Abstract
Background
Although withdrawal from analgesics with or without detoxification strategy represented a mainstay in medication overuse headache (MOH) management, recent evidence supports that it is no longer beneficial when CGRP-targeting monoclonal antibodies (CGRP-mAbs) are employed. However, MOH could be stratified into simple and complex MOH phenotypes according to different clinical parameters (i.e., amounts and class of analgesics, psychiatric comorbidities, history of relapse after withdrawal, symptoms of central sensitization, and maladaptive anticipatory response to pain experience). Herein, we explored the effectiveness of CGRP-mAbs plus detoxification strategy compared to CGRP- mAbs preventive treatment alone in patients with either simple or complex MOH phenotypes.
Methods
This is a six-month observational study including chronic migraine patients with MOH treated with subcutaneous CGRP-mAbs. Patients were stratified based on both MOH complexity and detoxification strategy to evaluate differences in the changes of monthly headache days, pain intensity and duration, and monthly days with acute medication intake after the first, third and sixth month of preventive treatment with CGRP-mAbs.
Results
Two hundred patients with migraine and MOH were recruited. A significant reduction of headache attacks frequency, intensity, duration and monthly days with acute medication intake has been observed both in patients sub-classified as complex MOH (58.5%) and in those with simple MOH (41.5%) after the first, third and sixth month of preventive treatment with CGRP-mAbs (p < 0.001). Furthermore, stratifying patients based on the MOH complexity and detoxification strategy, no differences were found in the reduction of monthly headache days as well as in other parameters of disease severity (p > 0.05).
Conclusion
Our findings might suggest a change in the mind-set of clinicians, still considering the withdrawal with or without detoxification strategy as a “conditio sine qua non” in patients with MOH, towards a novel approach where the reduction of analgesics intake represents the natural consequence of CGRP-mAbs effectiveness.
Introduction
Medication overuse headache (MOH) denotes a chronic migraine (CM) complication, due to interaction between frequent and excessive use of symptomatic drugs (the so-called “medication overuse”) and a susceptible migraine patient, leading to the paradoxical perpetuation and persistence of headache attacks (1,2). Experimental evidence suggests that medication overuse may underpin and promote central sensitization that, in turns, underlies interictal hyperalgesia and cutaneous allodynia, increased frequency of migraine attacks (up to clinical phenotype of chronic migraine) as well as a poor response to both preventive and symptomatic treatments (3–5). Along with genetic predisposition, numerous factors are known to precipitate and worsen MOH, such as the overuse of opioids or analgesics in combination, anxious and depressive symptoms, as well as some maladaptive behaviors (such as impulsivity, sensation seeking, emotional dysregulation, and cephalalgiaphobia) (6–9). Withdrawal from overused analgesics is one of the milestones of MOH management aiming to reduce headache frequency and improve responsiveness to acute or preventive treatments, and detoxification strategy (detox strategy) has been considered among the most effective strategies to achieve this result (10–12). However, nowadays, there is much evidence that seems to support the positive effects of CGRP-targeting monoclonal antibodies (CGRP-mAbs) in patients with MOH regardless of detox-related withdrawal from overused analgesics (13,14). None of the studies specifically compared the efficacy of CGRP-mAbs with or without the withdrawal strategy (before or during the preventive treatment), and patients were allowed to take acute headache medication as needed throughout the trial, indirectly suggesting that detox strategies are no longer needed in the current migraine therapeutic scenario, as more recently supported by a real-world experience (15). Nevertheless, all these observations lack important details about the different MOH phenotypes and, consequently, the role of detox strategies in patients with simple or complex MOH concomitantly beginning CGRP-mAbs preventive treatment (i.e., CGRP-mAbs plus detox strategy). Indeed, patients with CM + MOH are characterized by different healthcare difficulties and prognoses depending on often overlooked clinical parameters such as the amount of monthly acute medications, the classes of drugs overused, the presence of (pre-existing or concomitant) psychiatric comorbidities, as well as the history of MOH relapse after drug withdrawal. Altogether, the abovementioned clinical parameters may allow a MOH stratification into simple (i.e., MOH Type I) and complex (i.e., MOH Type II) MOH (16–18).
We hypothesize that CGRP-mAbs plus detox strategy could still represent a benchmark in selected patients with more severe MOH phenotypes. Therefore, in the present study, we explored the effects of CGRP-mAbs plus detox strategy compared to CGRP-mAbs preventive treatment alone in patients with CM + simple MOH versus CM + complex MOH at 1, 3 and 6 months. Furthermore, we investigated whether symptoms known to affect MOH severity such as “proxy/ies” of central sensitizationas well as maladaptive behaviors (specifically, the aberrant anticipatory response to pain experience such as cephalalgiaphobia) may play as outcomes predictors of CGRP-mAbs plus detox strategy.
