Abstract
Objective
To compare clinical characteristics, treatment patterns, and patient-reported outcome measures (PROMs) in persons with persistent post-traumatic headache (PTH) and those with episodic or chronic migraine (with and without medication-overuse headache).
Methods
This cross-sectional study included adults with persistent PTH following mild traumatic brain injury and adults with episodic or chronic migraine. Information on headache features, treatment patterns, and PROMs were collected via semi-structured interviews and standardized questionnaires, including the HIT-6, MIDAS, HADS, ASC-12, CSI, WHODAS-2.0, NDI, and ODI.
Results
A total of 132 participants with persistent PTH and 751 with migraine were enrolled. On average, participants with persistent PTH reported 27.2 ± 9.3 monthly headache days, and 93.9% had a migraine-like phenotype. Compared with episodic migraine, the persistent PTH group had higher MIDAS scores (72.5 [IQR, 28.0–156.0] vs. 40.0 [IQR, 20.0–62.8]; P < 0.001), HADS-Anxiety (6.0 [IQR, 3.3–9.0] vs. 5.0 [IQR, 3.0–7.0]; P = 0.003), higher HADS-Depression scores (6.0 [IQR, 3.0–9.0] vs. 3.0 [IQR, 1.0–5.0]; P < 0.001), CSI (67.2 [SD, 15.0] vs. 57.4 [SD, 12.5]; P = 0.002), WHODAS-2.0 (41.0 [IQR, 15.5–68.5] vs. 22.0 [IQR, 10.0–35.0]; P < 0.001), and NDI (21.0 [IQR, 16.0–27.0] vs. 14.5 [IQR, 8.0–22.0]; P = 0.007). No marked differences were noted between the PTH group and chronic migraine groups.
Conclusions
Persons with persistent PTH show similar disability and headache profiles to those with chronic migraine. These findings underscore the need for targeted management strategies, similar to those used in chronic migraine care.
This is a visual representation of the abstract.
Keywords
Introduction
Persistent post-traumatic headache (PTH) is a prevalent and disabling sequela of traumatic brain injury (TBI), yet it remains underrecognized and inadequately characterized in clinical practice (1–3). Although persistent PTH can result from TBI of any severity, mild TBI (mTBI) is the most predominant cause of injury (4,5). Epidemiologic data indicate that a considerable proportion of persons sustaining an mTBI continue to experience PTH one-year post-injury (6,7). Moreover, lifetime prevalence estimates suggest that PTH afflicts 4.7% of men and 2.4% of women (8,9), underscoring a substantial impact on public health and the need for greater clinical attention.
Although the International Classification of Headache Disorders, 3rd edition (ICHD-3), provides diagnostic criteria for persistent PTH (10), detailed clinical profiles of affected patients are limited (4–6). Clinically, most patients with persistent PTH report near-daily to daily headache resembling migraine, with a smaller subset presenting features of pure tension-type headache (TTH) (9,11–13). In support, one study found that 91 (91%) of 100 patients with persistent PTH after mTBI experienced recurrent migraine-like headache (14), underscoring considerable clinical overlap with primary headache disorders. Furthermore, both persistent PTH and migraine are associated with psychiatric comorbidities, cutaneous allodynia, and disruptions in daily functioning (15–19). However, it remains unclear whether persons with persistent PTH experience similar levels of disability and comorbidities as those with episodic or chronic migraine.
A comprehensive assessment of comorbidities and levels of disability in persons with persistent PTH is essential for guiding optimal clinical management. To address this gap, we conducted the first study to compare the clinical features, treatment patterns, and patient-reported outcomes (PROMs) between adults with persistent PTH and those with episodic or chronic migraine. We hypothesized that persons with persistent PTH would exhibit clinical characteristics and disability profiles similar to those with chronic migraine, including those with medication-overuse headache (MOH).
Methods
This study was approved by the relevant ethics committee of Capital Region, Denmark (Identifier: 20019411) and conducted in accordance with the principles of the Declaration of Helsinki, with later revisions (20). All participants provided written informed consent before enrollment.
Design and participants
We conducted a cross-sectional observational study at a single center from September 2020 to June 2023. Eligible participants were adults aged 18 to 65 years. The persistent PTH group was mainly recruited from the outpatient clinic of a national referral hospital. The migraine group was derived from the prospective Registry for Migraine (REFORM) study, detailed elsewhere (21).
The persistent PTH group included persons who had experienced a mTBI at least 12 months prior to enrollment. Eligibility required a diagnosis of persistent PTH attributed to mTBI in accordance with the ICHD-3 criteria for persistent headache attributed to mild traumatic injury to the head. In addition, participants were required to report a minimum of four headache days per month during the three months preceding enrollment (10). The key exclusion criteria were a history of primary or secondary headache disorders before the mTBI (except for infrequent episodic TTH) and any history of moderate-to-severe TBI, whiplash injury, or craniotomy. Participants using preventive headache medication(s) were eligible if their dosage had remained stable for at least two months prior to enrollment.
The migraine population comprised individuals diagnosed with migraine without aura, migraine with aura, or chronic migraine (with or without MOH), based on ICHD-3 criteria (10). Participants needed a history of migraine for at least one year and had to report four or more migraine days per month in the three months before enrollment. The main exclusion criteria included any history of cluster headache, hemiplegic migraine, or persistent PTH. The full list of eligibility criteria has been published elsewhere (21).
