Abstract
Background
Few studies have examined plasma calcitonin gene-related peptide (CGRP) levels in individuals with vestibular migraine (VM), with inconsistent findings. Additionally, salivary CGRP levels in VM have not been reported.
Methods
Interictal plasma and salivary CGRP levels were measured using an enzyme-linked immunosorbent assay in participants with VM corresponding to episodic migraine (VM, n = 81), chronic migraine without vestibular symptoms (CM, n = 73) and healthy controls (HC, n = 59).
Results
Plasma CGRP levels in VM with episodic migraine participants were significantly lower than those in participants with CM (median = 37.1 pg/mL, interquartile range (IQR) = 22.4–60.4 pg/mL vs. median = 74.6 pg/mL, IQR = 49.6–101.6 pg/mL; p < 0.001) but did not significantly differ from levels in HC. Similarly, salivary CGRP levels were also significantly lower in VM compared to CM (median = 54.5 pg/mL, IQR = 37.0–83.4 pg/mL vs. median = 72.0 pg/mL, IQR = 56.5–96.2 pg/mL; p = 0.036), with no significant difference observed between VM and HC. Receiver operating characteristic analysis showed that plasma CGRP levels effectively differentiated CM from VM corresponding to episodic migraine, achieving an area under the curve of 0.88. No significant correlations were found between plasma or salivary CGRP levels and clinical features of CM and VM.
Conclusions
Interictal plasma and salivary CGRP levels are unlikely to serve a biomarkers for VM.
This is a visual representation of the abstract.
Introduction
The link between migraine and vertigo has been acknowledged since ancient Greece (1). The term “vestibular migraine” (VM), first introduced in 1999, is now recognized as a distinct subtype of migraine that predominantly affects the vestibular system, essential for balance and spatial orientation (2). VM is the second most common cause of recurrent vertigo, representing 6–12.5% of patients in dizziness clinics and 9% of patients in migraine clinic cohorts (2,3). Criteria for VM diagnosis were jointly established by the International Headache Society and Bárány Society, which were then incorporated into the International Classification of Headache Disorders (ICHD-3) in 2012 (4,5).
Vestibular symptoms present in up to 60% of migraine patients (6). Chronic migraine (CM), defined as headaches occurring on 15 or more days per month over three months, is often associated with a higher prevalence of comorbid conditions, including vertigo. Both CM and VM typically present with severe headaches, photophobia or phonophobia, audiovestibular symptoms, and occasionally visual disturbances. Vestibular symptoms may occur with or without concurrent headaches, and the frequent presence of these symptoms in CM can complicate diagnosis due to overlapping diagnostic criteria (7). Although diagnostic tools such as brain imaging and vestibular function tests are frequently employed, they are often inadequate for distinguishing CM from VM, especially given the diverse and overlapping symptomatology of VM.
Calcitonin-gene-related peptide (CGRP) is a neuropeptide known for its vasodilatory and nociceptive properties and has been implicated in the pathophysiology of migraine (8). CGRP infusion induces migraine-like headaches, and elevated CGRP levels during migraine attacks are well documented (9). Increased interictal CGRP levels have been detected in individuals with CM and distinguish CM from EM (10). Following successful acute treatment, the increased blood CGRP found during a migraine attack decreases, but the decline is slower when treatment is ineffective (11). These findings have led to the consideration of interictal blood CGRP level as a potential biomarker for CM (12,13). In addition to its established role in migraine pathophysiology, CGRP also plays a crucial role in the functioning of the vestibular system (14). CGRP-immunoreactive efferents have been identified in both the maculae and cristae, where they form direct contacts with afferents surrounding type I vestibular sensory cells (15,16). Studies on CGRP knockout mice demonstrated a significant reduction (50%) in vestibulo-ocular reflex gain, underscoring the importance of CGRP in vestibular function (17). Moreover, treatment with anti-CGRP monoclonal antibodies not only alleviates headache symptoms, but also has been shown to significantly reduce vestibular symptoms in migraine patients, suggesting CGRP's influence on both systems (18). These observations raise the possibility that plasma CGRP levels may reflect vestibular system involvement in disorders like VM. Considering these reports, it is plausible that plasma CGRP levels in VM, particularly with episodic migraine, previously reported to be lower than in CM, might show alterations.
