Abstract
Background
This post-hoc analysis from three phase 3 treatment trials of rimegepant 75 mg — an oral small molecule calcitonin gene-related peptide receptor antagonist for acute and preventive treatment of migraine — assessed efficacy in adults with migraine based on triptan treatment experience.
Methods
Participants were assigned to one of four groups based on triptan treatment experience: insufficient response (e.g. lack of efficacy and/or poor tolerability) to 1 triptan, insufficient response to ≥2 triptans, current triptan users, and triptan-naïve participants. The co-primary efficacy endpoints were pain freedom and most bothersome symptom freedom at two hours postdose.
Results
In the three trials (N = 3507; rimegepant n = 1749, placebo n = 1758), 1235 (35.2%) participants had a history of insufficient response to 1 triptan (n = 910 [25.9%]) or ≥2 triptans (n = 325 [9.3%]), and 2272 (64.8%) had no history of insufficient response to triptans (current use = 595 [17.0%], naïve = 1677 [47.8%]). Rimegepant was effective on the co-primary endpoints in all subgroups (p ≤ 0.013), except for freedom from the most bothersome symptom in the triptan-naïve group (p = 0.06). No differences on co-primary endpoints were found in pairwise comparisons of rimegepant-treated participants.
Conclusions
Rimegepant was effective for the acute treatment of migraine in adults with a history of insufficient response to 1 or ≥2 triptans and in current triptan users. Efficacy on co-primary endpoints did not differ based on the number of insufficient triptan responses.
Trial registration: Clinicaltrials.gov: NCT03235479, NCT03237845, NCT03461757
Introduction
Sumatriptan and other serotonin 5-HT1B/1D receptor agonists (triptans) have been among the most widely prescribed medications in the United States for the acute treatment of migraine for decades (1). While many of those who receive a prescription for a triptan obtain sufficient relief, others may not achieve an initial response at two hours, may respond at two hours and then relapse within 24 hours of the initial dose (up to 40%), or may experience adverse events (AEs) (2). Anticipation of AEs may lead patients with migraine to delay or forgo acute treatment (3). Cardiovascular contraindications and precautions can further complicate triptan prescribing. Population-based surveys show that triptans are contraindicated in almost 10% of the total US migraine population (∼3.5 million people) (4,5) and in 14.5% of commercially-insured people with migraine in Germany (6). Frequent use of triptans may also lead to medication-overuse headache, particularly in those with high frequency episodic and chronic migraine (7,8).
These therapeutic limitations help to explain why one third of adults with migraine are dissatisfied with their usual acute treatment (9), most often due to incomplete and inconsistent relief, delayed onset of therapeutic effects, recurrence of symptoms after an initial response, or AEs (9–13). Nearly 90% of people are willing to try a different acute medication (9), yet poor efficacy and/or tolerability are associated with discontinuation of acute prescription medication and, frequently, with resumption of nonprescription medication use for subsequent attacks (14,15).
If response to the first triptan prescribed is insufficient, prescribing a second triptan remains relatively uncommon in primary care practice (<15%) (1,16–19). In the United States, if an initial triptan is not refilled, the most commonly prescribed prescription acute treatments are opioids (∼42%), nonsteroidal anti-inflammatory agents (in 22%), and barbiturate combinations (9%) (19). When a second triptan is prescribed, it is often unsuccessful (20), although some people with insufficient response/tolerability may benefit from a switch to a different triptan or the addition of a nonsteroidal anti-inflammatory drug (21). In a large population study, switching within the triptan class was not associated with improvement in headache-related disability and appeared to increase primary care costs (22,23). The American Headache Society has suggested that patients who do not respond to or tolerate oral triptans may benefit from acute treatment with agents that have a novel mechanism of action, including gepants (1).
