Abstract
Objective
To determine whether intravenous infusion of substance P elicits headache and arterial dilation in healthy adults.
Methods
In a double-blind, placebo-controlled, two-way crossover study, we randomly allocated healthy adults to receive either substance P (1.5 pmol/kg/min) or placebo (isotonic saline) by 20-minute intravenous infusion on separate experiment days. The primary endpoint was the incidence of headache within 12 hours after the infusion. Other endpoints included the area under the curve (AUC) for headache intensity scores over the 12-hour observation period and AUC changes in the superficial temporal artery (STA) diameter from baseline to 120 minutes post-infusion.
Results
Twenty-one participants underwent randomization and completed both experiment days. Headache was reported by 15 (71%) of 21 participants following substance P, compared with two (10%) after placebo (p < 0.001). The AUC for headache intensity scores was significantly higher after substance P infusion than after placebo (p = 0.03). In addition, there was a significant STA diameter increase with substance P, compared with placebo (p = 0.005).
Conclusions
Among healthy adults, substance P infusion elicited headache and arterial dilation. These findings support a role for substance P in headache pathogenesis and encourage further investigation in migraine and other headache disorders. Additional research is also needed to explore new therapeutic strategies targeting substance P or its downstream signaling.
Trial Registration:
CT identifier: NCT06632080.
This is a visual representation of the abstract.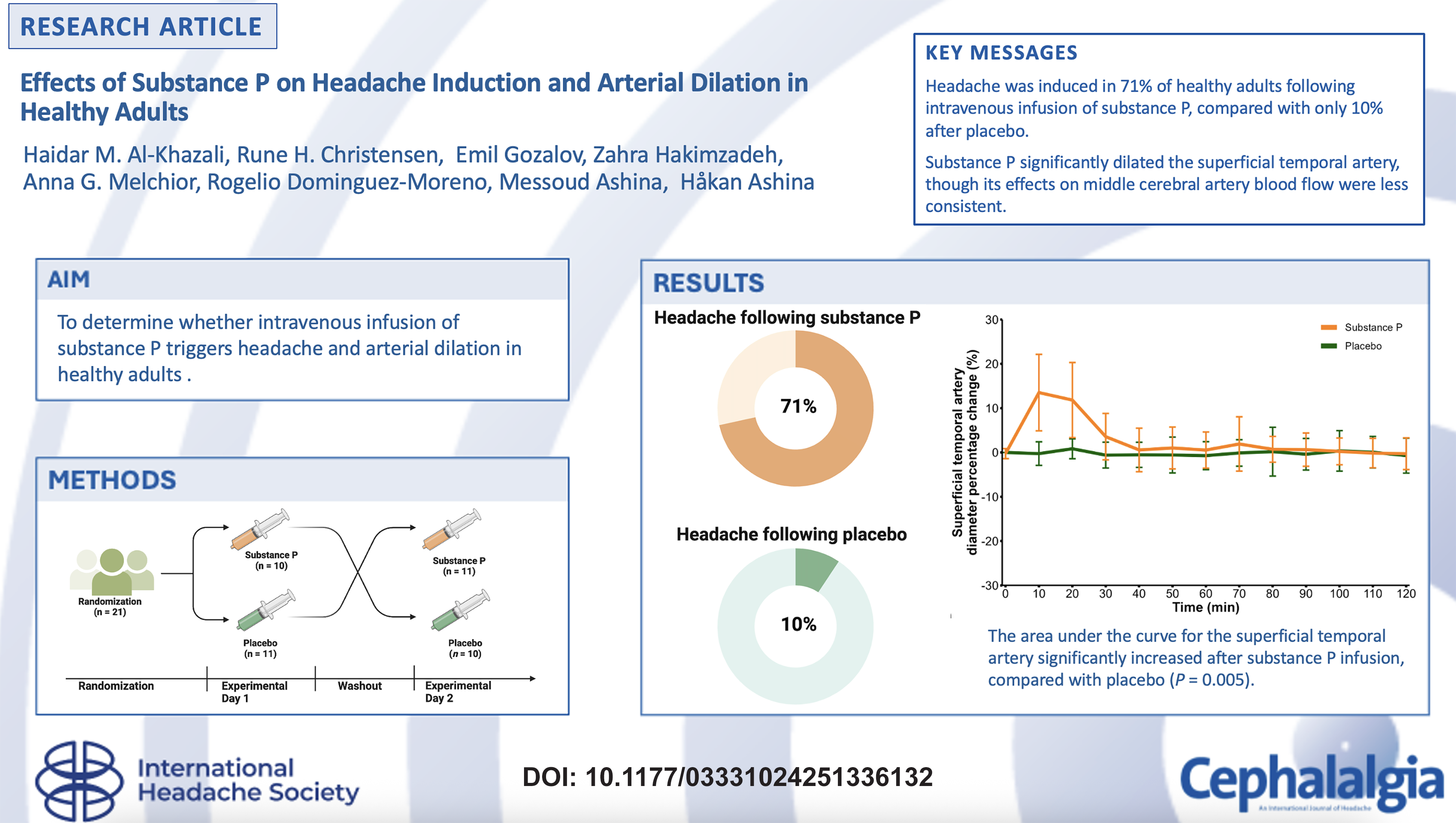
Introduction
Headache is among the most prevalent neurological complaints in clinical practice, with a phenotypic presentation that varies considerably among those affected (1). Despite this heterogeneity, ample human experimental evidence has demonstrated that headache disorders share key pathophysiologic mechanisms (2–9). Central to these mechanisms is the trigeminovascular system, which underscores the intricate interplay between primary afferent neurons of the trigeminal nerve and the meningeal vasculature (10). Through the release of vasodilatory and pro-inflammatory neuropeptides, this system is assumed to initiate and perpetuate a range of headache phenotypes (10).
Within the trigeminovascular system, several neuropeptides – including calcitonin gene-related peptide (CGRP), pituitary adenylate cyclase-activating polypeptide (PACAP) and substance P – have been identified as possible pathogenic mediators of head pain (10). Substance P, in particular, has been extensively studied in preclinical models for its ability to promote neurogenic inflammation, elicit mast cell degranulation and modulate nociceptive transmission (11). Notably, the application of substance P on trigeminal ganglion slices depolarizes neurons (12,13), providing further mechanistic support for a causal relationship between this peptide and headache-related nociceptive pathways.