However, research on CGRP levels in individuals with VM is limited and has produced inconsistent results (19,20). The non-invasive nature of saliva collection and the established presence of CGRP in human saliva, which suggests its role in trigeminovascular activation in migraine (21), make saliva a promising medium for monitoring neuropeptide biomarkers like CGRP (22). Given this background, we hypothesized that interictal CGRP levels might be distinctly altered in individuals with VM corresponding to episodic migraine, potentially serving as a unique biomarker for this condition. The present study aims to assess both plasma and salivary CGRP levels in patients with VM with episodic migraine frequency and CM during interictal periods, compared to healthy controls, to explore CGRP's potential as a biomarker for VM.
Methods
Participants
Participants with VM and CM were recruited at the Headache and Dizziness Clinic of Jeonbuk National University Hospital from May 2022 to September 2024. Eligibility for the VM group required a diagnosis according to the criteria established by International Classification of Headache Disorders, 3rd edition (ICHD-3) and the Bárány Society, with participants aged between 20 and 70 years (4). The CM participants had to meet the ICHD-3 criteria for CM (code 1.3) within the same age range (4,5). Healthy controls (HC), recruited through advertisements, were eligible if they had not experienced headache attacks in the past year and had no history of migraine. Exclusion criteria included abnormalities on neurological examination, a history of vestibular disorders other than VM, medication-overuse headache, use of anti-CGRP monoclonal antibody or CGRP receptor antagonists, or concurrent medical or psychiatric conditions. To ensure diagnostic clarity, participants with chronic headache disorders and dizziness symptoms meeting both CM and VM criteria were excluded. Consequently, we included only VM participants corresponding to EM, defined as a headache frequency of fewer than 15 days per month, while those corresponding to CM, defined as a headache frequency of 15 or more days per month, were excluded. Exclusion criteria also included pregnancy and individuals who declined participation.
Clinical assessments
Clinical assessments included scores of the Headache Impact Test-6 (HIT-6), Dizziness Handicap Inventory (DHI) (23), Patient Health Questionnaire-9 (PHQ-9) (24), Generalized Anxiety Disorder-7 (GAD-7) scale (25), and visual analog scale (VAS) for pain and vertigo. Additional clinical data, including headache frequency, headache duration, severity, aura, quality of headache (pulsating or non-pulsating) and side of headache (unilateral or bilateral), were collected. Information on vestibular symptoms, such as the frequency and duration of vertigo episodes and the presence of nausea or vomiting and other autonomic symptoms, was also gathered. Information on the use of medications for acute and prophylactic management of headache and vertigo was collected. Medications used to manage acute migraine included triptans, non-steroidal anti-inflammatory drugs, and combination analgesics. Prophylactic medications included antidepressants, anti-epileptic drugs, beta blockers, calcium channel blockers and onabotulinumtoxinA (BOTOX®; AbbVie, North Chicago, IL, USA).
Assessment of CGRP level
Sample collection and storage
To ensure accurate CGRP level measurements, we conducted sample collection during interictal periods: 72 hours after the cessation of typical attacks. For VM, sample collection was performed in the absence of headache and vestibular symptoms, whereas sampling for CM was conducted at least two days after the typical migraine episode had resolved, ensuring that participants were either headache-free or experiencing only mild headache at the time of sampling. To minimize the impact of circadian rhythms on CGRP levels, all samples were collected in the morning, between 9.00 am and 11.00 am. Participants continued their usual prophylactic medication regimen. Venous blood was collected from participants from the antecubital vein into 3 ml EDTA tubes (V-TUBE™; AB Medical, Inc., Seoul, Korea). The samples were centrifuged at 3000 rpm (~1006
For saliva collection, participants were instructed to fast for two hours prior to minimize potential contaminants. The collection process began with an initial mouth rinse with tap water, followed by stimulation of salivation through the application of 2% citric acid to the tongue. Early saliva samples were discarded to maintain consistency, with citric acid stimulation repeated every 30 seconds until 5 ml of saliva was collected. Samples were immediately stored at −80°C to preserve their integrity for enzyme-linked immunosorbent assay (ELISA) analysis (27). To ensure CGRP level stability, all plasma samples were analyzed within one month of storage. We also conducted comparative analyses of CGRP concentrations across different storage durations, confirming that there were no significant differences related to the timing of sample collection.
