Abstract
Background
The Migraine Disability Assessment (MIDAS) is widely used. However, there are limited data on how much a reduction in the MIDAS score indicates a change that matters to the patient.
Methods
Data from the DMKG (i.e. German Migraine and Headache Society) Headache Registry were used to determine the minimal important difference (MID) of the MIDAS, using the Patient Global Impression of Change (PGIC) as anchor and applying average change and receiver operating characteristic curve methods.
Results
In total, 1218 adult migraine patients (85.6% female, 40.2 ± 12.8 years, baseline MIDAS 44.2 ± 47.4, follow-up MIDAS 36.5 ± 45.3) were included. For patients with baseline MIDAS >20 (MIDAS grade IV, n = 757), different methods using PGIC “somewhat improved” as anchor yielded percent change MIDs of the MIDAS between −29.4% and −33.2%. For baseline MIDAS between 6 and 20 (grades II and III, n = 334), using PGIC “much improved” as anchor, difference change MIDs were between −3.5 and −4.5 points.
Conclusions
Based on the above results, we estimated the MID of the MIDAS at −30% for patients with a baseline MIDAS >20, and at −4 points for those with a baseline MIDAS of 6–20, for a tertiary headache care population.
Trial Registration
The DMKG Headache Registry is registered with the German Clinical Trials Register (DRKS 00021081).
Introduction
The Migraine Disability Assessment (MIDAS) (1) is one of the most frequently used patient-reported outcome measures (PROM) of migraine-associated disability (2) and a recommended outcome parameter for clinical migraine trials (3,4). Interpretation is facilitated by the MIDAS grades, ranging from little or no disability (grade I) to severe disability (grade IV). 1 Although the MIDAS has been shown to be responsive to treatment effects (5,6), there is little evidence on what would be the minimal change in MIDAS to indicate an improvement that matters to the patient, in other words a ‘minimal clinically important difference’ (MCID), now more often termed ‘minimal important difference’ (MID) (7,8). One previous study on 87 patients with chronic or high-frequency episodic migraine determined an MID of the modified MIDAS (with a 1-month recall period) of −4.5 points based on a Patient Global Impression of Change (PGIC) rating of at least “somewhat better”, which tentatively might be extrapolated to −13.5 points on the standard MIDAS (3-month recall period) (9). An analysis from the American Migraine Prevalence and Prevention (AMPP) study, published only as abstract, suggested that a MIDAS change by −5 points might be used as the MID, corresponding to a 25% decrease in headache days per month in this population-based cohort (10). It has been emphasized that MID values vary with population and context (11), which may explain the differences between the above results and shows that additional research is needed. Furthermore, a relative change score (e.g. reduction in percent of baseline) might be better suited to capture MIDAS improvement than an absolute change score (i.e. difference from baseline) given that the MIDAS has a very wide range (0 to 279 points). Here, we used data from the headache registry of the German Migraine and Headache Society (DMKG) to (i) examine the suitability of relative vs. absolute MIDAS change scores for MID determination and (ii) estimate the MID of the MIDAS in this population.
Methods
The DMKG Headache Registry is conducted in accordance with the Declaration of Helsinki. It was approved by the leading ethics committee of the Ludwig-Maximilians-University Munich (Nr. 20-004), and by the ethics committee of each participating center. The registry complies with European and German Data Protection laws and is registered with the German Clinical Trials Register (DRKS 00021081). Written informed consent was obtained from each participant.
The DMKG Headache Registry has been recruiting patients since June 2020. Detailed methods have been published (12). At the time of the present analysis (data closure 31 January 2024), 25 DMKG-accredited centers had contributed data (17 private practices, eight outpatient clinics; for a list, see Appendix).
We included all adult patients having a complete baseline visit (V1) and a complete first follow-up visit (V2) within the registry, an ICHD-3 (13) diagnosis of either migraine without aura, migraine with aura or chronic migraine, MIDAS scores available for both visits and PGIC (14) scores available for V2 (n = 1218).
The datasets analysed during the present study are available from the German Migraine and Headache Society on reasonable request (note that access to data principally suitable for conducting additional analyses will be reviewed by the Headache Registry's Scientific Steering Committee).