Methods
Study population
The present study is an observational, prospective, single center, study to evaluate whether CGRP-mAbs plus detox strategy could have different effects in patients with CM + simple MOH compared to patients with CM + complex MOH. To this aim, we analyzed clinical data from 200 patients with CM + MOH (according to the Classification of the International Headache Society criteria) (2) who were followed up for at least six months in the Headache Center of the Department of Neurology at the University of Campania “Luigi Vanvitelli” between January 2021 and September 2023 and who filled-in papery standardized headache diaries at baseline and in the course of following six months.
All patients were aged between 18 and 75 years and had failed at least three oral preventive medication classes from among propranolol or metoprolol, topiramate or valproate, amitriptyline or onabotulinumtoxin-A, due to lack of efficacy, intolerable side effects, or contraindications.
Lack of efficacy was defined as <50% of reduction in headache days/month in the frequency of headaches after at least three months of treatments as recommended by the European Headache Federation guidelines (6 months for onabotulinumtoxin-A) (19). Tolerability failure was defined as documented discontinuation due to adverse events at any time of the treatment. Patients were treated with CGRP-mAbs but other standard-of-care preventive oral medication (alone or in combination) were allowed if taken at a stable dose for at least three months before starting CGRP-mAbs preventive treatment.
The patients’ sample included subjects who underwent a detox strategy before starting CGRP-mAbs and patients who started CGRP-mAbs without detox strategy. Detox strategy was performed as an outpatient treatment as follows: From day 1, the overused analgesics were discontinued and patients were treated for six days with parenteral intravenous administration of 500 mL physiological solution 0.9% associated with betamethasone (4 mg for the first three days and 2 mg for the following three days), metoclopramide 20 mg per day and S-adenosylmethionine 400 mg per day. On Day 6, patients received the first administration of CGRP-mAbs and they were allowed to take only acetaminophen 1000 mg (up to three intakes per day) as rescue medication. All patients received subcutaneous (s.c.) injections of monthly galcanezumab 120 mg (with loading dose of 240 mg), or monthly fremanezumab 225 mg, or erenumab 140 mg every 28 days. At the first administration (T0), at the end of the first (T1), third (T2) and the sixth (T3) month of CGRP-mAbs treatment, the headache diaries were analyzed.
By reviewing the standardized paper headache diaries the following variables have been collected at baseline (i.e., the three months before starting the treatment with CGRP-mAbs) and during the treatment (i.e., the first, the third and the 6 months of treatment with CGRP-mAbs): i) headache frequency (defined as the monthly mean of headaches with or without migraine features) ii) headache intensity (by 1–10 numerical rating scale – NRS), iii) response to painkiller (2 h pain-freedom), iv) analgesics intake/month, and v) days with analgesics intake/month. Furthermore, all patients underwent a semi-structured extensive interview (at baseline) aimed to explore parameters of disease severity as migraine history duration (in years), anticipatory use of symptomatic medication/s (cephalalgiaphobia), the presence of comorbidities as depression and anxiety (assessed by Structured Clinical Interview for DSM-V Axis I Disorders) and cutaneous allodynia (by means of ASC-12). During the six-month period of observation, all adverse events (AE) related to the drug were recorded and used as safety measures.
The paper headache diary consisted of a table with the days of the month (up to 31 days) on the abscissa and the hours of the day (up to 24 h) on the frames. The patient could mark the onset and the end of every headache episode and the time of pain-killer intake. In case of headaches present at both the time of falling asleep and waking up, the night hours were considered as attack hours. Moreover, the patient inserted the intensity of the attacks. Finally, patients were instructed to identify “crystal clear days” (days without headache and other migraine symptoms) or “unclear days” (days without headache but with minimal migraine symptoms) (19).
The protocol was reviewed and approved by the Ethical Committee of the University of Campania “Luigi Vanvitelli”. Each patient gave written informed consentfor participation in the study and the analysis and publication of the protocol data.
Study design
Primary outcomes
In the first analytic step, patients were sub-classified as complex MOH (MOH type2) according with the presence of at least one among: i) ongoing psychiatric comorbidities (e.g., mood disorder or anxiety disorder) assessed by Structured Clinical Interview for DSM-V Axis I Disorders; ii) relapse into medication overuse following previous treatments with detox strategy (at least one); iii) daily use of multiple doses of symptomatic medication/s for each treated attack (≥2 doses/day of triptans, ergots, NSAIDs or daily analgesics in combination). Patients who did not fall into this category were classified as simple MOH. Then, patients with CM + MOH were divided in to four groups: i) patients with MOH type 1 treated with CGRP-mAbs with detox strategy; ii) patients with MOH type 1 treated with CGRP-mAbs without detox strategy; iii) patients with MOH type 2 treated with CGRP-mAbs with detox strategy; iv) patients with MOH type 2 treated with CGRP-mAbs without detox strategy (see Figure 1 for further clarification).