ELISA procedure
Plasma and salivary CGRP levels were determined using a commercial ELISA kit (CGRP ELISA Kit #A05481; Bertin, Montigny-le-Bretonneux, France), which utilizes a double-antibody sandwich method ensuring complete cross-reactivity with human α- and β-CGRP forms to measure total CGRP concentration. The procedure begins with a 96-well plate pre-coated with mouse monoclonal antibodies targeting the N-terminal of CGRP. CGRP tracer antibodies, which bind the C-terminal and are conjugated with acetylcholinesterase, are used to convert Ellman's reagent into a yellow ionized product that is measurable. The assay's sensitivity is calibrated to detect concentrations as low as 2 pg/mL. For assay preparation, 100 µl of each coded and anonymized plasma or saliva sample was applied to the appropriate wells. CGRP-free plasma, essential for standard curve calibration, was prepared using CGRP Affinity Sorbent (#A19482; Bertin). This sorbent removes CGRP from pooled plasma samples, which were then used to reconstitute the lyophilized CGRP standard and prepare serial dilutions as outlined in the Bertin ELISA kit manual.
Post-application, the plate underwent washing before the addition of CGRP standards, tracer solutions, and samples. The plate was incubated at 4°C for 16–20 hours, followed by another washing step. Ellman's reagent was then added, and the plate's absorbance was measured at 405 nm to determine CGRP concentrations, adjusted for sample dilution, and reported as pg/mL.
Statistical analysis
Categorical variables are summarized as frequencies and percentages (number of cases/total number in group, %). For continuous variables, normality was assessed using the Kolmogorov–Smirnov test. If normally distributed, continuous variables are reported as the mean ± SD, whereas non-normally distributed variables are expressed as median and interquartile range (IQR). Chi-squared tests were used to analyze categorical variables, including demographic and clinical characteristics. For comparisons of continuous variables, analysis of variance (ANOVA) or analysis of covariance (ANCOVA) was applied when parametric assumptions were met. If assumptions were violated, Quade's non-parametric ANCOVA was used. In particular, plasma and salivary CGRP levels were compared using Quade's non-parametric ANCOVA, adjusting for sex, age and scores on the GAD-7 (anxiety) and PHQ-9 (depression) scales. As an observational study, this analysis did not have a predefined sample size based on statistical power calculations. CGRP levels, treated as non-parametric data, are presented as median with IQR, and differences between groups were assessed using the Kruskal–Wallis test. Post-hoc pairwise comparison and Bonferroni correction for multiple comparisons were performed. p < 0.05 was considered statistically significant. All data analyses were performed using SPSS, version 29.0 (IBM Corp., Armonk, NY, USA), except the receiver operating characteristic (ROC) curve used to determine the CGRP levels in plasma and saliva that best differentiated CM and VM, which were conducted using Prism, version 9.5.1 (GraphPad Software Inc., San Diego, CA, USA).
Ethical approval and patient consent
This prospective observational cohort study received approval and oversight from the Institutional Review Board of Jeonbuk National University Hospital (IRB No. 2021-12-012-002). All participants provided their written informed consent. All methods were carried out in accordance with relevant guidelines and regulations including the Declaration of Helsinki and its following amendments.