Trained personnel verified the eligibility of all participants through reviews of electronic medical records. A complete account of inclusion and exclusion criteria is provided in Online Supplemental Tables 1 and 2.
Demographics Among persons with persistent post-traumatic headache and migraine subgroups.

Data are mean (SD), median (IQR), or no (%).
PPTH: persistent PTH; EM: episodic migraine; CM: chronic migraine; MOH: medication-overuse headache; no: number.
Clinical characteristics Among persons with persistent post-traumatic headache and migraine subgroups.

Data are mean (SD), median (IQR), or no (%).
PPTH: persistent PTH; EM: episodic migraine; CM: chronic migraine; MOH: medication-overuse headache; n: number.
Procedures
Potential participants were initially screened via telephone. Those who met preliminary eligibility criteria were invited for in-person, semi-structured interviews. During these interviews, two physicians (H.M.A. and R.H.C.) conducted thorough examinations and detailed questioning. The participants were then provided with electronic PROMs using the Research Electronic Data Capture (REDCap) software and were instructed to complete these within 24 h.
Two physicians (H.M.A. and R.H.C.) collected sociodemographic data, clinical characteristics, and comprehensive medical histories. Information gathered included details on headache frequency, phenotype, intensity, location, accompanying symptoms, autonomic features, trigger factors, comorbidities, and treatment.
Outcome measures
The Headache Impact Test-6 (HIT-6) was used to evaluate the impact of headache on daily functioning (22) and the Migraine Disability Assessment (MIDAS) was employed to assess headache-related disability (23). Detailed descriptions of these instruments, including scoring and interpretation, are available in the Online Supplementary Appendix.
Hospital Anxiety and Depression Scale
The Hospital Anxiety and Depression Scale (HADS) is a self-administered, 14-item questionnaire designed to screen for anxiety and depression over the past seven days (24). It consists of two subscales — HADS-Anxiety and HADS-Depression — each containing seven items. Participants rate each item on a four-point Likert scale, resulting in subscale scores ranging from 0 to 21. Scores of 0 to 7 on either subscale indicate no risk of anxiety or depression; scores of 8 to 10 suggest a possible risk; and scores of 11 or higher denote a probable risk.
Allodynia Symptom Checklist
The Allodynia Symptom Checklist-12 (ASC-12) assesses the presence and severity of cutaneous allodynia (25). The questionnaire comprises 12 items, each contributing to a total score between 0 and 24. Scores are categorized as mild allodynia (3–5), moderate allodynia (6–8), or severe allodynia (9 or higher). In this study, participants were classified as having cutaneous allodynia if they scored 3 or higher, and as not having cutaneous allodynia if they scored between 0 and 2.
Central Sensitization Inventory
The Central Sensitization Inventory (CSI) is a self-administered questionnaire designed to identify symptoms associated with central sensitization (26). It comprises 25 items, each rated on a scale from 0 (“never”) to 4 (“always”), yielding a total score between 0 and 100. A total score exceeding 40 indicates the presence of central sensitization symptoms.
World Health Organization Disability Assessment Schedule 2.0
The World Health Organization Disability Assessment Schedule 2.0 (WHODAS 2.0) assesses difficulties in daily activities due to health conditions over the past 30 days (27). We used the 12-item short version (WHODAS-2.0), in which each item is rated on a five-point scale from 1 (“none”) to 5 (“extreme” or “cannot do”). The total score ranges from 12 to 60, with higher scores indicating greater levels of disability.
Neck Disability Index
The Neck Disability Index (NDI) is a 10-item questionnaire used to assess self-reported disability in persons with neck pain (28). It evaluates various aspects of daily life, including pain intensity, personal care, lifting, reading, headache, concentration, work, driving, sleep, and recreational activities. Each item is scored from 0 to 5, yielding a total score between 0 and 50. Higher scores indicate greater disability. Disability levels are categorized as follows: no disability (0–4), mild disability (5–14), moderate disability (15–24), severe disability (25–34), and complete disability (35–50).
Oswestry Disability Index
The Oswestry Disability Index (ODI) is a self-administered questionnaire that assesses disability related to low back pain (29). It covers multiple dimensions of daily function, including pain intensity, physical activity, personal care, lifting, walking, sitting, standing, sleeping, and participation in recreational activities. Each of the 10 items is scored from 0 to 5, resulting in a total score ranging from 0 to 50. Higher scores reflect greater levels of disability. Disability levels are categorized as: no disability (0–4), mild disability (5–14), moderate disability (15–24), severe disability (25–34), and complete disability (35–50).
Case definitions in the persistent PTH group
In the persistent PTH group, headache phenotypes were classified based on ICHD-3 criteria (10). A migraine-like headache was defined as a headache lasting at least 4 h and meeting ICHD-3 criteria C and D for migraine without aura (10). Specifically, participants had to experience at least two of the following pain features: unilateral location, pulsating quality, moderate or severe intensity, or aggravation by or causing avoidance of routine physical activity. In addition, they needed to have at least one of the following during the headache: nausea/vomiting, or both photophobia and phonophobia. For participants reporting nausea/vomiting, photophobia, or phonophobia, these symptoms were considered as accompanying symptoms only if their severity increased as the pain intensity worsened.