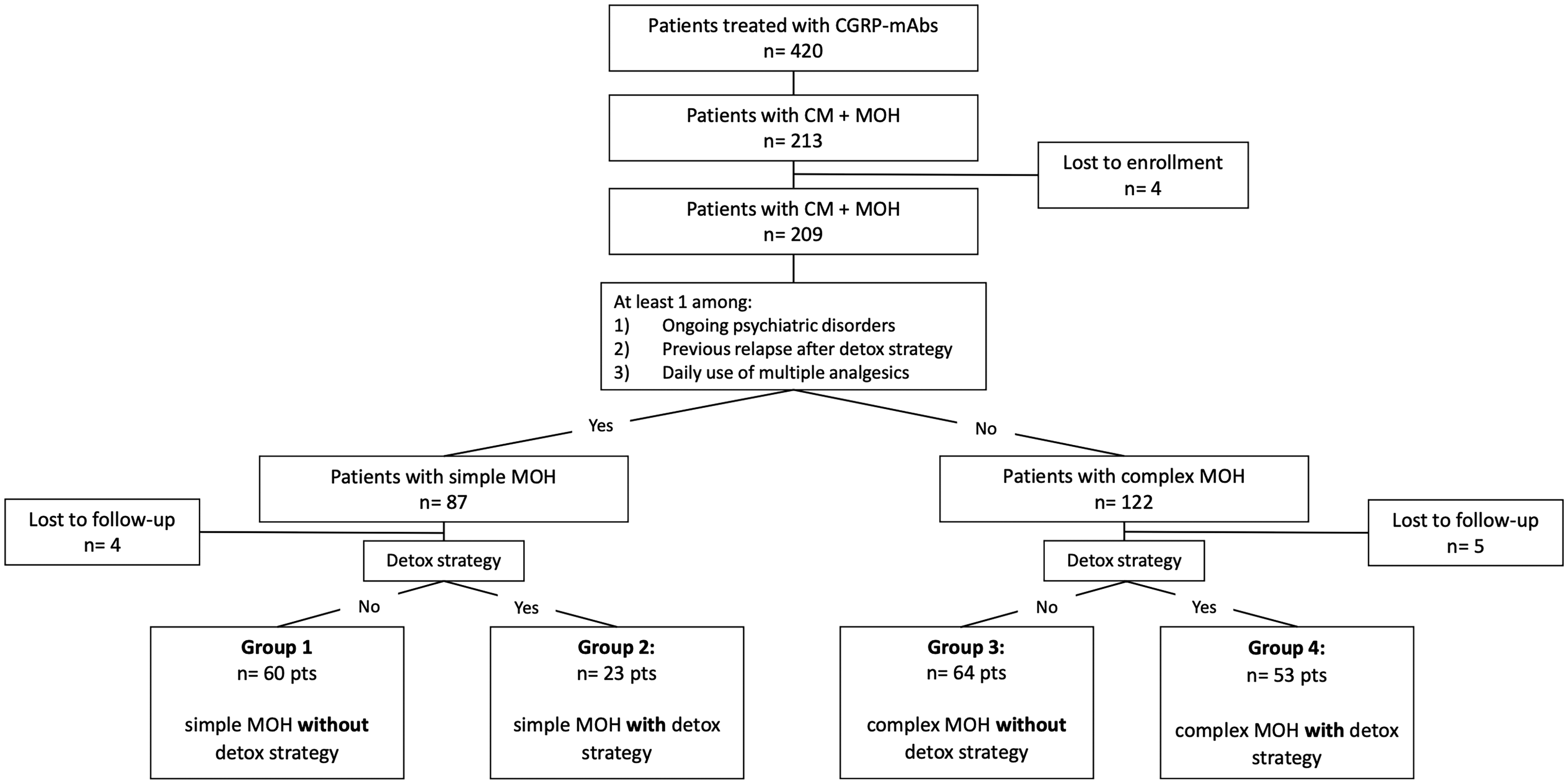
Flow-chart of participants’ subgroups obtained by stratifying patients with chronic migraine with simple (type 1) or complex (type 2) MOH and according to detox strategy. Missing data constituted the main explanation for patient exclusion. At a minimum, treatment information at months 0 and 6 were required for inclusion.
The primary endpoints of the study were to evaluate changes in monthly headache days, mean pain intensity, duration of headache attacks and the days with acute medication intake in the four groups under examination at the end of the first (T1), third (T2) and sixth (T3) month of CGRP-mAbs preventive treatment compared with the baseline (T0).
Secondary outcomes
In the second analytic step, all the patients’ samples were subclassified based on detox strategy (YES/NO) and according with symptoms “proxy” of central sensitization as: i) cutaneous allodynia (present or not based on ASC-12); ii) response to symptomatic drugs intake (i.e., 2 h pain-freedom after acute medication intake); iii) baseline crystal clear days (present or absent); and iv) cephalalgiaphobia (present or absent) (among maladaptive behaviors). The secondary endpoint of the study was to evaluate putative changes in the frequency of headache days per month in the groups under examination at the end of the first (T1), third (T2) and sixth (T3) month of CGRP-mAbs preventive treatment compared with the baseline (T0) (see Online Supplementary Material 1 for further clarifications).