Results
Demographic and clinical characteristics
This study evaluated CGRP levels in plasma and saliva among three groups: 81 with VM, 73 patients with CM without vestibular symptoms and 59 healthy controls (Table 1). The age and sex distributions did not differ significantly among these groups.
Demographic and clinical characteristics of patients with chronic or vestibular migraine and healthy controls.

Categorical variables were analyzed using the chi-squared test; parametric quantitative variables were analyzed using ANOVA; non-parametric quantitative variables were analyzed using the Mann–Whitney U test. IQR = interquartile range; ANOVA = analysis of variance.
Regarding clinical characteristics, headache frequency was significantly higher in the CM group compared to the VM group corresponding to EM (median = 17.0 days/month, IQR = 15.0–21.0 days/month vs. median = 10.0 days/month, IQR = 7.0–12.5 days/month, p < 0.001). CM patients also experienced more frequent migraine attacks compared to VM patients (median = 11.5 days/month, IQR = 9.0–15.0 days/month vs. median = 9.0 days/month, IQR = 4.3–12.0 days/month, p = 0.004). However, headache severity, duration, and frequencies of pulsating pain, photophobia and phonophobia did not differ significantly between CM and VM groups. There was also no significant difference in disease duration or headache impact (HIT-6 scores) between the two groups. The VM group reported significantly higher levels of depression (PHQ-9: median = 8.0, IQR = 5.0–13.0 vs. median = 5.0, IQR = 2.0–7.0, p < 0.001) and anxiety (GAD-7: median = 7.5, IQR = 3.3–13.3 vs. median = 1.5, IQR = 0–4.25, p < 0.001) than the CM group.
Treatment modalities differed significantly between the CM and VM groups (Table 1). A higher proportion of CM patients used acute analgesics compared to VM patients (71.2% vs. 49.4%, p = 0.006) and they also used these medications more frequently (median = 6.0 days/month, IQR = 3.0–17.0 days/month vs. median = 1.0 days/month, IQR = 0–6.5 days/month, p = 0.001). Among the types of acute analgesics, CM patients were more likely to use triptans (27.4% vs. 11.1%, p = 0.010) and combination analgesics (21.9% vs. 7.4%, p = 0.010) than VM patients. Additionally, a significantly larger proportion of CM patients used prophylactic medications compared to VM patients (79.5% vs. 63.0%, p = 0.025), with anti-epileptic drugs being more commonly prescribed to CM patients (72.6% vs. 49.4%, p = 0.003).
Plasma and salivary CGRP levels
Plasma and salivary CGRP levels varied significantly among the groups (Figure 1 and Table 2). Plasma CGRP levels were notably higher in the CM group compared to the VM group (median = 74.6 pg/mL, IQR = 49.6–101.6 pg/mL vs. median = 37.8 pg/mL, IQR = 22.4–60.4 pg/mL, p < 0.001) and HC (median = 37.5 pg/mL, IQR = 27.7–57.4 pg/mL, p < 0.001). Similarly, salivary CGRP levels were significantly elevated in the CM group relative to HC (median = 72.0 pg/mL, IQR = 56.5–96.2 pg/mL vs. 40.9 pg/mL, IQR = 34.9–71.0 pg/mL, p = 0.001) and the VM group (median = 54.5, IQR = 37.0–83.4 pg/mL, p = 0.036). Notably, there was no significant difference in CGRP levels between the VM group and HC for either plasma or saliva.

Comparisons of plasma (a) and salivary (b) concentrations of CGRP among VM, CM and HC groups. Data are presented as the median (IQR). (a) The plasma CGRP level was found to be significantly greater in the CM group than in the VM corresponding to EM (p < 0.001) and HC groups (p < 0.001). (b) The salivary CGRP concentration was significantly greater in the CM group than in the VM corresponding to EM (p = 0.036) and HC group (p = 0.001).
Comparison of interictal CGRP concentrations between patients and control groups.