Measures
MIDAS
The MIDAS uses five items to measure headache-related disability within the last three months in the areas (i) work/school, (ii) household and (iii) family/social/leisure activities. It assesses number of days with complete disability (three items), and with productiveness reduced by half or more (two items). The MIDAS score (item sum) ranges from 0 to 279. Four grades of disability have been defined: grade 1 (MIDAS score 0–5) little or no disability; grade 2 (6–10) mild disability; grade 3 (11–20) moderate disability; and grade 4 (≥21) severe disability.
We defined an absolute and a relative change score for the MIDAS:
ΔMIDAS = MIDASV2 – MIDASV1
(MIDAS difference score) %MIDAS = (MIDASV2 – MIDASV1)/MIDASV1*100 (MIDAS percent change score)
Note that negative change scores indicate improvement from V1 to V2.
PGIC
In the PGIC, patients rated the change of their headache condition aver the past three months, on a seven-point Likert scale (very much improved, much improved, somewhat improved, unchanged, somewhat worse, much worse, very much worse).
Statistical analysis
Statistical analysis was performed with SPSS (IBM Corp., Armonk, NY, USA). Descriptive statistics included the mean ± SD, numbers and percentages, and median and interquartile range (IQR) as appropriate.
The MIDAS has a wide range (0 to 279 points). For patients with a MIDASV1 grade I (0–5 points, little or no disability), determination of a MID is not useful because there is little room for improvement. We therefore based analysis on patients with MIDASV1 grades II–IV (6–279 points).
Suitability of relative (ΔMIDAS) vs. absolute (%MIDAS) change scores for MID determination
Given the wide range of the MIDAS, we hypothesized that there is a large dependence of ΔMIDAS values from MIDASV1 scores, limiting usability of an MID based on the ΔMIDAS because the MID ideally should be a threshold applicable independent of baseline scores. We also hypothesized that %MIDAS values would show a smaller correlation with MIDASV1 scores, making them better suited for MID determination. Also from a clinical point of view, an improvement by 10 points is likely to have a larger meaning for a patient with a baseline MIDAS of 30 compared to a patient with a baseline MIDAS of 150, which would be better represented by %MIDAS (−33% in the first case and −6.6% in the second). However, %MIDAS values potentially get very large for small MIDASV1 scores (an extreme example would be an increase from 10 to 270 points, corresponding to a %MIDAS of 2600%), which would severely bias average values (that are used for the average change MID determination method, see below). Also, %MIDAS values get very coarse for small MIDASV1 scores. To investigate these effects in more detail, we calculated and plotted correlations between MIDASV1 scores and ΔMIDAS and %MIDAS, respectively.
Determination of the MID of the MIDAS
It has been emphasized that MID values crucially depend on the employed method. Therefore, a careful choice of methods is important and if possible, results of several approaches should be considered in the final estimation of MID (11,15,16). Anchor-based methods using clinical or patient-based indicators and longitudinal data are considered first choice but may be supplemented by distribution-based methods that statistically estimate MID from baseline data. 11 Here, we used two anchor-based methods and two distribution-based methods (17) and several sensitivity analyses as detailed below.
Anchor-based: Average change approach
The PGIC was used as clinical anchor. First, it was verified if there was a correlation between the anchor and the MIDAS change score (8). Where possible, the “somewhat improved” compared to “unchanged” PGIC scores were used as anchor points as has been recommended (6,17) to capture the
Anchor-based: ROC curve approach
We used ROC curve analysis as a second anchor-based method for MID determination. The PGIC (same anchor points as above) was used as gold standard, and the MIDAS change score as the diagnostic test. The area under the curve (AUC) was used as a criterium for test quality. As sensitivity and specificity are of equal importance for the purpose of MID determination, we determined the MID as the cut-off value providing the maximum product of sensitivity and specificity (this is also the point where sensitivity equals specificity; where no such point could be found in the data, the nearest approximation was used) (18).
Distribution-based: SEM approach
A commonly used approximation for the MID is the standard error of the mean (SEM), calculated as SEM = SD*SQRT(1 −
Distribution-based: Effect-size approach
Another frequently used approach to estimate the MID is to calculate the 0.5*SD (equaling the change corresponding to an effect size (Cohen's
Sensitivity analysis
Because the DMKG Headache Registry is purely observational, without fixed intervals between visits, we anticipated a large variability of the interval between V1 and V2. We therefore performed a sensitivity analysis, limiting analysis to patients with an interval of 8–16 (12 ± 4) weeks between V1 and V2. As a second sensitivity analysis, we limited analysis to patients with an actual change (in drug or dose) of preventive migraine medication between V1 and V2.