Despite these compelling preclinical insights, the precise role of substance P in the human experience of headache remains poorly understood. An outstanding question is whether intravenous (IV) infusion of substance P can trigger headache in humans, thus mirroring the headache-inducing properties documented for CGRP and PACAP (14). To address this knowledge gap, we conducted the first randomized, double-blind, placebo-controlled, two-way crossover study examining the effects of IV infusion of substance P in healthy adults. Our objectives were to: (i) determine the incidence and intensity of headache following substance P infusion; (ii) measure changes in superficial temporal artery (STA) diameter; and (iii) examine concomitant alterations in middle cerebral artery blood flow velocity (VMCA).
Methods
This study was approved by the Scientific Ethics Committees for the Capital Region of Denmark (Identifier: H-23030963; Approval Date: August 28, 2023). All participants provided written informed consent before the commencement of any study-related assessments or procedures. The study was registered with ClinicalTrials.Gov (Identifier: NCT06632080) and adhered to the principles of the Declaration of Helsinki with subsequent revisions (15). All authors affirm that the manuscript is an accurate and transparent account of the study being reported. The full study protocol is available in the supplemental material.
Design
This study applied a randomized, double-blind, placebo-controlled, two-way crossover design, enrolling healthy adults. All assessments and procedures were conducted at the research facilities of a national referral hospital. The study was conducted between October 2024 and January 2025. All participants were recruited using an online advertisement (https://forskningnu.dk) appearing on a digital platform dedicated to the recruitment of participants for research.
Trained personnel screened potential participants by phone, using a standardized checklist of inclusion and exclusion criteria. Additional verification was performed through a review of electronic medical records. Eligible participants were then scheduled for two separate experiment days, during which they were randomly allocated to receive either IV substance P or IV placebo (Figure 1). A minimum washout period of one week was chosen to minimize the risk of carryover effects.