Statistical analysis
All demographic and clinical data were checked for normality using Shapiro–Wilk test. The sample size was calculated a priori by means of G*Power software (version 3.1.9.7). For the ANOVA test, using a medium effect size of 0.30, considering four groups, setting alpha error to 0.05 and a power of 0.95, we obtained a sample size needed of at least 196 patients. Continuous variables conforming normal distribution are reported as mean ± standard deviation (SD) while continuous data not conforming normal distribution and categorical variables are expressed as median ± interquartile range, rates values are reported as subjects-counts and percentage. We used one-way ANOVA test to compare continuous variables conforming to normal distribution, Pearson's chi-square test to compare categorical variables, while non-parametric Kruskal-Wallis test was used for data not conforming to normal distribution. Hypothesis testing was 1-tailed and results have been considered statistically significant if p < 0.05. Bonferroni correction for multiple comparison was applied. All analyses were performed using STATA version 16 (StataCorp, College Station, TX, USA).
Results
Demographic and baseline headache characteristics
The study population consisted of 200 patients with migraine and MOH with previous treatment failures of at least three preventive medication classes according the Italian Medicines Agency (AIFA) provisions for the CGRP-mAbs preventive treatment in reimbursement regimen. The majority of patients were female (81%), with a mean age of 48.9 ± 11.8 years. The average time since migraine onset was 29.1 (± 13.8) years. The average frequency of headache days/month was 22.2 (± 5.3). Demographic and baseline headache characteristics of patients included in the study are reported in Table 1.
Baseline demographic and clinical features of patients with simplex and complex MOH.

MOH: medication overuse headache; BMI: body mass index; MHD: monthly headache days; NRS: numerical rating scale; *median ± interquartile range; § mean ± standard deviation.
A significant reduction of headache attacks frequency, intensity, duration and monthly days with acute medication intake has been observed both in patients sub-classified as complex MOH (58.5%) and in those with simple MOH (41.5%) after the first, third and sixth month of preventive treatment with CGRP-mAbs compared with baseline (p < 0.001). However, no differences were found in the two groups (i.e.,: patients with MOH type 1 and patients with MOH type 2) in the reduction of headache days per month as well as in the other parameters of disease severity such as headache intensity, headache duration, and days with monthly acute medication intake, after 1, 3 and 6 months of treatment with CGRP-mAbs (see Table 2 for further information).
Treatment related changes in clinical parameters of disease severity in patients with simplex and complex MOH.

MOH: medication overuse headache; BMI: body mass index; MHD: monthly headache days; NRS: numerical rating scale; *median ± interquartile range; § mean ± standard deviation; In bold statistically significant value.
Primary endpoints
After the stratification of patients based on both the diagnosis of MOH types (i.e., MOH types 1 and types 2) and detox strategy, no differences were found in the four groups (i.e.,: patients with MOH type 1 performing or not performing detox strategy and patients with MOH type 2 performing or not performing detox strategy) in the reduction of headache days per month as well as in the other parameters of disease severity such as headache intensity, headache duration, and days with monthly acute medication intake, after 1, 3 and 6 months of treatment with CGRP-mAbs (see Figure 2 and Tables 3 and 4 for further information). Once again, a significant reduction in headache attacks frequency, intensity, duration and monthly days with acute medication intake has been observed in all groups after the first, third and sixth month of preventive treatment with CGRP-mAbs compared with baseline (p < 0.001).

Diagrams showing headache attacks A) frequency, B) duration and C) intensity as well as D) days with symptomatic medication intake in the participants’ groups obtained by stratifying patients with chronic migraine based on simple (type 1) and complex (type 2) MOH and according to detox strategy.
Baseline demographic and clinical features of the four groups under examination obtained stratifying patients with MOH based on both MOH complexity and detox strategy.

MOH: medication overuse headache; BMI: body mass index; MHD: monthly headache days; NRS: numerical rating scale; *median ± interquartile range; § mean ± standard deviation; In bold statistically significant value.
Treatment related changes in clinical parameters of disease severity in the four groups under examination obtained stratifying patients with MOH based on both MOH complexity and detox strategy.

MOH: medication overuse headache; MHD: monthly headache days; NRS: numerical rating scale.
*median ± interquartil range; § mean ± standard deviation
Secondary endpoints
In the subsequent steps, no differences were found in the reduction of headache attacks per month after 1, 3 and 6 months of treatment with CGRP-mAbs in the groups obtained considering both detox strategy (YES/NO) and presence or absence of each of the following parameters: interictal cutaneous allodynia, crystal clear days, loss of response to acute medication (altogether as “proxy” of central sensitization), and cephalalgiaphobia (see Figure 3 for further information).