Results
Characteristics of the study population (n = 1218) are shown in Table 1. The MIDAS scores at baseline (V1) and follow-up (V2) were 44.2 ± 47.4 and 36.5 ± 45.3, respectively. PGIC distributions are shown below. The interval between V1 and V2 amounted to a median of 13 weeks (IQR 9–19 weeks). As stated in the methods, further analysis was based on patients with a MIDASV1 > 5 (n = 1091).
Characteristics of the study population at baseline visit (V1, n = 1218).

Average within past three months. MIDAS: Migraine Disability Assessment.
Suitability of relative vs. absolute MIDAS change scores for MID determination
As hypothesized, there was a relevant correlation between ΔMIDAS and MIDASV1 (ρ = −0.395,
In summary, we decided to determine two separate MIDs for two ranges of baseline values: based on %MIDAS values for baseline MIDAS >20 (MIDAS grade IV) and based on ΔMIDAS values for baseline MIDAS between 6 and 20 (MIDAS grades II and III).
MID of the %MIDAS for baseline MIDAS scores >20
There were 757 patients with MIDASV1 > 20. Table 2 shows the MIDAS change scores at the different PGIC levels for the anchor-based, average change approach. The average %MIDAS in the PGIC “somewhat improved” group (−29.4 ± 50.9%, n = 214) was significantly larger than that in the PGIC “unchanged” group (−5.1 ± 67.0%, n = 182, Mann–Whitney,
Baseline MIDAS >20 (n = 757): %MIDAS values at the different PGIC levels.
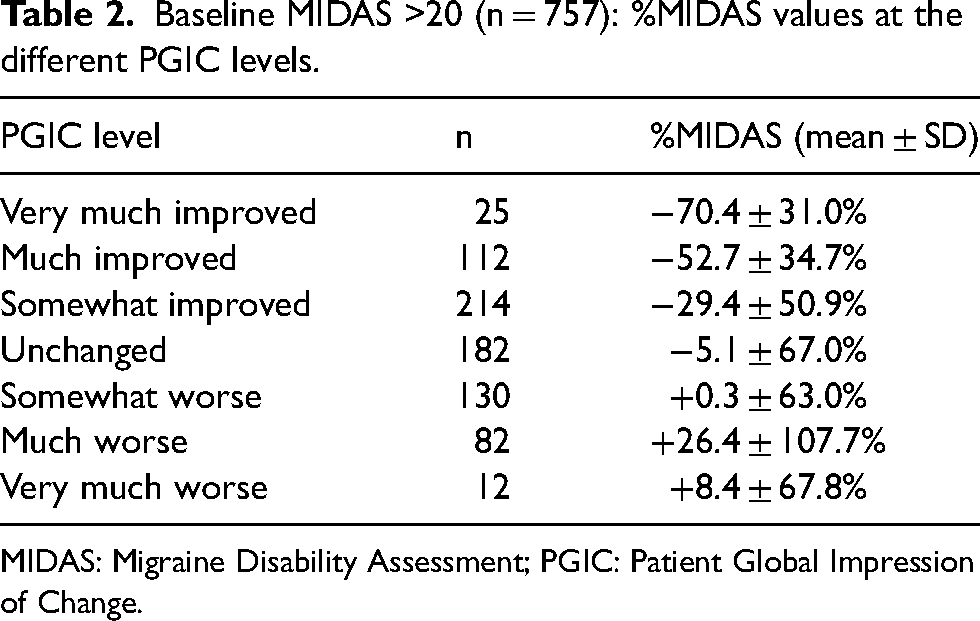
MIDAS: Migraine Disability Assessment; PGIC: Patient Global Impression of Change.
Using the ROC curve approach for differentiating between patients with a PGIC of “somewhat better” and “unchanged” resulted in an AUC of 0.635 (Figure 1) and a cut-off value for %MIDAS of −30.0% (sensitivity and specificity both 0.62). Thus, the MID of the %MIDAS determined by this method was −30.0%.