Study flow diagram. n, number.
Participants
Eligible participants were adults aged 18–65 years, with a body weight of 50–100 kg. Individuals were excluded if they had a personal history of any primary headache disorder – other than infrequent episodic tension-type headache – or if they had a first-degree relative with a primary headache disorder. Individuals with clinically significant neurologic, psychiatric or other medical conditions (including hypersensitivity or autoimmune conditions) were also excluded to minimize confounding influences. A complete account of the inclusion and exclusion criteria is provided in in the supplementary material (Tables S1 and S2).
Randomization and blinding
The experimental drug, substance P, was purchased from Tocris Bioscience (Bristol, UK). A continuous 20 mL IV infusion was chosen, delivering 1.5 pmol/kg/min over 20 minutes. This dose matched that used in several previous human studies (16–20). An equivalent volume of isotonic saline served as the placebo. Independent pharmacy staff performed drug preparation, randomization and allocation concealment to maintain blinding.
Block randomization was used in groups of four participants to ensure balanced allocation. Within each block, two participants received substance P and two received placebo. The sequence was generated by a computer-based random number program. Of the 21 participants, five blocks comprised four participants each, whereas the final block included a single participant. For safety oversight, the randomization code was kept in sealed envelopes and opened only if clinically indicated, thus preserving investigator blinding.
Experiment days
Participants were required to abstain from analgesics, antihistamines, and anti-inflammatory drugs for 48 hours and to avoid methylxanthine-containing foods and beverages (e.g. coffee, tea, cocoa) for 12 hours before infusion.
On both experiment days, trained personnel conducted a physical and neurologic examination to confirm continued eligibility. The participants were then informed that substance P could induce a mild, transient headache, although no details were given regarding its onset, duration or associated features.
After these initial assessments, a peripheral venous catheter was inserted into the antecubital vein for drug infusion. The participants rested supine for 15 minutes to minimize hemodynamic variability. Baseline measurements included vital signs (blood pressure and heart rate) and a 12-lead electrocardiogram. High-resolution ultrasonography was used to assess the STA diameter, whereas transcranial Doppler was applied to measure VMCA. The participants were also asked to confirm that they were free of headache at baseline.
Following these measurements, the assigned infusion (substance P or placebo) was administered continuously over 20 minutes. STA diameter and VMCA were measured at pre-specified intervals for 120 minutes post-infusion and vital signs were monitored every 10 minutes to ensure safety. At the end of the 120-minutes observation, participants were discharged with instructions to complete a standardized headache diary for the subsequent 10 hours. The diary captured headache presence or absence, headache intensity and any use of rescue headache medications. Headache intensity was rated on an 11-point numeric rating scale, ranging from 0 (“no pain”) to 10 (“worst imaginable pain”). In brief, each participant contributed headache-related data over a 12-hour observation period, encompassing 2 hours in-hospital phase plus an additional 10 hours at home.
Measurements of STA diameter and VMCA
A high-resolution ultrasound system (Dermascan C unit, 20 MHz; Cortex Technology, Aalborg, Denmark) was used to measure the STA diameter. The frontal branch of the STA was identified approximately 1–2 cm lateral to the temporal hairline. Each measurement consisted of four consecutive ultrasound scans at a single site, and the mean of these scans was recorded. Measurements were performed at baseline (i.e. time of infusion start) and then every 10 minutes until 120 minutes post-infusion. To minimize bias, one trained operator, blinded to treatment allocation, performed all assessments on both experiment days for the same participant.
Simultaneous bilateral VMCA measurements were obtained using a 2-MHz TCD probe (DWL Doppler System, Singen, Germany) positioned at the temporal acoustic window. The depth was set to 50 mm, gain to 38, sample volume to 8, power to 420 and filter to 100 for all measurements. Before recordings, the mean VMCA was determined with equal flow on both sides using the same MCA branch (middle, posterior or anterior), which was maintained for all subsequent recordings across both experiment days. The insonation angle remained consistent through all time points. Measurements were taken at baseline and every 10 minutes up to 120 minutes post-infusion. Data from sides were averaged to yield a single VMCA value per participant per time point. To minimize bias, a single trained operator conducted all measurements on both experiment days.