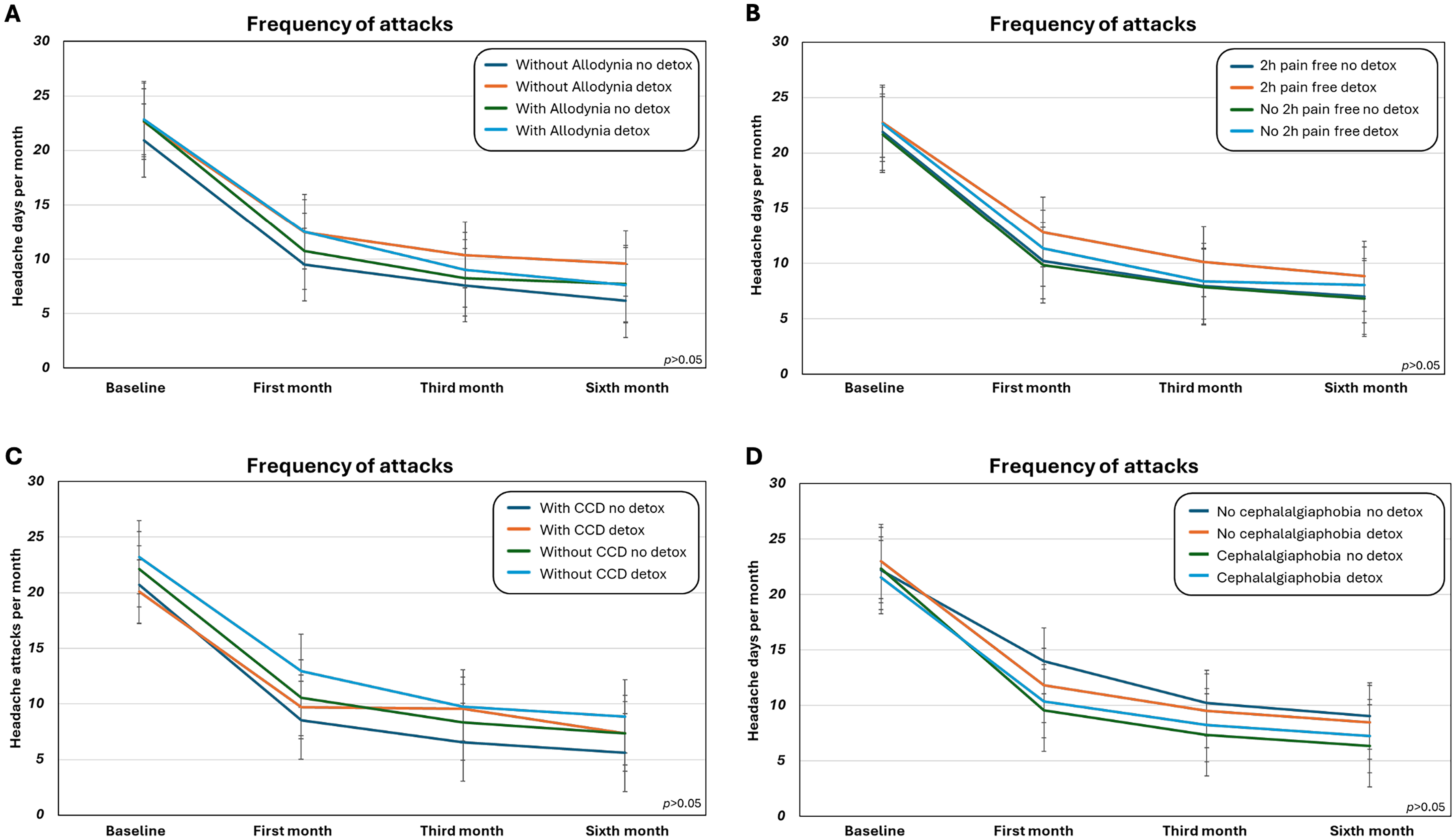
Diagrams showing monthly headache attacks frequency in the participants’ subgroups obtained by stratifying patients with chronic migraine and MOH based on A) cutaneous allodynia and detox strategy, B) response to symptomatic drugs (pain free at 2 h) and detox strategy, C) presence of crystal clear days (CCD) and detox strategy, and D) cephalalgiaphobia and detox strategy.