ROC curve. The graph illustrates the receiver operating characteristic curve for MID determination using Patient Global Impression of Change (PGIC) “somewhat improved” vs. “unchanged” as gold standard and the %MIDAS as diagnostic test, in patients with MIDASV1 > 20. MID: minimal important difference; MIDAS: Migraine Disability Assessment.
We also performed two sensitivity analyses. First, we limited analysis to patients with an actual change in preventive migraine medication between V1 and V2 (n = 423). The average change method resulted in a MID of −33.2%, and the ROC curve method resulted in −31.3%. Second, we limited analysis to patients with 12 ± 4 weeks between V1 and V2 (n = 398). In this group, the average change MID was −31.0% and the ROC curve MID was −32.1%. Detailed results are provided in the Supplementary material (Tables S1 and S2).
Distribution-based methods cannot be used on relative change scores such as the %MIDAS.
Table 3 summarizes the MID of the MIDAS determined by the different methods. In view of these results, we propose that a reduction of MIDAS scores by 30% might be a useful MID estimate for the group with a baseline MIDAS of >20 (Figure 2).

Overview of study results. MIDAS, Migraine Disability Assessment.
Baseline MIDAS of >20: values for MID of the MIDAS determined with different methods and in different subgroups.

PGIC “somewhat improved” vs. “unchanged”. MID: minimal important difference; MIDAS: Migraine Disability Assessment; ROC: receiver operating characteristic.
MID of the ΔMIDAS for baseline MIDAS scores 6 to 20
In this subset, there were 334 patients. Table 4 shows the MIDAS change scores at the different PGIC levels. There was a significant correlation of the anchor (PGIC) with ΔMIDAS values (ρ = 0.300,
Baseline MIDAS of 6 to 20 (n = 334): ΔMIDAS values at the different PGIC levels.

MIDAS: Migraine Disability Assessment; PGIC: Patient Global Impression of Change.
Again, we performed two sensitivity analyses: once in the subgroup of patients with a change in preventive medication between V1 and V2 (n = 105) and once for the subgroup of patients with 12 ± 4 weeks between V1 and V2 (n = 157). Detailed results are provided in the Supplementary material (Tables S1 and S2), and the resulting MID values were between −3.5 and −4.5 points (Table 5).
Baseline MIDAS of 6 to 20: values for MID of the MIDAS determined with different methods and in different subgroups.

PGIC “much improved” vs. “unchanged”. MID: minimal important difference; MIDAS: Migraine Disability Assessment; ROC: receiver operating characteristic; SEM: standard error of the mean.
Next, we used distribution-based methods to estimate the MID of the MIDAS, using the entire group with MIDASV1 scores of 6 to 20 (n = 334), which had a mean ± SD of 13.4 ± 4.5. Using Cronbach's alpha 1 of 0.83, the SEM amounted to −1.9 points. The MID corresponding to an effect size of d = 0.5 was −2.3 points.
Table 5 summarizes the MID of the MIDAS determined by the different methods. In view of these results, and giving more weight to the anchor-based methods as recommended, 11 we propose that a reduction in the MIDAS score by 4 points might constitute a useful MID estimate for the group with a baseline MIDAS between 6 and 20 (Figure 2).
Exploratory analysis: MID of the ΔMIDAS for baseline MIDAS scores 11 to 20
Because the American Headache Society recommends a cut-off of −5 points for the group of patients with baseline MIDAS scores between 11 and 20, we performed an exploratory analysis in this group (n = 228). Detailed results and a summary are provided in the Supplementary material (Tables S1 to S4). Shortly, anchor-based methods gave MID values between −4.0 and −6.8 points, which distribution-based methods resulted in −1.3 to −1.6 points. In summary, the cut-off of −5 points appears to be a good estimation of the MID of the MIDAS in this group.
Discussion
The major result of the present study is that, within a population of migraine patients from tertiary headache care,
The MID of the MIDAS can be estimated at −30% in the group with baseline MIDAS scores >20. The MID of the MIDAS can be estimated at −4 points in the group with baseline MIDAS scores between 6 and 20 (Figure 2).
The present data confirm that the MIDAS is responsive to patients’ perception of change of their migraine disorder, as assessed by the PGIC, and suggest that the MID of the MIDAS might be best captured by using different methods according to the baseline MIDAS.