CGRP = calcitonin gene-related peptide; KW = Kruskal–Wallis test; W = Wilcoxon signed rank test; IQR = interquartile range; CM = chronic migraine; VM = vestibular migraine; HC = healthy controls.
*Adjusted by Bonferroni correction for multiple comparisons.
The diagnostic potential of CGRP levels for distinguishing CM from VM was further assessed via ROC curve analysis (Figure 2). Plasma CGRP levels displayed a significant discriminative ability to differentiate CM from VM corresponding to EM, with an area under the curve (AUC) of 0.88 (95% confidence interval (CI) = 0.80–0.96; p < 0.001). Salivary CGRP levels had a more moderate discriminative capacity, with an AUC of 0.65 (95% CI = 0.53–0.79; p = 0.015). The Youden index indicated an optimal cutoff value of 39.7 pg/mL for plasma CGRP, yielding 96.3% sensitivity and 76.2% specificity, whereas a cutoff of 48.4 pg/mL for salivary CGRP achieved 86.4% sensitivity and 50.0% specificity.

Receiver operating characteristic curve analyses for plasma (red line) and salivary (purple line) CGRP levels in CM and VM corresponding to EM participants. Plasma CGRP levels (red line) demonstrated a high discriminatory ability for differentiating CM from VM, with an AUC of 0.88 (95% CI = 0.80–0.96), indicating significant effectiveness as a diagnostic marker (p < 0.001). Salivary CGRP levels (purple line) showed a more limited ability to differentiate between CM and VM, with an AUC of 0.65 (95% CI = 0.53–0.79), suggesting lower discriminatory capability compared to plasma CGRP. AUC = area under the curve; CGRP = calcitonin-gene-related peptide; CI = confidence interval; CM = chronic migraine; EM = episodic migraine; HC = healthy control; VM = vestibular migraine.
Correlations between plasma and salivary CGRP levels and clinical features
The correlations between plasma and salivary CGRP levels and various clinical features, including headache and migraine frequency, headache duration, headache severity, vertigo frequency, vertigo episode duration, intensity of vertigo, monthly analgesic use, HIT-6 score, PHQ-9 score, GAD-7 score and disease duration, were analyzed using Spearman's correlation (Table 3). The results showed no significant correlations between plasma and salivary CGRP levels and any of these migraine, vestibular, and psychiatric features in both the CM and VM groups, as well as in a combined analysis.
Spearman's rank correlation coefficients for correlations between saliva, plasma CGRP levels and clinical characteristics of vestibular and chronic migraine participants.
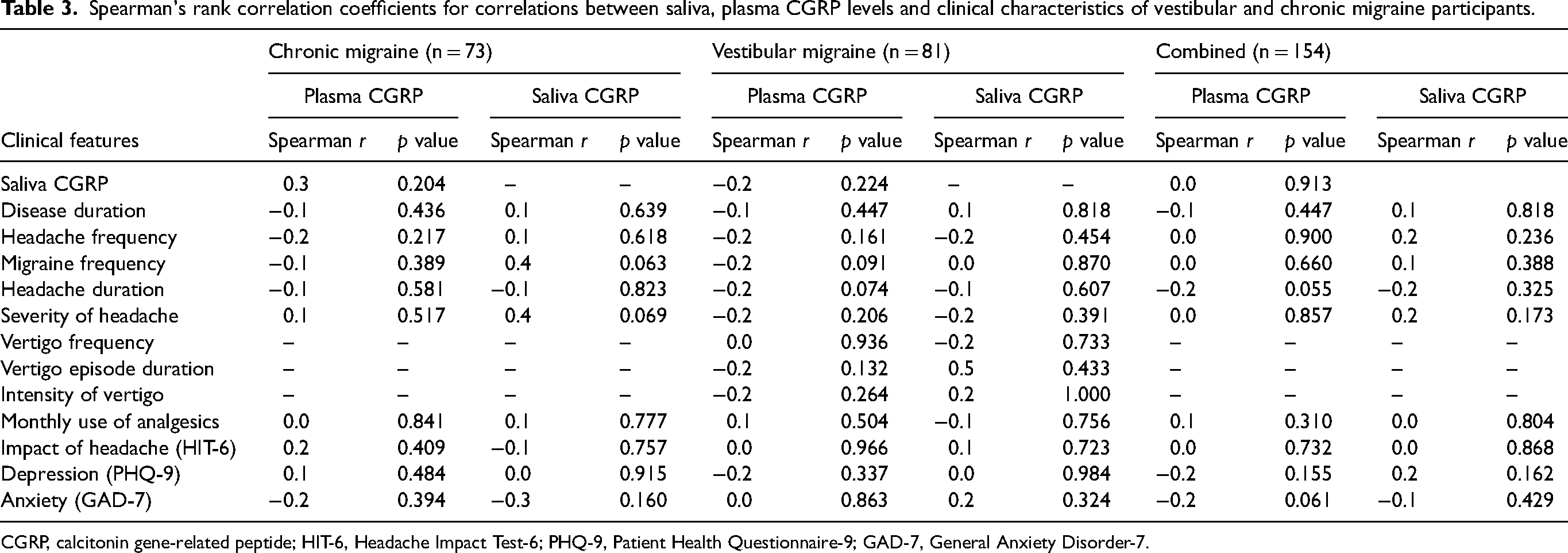
CGRP, calcitonin gene-related peptide; HIT-6, Headache Impact Test-6; PHQ-9, Patient Health Questionnaire-9; GAD-7, General Anxiety Disorder-7.