Discussion
In the present study, we demonstrated that preventive treatment with CGRP-mAbs plus detox strategy does not result in a more effective or quicker response compared to preventive treatment with CGRP-mAbs alone in patients with CM stratified in simple (type 1) or complex (type 2) MOH phenotypes. Indeed, no differences were found either in the frequency of headache days per month or in other parameters of disease severity (headache intensity and duration, and days with acute medication intake) after 1, 3 and 6 months of treatment with CGRP-mAbs. Moreover, the effectiveness of preventive treatment with CGRP-mAbs plus detox strategy is independent from clinical features of MOH severity – considering symptoms recognized as “proxies” of central sensitization as well as aberrant anticipatory response to pain experience (i.e., cephalalgiaphobia) in these patients. Among the challenges that MOH imposes, treatment concerns are the most impactful especially considering the poor effectiveness of preventatives, based on repositioning drugs, in these patients, as recently demonstrated by a systematic review and meta-analysis showing no superiority of oral preventive treatments compared with placebo (19). Nevertheless, although the therapeutic approach to MOH is still a matter of debate, withdrawal from overused acute analgesics and concomitant preventive treatment seem should be considered the two pillars of care for patients with MOH. Indeed, some agreement had been reached about the withdrawal from overused analgesics as a prerequisite to improve responsiveness to preventive and acute treatments (11,20–22) as demonstrated in the last few years when concomitant withdrawal and preventive treatment have been proven to be superior to preventive treatment alone or to withdrawal with deferred optional preventive treatment (23). The strategies employed to withdraw from overused acute medication varies considerably between studies as a consequence of the differences within and across countries, ranging from structured pharmacological programs including hydration, corticosteroids, neuroleptic agents, tranquilizers, or antiemetics (the so-called “detoxification”) to simply advice or education alone about the necessity of interrupting the overused acute medication (24,25).
However, recent evidence rising from both randomized controlled trials and real-world experiences support the role of preventive treatment with CGRP-mAbs as effective in CM + MOH regardless of withdrawal from overused analgesics (12–15,26). More specifically, post-hoc analysis of phase III trials focusing on patients with CM + MOH, reported that CGRP-mAbs (erenumab and fremanezumab) are effective in reducing the frequency of monthly headache attacks in the absence of a formal withdrawal strategy (13,14). Similarly, a very recent retrospective real-world analysis performed on a large sample of 291 patients with and without MOH receiving CGRP-mAbs without a detox strategy confirmed the effectiveness of CGRP antibody (27). Finally, a further real-world investigation comparing patients with CM + MOH receiving CGRP-mAbs who underwent an in-hospital detox strategy with a sample of patients with CM + MOH receiving CGRP-mAbs who did not perform detox strategy found no differences in treatments response (15).
On the other hand, the term MOH appears to be a “cauldron” containing different phenotypes that, although not contemplated in the ICHD-III, would be appropriately identified based on complicating variables, putatively affecting treatment strategies and prognosis (16–18). In line with this point of view, some authors have pragmatically suggested to subdivide MOH into two clinical subtypes as simple MOH (type I) and complex MOH (type II) based on the overuse of analgesics in combination, opioid/barbiturate-containing medications, psychiatric comorbidities, and history of relapse after withdrawal of overused analgesics (16–18,24). Moreover, symptoms of central sensitization such as cutaneous allodynia, crystal clear days, response to treatments (e.g., loss of response to symptomatic drugs over time), as well as maladaptive behaviors such as cephalalgiaphobia may allow us to further characterize patients belonging to the wide spectrum of MOH. Accordingly, differences in MOH severity may reverberate in clinical practice: for instance, in patients with simple MOH advice and education are as effective as a detox strategy in achieving withdrawal of the overused analgesics, whereas in complex/complicated MOH patients a detox strategy is significantly more effective than advice alone (24,28,29).
Herein, we found no differences in the effectiveness of preventive treatment with CGRP-mAbs in patients with MOH either simple or complex, independently of the detox strategy, suggesting that CGRP-mAbs are effective independently of the class of symptomatic drug overused, the psychiatric comorbidities, and the history of MOH relapse (all parameters able to stratify patients with simple or complex MOH).
Furthermore, we have explored the effectiveness of CGRP-mAbs plus detox strategy on patients with MOH considering cutaneous allodynia, crystal clear days, loss of response to symptomatic drugs over time and cephalalgiaphobia, altogether known to be clinical hallmarks of more severe MOH phenotypes and negative predictors of response to preventive treatments (8,30,31). Indeed, the sensitization phenomenon is underpinned by a heightened sensitivity and hyperexcitability of central nervous system, affecting pain perception and modulation, clinically resulting in ictal and interictal allodynia, hyperalgesia, and pain perpetuation with poor response to both symptomatic drugs and traditional “repositioning” preventive treatments, as well as the absence of “crystal clear” days (32–35). Similarly, obsessional and drug-seeking behaviors frequently exhibited by patients with MOH are led by certain maladaptive conditions, encompassing impulsivity, emotional dysregulation, and especially anticipatory anxiety and fear of headache (26,36,37). In particular, the fear of having headache attacks experienced during a pain-free period leading patients to preemptively overuse analgesic medications, known as cephalalgiaphobia, seems to strongly contribute to medication overuse headache and represents a well-known predictor of poor treatment outcomes in MOH patients.