Endpoints
The primary endpoint was the difference in headache incidence from baseline to 12 hours post-infusion initiation, comparing substance P and placebo. The secondary endpoint was the difference in the area under the curve (AUC) for headache intensity scores across the same 12-hour observation period. The tertiary endpoints, quantifying potential vascular effects, included differences in the AUC for percentage changes in both STA diameter and VMCA velocity from baseline to 120 minutes post-infusion.
Statistical analysis
For sample size calculations, we used McNemar's test, which is appropriate for a paired dichotomous primary endpoint. We assumed that 50% of participants would experience headache exclusively after IV substance P, whereas only 10% would report headache exclusively after IV placebo. Based on these assumptions, we determined that 21 participants would provide 80% power at p < 0.05 (one-sided). As specified in the protocol, any participant who withdrew before completing both experimental days would be replaced to maintain the target sample size.
Descriptive statistics were used to summarize baseline characteristics of the study population. Continuous variables were reported as the mean ± SD or medians with interquartile ranges, as appropriate. Categorical variables were presented as absolute counts with percentages. The primary endpoint was analyzed using McNemar's test. For the secondary endpoint, we computed AUC values for headache intensity scores using the trapezium rule. The calculated AUC values were then corrected for baseline scores and analyzed using the Wilcoxon signed-rank test. In addition, we calculated AUC values for the percentage change in mean STA diameter, VMCA, heart rate and mean arterial pressure. Paired t-tests were then used to compare these hemodynamic responses between substance P and placebo. All statistical analyses were performed using R statistical software, version 4.1.0 (R Foundation, Vienna, Austria).
Results
In total, 50 individuals were screened for eligibility from October 2024 to January 2025. Of these, 21 were randomized and completed both experiment days (Figure 1). The mean ± SD age was 34.1 ± 13.2 years, and most participants (n = 13; 62%) were female. The participants’ body mass index was 24.5 ± 4.2 kg/m2. Table 1 summarizes the participants’ demographics and baseline characteristics.
Baseline characteristics and hemodynamic variables.

Data are n (%) or mean ± SD.
BMI = body mass index; MCA = middle cerebral artery; STA = superficial temporal artery; bpm = beats per minute.
Headache
Fifteen (71%) of 21 participants experienced headache within 12 hours after receiving substance P, compared two (10%) participants after placebo (p < 0.001). Furthermore, 13 participants developed headache exclusively after substance P, whereas none did so after placebo. During the 120-minute in-hospital phase, 13 participants reported headache following substance P, compared with only one participant after placebo.
In participants with substance P-induced headache, the median time to onset was 20 (interquartile range = 10–30) minutes. One participant experienced a migraine-like headache, characterized by moderate pain intensity, throbbing quality, and accompanying symptoms of nausea, photophobia and phonophobia. Table 2 presents a summary of headache features and associated symptoms induced by substance P and placebo.
Clinical characteristics of headache induction and associated symptoms in healthy participants following substance P and placebo.

No., number; Bilat, bilateral; Unilat, unilateral; Throb, throbbing; Pres, pressing; NA, not applicable.
Localization: unilateral or bilateral/pain intensity (11-point numeric scale: 0 = no headache, 10 = worst imaginable)/headache quality (throbbing, pressing, or both)/aggravation by routine physical activity (+ denotes presence, – denotes absence).
Associated symptoms: vomiting, nausea, photophobia and phonophobia, respectively.
Migraine-like headache: meets criteria C, D and E for migraine without aura per the International Classification of Headache Disorders, 3rd edition (1).
The AUC values for headache intensity scores were significantly higher across the 12-hour observation period after substance P, compared with placebo (p = 0.03) (Figure 2). The median time to peak headache intensity was 30 (interquartile range = 10–60) minutes after infusion start with substance P.