Based on the analysis of the correlation between MIDASV1 and ΔMIDAS and %MIDAS, we decided to base determination of the MID on relative change scores (%MIDAS) in patients with a baseline MIDAS >20 points and on absolute changes scores (ΔMIDAS) for MIDAS baseline scores between 6 and 20. Although the cut-off of 20 is somewhat arbitrary, it was oriented at MIDAS grades, with MIDAS >20 corresponding to MIDAS grade IV (severe disability). 1 It might appear to be a disadvantage to have two different MIDs for different ranges of baseline measures. However, it has been emphasized before that the MID can depend of baseline values (8,17), and the alternative would be to have a less accurate MID covering the entire range. We consider that the present strategy to assign different MID values to different MIDAS grades (which are well-known to clinicians) is a good compromise between simplicity and accuracy.
However, there is an additional difference between the MIDs determined for the two ranges of baseline values. Although the MID of the baseline MIDAS >20 group was estimated to reflect the “somewhat improved” PGIC rating, the MID of the baseline MIDAS 6–20 group had to be based on the “much improved” PGIC rating. The reason for this was that the MIDAS difference score corresponding to the “somewhat improved” group was positive (i.e. reflecting worsening of MIDAS scores) and not significantly different from the “unchanged” group. This finding can be interpreted in different ways. One possibility would be that the MIDAS is not sufficiently sensitive to detect small differences in less severely affected patients and/or that our sample of less severely affected patients was too small. Alternatively, for less severely affected patients, a “somewhat improved” rating might not be clinically significant (and therefore not reflected by improved MIDAS scores). Nonetheless, it must be mentioned that there is a risk that the present results overestimate the true MID in the 6–20 baseline group.
Our findings deserve particular attention because there are only two previous studies on the MID of the MIDAS. The first, on 87 patients under physical therapy for migraine prevention, determined an MID of the modified (1 month recall period) MIDAS of −4.5 points, which might tentatively be extrapolated to −13.5 points on the standard (three months) MIDAS. 9 Patients were severely affected, with 12.6 ± 7.6 headache days per month and a baseline standard MIDAS of 60.6 ± 28.2. As a result of the limited number of subjects, the MID calculation was based on all patients with a rating equal to or above “somewhat better” (i.e. including patients with large improvements), which might lead to overestimation of the MID. 11 Because the study used absolute change scores, results are difficult to compare to the present study. However, using mean values, an improvement by 13.5 points from a baseline score of 60.6 would correspond to −22.4%, which is not too far from our result of −30%. The second study, comprising an analysis from the AMPP study, was published only as abstract. This investigated 2442 participants with migraine and ≥4 headache days per month from the general population. The MID of the MIDAS was estimated at −5 points, using a 25% decrease of headache days as an anchor. 10 The results are difficult to compare with those of the present study because of the different anchor, as well as because of a lower average baseline MIDAS score (26.4 points), implying that a substantial part of the patients likely fell into the baseline MIDAS 6–20 range. MID values are known to depend on the study population and context, which may account for part of the differences. Also, both previous studies did not address the problem of the dependence of absolute differences from baseline scores.
Interestingly, the American Headache Society, based on expert opinion, states that a clinically meaningful improvement of MIDAS scores should require a decrease by ≥30% in patients with a baseline MIDAS of >20, and by ≥5 points in those with a baseline MIDAS of 11–20 (19), which is very similar to the presently determined MID values. Part of the AHS recommendation (≥30% for MIDAS baseline >20) has also been adopted by the German guideline on migraine treatment (20).
Strengths and limitations
The large sample size is an important strength of the present study because it allowed determination of the MID using the group of PGIC “somewhat improved”, at least for the baseline MIDAS > 20 group. It has been emphasized that including patients with larger improvements or pooling groups (e.g. all those who rated “somewhat improved” or better in the PGIC), as done in some studies in the headache field (9,21), will lead to overestimation of the MID (11). It must be admitted that numbers of patients were rather low in some of the sensitivity analyses, especially those in the MIDASV1 6–20 point subgroups with a change in preventive medication or with 12 ± 4 weeks between visits. This probably reflects the fact that less severely affected patients are less likely to start or change their migraine prevention, and visit their headache specialist less frequently. However, despite of the lower numbers in these analyses, results corroborated those of the main analysis.