One such gepant, rimegepant, is an orally administered small molecule calcitonin gene-related peptide receptor antagonist indicated for the acute treatment of migraine and preventive treatment of episodic migraine (24). Rimegepant has demonstrated efficacy and safety in three randomized, placebo-controlled phase 3 trials in the acute treatment of migraine (25–27), each of which enrolled individuals with and without a history of triptan use. This study analyzed current and lapsed triptan users and the reasons for discontinuation, as well as those who had never tried a triptan. Because rimegepant acts as an antagonist at calcitonin gene-related peptide receptors and triptans act as agonists at serotonin receptors, we hypothesized that history of an insufficient response to triptans would not meaningfully affect response to acute treatment with rimegepant. We further hypothesized that the efficacy of rimegepant would be consistent regardless of history of triptan insufficient response or current triptan use. To test these hypotheses, we conducted a post-hoc analysis to compare the efficacy of rimegepant with placebo in the acute treatment of migraine in subgroups of participants based on historical and current triptan experience.
Methods
Ethics
This subgroup analysis is based on the results of three clinical trials of rimegepant for the acute treatment of migraine, each of which was prospectively registered at clinicaltrials.gov (NCT03235479, NCT03237845, NCT03461757). All three trials were conducted in accordance with the principles of the Guidelines for Good Clinical Practice, the Declaration of Helsinki, and all applicable local regulations. The protocols were approved by independent ethics committees and/or institutional review boards at each study center. Before any study-related procedures were undertaken, participants in all three trials provided written informed consent to participate.
Study conduct
This analysis was based on pooled data from three methodologically similar, double-blind, randomized, multicenter phase 3 trials comparing the safety and efficacy of a single 75 mg dose of rimegepant with placebo in the acute treatment of a single migraine attack of moderate to severe pain intensity. The duration of participation in each of the three trials was approximately 11 weeks, including a screening period of three to 28 days, an acute treatment phase of up to 45 days, and an end-of-treatment visit.
Upon study entry, participants were entered into an interactive web response system. After screening and within three to 28 days of signing informed consent, participants returned to study centers. They were randomized to study treatment and provided with an electronic diary. Study personnel instructed participants on the correct use of the electronic diary prior to discharge from the site.
Participants were given a single dose of study medication: rimegepant 75 mg oral tablet or matching placebo in two studies (25,27) and rimegepant 75 mg orally disintegrating tablet or matching placebo in the third (26). They were and instructed to treat a migraine attack with pain of moderate or severe intensity after answering electronic diary questions about their current pain and symptoms and identifying their currently most bothersome symptom (MBS) from among the migraine-associated symptoms of phonophobia, photophobia, and nausea. Participants completed the electronic diary for up to 48 hours after taking study medication. Pain intensity, the presence or absence of migraine-associated symptoms, and functional disability rating were assessed immediately before dosing and at 15, 30, 45, 60, and 90 minutes and at two, three, four, six, eight, 24, and 48 hours postdose.
After completion of the 48-hour assessment, participants returned to the study center within seven days of the treated migraine attack (plus up to two days if necessary) for review of the electronic diary, assessment of medication compliance, and monitoring of tolerability and safety. Participants who had not experienced a migraine attack of moderate or severe pain intensity within 45 days of randomization were instructed to return unused study medication and the electronic diary to the study center.
Participants
The study population included men and women aged 18 years and older who had migraine with or without aura according to the criteria of the International Classification of Headache Disorders, 3rd Edition, beta version (28); a one-year history of attacks lasting four to 72 hours if untreated, with the age of onset prior to 50 years of age; at least two and not more than eight migraine attacks of moderate or severe pain intensity per month, as well as fewer than 15 days with migraine or nonmigraine headache within the last three months. Participants had to be able to distinguish migraine attacks from attacks of tension-type and cluster headache, and those taking preventive migraine medication had to be on a stable dose for ≥3 months before screening. If they met all other criteria for inclusion, participants with contraindications to triptans could be included. Women of childbearing potential and nonsterile men could participate if they were using two acceptable methods of contraception to avoid pregnancy throughout the study, and the women had a negative serum or urine pregnancy test (minimum sensitivity 25 IU/L or equivalent units of human chorionic gonadotropin) at baseline and were not breastfeeding.