A chronic migraine-like headache phenotype was defined by headache occurring on 15 or more days per month for over three months, with at least eight days per month fulfilling ICHD-3 criteria C and D for migraine without aura (10).
A TTH-like phenotype was defined by headache meeting ICHD-3 criteria A through D for either infrequent episodic TTH, frequent episodic TTH, or chronic TTH (10). These criteria include features such as bilateral location, pressing or tightening quality, mild or moderate intensity, and not being aggravated by or causing avoidance of routine physical activity. Furthermore, these headache episodes were characterized by the absence of nausea/vomiting and no more than one of photophobia or phonophobia.
Case definitions in the migraine population
The participants in the migraine population were assigned to one of the following mutually exclusive groups: (I) episodic migraine, (II) chronic migraine without MOH, or (III) chronic migraine with MOH. Episodic migraine was defined as having fewer than 15 headache days per month. Conversely, chronic migraine was characterized by headache occurring on 15 or more days per month for more than three months, with at least eight days per month meeting the criteria for migraine without aura (10).
The chronic migraine with MOH group comprised individuals who met the criteria for both chronic migraine and medication-overuse. Specifically, these participants had used combination analgesics, triptans, ergot alkaloids, or opioids on ten or more days per month, or simple analgesics on 15 or more days per month, within the past three months. Inclusion in this group required fulfillment of both the chronic migraine and medication-overuse ICHD-3 criteria (10).
Statistical analysis
A sample size calculation was not conducted, as the current study is exploratory in nature. Descriptive statistics were used to summarize demographic and clinical characteristics of the study population. For normally distributed continuous variables, we reported means with standard deviations (SD); for non-normally distributed data, we presented medians with interquartile ranges (IQR). The normality of continuous variables was evaluated using visual inspection of QQ-plots and histograms. Group comparisons for continuous data were performed using the Welch t-test for normally distributed data, as it provides greater robustness in cases where there is unequal variance between groups (heteroskedasticity) (30), which we expected in this dataset. We chose the Welch test over the Student's t-test due to its ability to provide more accurate results in such conditions, even though the latter is more commonly used. If the data distribution was non-normally distributed, we applied the Wilcoxon rank-sum test. Categorical variables were summarized as counts and percentages, and we compared groups using Chi-squared tests. Missing data were handled via complete case analysis. Statistical significance was defined as a two-sided P value less than 0.05. To reduce the risk of type I error in the analyses involving PROMs, we controlled the false discovery rate with the Benjamini-Hochberg procedure. To distinguish persistent PTH from migraine, we performed a binomial multivariable logistical regression analysis. The model included unilateral headache, higher pain intensity, pulsating headache, nausea, monthly headache days, age, and sex as predictors. Multicollinearity was assessed using the Variance Inflation Factor, with a threshold of 5. Moreover, we tested linearity to the logit of continuous variables using Box-Tidwell's test. The model was fitted using the “binomial” family, and odds ratios (OR) with 95% confidence intervals (95% CI) were calculated to determine the strength and direction of associations. This approach allowed us to identify key variables linked to the likelihood of having persistent PTH versus migraine. All statistical analyses were performed using R statistical software (version 4.2.0).
Results
From September 2020 to June 2023, we enrolled 132 participants with persistent PTH and 751 with migraine. The persistent PTH group had a mean age of 40.6 ± 1.6 years, and most were female (n = 106; 76.5%). Incidental falls were the most frequent cause of mTBI (24.2%, [n = 32]), followed by road traffic accidents (20.5%, [n = 27]) and sports-related injuries (20.5%, [n = 27]). The median time since mTBI was 8.9 ± 3.0 years. Detailed characteristics of the persistent PTH group are provided in Table 1.
In the migraine population, the mean age was 43.8 ± 12.2 years, with 88.8% (n = 667) participants being female (Table 1). Among these participants, 30.2% (n = 227) had episodic migraine, 38.3% (n = 288) had chronic migraine without MOH, and 31.4% (n = 236) had chronic migraine with MOH. Compared with the migraine groups, participants with persistent PTH were younger than those with episodic migraine (P = 0.003) and chronic migraine with MOH (P = 0.001), but not significantly younger than those with chronic migraine without MOH (P = 0.218). The proportion of females was significantly lower in the persistent PTH group, compared with all three migraine groups (All P < 0.05).
Clinical characteristics
The participants with persistent PTH reported an average of 27.2 ± 9.3 headache days per month (Table 2). Almost all (93.9%, [n = 124]) met the criteria for a migraine-like phenotype, while the remaining participants (6.1%, [n = 8]) had a TTH-like phenotype (Online Supplemental Tables 3–6).
In the migraine population, participants with episodic migraine experienced an average of 12.5 ± 3.3 monthly headache days (Table 2). Those with chronic migraine without MOH reported a mean of 22.5 ± 6.8 days, and participants with chronic migraine with MOH averaged 22.2 ± 6.4 days. The number of monthly headache days was significantly higher in the persistent PTH group, compared with all three migraine groups (All P < 0.01).
Among participants with persistent PTH, most (72.0% [n = 95]) reported experiencing headache of bilateral location (Table 2). The corresponding figures were 44.5% (n = 101) in the episodic migraine group, 39.6% (n = 114) in the chronic migraine without MOH group, and 40.7% (n = 96) in the chronic migraine with MOH group (All P < 0.001).
