Abstract
Background
The association between insufficient sleep and migraine is largely unexplained. In this blinded cross-over study we investigate whether insufficient sleep disturbs processing of nociceptive signals more in individuals with migraine compared to those without migraine.
Methods
Evoked potentials to nociceptive laser stimuli and high density electrical dermal stimuli were recorded in migraine and control subjects after two nights of habitual sleep and after two nights of sleep restriction (4 h sleep/night). Researchers conducting the measurements and data processing were blinded to diagnosis and sleep condition. Both recordings were interictal in 21 migraine subjects. Amplitudes, amplitude habituation and latencies were compared to measurements in 31 controls.
Results
Electrically induced N2P2 amplitude showed more habituation after sleep restriction compared to habitual sleep in the migraine group (p < 0.025). The migraine group also had less N2P2 amplitude habituation after habitual sleep compared to the control group (p < 0.035). We found no effect of sleep restriction on N2P2 amplitude habituation to laser stimulation, and no effect of sleep restriction on N2P2 amplitudes.
Conclusion
Insufficient sleep may slightly increase cortical inhibitory responses in migraine. Our findings support that migraine is associated with a vulnerability for insufficient sleep between attacks.
Introduction
A clinical relationship between sleep and migraine is well known. Sleep can ease migraine headache, migraine attacks may start during or after sleep, and many migraine subjects report disturbed sleep as a migraine trigger (1). People with migraine also have reduced sleep quality, more daytime sleepiness and more severe sleep disturbances compared to headache-free individuals (2), while insomnia is associated with an increased risk of developing migraine (3). This multifaceted relationship between sleep and migraine is still not fully understood, and in this study we investigate how nociceptive signalling is influenced by insufficient sleep in migraine.
Migraine may be characterized by general increased responsivity to sensory stimuli and altered cortical excitability (4). Increased pain sensitivity has been reported in migraine patients between attacks (5,6). Interestingly, sleep disturbances are also associated with increased pain sensitivity. The mechanisms are not fully understood but may involve proinflammatory factors directly affecting nociceptors, enhanced pain transmission at the spinal level, as well as effects on the mesolimbic system (7).
Sleep restriction increases pain sensitivity in healthy subjects (8) and is a promising model for investigating the effect of insufficient sleep in migraine (9). Laser evoked potentials (LEP) and nociceptive somatosensory evoked potentials (nSEP) to high-density electrical stimulation are objective measurements of pain-related activity (10). Studies of LEP and nSEP in healthy subjects indicate altered pain-related activity following sleep restriction (11,–13). Some studies have reported increased LEP amplitude and reduced LEP amplitude habituation in migraine subjects compared to controls (14,15), while this was not reproduced in another study (16). This indicates that alterations in pain processing in migraine between attacks may be subtle. Furthermore, data from TMS and EEG indicate an effect of sleep restriction on inhibitory systems in migraine subjects (9,17). Accordingly, it is of great interest to investigate if sleep restriction increases these possible and subtle differences in nociceptive processing in migraine.
In the present study we aimed to better explain the relationship between insufficient sleep and pain physiology in migraine. We hypothesized that sleep restriction will increase abnormal pain processing in migraine between attacks. The primary objectives were to measure if sleep restriction has different effects on LEP and nSEP amplitude and amplitude habituation in interictal migraine subjects compared to controls. The effect of sleep restriction on LEP and nSEP in migraine has not been investigated previously. Secondary objectives were to measure the effect of sleep restriction on LEP and nSEP latencies. We also intended to explore if significant primary variables correlated with clinical measures regarding headache burden, insomnia, depression and anxiety.
Methods
Subjects
Migraine subjects and headache-free controls matched for sex and age were recruited through advertisement within the Norwegian University of Science and Technology (NTNU). In this study 289 subjects were considered for inclusion in the migraine group, and 215 were considered not eligible based on exclusion criteria after telephone screening by a study nurse. Exclusion criteria were co-existing tension type headache, sleep disorders, hypertension, infectious, metabolic, endocrine, neuromuscular or connective tissue disease, acute or chronic pain, recent injury affecting function, symptomatic heart disease, cardiovascular medication, pulmonary disease affecting function, pulmonary medication, cerebrovascular disease, neurologic or psychiatric disease with decreased function, neoplastic disease, neuroleptic, antiepileptic or analgesic medication, antidepressants, medicine which could affect neural, vascular or muscular function, pregnancy, implantations, previous craniotomy or cervical neurosurgery, drug abuse, or body mass index below 17 or above 35.After the telephone screening, 74 subjects were examined in-person by a neurologist for confirmation of the diagnosis according to the International Classification of Headache Disorders-III beta criteria for migraine with or without aura (18), and 55 migraine subjects were included. Ten of these dropped because it was not practically possible for them to complete two nights of sleep restriction and/or all three laboratory visits. In addition, one patient dropped out because of an injury. Measurements from two additional migraine subjects were not included in analysis because they had not completed headache diaries. Another migraine subject did not complete nSEP measurement on test day 2. Measurements in migraine subjects were classified as interictal if there was no migraine headache in the 24 h before and after the measurement. Twenty-one migraine subjects had interictal nSEP and LEP measurements after both sleep conditions and were included in primary analyses (Figure 1). Thirty-one controls matched for age and sex were included in the study by telephone interview. The subjects were instructed not to use any form of caffeine or nicotine product after midnight on the days with testing.