Participants were excluded if they had any medical condition that might interfere with study assessments of efficacy and safety or expose them to undue risk of AEs. Participants were also excluded if they had been treated for or showed evidence of alcohol or drug abuse within the past 12 months; had a history of drug or other allergy that made them unsuitable for participation; or had electrocardiogram or laboratory test findings that raised safety concerns.
Additional criteria for participation are available in the study protocols, which are available at clinicaltrials.gov.
Assessments
The co-primary efficacy endpoints were pain freedom and freedom from the MBS associated with migraine (i.e. phonophobia, photophobia, or nausea) at two hours postdose. Pain freedom was assessed using the proportion of participants who reported no pain at two hours postdose. Pain intensity was measured with a 4-point rating scale (0 = none, 1 = mild, 2 = moderate, 3 = severe). Freedom from the MBS was assessed using the proportion of participants in whom the MBS reported immediately before dosing was absent at two hours postdose. Associated symptom status was reported postdose as present or absent for each symptom (nausea, phonophobia, and photophobia).
The key secondary efficacy endpoint of pain relief was assessed using the proportion of participants who reported mild or no pain at two hours postdose.
Statistical analysis
The statistical methods used in the individual trials have been published previously (25,26). The efficacy population included randomized participants who had a migraine attack with pain of moderate or severe intensity at the time of study medication dosing, took study medication, and had postdose efficacy data.
Four subgroups based on historical and current triptan experience were analyzed for efficacy: (1) participants reporting a history of insufficient response to 1 triptan; (2) participants reporting a history of insufficient response to ≥2 triptans; (3) participants reporting current use of triptans and no history of triptan insufficient response; and (4) participants reporting no current use of triptans and no history of triptan insufficient response, that is, triptan-naïve participants. Note that subgroups (1) and (2) are referred to as “triptan insufficient responder” subgroups. A history of insufficient response to triptans was defined as a self-reported history of discontinuation of any medication in the triptan class (i.e. any formulation of almotriptan, eletriptan, frovatriptan, naratriptan, rizatriptan, sumatriptan, or zolmitriptan, or the combination sumatriptan-naproxen) for any reason, including lack of efficacy and/or tolerability. Current use of triptans was defined as self-reported use of triptan medication at screening. Participants with a history of triptan insufficient response were asked to choose at least one of six possible reason(s) for discontinuation (Table 1); these choices were based on previously published reasons for dissatisfaction with medications used for the acute treatment of migraine (9–13). Participants quantified the frequency of each reason for discontinuation using one of the following three options: most or all of the time; some of the time; or rarely.
Reasons for historical triptan insufficient response.a

aVerbatim reasons provided to participants for self-reporting history of triptan insufficient response at screening. They were required to choose ≥1 reason.
bLack of efficacy.
cIntolerability.
In each of the four triptan experience subgroups, treatment comparisons of three efficacy endpoints (co-primary endpoints of pain freedom and MBS freedom at two hours postdose; key secondary endpoint of pain relief at two hours postdose) were performed using Cochran-Mantel-Haenszel tests stratified by preventive migraine medication use at randomization (yes, no). Participants with missing data or rescue medication use before or at the time of assessment were classified as treatment non-responders. The 12 treatment comparisons were tested using the Hochberg procedure at a significance level of alpha ≤0.05 (29).
To evaluate the consistency of rimegepant treatment effects, the co-primary efficacy endpoints were compared pairwise between triptan experience subgroups in rimegepant-treated participants. Comparisons were performed using the logit link function in logistic regression models that included class predictor variables for triptan experience (four levels: triptan-naïve, current triptan use, insufficient response to 1 triptan, insufficient response to ≥2 triptans) and preventive migraine medication use (yes, no). Because these analyses were intended to show a lack of difference between subgroups, no multiplicity adjustment was made.
Statistical analyses were performed using SAS, version 9.4 (SAS Institute, Cary, NC, USA).
Results
Participants
Of the 3507 participants in the three trials (rimegepant n = 1749, placebo n = 1758), 910 (25.9%) had a history of insufficient response to 1 triptan, and 325 (9.3%) had a history of insufficient response to ≥2 triptans (Figure 1). The remaining 2272 (64.8%) participants had no history of triptan insufficient response; 595 (17.0%) were current triptan users, and 1677 (47.8%) were triptan-naïve.