In terms of headache quality, the proportion of participants with persistent PTH reporting pulsating headache (68.2% [n = 90]) did not differ significantly from that of participants with episodic migraine (67.0% [n = 152]; P = 1.00) or those with chronic migraine without MOH (75.7% [n = 218]; P = 0.098). In contrast, the frequency of pulsating headache was significantly higher among participants with chronic migraine with MOH (77.5% [n = 183]) compared with those with persistent PTH (P = 0.046).
Headache aggravation by routine physical activity was common across all groups, with 79.5% (n = 105) of persistent PTH participants reporting this complaint, similar to those with episodic migraine (86.3%, [n = 196]; P = 0.124) and chronic migraine with MOH (87.7%, [n = 207]; P = 0.052) (Table 2). Participants with chronic migraine without MOH had the highest rate of headache aggravation by physical activity (90.7%, [n = 261]; P = 0.003).
Typical headache intensity was reported as moderate-or-severe by nearly all participants with persistent PTH (97.8% [n = 129]). Comparable figures were seen in episodic migraine (95.6% [n = 217]), chronic migraine without MOH (98.7% [n = 284]), and chronic migraine with MOH (98.7% [n = 233]). No significant differences emerged among the four groups in terms of self-reported headache intensity (All P > 0.05; Table 2).
Photophobia and phonophobia were prevalent among participants with persistent PTH and all migraine groups (Table 2). In the persistent PTH group, 93.2% (n = 123) reported photophobia, which did not differ significantly from the migraine groups (All P > 0.05). Similarly, phonophobia was reported by 96.2% (n = 127) among participants with PTH, mirroring the rates observed in all migraine groups (all P > 0.05). By contrast, nausea was reported by 75.0% (n = 99) of persistent PTH participants, a lower relative frequency than in each migraine subgroup (All P < 0.001). Vomiting was also significantly less frequent in participants with persistent PTH (31.1%, n = 41) compared with all migraine groups (All P < 0.001).
Multivariable logistic regression analysis
Multivariable logistic regression identified several factors associated with a lower likelihood of persistent PTH, compared with migraine. Unilateral headache (OR, 0.30; 95% CI, 0.19–0.46; P < 0.001), higher pain intensity (OR, 0.65; 95% CI, 0.56–0.74; P < 0.001), and nausea (OR, 0.49; 95% CI, 0.28–0.87; P = 0.013) were associated with a reduced likelihood of persistent PTH.
In contrast, younger age (OR 0.98; 95% CI, 0.96–0.99; P = 0.039) and female sex (OR, 1.99; 95% CI, 1.14–3.43; P = 0.014) were associated with a higher likelihood of persistent PTH. In addition, a higher number of monthly headache days (OR, 1.03; 95% CI, 1.02–1.04; P < 0.001) increased the probability of persistent PTH, compared with migraine.
Treatment patterns
Among participants with persistent PTH, 89.4% (n = 118) reported using acute headache medications, with an average of 12.1 ± 8.1 days of acute medication use. In contrast, 96.9% (n = 220) of participants with episodic migraine used acute treatments, averaging 10.5 ± 6.0 days of acute medication use (P < 0.001). Participants with chronic migraine without MOH reported a mean of 8.8 ± 4.5 days of acute rescue medication use (P < 0.001), while those with chronic migraine and MOH reported a mean of 17.6 ± 6.1 days of acute medication use (P < 0.001). Table 3 provides detailed information on current and prior failures in acute headache medication use.
Acute headache medication overview Among persons with persistent post-traumatic headache and migraine subgroups.
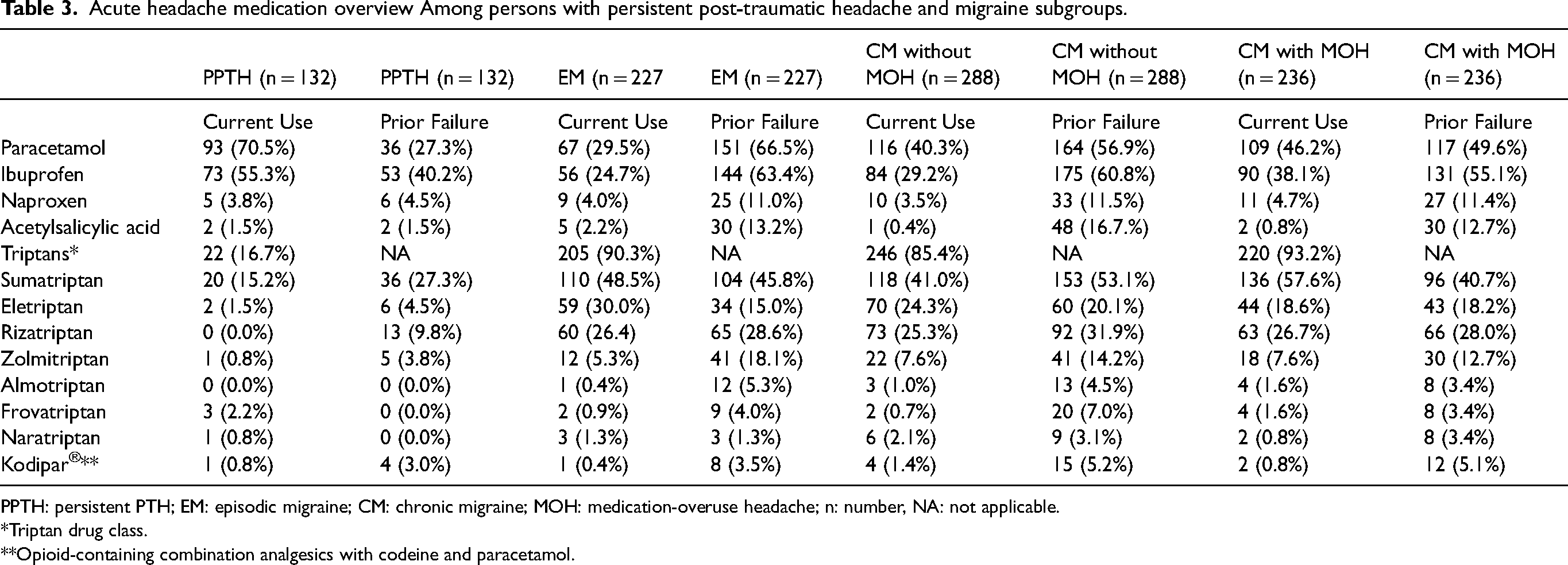
PPTH: persistent PTH; EM: episodic migraine; CM: chronic migraine; MOH: medication-overuse headache; n: number, NA: not applicable.