In the present study, no differences in the effectiveness (in terms of reduction of headache days per month) were found between patients with CM + MOH treated with preventive treatment with CGRP-mAbs plus detox strategy compared to patients with CM + MOH treated with preventive treatment with CGRP-mAbs without detox strategy after the first, the third and the sixth administrations, considering the presence of symptoms of central sensitization as well as cephalalgiaphobia.
According to the present findings, we suggest that preventive treatment with CGRP-mAbs is effective in patients independently of withdrawal from overused analgesics (38,39). In other terms, the reduction of the number of headache episodes by effective preventive treatments, such as CGRP-mAbs, will consequently reduce the number of symptomatic drugs, independently of detox strategies, acting as a barrier to the cascade of events leading to MOH.
The present study is not exempt from limitations, mainly related to the well-known evidence shortcomings primarily affecting a more precise MOH sub-classification (beyond the number of days with analgesics intake) and to the lack of evidence-based standardized therapeutic detox strategy protocols. Specifically, due to lack of defined criteria to sub-classify patients with CM and simple or complex MOH, we applied a set of “features” representing the best combination of the clinical parameters extensively described in MOH dedicated literature.
Moreover, the detox strategy used in the present study is just one of many and, considering that no definite therapeutic recommendations are available, it only reflects good clinical practice suggestions where hydration, corticosteroids, and antiemetics represent the core of detox strategy (25). Obviously, we cannot exclude that different strategies (e.g., longer duration of detox treatment, different class drugs, or inpatient/outpatient management) might address divergent results, although it 's noteworthy that the type of withdrawal strategy (inpatient versus outpatient, advice alone versus pharmacological treatments) does not seem to influence the effects derived from the institution of an effective preventive and abortive regimen.
Furthermore, due to its observational, non-randomized, and open-label nature, the present study mirrors the real-world clinical practice. Therefore, although those patients who practiced the detox strategy and those who did not, did not show significant differences at baseline in term of both demographic and disease severity features (see Table 5 for further information), it is not possible to exclude that patients treated with detox strategy according to clinical judgment could have shown peculiar clinical characteristics, not detectable by the employed conventional questionnaires. Finally, although long-term effectiveness of the two different therapeutic strategies (i.e., CGRP mAbs with or without detox strategies) goes beyond the aims of the present study, we are aware that the six-month observation period might be not adequate enough to assess the sustainability of reduced analgesic intake in patients with complex MOH phenotypes using CGRP-mAbs.
Baseline demographic and clinical features of patients with MOH treated or not with detox strategy before CGRP-mAbs administrations.
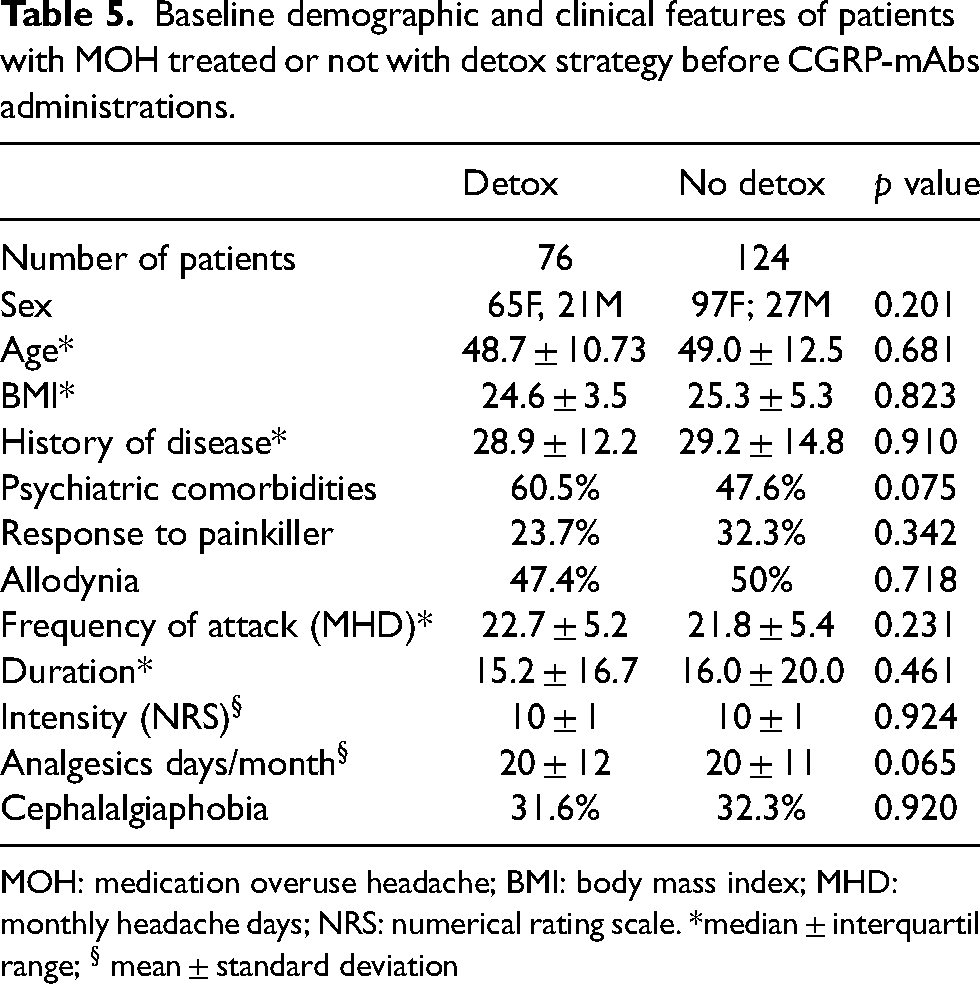
MOH: medication overuse headache; BMI: body mass index; MHD: monthly headache days; NRS: numerical rating scale. *median ± interquartil range; § mean ± standard deviation
Conclusions
Altogether, our findings do not support our original hypothesis of CGRP-mAbs plus detox strategy as a therapeutic benchmark in selected patients with more severe MOH phenotypes. Contrariwise, our results might suggest a change in the paradigm that still considers the withdrawal strategy as a “conditio sine qua non” in patients with CM + MOH towards a novel treatment paradigm in which CGRP-mAbs alone are able to exert their effectiveness without the need of detox strategies.