Participants in rimegepant clinical trials and triptan experience subgroups.
Demographics and baseline characteristics of the triptan insufficient response subgroups were generally consistent with those of current triptan users (Table 2). Triptan-naïve participants were slightly younger, with fewer years since migraine onset (16 vs 22–24) and a shorter mean duration of untreated attacks (27 hours vs 34–36 hours) than those in the other three subgroups (Table 2). Triptan-naïve participants also had a shorter mean duration of untreated attacks (27 hours vs 34–36 hours); however, the frequency of moderate or severe monthly migraine attacks during the three months prior to screening was consistent across all four subgroups (4.6–4.7). Furthermore, triptan-naïve participants were less likely to use a preventive migraine medication (6%) than current triptan users (23%), and those who had treatment insufficient response with 1 triptan (23%) and ≥2 triptans (32%).
Demographics and migraine history in the triptan experience subgroups.

aIncludes American Indian or Alaska Native, Asian, Multiple, and Native Hawaiian or Other Pacific Islander.
As shown in Table 3, the most common agents in the historical triptan insufficient response subgroups were sumatriptan (1 triptan: 73.0% [664/910]; ≥2 triptans: 82.5% [268/325]) and rizatriptan (1 triptan: 15.4% [140/910]; ≥2 triptans: 62.8% [204/325]). The self-reported reasons for an insufficient response to oral sumatriptan and oral rizatriptan are shown in Table 4. Participants in both triptan insufficient response subgroups cited inconsistent relief as the most common reason for insufficient response to oral sumatriptan (70% to 76%) and oral rizatriptan (52% to 77%). The second most common reason for insufficient response was slow onset of relief (41% to 71%). Participants in both triptan insufficient response subgroups also frequently cited side effects, recurrence of pain, and lack of treatment effects on other migraine-associated symptoms as reasons for an insufficient response (Table 4).
Most common triptansa historically associated with an insufficient response.

aAll routes of administration.
Reasons for historical insufficient response to oral sumatriptan and oral rizatriptan in the triptan insufficient response subgroups.

Efficacy
Results for the co-primary efficacy endpoints are presented in Figure 2. Rimegepant was more effective than placebo at two hours postdose among participants with a history of insufficient response to 1 triptan and ≥2 triptans for pain freedom (1 triptan: 20.7% vs 12.4%, p < 0.001; ≥2 triptans: 20.0% vs 10.2%, p = 0.013) and MBS freedom (1 triptan: 36.2% vs 24.4%, p < 0.001; ≥2 triptans: 43.0% vs 21.5%, p < 0.001). Rimegepant was also more effective than placebo among current triptan users for pain freedom (20.4.% vs 6.8%, p < 0.001) and MBS freedom (37.2% vs 22.2%, p < 0.001) at two hours postdose. Among triptan-naïve participants, rimegepant was more effective than placebo for two-hour pain freedom (19.6% vs 14.7%, p = 0.007) and not significantly different than placebo for two-hour MBS freedom (35.0% vs 30.7%, p = 0.06). For the key secondary endpoint of pain relief at two hours postdose, rimegepant was more effective than placebo among participants with a history of insufficient response to 1 triptan and ≥2 triptans (1 triptan: 58.4% vs 42.8%, p < 0.001; ≥2 triptans: 69.5% vs 36.6%, p < 0.001), as well as current triptan users and triptan-naïve participants (current: 61.8.% vs 41.3%, p < 0.001; triptan-naïve: 54.4% vs 47.0%, p = 0.002).