*Triptan drug class.
**Opioid-containing combination analgesics with codeine and paracetamol.
Current use of paracetamol and ibuprofen was significantly more frequent in persistent PTH, compared with the migraine subgroups (All P < 0.05; Table 3). In contrast, triptan use was significantly lower in the persistent PTH group, as compared with the migraine subgroups (All P < 0.001; Table 3).
Regarding preventive headache medications, 38.7% (n = 51) of participants with persistent PTH reported current use. In the episodic migraine group, 61.2% (n = 139) of participants used preventive treatments (P < 0.001). For chronic migraine without MOH, 53.1% (n = 153) reported using preventive medications (P = 0.008), and in the chronic migraine with MOH group, 50.8% (n = 120) were on preventive treatments (P = 0.03). Table 4 provides a comprehensive overview of current and past preventive medication use.
Pharmacological interventions for preventive treatment of headache in individuals with persistent post-traumatic headache and migraine subgroups.

PPTH: persistent PTH; EM: episodic migraine; CM: chronic migraine; MOH: medication-overuse headache; n: number.
*Compare current usage patterns in the PPTH group with those in the migraine subgroups.
The three most commonly used preventive medications among participants with persistent PTH were amitriptyline, candesartan, and OnabotulinumtoxinA. Amitriptyline was used by 6.1% of participants in both the persistent PTH (n = 8) and episodic migraine groups (n = 14) (P = 1.000), with no use reported in the chronic migraine groups (both P > 0.05). Candesartan was used by 9.8% (n = 13) of persistent PTH participants and 23.8% (n = 54) of episodic migraine participants (P = 0.001), but none of the chronic migraine participants reported using it (both P > 0.05). OnabotulinumtoxinA had broader use, with 8.3% (n = 11) of persistent PTH participants, 15.0% (n = 34) of episodic migraine (P = 0.071), 13.5% (n = 39) of chronic migraine without MOH, and 10.6% (n = 25) of chronic migraine with MOH participants reporting current use (both P > 0.05).
Patient-Reported Outcome Measures
Headache Impact Test-6
The participants with persistent PTH had median HIT-6 scores similar to those with episodic migraine (63 [IQR, 59.5–66.0] vs 63 [IQR, 61.0–65.0]; P = 0.100) (Table 5, Figure 1A). However, their scores were significantly lower than those of participants with chronic migraine without MOH (64.0 [IQR, 63.0–66.0]; P = 0.007) and chronic migraine with MOH (65.0 [IQR, 62.0–66.0]; P = 0.003).