In this scenario, the withdrawal of overused analgesics is no longer a superimposed indication for patients, since the progressive reduction in the number of migraine episodes, as a direct consequence of the CGRP-mAbs efficacy, ties in with a gradually progressive reduction in acute medication intake, also avoiding the well-known withdrawal symptoms related to the abrupt interruption of overused drugs (e.g., headache, nausea, dizziness, and disordered sleep). Accordingly, although some clinicians tend to recommend abrupt discontinuation (as long as the dose and type of medication do not require a slow withdrawal for safety), treatment studies have not shown a significant difference in benefit between abrupt and slow discontinuation (40,41).
Therefore, a reappraisal of recommendations regarding the therapeutic approach to patients with MOH might be advantageous. Furthermore, considering on one hand the personal, therapeutic, and socio-economic burden related to CM + MOH and, on the other hand, the effectiveness of preventive treatment with CGRP-mAbs in these patients, the role of CGRP-mAbs as the first therapeutic strategy of CM + MOH should be taken in consideration (42).
Finally, when the detox strategies disappear from clinical practice, we could hopefully assist in removing the term detoxification, that represents another source of stigmatization, implying the concept of “addiction”, in patients with CM + MOH (43). Indeed, although is not always possible to determine whether a refractory or severe headache pattern tends to precipitate the symptomatic medication overuse or if the increased symptomatic medication use led to the MOH, we all should be acquainted with the fact that actual addiction and medication dependence do not appear as a general rule in patients with MOH.
Clinical implications
When stratifying patients based on the complexity of MOH and the detoxification strategy used, no differences were observed in the parameters of disease severity with CGRP-mAb preventive treatment. The effectiveness of CGRP-mAbs, regardless of whether a detoxification strategy was applied, was independent of clinical “proxies” for central sensitization and aberrant anticipatory responses to pain experiences. Currently, withdrawal from overused analgesics could be considered a natural consequence of CGRP-mAb effectiveness in patients with MOH.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024251329808 - Supplemental material for No additional benefit with detoxification strategies: A real world experience in 200 patients with chronic migraine and either simple or complex MOH treated with CGRP monoclonal antibodies
Supplemental material, sj-pdf-1-cep-10.1177_03331024251329808 for No additional benefit with detoxification strategies: A real world experience in 200 patients with chronic migraine and either simple or complex MOH treated with CGRP monoclonal antibodies by Marcello Silvestro, Ilaria Orologio, Pasquale Sozio, Valentina Dortucci, Francesca Trojsi, Mattia Siciliano, Gioacchino Tedeschi, Alessandro Tessitore and Antonio Russo in Cephalalgia
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.S. has received speaker honoraria from Novartis, Pfizer, AbbVie, Teva and Lilly. A.T. has received speaker honoraria from Novartis, Schwarz Pharma/UCB, Lundbeck, Abbvie and Glaxo. G.T. has received speaker honoraria from Sanofi-Aventis, Merck Serono, Bayer Schering Pharma, Novartis, Biogen-Dompé AG, Teva and Lilly; has received funding for travel from Bayer Schering Pharma, Biogen-Dompé AG, Merck Serono, Novartis and Sanofi Aventis; and serves as an associate editor of Neurological Sciences. A.R. has received speaker honoraria from Allergan, Lilly, AbbVie, Pfizer, Novartis and Teva and serves as an associate editor of Frontiers in Neurology (Headache Medicine and Facial Pain session). All other authors have nothing to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