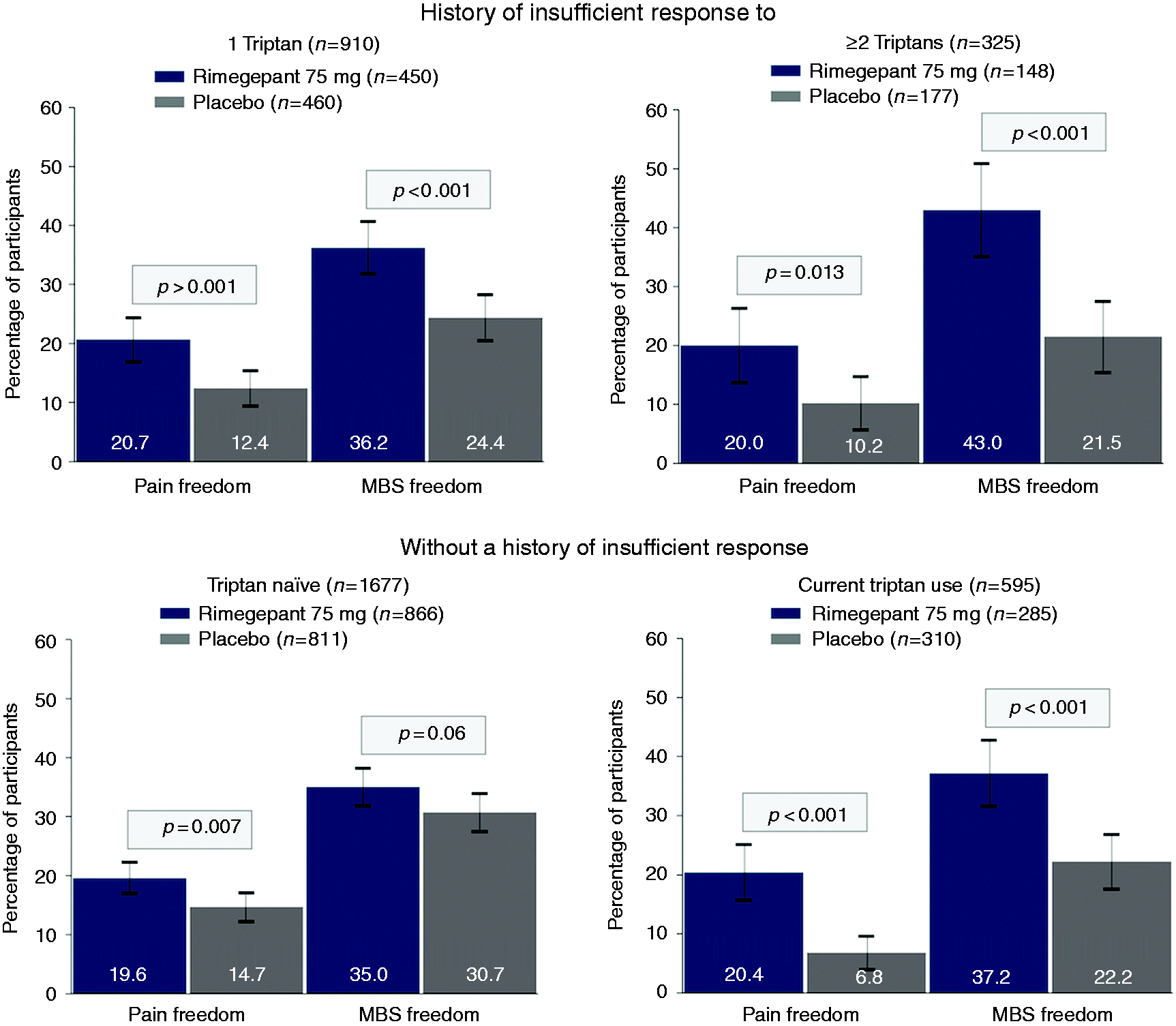
Co-primary efficacy endpoints at 2 hours postdose by triptan experience subgroups.MBS: most bothersome symptom.
As shown in Table 5, six pairwise comparisons of the four triptan experience subgroups were performed in rimegepant-treated participants for each co-primary endpoint using logistic regression. For each co-primary endpoint, none of the pairwise comparisons showed statistically significant differences between subgroups, given all 95% confidence intervals for odds ratios captured 1.
Co-primary efficacy endpoints compared pairwise between triptan experience subgroups in rimegepant-treated participants.