Individual headache intensity scores following substance P and placebo in healthy adults. NRS = Numeric Pain Rating Scale (11-point scale, 0 = no headache, 10 = worst imaginable headache). Headache intensity over 12 hours in healthy adults after intravenous infusion of substance P (left side) or placebo (isotonic saline) (right side). Individual headache intensity scores for substance are shown as colored lines.
Among participants who experienced headache after substance P, all reported bilateral headache (n = 15; 100%), most described the pain as pressing (n = 12; 80%) and nearly all rated it as mild (n = 13; 86%). At peak headache, one-third of participants (n = 5; 33%) noted pain aggravation with routine physical activity and more than half (n = 8; 53%) reported photophobia. Three participants (20%) experienced phonophobia and six (40%) developed nausea or vomiting.
Hemodynamic responses
During the 120-minute in-hospital phase, the AUC values for percentage change in STA diameter was significantly greater with substance P than with placebo (p = 0.005) (Figure 3A). By contrast, the AUC values for percentage change in VMCA from baseline to 120 minutes did not differ between the two days (p = 0.13) (Figure 3B). However, post-hoc analyses revealed significant reduction in VMCA at both 30 and 60 min after the start of substance P infusion, compared with placebo (AUC0–30 min, p = 0.001; AUC0–60 min, p = 0.03). Heart rate and mean arterial pressure remained stable, with no differences noted from baseline to 120 minutes for either day (All p > 0.05) (Figure 3C and D).

Hemodynamic responses following substance P and placebo in healthy adults. Hemodynamic changes from baseline to 120 minutes after substance P (orange) and placebo (green) infusion. Percentage changes from baseline are shown for mean superficial temporal artery diameter (A), middle cerebral artery velocity (B), heart rate (C) and mean arterial blood pressure (D). Error bars indicate 95% confidence intervals.
Adverse events
Following IV administration of substance P, the most frequently reported adverse events were flushing (n = 20), sensations of warmth (n = 18), palpitations (n = 10) and neck pain (n = 2). In contrast, IV placebo administration resulted in flushing in one participant, sensations of warmth in two participants and palpitations in one participant.
Discussion
This randomized, double-blind, placebo-controlled, two-way crossover study is the first to demonstrate that IV substance P elicits headache and STA dilation in healthy adults. Specifically, 15 (71%) of 21 participants developed headache within 12 hours after receiving substance P, compared with only two (10%) following placebo. Headache onset was often rapid, arising within 30 minutes, and typically mild and pressing. Substance P infusion also caused a significant increase in STA diameter, whereas VMCA changes were less consistent. These findings align with preclinical research implicating substance P in nociceptive transmission, neurogenic inflammation and dilation within the trigeminovascular system (11,21). Collectively, our results underscore that targeting substance P signaling might offer novel therapeutic opportunities in headache disorders. Given the headache-eliciting properties of substance P, further experimental studies are warranted in patients with headache disorders.
Similarities with other peptidergic headache triggers
Our findings reinforce the mechanistic importance of neuropeptides in headache pathogenesis and position substance P alongside CGRP and PACAP. Both CGRP and PACAP reliably induce headache and arterial dilation in healthy adults. For example, an experimental study reported that seven (64%) of 11 healthy adults experienced headache after CGRP infusion (2). Substance P shares these properties and is co-expressed with CGRP in primary afferent neurons of the trigeminal ganglion (22–25). This co-expression might amplify nociceptive signaling and promote the release of additional pro-inflammatory mediators. Notably, substance P appears consistently co-expressed with CGRP in dural nerve fibers and trigeminal ganglion neurons (26), whereas CGRP can exist in some trigeminal neurons without substance P expression (26). These expression patterns should be further studied to determine whether these neuropeptides converge on shared trigeminovascular pathways, potentially informing novel therapeutic strategies. Since CGRP and PACAP also trigger migraine attacks in people with migraine, an important next step is to test whether IV substance P elicits migraine attacks as well.