Recall bias is an important limitation of the PGIC, which assesses change retrospectively. It has been reported that PGIC ratings are often more closely correlated with the present state than with the extent of improvement or deterioration (11). Nonetheless, PGIC is a preferred anchor because it captures the patients’ perspective and the global change with respect to the disorder in question (16,17). The PGIC has a time frame of three months, whereas some of the patients in the present analysis had a shorter or longer time between visits, which might be another source of bias. However, limiting analysis to patients with a visit distance of approximately three months produced similar results.
Furthermore, it is important to consider that the MID may vary with population and context (8,11). Thus, the present results stemming from patients attending specialized headache care may not generalize to less severely affected patients in primary care. However, the MID does not depend on treatment and can be assessed from observational studies (11). Nonetheless, we performed a sensitivity analysis in patients with a change in preventive medication, which gave largely similar values compared to the total group.
It must be noted that the group distinction achieved by the MID was by no means absolute. There was considerable overlap in MIDAS scores between the “unchanged” and “somewhat improved” groups, also indicated by the limited AUC values (0.635–0.753; for a diagnostic test, an AUC >0.7 is recommended) (18). However, this is the trade-off for determining a “minimal” important difference and has also been reported by others (9). On the other hand, using the average change approach to determine the MID by definition excludes approximately half of the patients with a PGIC rating of “somewhat improved” group. In summary, it is important to note that the MID is not a diagnostic test but rather an estimation of the minimal change of a PROM that would matter to the patient.
Finally, the present study focused on improvement of the migraine disorder. The MID for deterioration would have to be determined separately.
Conclusions
The present data suggest that in the present population of migraine patients from specialized headache care, the MID of the MIDAS corresponds to −30% within the group with a baseline MIDAS of >20, and to −4 points within the group with a baseline MIDAS of 6–20.
Clinical implications
The minimal important difference of the Migraine Disability Assessement (MIDAS) was estimated at −30% for patients with a baseline MIDAS of >20, and at −4 points for those with a baseline MIDAS of 6–20. These results stem from a tertiary headache care population. They corroborate the expert-opinion based recommendations for determination of preventive medication efficacy of the AHS and the German guideline.
Supplemental Material
sj-docx-1-cep-10.1177_03331024241261077 - Supplemental material for Minimal important difference of the Migraine Disability Assessment (MIDAS): Longitudinal data from the DMKG Headache Registry
Supplemental material, sj-docx-1-cep-10.1177_03331024241261077 for Minimal important difference of the Migraine Disability Assessment (MIDAS): Longitudinal data from the DMKG Headache Registry by Ruth Ruscheweyh, Stefanie Förderreuther, Tobias Freilinger, Charly Gaul, Gudrun Goßrau, Tim Patrick Jürgens, Torsten Kraya, Lars Neeb, Victoria Ruschil, Jörg Scheidt and Thomas Dresler in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024241261077 - Supplemental material for Minimal important difference of the Migraine Disability Assessment (MIDAS): Longitudinal data from the DMKG Headache Registry
Supplemental material, sj-pdf-2-cep-10.1177_03331024241261077 for Minimal important difference of the Migraine Disability Assessment (MIDAS): Longitudinal data from the DMKG Headache Registry by Ruth Ruscheweyh, Stefanie Förderreuther, Tobias Freilinger, Charly Gaul, Gudrun Goßrau, Tim Patrick Jürgens, Torsten Kraya, Lars Neeb, Victoria Ruschil, Jörg Scheidt and Thomas Dresler in Cephalalgia
Footnotes
Acknowledgements
First, we thank all of the participating patients and headache centers. In addition, we thank Dirk Reinel, Johannes Drescher and Florian Wogenstein at the software development company “smartlytic” (Hof, Germany) and Yannic Siebenhaar at the Institute for Information Systems at the University of Applied Sciences Hof (Germany) for their excellent technical development of the DMKG Headache Registry, as well as Beatrix Weber and Katharina Diersch at the University of Applied Sciences Hof (Germany) and Lukas Wagner (HK2 Comtection GmbH) and Jonas Jacobsen (HK2 Rechtsanwälte), both Berlin, Germany for excellent management of the legal and data protection aspects. Moreover, we thank the “Münchner Studienzentrum” at the Technical University of Munich, Germany for outstanding organisational support. Finally, we thank the above-mentioned pharmaceutical companies for their financial support. The following centers contributed data to the present analysis: Dr Marziniak, München; Dr Goßrau, Dresden; Dr Rambold, Mühldorf, Dr Kukowski, Hildesheim, Dr Peikert, Bremen, Dr Weber, Fürstenfeldbruck; Dr Friedrich, Ravensburg; Dr Förderreuther, München; Dr Thilmann, Mannheim; Dr Malzacher, Reutlingen; Dr Menekes, Stuttgart, Dr Ruscheweyh, München, Dr Lewis, Stuttgart, Dr Weber, Erlangen, Dr Ermeling-Heuser, Bonn, Dr Fleischmann, Greifswald, Dr Rimmele, Rostock, Dr Erbacher, Straubing, Dr Gendolla, Essen, Dr Kraya, Leipzig, Dr Gesmann, Herford, Dr Eckle, Ulm, plus three centers that in the meantime have migrated or stopped recruitment.