In addition, inner-individual correlations between plasma and salivary CGRP levels were assessed separately in the CM group, VM group, and a combined cohort. The scatter plots revealed no significant correlation between plasma and saliva CGRP levels within subjects across all groups, indicating a lack of direct association between plasma and saliva measurements within individuals in the context of CGRP levels (Figure 3).

Inner-individual correlation between plasma and salivary CGRP levels in CM, VM and combined groups. Scatter plots of plasma versus salivary CGRP levels in subjects with CM, VM corresponding to EM, and combined groups. No significant correlations are observed within individuals across all groups, as indicated by linear regression lines. CGRP = calcitonin-gene-related peptide; CM = chronic migraine; EM = episodic migraine; HC = healthy control; VM = vestibular migraine.
Discussion
The major findings of the present study were as follows: (i) the interictal plasma and salivary CGRP levels in participants with VM corresponding to EM were not significantly different from those in HC; (ii) interictal plasma and salivary CGRP levels were significantly elevated in participants with CM compared with both VM participants and HC; and (iii) there was no significant association between headache and vestibular characteristics of CM and VM and either plasma or salivary CGRP levels.
The pathophysiology of VM is not well understood but is considered to involve mechanisms similar to those involved in migraine. Interactions among the trigeminovascular system (TVS), nociceptive brainstem centers, thalamocortical networks and the vestibular system may underlie VM symptoms (28). Evidence from animal models suggests that migraine-related neural sensitization extends from the trigeminal nuclei to the vestibular nuclei, potentially precipitating vestibular dysfunction (29). The role of CGRP in the vestibular system may also influence plasma CGRP levels, further complicating the interpretation of CGRP levels in VM (14). Furthermore, reciprocal connections between the brainstem vestibular nuclei and structures modulating trigeminal nociceptive inputs, such as the rostral ventromedial medulla, ventrolateral periaqueductal gray matter, locus coeruleus and nucleus raphe magnus, are crucial for understanding the pathophysiology of VM (30). Elevated CGRP levels in the trigeminal and vestibular nuclei suggest a significant role for CGRP in chronic sensitization associated with VM, potentially increasing vestibular system sensitivity and contributing to VM symptoms (29). However, the role of CGRP in VM remains unclear because previous studies have not consistently shown significant differences in CGRP levels between VM participants and HC, indicating a gap in our understanding (Table 4) (19,20).
Review of previous studies on accessing CGRP levels in vestibular migraine patients.