Hypothesized mechanisms of substance P–induced headache
Mounting preclinical evidence implicates substance P in the pathogenesis of head pain (22,24,25,27,28). Preclinical experiments have demonstrated that trigeminal ganglion neurons can produce substance P (22,23). Once synthesized, it is packaged into dense-core vesicles and transported to both peripheral and central nerve terminals (29). The primary actions of substance P occur through binding to neurokinin-1 receptors (NK1-R), which are widely expressed in the trigeminovascular system, including on endothelial cells and neurons (11). Substance P also binds – with much lower affinity – to neurokinin-2 (NK2-R) and neurokinin-3 (NK3-R) receptors (11). Activation of these receptors facilitate distinct signaling cascades that broaden its effects (11). In light of these receptor interactions and the available preclinical evidence, we propose four principal mechanisms by which substance P might cause head pain.
One proposed mechanism of action is that substance P binds to its NK1-Rs on meningeal nociceptors (30,31), leading to their activation. Although direct in vivo evidence is missing, it has been demonstrated that application of substance P on trigeminal ganglion slices can depolarize the cell bodies of neurons in a dose-dependent manner (12,13). This excitatory effect might extend to peripheral terminals, including those innervating meningeal tissues. Therefore, it is essential to determine whether systemic administration of substance P activates and sensitizes meningeal nociceptors in vivo. In this context, rodent studies using meningeal nociceptor electrophysiological recordings would be invaluable.
A second plausible mechanism involves substance P-induced nitric oxide (NO) release in the meningeal vasculature (32–34). Substance P can bind to its NK1-R on endothelial cells, prompting NO production (32–34), a known molecular headache trigger (14). Human provocation studies have demonstrated that IV NO donors cause headache in healthy adults (35,36), migraine attacks in people with migraine (37) and tension-type headache in those with chronic tension-type headache (38). Evidence from randomized clinical trials has also shown that non-selective inhibition of NO synthase (NOS) with
A third possible mechanism involves substance P-mediated release of pro-inflammatory mediators from mast cells. Compelling evidence indicates that substance P can bind to the Mas-related G protein-coupled receptor X2 (MRGPRX2) and its rodent orthologues (43). Activation of MRGPRX2 causes the release of histamine (44), a known molecular headache trigger and arterial dilator in humans (45). Future research should explore novel drugs targeting MRGPRX2, given that it can also be activated by PACAP (46).
A final mechanistic consideration is the action of substance P on the spinal trigeminal nucleus. Preclinical studies indicate that substance P can be released from the central terminals of primary afferent neurons (47), where it binds to NK1-R on second-order neurons (48), facilitating excitatory nociceptive transmission. Given our study's systemic route of administration, this mechanism assumes that exogenous substance P crosses the blood–brain barrier to reach NK1-R in the spinal trigeminal nucleus. Some evidence suggests that substance P can alter blood-brain barrier permeability, enabling its CNS entry (49). In addition, substance P is known to potentiate the activity of glutamate (50,51), the principal excitatory neurotransmitter in nociceptive pathways, potentially amplifying pain signals.
Taken together, our findings underscore the importance of elucidating peptidergic signaling in headache pathogenesis. Although NK1-R antagonists failed in small clinical trials for acute and preventive migraine treatment (52–54), novel strategies might still hold therapeutic promise. These strategies could include optimized formulations or combination therapies that simultaneously target multiple G protein-coupled receptors. Downstream inhibitions of effectors such as eNOS might further refine therapeutic efficacy and reduce off-target effects. In addition to these approaches, substance P interacts with receptors beyond NK1-R, including MRGPRX2 on mast cells (43). This broader receptor engagement implies that MRGPRX2-mediated signaling could represent an unexplored drug target for headache disorders. Furthermore, it is plausible that substance P causes head pain via multiple receptors, suggesting that its ligand might achieve broader inhibition of pro-nociceptive actions compared with single-receptor antagonism. Future investigations should clarify the molecular interactions among these signaling pathways to support the design of targeted therapies for headache disorders.