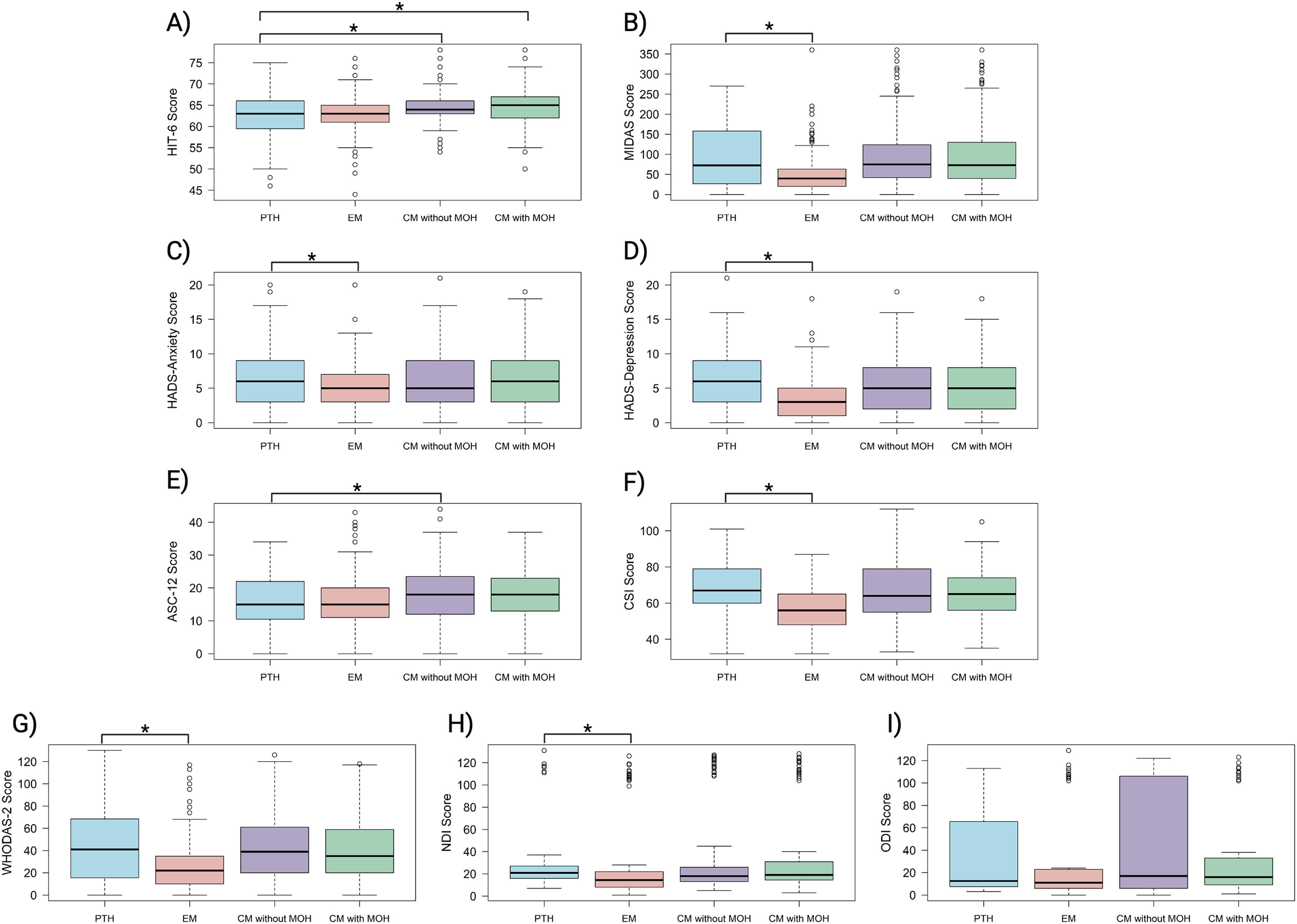
Patient reported outcome measures in persistent post-traumatic headache and migraine subgroups.
Patient reported outcome measures in persistent post-traumatic headache and migraine subgroups.

PPTH: persistent PTH; EM: episodic migraine; CM: chronic migraine; MOH: medication-overuse headache; n: number; HIT-6: Headache Impact; MIDAS: Migraine Disability; HADS: Hospital Anxiety and Depression Scale; ASC-12: Allodynia Symptom Checklist-12; CSI: Central Sensitization Inventory; WHODAS-2.0: World Health Organization Disability Assessment Schedule 2.0; NDI: Neck Disability Index and ODI: Oswestry Disability Index.
Migraine Disability Assessment
The median MIDAS score for the persistent PTH group was significantly higher than that of the episodic migraine group (72.5 [IQR, 28.0–156.0] vs 40.0 [IQR, 20.0–62.8]; P < 0.001) (Table 5, Figure 1B). The MIDAS scores in the persistent PTH group were comparable to those in the chronic migraine without MOH group (75.0 [IQR, 42.0–123.2]; P = 0.805) and the chronic migraine with MOH group (73.0 [IQR, 40.0–130.0]; P = 0.981).
Hospital Anxiety and Depression Scale
The median HADS-Anxiety scores were significantly higher in the persistent PTH group, compared with the episodic migraine group (6.0 [IQR, 3.3–9.0] vs 5.0 [IQR, 3.0–7.0]; P = 0.003) (Table 5, Figure 1C). Likewise, the HADS-Depression scores were also higher in the persistent PTH group than in the episodic migraine group (6.0 [IQR, 3.0–9.0] vs 3.0 [IQR, 1.0–5.0]; P < 0.001) (Figure 1D). No significant differences were found between the persistent PTH group and the chronic migraine groups on either HADS subscale.
Allodynia Symptom Checklist-12
The mean ASC-12 scores were significantly lower in the persistent PTH group compared with the chronic migraine without MOH group (16.0 [SD, 7.6] vs 18.4 [SD, 8.3]; P = 0.042) (Table 5, Figure 1E). There were no significant differences between the persistent PTH group and those in the episodic migraine group or the chronic migraine with MOH group.
Central Sensitization Inventory
The mean CSI scores were significantly higher in participants with persistent PTH, compared to those with episodic migraine (67.2 [SD, 15.0] vs. 57.4 [SD, 12.5]; P = 0.002) (Table 5, Figure 1F). In contrast, the mean CSI scores were similar between the persistent PTH group and both chronic migraine groups.
World Health Organization Disability Assessment Schedule 2.0
The mean health-related disability scores, measured using WHODAS 2.0, were significantly higher in the persistent PTH group than in those with episodic migraine (41.0 [IQR, 15.5–68.5] vs 22.0 [IQR, 10.0–35.0]; P < 0.001) (Table 5, Figure 1G). Conversely, the WHODAS scores were comparable between the persistent PTH group and the chronic migraine groups.