CGRP = calcitonin-gene related peptide; ELISA = enzyme-linked immunosorbent assay; F = female; VM = vestibular migraine; HC = healthy controls.
Our detailed comparison of CGRP levels among individuals with VM and those with CM without vestibular symptoms revealed certain distinctions, enriching the ongoing dialogue regarding migraine pathophysiology. CM affects 1–2% of the population globally and is associated with more frequent attacks, greater symptom severity, and more comorbidities than EM. Numerous studies report elevated plasma and salivary CGRP levels in CM, with CGRP playing a crucial role in CM pathophysiology by promoting neurogenic inflammation and central sensitization. By contrast, CGRP levels in individuals with EM tend to be similar to those in HC, although some studies report slight increases (31). Although the role of CGRP as a biomarker is challenging due to variability in serological levels influenced by factors such as protease degradation, timing of sample collection, biological variability, circadian rhythms and methodological differences (10), our findings demonstrate consistently elevated plasma and salivary CGRP levels in CM participants even during interictal periods. This supports the hypothesis that CGRP is involved in sustained nociceptive signaling and chronic pain in CM. On the other hand, the similarity in plasma and salivary CGRP levels between VM participants and HC suggests a less prominent role for CGRP in VM pathophysiology. The main pathophysiological mechanisms of VM are centered in the vestibular system, and sustained release of CGRP may play a lesser role. The pathophysiology of VM also differs from CM in sensory processing. Neuroimaging studies have revealed altered modulation within vestibular, brainstem and cortical networks in VM, with dizziness and disorientation linked to abnormal sensory integration (28). Evidence points to central dysfunctions in vestibular processing, with VM patients exhibiting abnormal perception thresholds (32). Structural and functional imaging shows gray matter abnormalities in pain and visual processing regions in VM, correlating with disease duration and headache intensity, and suggesting abnormal brain sensitization and disrupted sensory processing (28). Our results found no correlation between CGRP levels and migraine headache or vestibular symptoms, including their frequency or duration, in VM patients. However, to explore this hypothesis further, subsequent research could examine CGRP levels in a larger sample of VM patients, both with episodic and chronic migraines, who experience dissociated migraine and vestibular symptoms.
Similarly, the CM group experienced more frequent migraine headaches, whereas the VM corresponding to EM group exhibited more pronounced psychiatric symptoms of depression and anxiety. No significant correlations were observed between CGRP levels and either migraine headache frequency or psychiatric symptoms in both CM and VM groups, as well as in a combined analysis (Table 3). Previous studies have reported that reductions in blood CGRP levels in individuals with CM in response to treatment, along with a reduction in headache frequency (33). Another study found that salivary CGRP levels were associated with headache frequency in CM and that effective treatment with anti-CGRP monoclonal antibodies correlated with reductions in both headache frequency and salivary CGRP levels (34). In recent years, the relationship between the immune system and psychiatric disorders has also gained attention, with evidence suggesting that the severity of depressive symptoms may be modulated by inflammatory processes (35). Notably, elevated brain levels of CGRP have been identified in animal models of depression, although antidepressants appear to have little effect on brain CGRP levels (36). Despite these findings, our study did not identify significant correlations between plasma and salivary CGRP levels and clinical characteristics such as migraine frequency or psychiatric disorders in CM and VM corresponding to EM groups, or in combined analyses (Table 3). Possible reasons for these discrepancies include variability in CGRP measurement methods, limited sample sizes and differences in sampling techniques. To our knowledge, the relationship between CGRP levels and both headache frequency and psychiatric disorders in untreated CM and VM corresponding to EM individuals has not yet been fully established and warrants further investigation. The salivary glands, innervated by the third branch of the trigeminal nerve and close to the TVS, allow less invasive detection of CGRP release during migraine episodes (37). Salivary CGRP levels is considered a reliable and sensitive marker of TVS activation (38). A 30-day study tracking salivary CGRP levels reported greater interictal CGRP levels in patients with EM compared than in HC (39). In the present study, we employed a standard gustatory stimulus by applying citric acid to the sides and tip of the tongue several times per minute. This method stimulates saliva secretion at a constant flow rate from the submandibular and sublingual glands, which are innervated by trigeminal nerves containing CGRP. Despite our similar CGRP levels in plasma and saliva within each migraine group, our investigation indicated that salivary CGRP levels were higher in CM patients compared to those with VM and HCs (Table 2).