CI, confidence interval; MBS, most bothersome symptom.
Comparisons used logistic regression models in rimegepant-treated participants; models included class predictors variables for triptan experience (4 levels: triptan-naïve, current triptan use, insufficient response to 1 triptan, insufficient response to ≥2 triptans) and preventive migraine medication use (yes, no).
Discussion
This post-hoc analysis of pooled data from three phase 3 clinical trials evaluated the efficacy of rimegepant versus placebo in the acute treatment of migraine based on historical and current experience with medications in the triptan class. Results demonstrated that rimegepant was effective in the acute treatment of migraine in participants with a history of triptan insufficient response to 1 or ≥2 triptans, as well as in current triptan users. There were also no significant differences in the co-primary endpoints between the four subgroups in rimegepant-treated participants.
These results indicate that a history of insufficient response to 1 or ≥2 triptans does not influence the efficacy of rimegepant. Higher placebo response rates were observed in the triptan-naïve subgroup, which may have contributed to the lack of significant difference from placebo (p = 0.06) on the two-hour MBS freedom endpoint in that subgroup. High placebo response rates have also been reported in triptan-naïve subgroups from trials of other medications, including triptans and gepants (30–32). It is possible that participants in the triptan-naïve subgroup had higher expectations of success with study treatment, which has been shown to enhance the placebo effect and increase placebo response rates (33). It is also possible that highly placebo-responsive patients had their treatment needs met by prior therapy and are therefore less likely to enroll in a clinical trial. Additionally, since placebo response tends to be lower in people with severe migraine headache pain (33,34), the attack characteristics of the triptan-naïve subgroup — shortest migraine history, shortest mean duration of untreated attacks, and lowest rate of preventive medication use — suggest that these participants may have less severe attacks than current triptan users and those in the triptan insufficient response subgroups. The lower placebo response rates in participants with current triptan use and a history of triptan insufficient response may reflect a lowering of expectations based on prior experience.
The most common reasons participants provided for triptan insufficient response align with previous research about the attributes of acute medication most valued by people with migraine (9–13), confirming the persistence of unmet needs in the acute treatment of migraine and suggesting therapeutic opportunities with rimegepant. For example, slow onset of action was frequently cited as a reason for triptan insufficient response; rimegepant has demonstrated pain relief and a return to normal function within one hour postdose, which may represent a meaningful improvement for many patients (26). Rimegepant has also consistently demonstrated favorable safety and tolerability, which may benefit patients whose insufficient response is due to AEs.
The efficacy findings from this analysis are comparable to those seen in the individual rimegepant trials and consistent with the results of post-hoc analyses of the effects of prior triptan exposure on the efficacy of ubrogepant, lasimiditan, and triptans (30,32,35,36). This analysis differs from previous work by distinguishing between participants who had a history of insufficient response to 1 triptan and participants who had a history of insufficient response to ≥2 triptans.
The preponderance of evidence suggests that once patients have an insufficient response to a triptan, outcomes tend to improve by switching to a different class of medication and worsen by use of a second agent in the triptan class. For example, in the American Migraine Prevalence and Preventive study, a second triptan was associated with increased migraine-related disability (+10.4 on the Migraine Disability Assessment), and in the OVERCOME study, observational data showed that the effectiveness of second triptan is more likely to be poor or very poor compared with a first triptan (20,37). These results reflect the results of real-world triptan switches. Taken together with the main insight from the present analysis — that there was no difference in the efficacy of rimegepant between participants who had a history of insufficient response to 1 triptan and participants who had a history of insufficient response to ≥2 triptans — the data challenge the rationale for requiring insufficient response to multiple triptans before considering alternative treatment options.
This analysis has strengths and limitations. To the extent that the large clinical trial sample is representative of real-world clinical practice, these findings should be generalizable to the migraine population eligible for acute prescription treatments. The single-attack design of the individual trials prevented analysis of the consistency rimegepant treatment effects across attacks, an important predictor of satisfaction with treatment (12). Another limitation is that this is a post-hoc analysis; the triptan-naïve subgroup was younger, had a shorter time since migraine onset, a shorter duration of untreated attacks, and a lower rate of preventive medication use than the overall population. These differences in characteristics could confound the association between treatment group and treatment outcomes. Finally, as the individual rimegepant trials included in this analysis did not include an active comparator, the results of this analysis cannot be used to make direct efficacy comparisons of rimegepant with triptans or other treatments.