Neck Disability Index and Oswestry Disability Index
The participants with persistent PTH had higher median NDI scores than those with episodic migraine (21 [IQR, 16–27] vs 14.5 [IQR, 8.0–22.0]; P = 0.007) (Table 5, Figure 1H). The NDI scores were similar between the persistent PTH group and both chronic migraine groups. Moreover, no significant differences were found in ODI scores between participants with persistent PTH, compared to all migraine groups (Figure 1I).
Discussion
In this cross-sectional study, we compared the clinical profiles, treatment patterns, and PROMs among adults with persistent PTH and those with episodic or chronic migraine (with or without MOH). Our principal finding is that, although the headache phenotype in persistent PTH is predominantly migraine-like, individuals with PTH display several distinct features. They report a higher frequency of bilateral pain, slightly lower mean headache intensity, and less frequent triptan use. The reduced use of triptans might primarily reflect differences in physician prescribing practices in addition to less marked efficacy.
Despite these differences, disability burdens, psychiatric comorbidities, and central sensitization indices in the persistent PTH group approximate those observed in people with chronic migraine. These results support our original hypothesis that persistent PTH shares many clinical and disability-related characteristics with chronic migraine. However, persistent PTH appears to differ in headache location, intensity, and triptan use. Taken together, our findings underscore the need for greater awareness and tailored management strategies for persistent PTH, particularly regarding appropriate therapeutic interventions for headache arising after mTBI.
Clinical characteristics and headache phenotype
Our findings reinforce earlier observational research showing that the clinical features of persistent PTH often overlap with primary headache disorders (7,9,11–14). In the present sample, 94% (124 of 132) of participants had a migraine-like phenotype, while the remaining eight participants met the criteria for a TTH-like phenotype. These proportions mirror those reported in a previous cross-sectional study, which found that 91 of 100 individuals with persistent PTH fulfilled migraine-like criteria (14). A recent 28-day diary study also noted similar trends, with 56 of 64 participants having a migraine-like phenotype (11).
Despite this strong overlap, our participants with persistent PTH reported substantially more monthly headache days (27.2 vs. 12.5) than those with episodic migraine. This finding underscores the disabling nature of persistent PTH and suggests that its burden can match or surpass that of episodic migraine. This is further underlined by our participants with episodic migraine having a relatively high burden of attacks, when compared to individuals with episodic migraine in the general populations. Clinically, it might therefore be more useful to compare persistent PTH with chronic migraine rather than episodic migraine, given the more similar frequency of headache days observed in these populations.
When examining headache intensity, our participants with persistent PTH reported moderate-to-severe pain at rates comparable to those with episodic or chronic migraine, including those with MOH. This similarity in pain severity underscores the debilitating nature of persistent PTH and further emphasizes the shared clinical manifestations between persistent PTH and migraine. Hence, clinicians are advised not to rely on headache intensity alone when differentiating persistent PTH from migraine.
Notably, our data also showed a higher frequency of bilateral pain in participants with persistent PTH, compared with both episodic and chronic migraine groups. Although bilateral headache is often associated with TTH (31), many participants still fulfilled migraine-like criteria. These results imply that persistent PTH might differ from migraine in its tendency toward bilateral pain. Such distinctions could reflect distinct pathophysiological processes, although this remains speculative.
Regarding associated symptoms, rates of photophobia and phonophobia in persistent PTH were comparable to those in both episodic and chronic migraine. This finding aligns with existing reports documenting clinical overlap between persistent PTH and migraine (14). However, nausea and vomiting were less prevalent in the persistent PTH group than in any migraine group. These observations might reflect different underlying neurobiologic pathways in persistent PTH versus migraine (32,33). While photophobia and phonophobia appear to be common to both disorders, the lower prevalence of nausea and vomiting in persistent PTH could aid clinicians in distinguishing it from migraine in clinical practice.
Treatment patterns
A novel aspect of our findings is the detailed characterization of treatment patterns in persistent PTH relative to episodic and chronic migraine. Our data show that people with persistent PTH more frequently use simple analgesics, such as paracetamol and ibuprofen, whereas triptans are used less frequently compared to those with migraine. This pattern might reflect limited awareness that PTH can present with migraine-like features (5,34). Clinicians might favor generic analgesics for what they perceive as “post-traumatic” headache, rather than considering triptans. In addition, an incomplete phenotype-based classification of persistent PTH might reduce the use of migraine-specific therapies. However, it remains important to note that persistent PTH is not migraine (35) and, in absence of robust randomized controlled trials, a degree of caution in prescribing migraine-specific medication might be justified (5,34,36).
Our results also indicate that fewer participants with persistent PTH receive preventive headache medications than those with episodic or chronic migraine. Several factors could contribute to this discrepancy. Healthcare providers might prioritize non-pharmacological interventions, such as physiological therapy or physical therapy for persistent PTH (37–39). Undertreatment might also occur if clinicians do not fully recognize the severity of persistent PTH and its associated comorbidities. These gaps underscore the need for well-designed randomized controlled trials that evaluate both conventional and novel preventive headache medications. Such research would help guide clinicians toward more effective management strategies and reduce the substantial burden that PTH imposes.