Limitations
This study has several limitations. First, the in-hospital phase lasted only one hour for feasibility purposes, after which participants were discharged and asked to document outcome data hourly until 12 hours post-infusion start. Environmental factors, such as certain foods or daily activities, might have influenced the subsequent onset of headache. Nevertheless, most participants reported experiencing headache during the in-hospital phase, suggesting that substance P is indeed a molecular headache trigger. Second, we restricted enrollment to healthy adults without any personal history of headache disorders. This approach ensured a homogenous study population but prevented us from examining whether substance P can induce headache differently in people with headache disorders. Third, we administered a single IV dose of substance P (1.5 pmol/kg/min). It remains unknown whether higher doses might have elicited headache of different character or intensity. Indeed, most participants experienced a transient, mild headache, which might, in part, reflect the relatively modest dose used for safety reasons. Fourth, although we measured STA diameter and VMCA, we did not record meningeal arterial dilation or direct markers of neurogenic inflammation. Thus, our data cannot confirm the exact site of nociceptor activation that led to headache. Fifth, although our homogeneous sample of healthy adults minimizes confounding factors, it also limits generalizability. The participants are thus not fully representative of the adult general population. Finally, while our findings imply a causal role for substance P in headache, further preclinical work is needed to clarify its underlying mechanism.
Conclusions
Our results demonstrate that IV substance P elicits headache and arterial dilation in healthy adults. These findings bolster the hypothesis that substance P might be a pathogenic driver in headache disorders, such as migraine, cluster headache and post-traumatic headache. Continued research is warranted to assess dose-response relationships, co-mediator interactions and the potential for targeting substance P signaling to treat headache disorders.
Clinical implications
Intravenous infusion of substance P induced headache in 15 (71%) 21 healthy adults, compared with only two (10%) following placebo.
Substance P administration significantly dilated the superficial temporal artery, although its effects on middle cerebral artery blood flow were less consistent.
These results support substance P as a potential mediator of head pain, suggesting that targeting its signaling might offer therapeutic promise for headache disorders.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251336132 - Supplemental material for Effects of substance P on headache induction and arterial dilation in healthy adults
Supplemental material, sj-docx-1-cep-10.1177_03331024251336132 for Effects of substance P on headache induction and arterial dilation in healthy adults by Haidar M. Al-Khazali, Rune H. Christensen, Emil Gozalov, Zahra Hakimzadeh, Anna G. Melchior, Rogelio Dominguez-Moreno, Messoud Ashina and Håkan Ashina in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024251336132 - Supplemental material for Effects of substance P on headache induction and arterial dilation in healthy adults
Supplemental material, sj-pdf-2-cep-10.1177_03331024251336132 for Effects of substance P on headache induction and arterial dilation in healthy adults by Haidar M. Al-Khazali, Rune H. Christensen, Emil Gozalov, Zahra Hakimzadeh, Anna G. Melchior, Rogelio Dominguez-Moreno, Messoud Ashina and Håkan Ashina in Cephalalgia
Footnotes
Author contributions
HMA, RHC, MA and HA were responsible for study concept and design. HMA, RHC, EG, ZH, AGM, RD, MA and HA were responsible for data acquisition and analysis. HMA, RHC and HA were responsible for drafting of the manuscript. HMA, RHC, EG, ZH, AGM, RD, MA and HA were responsible for critical revision of the manuscript. HA was responsible for Funding acquisition.
Data availability statement
Upon reasonable request, the necessary data and materials are available from the corresponding author to interested researchers for the purpose of academic scrutiny, reproducibility and further scientific investigation.
Declaration of conflicting interests
HMA has received personal fees from Pfizer, outside of the submitted work. RHC has received personal fees from Teva, outside of the submitted work. MA has received personal fees from AbbVie, Amgen, AstraZeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer and Teva, outside of the submitted work. MA is also a Primary Investigator for ongoing clinical trials sponsored by AbbVie, Lundbeck and Pfizer. MA also serves as an Associate Editor of Brain and The Journal of Headache and Pain. HA has received personal fees from AbbVie, Lundbeck, Pfizer and Teva, outside of the submitted work. HA also serves as an Editorial Board Member of The Journal of Headache and Pain. The remaining authors declare no potential conflicts of interest.
Ethical statement
All participants provided their written informed consent before the commencement of any study-related assessments or procedures.
Funding
This work was supported by a research grant from the Lundbeck Foundation (R403-2022-1352 to HA).
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