Author contributions
All authors made substantial contributions to the conception and design of the work and the interpretation of the data. RR and TD performed the data analyses. RR drafted the manuscript. All other authors substantially revised the manuscript. All authors read and approved the final version of the manuscript submitted for publication.
Declaration of conflicting interests
RR has received travel grants and/or honoraria for lectures or advisory boards from Allergan/AbbVie, Lilly, Lundbeck, Novartis, Pfizer and TEVA. GG has received honoraria from Lilly, Novartis Pharma, TEVA, Lundbeck, and research grants from Novartis Pharma during the last three years. She does not hold any stocks of pharmaceutical companies. TD has received honoraria for consulting and lectures from Hormosan Pharma, Novartis Pharma, and TEVA. TF received honoraria for advisory boards from Novartis, TEVA and Lundbeck and honoraria for lectures from Novartis, TEVA, Lilly, Bayer and UCB. SF has received honoraria from Allergan/AbbVie, Hormosan, Eli Lilly, Lundbeck, Novartis, Sanofi and TEVA. She served on Advisory Boards for Hormosan, Eli Lilly, Lundbeck, Novartis, Sanofi and TEVA. CG has received honoraria for consulting and lectures within the past three years from Abbvie, Lilly, Novartis Pharma, Hormosan Pharma, Grünenthal, Sanofi-Aventis, Lundbeck, Perfood, Reckitt, Chordate and TEVA. His research is supported by a grant of the German Research Foundation (DFG). He does not hold any stocks of pharmaceutical companies. TK has received travel grants and/or honoraria from Allergan/AbbVie, Hormosan, Lilly, Novartis and TEVA. LN has received honoraria for lectures and consulting within the past three years from Abbvie, Lilly, Lundbeck, Novartis, Perfood and TEVA. VR received the Soyka award as a research grant from Novartis. JS declares no conflict of interest. TPJ has received honoraria from Allergan/AbbVie, Grünenthal, Hormosan, Lilly, Lundbeck, Novartis, Sanofi and TEVA. He served on Advisory Boards and/or consulted for Allergan/AbbVie, Hormosan, Lilly, Lundbeck, Novartis and TEVA.TPJ has received honoraria from Allergan/AbbVie, Grünenthal, Hormosan, Lilly, Lundbeck, Novartis, Sanofi and TEVA. He served on Advisory Boards and/or consulted for Allergan/AbbVie, Hormosan, Lilly, Lundbeck, Novartis and TEVA.
Ethical statement
The DMKG Headache Registry is conducted in accordance with the Declaration of Helsinki. It was approved by the leading ethics committee of the Ludwig-Maximilians-University Munich (Nr. 20-004), and by the ethics committee of each participating center. Written informed consent was obtained from each participant.
Funding
The DMKG Headache Registry is funded by the German Migraine and Headache Society and by unrestricted grants or funding of specific analyses from companies with an interest in headache research (up to the time of submission of this work, Lilly, Teva, Novartis, Pfizer and Lundbeck have made contributions). Supporting companies were given the opportunity to comment on the research protocol, and their comments were considered where judged to be scientifically relevant. Apart from that, supporting companies did not influence the study design. Teva supported a previous version of the present analysis. None of the companies influenced data collection, analysis, interpretation or writing of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