This study had several limitations. First, while we included a relatively large sample size, we did not perform a formal sample size calculation, which may affect the statistical power and generalizability of our findings. Additionally, the absence of a group with EM without vestibular symptoms and a VM with CM group limits the scope of our conclusions. Future studies should include these groups to better contextualize our results and enhance our understanding of CGRP levels across different migraine subtypes. Second, the cross-sectional design restricts our ability to establish causality or temporal relationships between CGRP levels and migraine characteristics, highlighting the need for longitudinal studies. Third, our measurements of plasma and salivary CGRP levels were taken during interictal phases only, which may have underestimated differences between CM and VM because dynamic changes during acute attacks were not captured. Additionally, while we used established saliva collection methods, the lack of standardized protocols may have introduced inconsistencies in CGRP measurements; future research should standardize these procedures and explore alternative techniques to improve reliability and accuracy.
This study had several strengths. First, unlike previous studies that generally compared VM patients with either HC or non-VM migraine patients, our study uniquely assessed plasma CGRP levels across VM, CM and HC groups, offering a more comprehensive view of CGRP variations (13,14). Second, despite limited data on CGRP levels in VM, our study had a larger sample size than previous research, improving reliability with respect to identifying CGRP patterns. Third, by analyzing CGRP in saliva (a non-invasive, convenient biomarker source), our study was the first to assess salivary CGRP in VM patients, finding no significant differences from healthy controls.
In conclusion, the present study found that CGRP levels in both plasma and saliva of VM corresponding to EM participants were not significantly different from those of HC during the interictal period. By contrast, CM participants without vestibular symptoms exhibited higher interictal plasma and salivary CGRP levels compared to both VM and HC. These findings might underscore distinct pathophysiological mechanisms between VM corresponding to EM and CM, suggesting that CGRP plays a more significant role in the pathophysiology of CM than VM corresponding to EM. Despite the challenges of using plasma and salivary CGRP as biomarkers for VM, the present study offers valuable insights into the neurochemical differences between these migraine subtypes. Future research should investigate the temporal variability of CGRP levels and their correlation with clinical manifestations to further clarify the role of CGRP in migraine and vestibular symptoms.
Article highlights
Interictal plasma and salivary CGRP levels in VM corresponding to episodic migraine participants do not significantly differ from healthy controls but are elevated in chronic migraine. There is no significant relationship between interictal CGRP levels and clinical characteristics in participants with VM with EM, suggesting that CGRP levels may not serve as effective biomarkers for VM during interictal periods.
Footnotes
Acknowledgements
We express our sincere gratitude to all participants who generously contributed their time and effort to this study.
Author contributions
S-YO and MKC were responsible for the study design. S-YO, YB, J-JK and JC were responsible for the collection and analysis of data. All authors were responsible for data interpretation. YB, S-YO and MKC were responsible for manuscript preparation. S-YO, MKC and MD were responsible for manuscript review and revisions. All authors approved the final version of the manuscript submitted for publication.
Data availability
The data that have not been provided in the article may be shared upon request to the corresponding author by any qualified investigator for purposes of the replication of procedures and results
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical statement
All participants provided written informed consent. All methods were carried out in accordance with relevant guidelines and regulations including the Declaration of Helsinki and its following amendments.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a National Research Foundation of Korea (NRF) grant funded by the Korean government (Ministry of Science and ICT) (No. RS-2025-00553480) and the Fund of the Biomedical Research Institute, Jeonbuk National University Hospital.