Rimegepant has demonstrated efficacy in multiple placebo-controlled clinical trials for the acute treatment of migraine as well as a placebo-controlled trial for the preventive treatment of migraine, with tolerability and safety comparable to placebo (25–27). This post-hoc analysis extends its therapeutic potential by showing that rimegepant is effective in patients with a history of triptan insufficient response and in those currently using triptans.
Conclusions
Oral rimegepant 75 mg was effective for the acute treatment of migraine among participants with a history of triptan insufficient response and in current triptan users. The efficacy of rimegepant was consistent among those with insufficient response to 1 or ≥2 triptans and those who were triptan-naïve or currently using triptans.
Clinical implications
A post-hoc analysis of pooled data from three phase 3 clinical trials evaluated the efficacy of rimegepant versus placebo in the acute treatment of migraine based on historical and current experience with medications in the triptan class. Rimegepant was effective in the acute treatment of migraine in participants who had insufficient response to 1 triptan or ≥2 triptans, as well as in those who were current triptan users. There were no clinically meaningful differences in the co-primary endpoints between subgroups of rimegepant-treated participants. This study, along with previously published observational data, challenges the rationale for requiring that patients with migraine endure insufficient response to multiple triptans before considering alternative treatments.
Footnotes
Acknowledgements
The authors thank Chandra Abbott for assistance with the publication of this manuscript. Medical writing services were provided by Christopher Caiazza.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Richard B Lipton serves on the editorial board of Neurology and Cephalalgia and as senior advisor to Headache but is not paid for his roles on Neurology or Headache. He has received research support from the NIH. He also receives support from the Migraine Research Foundation and the National Headache Foundation. He receives research grants from Allergan, Amgen, Dr. Reddy’s Laboratories, and Novartis. He has reviewed for the NIA and NINDS and serves as consultant, advisory board member, or has received honoraria from Alder, Allergan, Amgen, Biohaven, Dr. Reddy’s Laboratories, electroCore, Eli Lilly, BDSI, eNeura Therapeutics, GlaxoSmithKline, Merck, Novartis, Pfizer, Teva, and Vedanta. He receives royalties from Wolff’s Headache (8th Edition, Oxford University Press, 2009) and Informa. He holds stock or options in Biohaven Pharmaceuticals and Manistee.
Andrew Blumenfeld serves as a consultant for Alder, Allergan, Amgen, Aeon, Avanir, Biohaven, ElectroCore, Eli Lilly, Equinox, Impel, Lundbeck, Novartis, Pernix, Promius, Supernus, Revance, Teva, and Theranica.
Christopher M Jensen, Robert Croop, Alexandra Thiry, Gilbert L’Italien, Beth A Morris, and Vladimir Coric, are employed by and hold stock/stock options in Biohaven Pharmaceuticals.
Peter J Goadsby reports, over the last 36 months, grants and personal fees from Eli-Lilly and Company, grant from Celgene, and personal fees from Aeon Biopharma, Allergan/Abbvie, Amgen, Biodelivery Sciences International, Biohaven Pharmaceuticals Inc., CoolTech LLC, Dr Reddys, Epalex, Impel Neuropharma, Lundbeck, Novartis, Praxis, Sanofi, Satsuma and Teva Pharmaceuticals, and personal fees for advice through Gerson Lehrman Group, Guidepoint, SAI Med Partners, Vector Metric, and fees for educational materials from CME Outfitters, Omnia Education, WebMD, and publishing royalties or fees from Massachusetts Medical Society, Oxford University Press, UptoDate and Wolters Kluwer, and for medicolegal advice in headache, and a patent magnetic stimulation for headache (No. WO2016090333 A1) assigned to eNeura without fee.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Biohaven Pharmaceuticals, Inc.