Patient-Reported Outcome Measures
The present study used a panel of PROMs to provide insight into shared features and subtle differences between persistent PTH and migraine. Although both HIT-6 and MIDAS evaluate headache-related disability, they quantify slightly distinct domains. In our sample, HIT-6 scores for persistent PTH were similar to those for episodic migraine but were lower than those for chronic migraine. By contrast, MIDAS scores in the persistent PTH group exceeded those in episodic migraine and approximated those in the chronic migraine groups. These findings might reflect the distinct assessment windows and focuses of HIT-6 and MIDAS: HIT-6 addresses the broad impact of headache across multiple life areas (22), whereas MIDAS quantifies the number of days with lost or reduced function (23). Concomitant factors such as psychiatric comorbidities or other post–mTBI sequelae could further contribute to the functional burden observed in patients with persistent PTH.
Psychiatric comorbidities also emerged as a key factor distinguishing PTH from episodic migraine. Rates of anxiety and depression symptoms in persistent PTH resembled those found in chronic migraine but exceeded those in episodic migraine. Observational data suggest that psychiatric symptoms and mild cognitive impairment often accompany PTH (16,40–42). Chronic migraine and other chronic pain disorders also exhibit higher rates of anxiety and depression, reinforcing the link between these psychiatric comorbidities and more disabling headache phenotypes (17,43,44). The high burden of psychiatric symptoms in PTH might be driven by frequent headache and by direct effects of mTBI on brain function (32). These findings underscore the importance of multidisciplinary care that addresses both persistent PTH and its associated comorbidities.
Our data further revealed a shared pattern of cutaneous allodynia in both persistent PTH and chronic migraine, implying a role for central sensitization in both disorders (5,45–47). Allodynia scores were slightly higher in chronic migraine, suggesting that certain pain-processing mechanisms might differ between the two disorders (32,33,48-50). The CSI also supported this notion, as participants with persistent PTH had higher scores than those with episodic migraine. These findings suggest that persistent PTH can involve more widespread symptoms tied to central sensitization, placing it closer to the chronic migraine spectrum (51). However, further research is needed to understand the relative contribution of repeated nociceptive input from PTH versus mTBI-related alterations.
Neck disability further distinguishes persistent PTH from episodic migraine. The participants with persistent PTH reported significantly higher NDI scores, consistent with prior work showing that neck pain is common in PTH and contributes to functional limitations (4,52). Within migraine populations, neck pain is about twice as frequent in chronic migraine, compared with episodic migraine (53), and mean NDI scores are significantly higher in chronic versus episodic migraine (54). Taken together, these findings suggest that persistent PTH and chronic migraine share overlapping mechanisms. The observed increased neck disability might reflect convergence of nociceptive pathways within the trigeminocervical complex (55).
Limitations
This study has several limitations. First, our cross-sectional design restricts the ability to infer causal relationships or to document longitudinal changes in the clinical evolution of persistent PTH. Second, our recruitment strategy might be susceptible to referral patterns and volunteer biases. Both participants with persistent PTH and those with migraine were largely recruited from an outpatient referral clinic, potentially selecting more severe and adversely affected cases. Third, we relied on self-report and recall-based interviews and PROMs to record information on medical history and measures of disability. Lastly, although missing data were minimal, gaps in responses could influence results.
Conclusions
Most people with persistent PTH exhibit migraine-like phenotypes, with disability levels that approximate those found in chronic migraine. However, persistent PTH remains underrecognized and undertreated, emphasizing the need for improved characterization and randomized controlled trials to guide effective clinical management. Furthermore, recognizing and addressing associated comorbidities might optimize outcomes and improve quality of life for this frequently overlooked patient population.
Key findings
The majority (94%) of participants with persistent PTH meet the criteria for migraine-like headache. Persistent PTH imposes a level of disability comparable to chronic migraine and higher than that of episodic migraine. Participants with persistent PTH report more frequent use of simple analgesics like paracetamol and ibuprofen, compared to those with migraine.
Footnotes
Author contributions
Concept and design: HA. Acquisition, analysis, or interpretation of data: HMA, RHC, HA. Drafting of the manuscript: HMA, RHC, HA. Critical revision of the manuscript for important intellectual content: HMA, RHC, HA. Statistical analysis: HMA, RHC, HA. Administrative, technical, or material support: MHA. Supervision: HA. All authors critically reviewed the manuscript and vouch for the accuracy and completeness of the data.
Availability of data and materials
Upon reasonable request, the corresponding author will provide the necessary data and materials to interested researchers for the purpose of academic scrutiny, reproducibility, and further scientific investigation.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HMA has received personal fees from Lundbeck and Pfizer that are unrelated to this manuscript. RHC has received personal fees from Teva that are unrelated to this manuscript. HA has received personal fees from AbbVie, Lundbeck, Pfizer, and Teva that are unrelated to this manuscript. HA is an Editorial Board Member of The Journal of Headache and Pain.
Ethics
This study was approved by the relevant ethics committee and conducted in accordance with the principles of the Declaration of Helsinki. All participants provided written informed consent before enrollment.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding from the Lundbeck Foundation (R403–2022–1352 to HA). Lundbeck Foundation, (grant number R403-2022-1352).
Supplementary material
Supplementary material for this article is available online.
