorbidities with MOH or FM. However, these analyses did not have sufficient powers for comparison. Calculated statistical powers by preventive medication, MOH, FM and use of anti-glutamatergic medications were 0.074, 0.056, 0.075 and 0.130, respectively. Nevertheless, we presented these results to provide more information on the glutamate levels of CM participants according to various conditions. Therefore, further studies with sufficient sample numbers for comparison of glutamate levels among CM participants according to these conditions are needed. Fourthly, we did not separately enrol CM participants whether they were headache-free or having a mild headache, and collected saliva of CM participants when their headache intensity was no more than mild. Owing to continuous or near-continuous headache in CM, individuals with CM often had difficulty in reporting whether they were headache-free or had mild headache, and some neuropeptide studies recruited participants regardless of headache status (17,36). Nevertheless, neuropeptide levels in individuals with CM may vary between the headache-free state and ictal state (37). Further studies on CM assessing glutamate level according to exact headache status are needed. Lastly, we did not evaluate the duration of migraine or CM. Disease duration is associated with brain damage in migraine (38). Therefore, it can affect the glutamate level of CM. Further study for the association between glutamate level and disease duration of CM will be needed.
As a strength, our study diagnosed EM and CM according to the most recent ICHD criteria. Additionally, we assessed glutamate levels in CM participants according to common clinical presentations, including comorbidity with MOH and FM, and in participants receiving preventive treatment. Balancing the limitation and strengths of our work, we can conclude that salivary glutamate of participants with CM was elevated compared to that of participants with CM and healthy controls. Our findings suggest that salivary glutamate level could be an indication for CM.
Footnotes
Article highlights
Salivary glutamate level of individuals with chronic migraine was significantly higher compared to that of individuals with episodic migraine or controls.
Salivary glutamate level of individuals with episodic migraine was marginally elevated from that of healthy controls.
Salivary glutamate level could be an indicator of chronic migraine.
Author contributions
JN designed the study, contributed to laboratory measurements, and helped to draft the manuscript. HL contributed to laboratory measurements, study design, and assisted with the writing of the manuscript. JK and JK analysed data, and assisted with the writing of the manuscript. MC contributed to the conceptualization and design of the study, collection of the clinical data, analysis and interpretation of the results, and writing of the manuscript.
Acknowledgments
We thank the Department of Biostatistics, Clinical Trial Center, Biomedical Research Institute, Pusan National University Hospital.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ji Hye Nam is an assistant research engineer on the immunological analysis team at Abion-CRO. Hun Seok Lee is a senior researcher at Seoul National University and a team manager of the immunological analysis team at Abion-CRO. In the past 24 months, Min Kyung Chu, MD, PhD was involved as a site investigator for a multicenter trial sponsored by Eli Lilly, worked an advisory member for Teva, and received lecture honoraria from Allergan Korea and Yuyu Pharmaceutical Company.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Hallym University Research Fund 2016 (HURF-2016-16).