Flow chart showing inclusion of migraine subjects and number of interictal recordings.
Subjects completed questionnaires with demographical data, depression, anxiety (Hospital Anxiety and Depression scale, HADS), insomnia severity scale (0–12) (19) and tendency to fall asleep (Epworth sleepiness scale, ESS) (20). Data on clinical migraine traits were collected in a semi-structured interview with a research nurse before the training day, including intensity, frequency and duration of headache, and frequency and intensity of photophobia and phonophobia.
Study design
The data presented in this study stems from a larger data collection conducted from May to December 2016, including pain thresholds and conditioned pain modulation (21,22). The study had a controlled, paired cross-over design. Researchers conducting the examination and data processing were blinded to diagnosis and sleep condition. The same protocol was repeated after two sleep conditions, habitual sleep and sleep restriction, in randomized order. Separate block randomization was applied for migraine subjects and controls to ensure the same number of migraine and control subjects had the sleep restriction and habitual sleep conditions first. Subjects arrived for a training day 2–14 days before their first test day. There were 7–14 days between the two test days (Figure 2).

Overview of study design. After inclusion, the order of sleep conditions was randomized. There were 2-14 days between the training day and first test day, and 7–14 days between the two test days. Migraine subjects completed headache diaries, and migraine subjects and controls completed sleep diaries from 14 days before first test day until 1 week after the second test day. Subjects wore an actigraph from the training day to the last test day.
For the sleep restriction condition, we instructed the subjects to sleep for 4 h per night for two consecutive nights directly before testing. For the habitual sleep condition, they were told to sleep for approximately 8 h per night for two nights. The subjects slept at home and were asked to not sleep at other times during the day. Wake up time was 7 AM for both sleep conditions.
Procedure
Migraine subjects filled out headache diaries and all subjects filled out sleep diaries from 14 days before the first test day until 1 week after the last test day. In addition to completing sleep diaries, all subjects wore an actigraph (Actiwatch Spectrum Plus, Philips Respironics, USA) from the training day to the last test day. The subjects filled out the Karolinska sleepiness scale (23) before and after testing on both test days. Behavioural alertness was measured by calculating reaction time from a computerized version of the 10 min psychomotor vigilance test (PVT) (24).
The training day was included to familiarize the test subjects with the experimental procedures and to minimize learning effects between examination days. On the training day, subjects were given information about the tests, the tests were performed in abbreviated versions, and height and weight were measured.
On the test days, the subjects arrived at the laboratory at 8 or 9:30 AM. The experiment started at the same time of day on both test days for each subject. During nSEP and LEP measurements, the subject was lying comfortably on an examination bench with the lights slightly dimmed. The subjects were instructed to lay still and relax, keep their eyes open and to fix their gaze on a preplaced marker in the centre of their vision, and to limit blinking for a few seconds after the LEP- and nSEP-stimulations. All stimulations were carried out on the right arm.
EEG-recording
LEPs and nSEPs were recorded using the actiCAP active electrode system with 32 channels placed according to the 10–10 system (25), using FCz as a common reference electrode, and AFz as ground electrode. EEG were recorded with 5000 Hz sampling rate, 0.1 microvolt resolution, low cutoff 0.0159 Hz (10 s), high cutoff 1000 Hz. A BrainAmp amplifier with 32 channels were used with the recording software Brainvision recorder, version 1.20.0701 (Brain Products GmbH, Munich, Germany). Electrodes were mounted on the nasion for offline re-referencing, and lateral to the right eye and superior and lateral to the left eye for detection of eye movements and blinking. Impedance was kept below 20 kΩ.
LEP
Laser stimulation was applied to the dorsum of the right hand, distal to the extensor line of the wrist, proximal to the metacarpophalangeal joints, and between the second and fourth metacarpal bones. The stimulation site was changed randomly with a minimum distance of 10 mm between consecutive stimuli to avoid skin damage, and peripheral nociceptor fatigue or sensitization.
The painful heat stimuli were generated by an infrared neodymium: yttrium-aluminumperovskite (Nd:YAP) laser (STIMUL 1340, DEKA M.E.L.A SRL, Calenzano (FI), Italia), using a wavelength of 1340 nm. The laser stimuli were transmitted through an optic fiber with an energy ranging from 2 to 6.5 J (4.0–12.9 J/cm2, using a laser beam diameter of 8 mm (area ≈ 50 mm2) and a duration of 6 ms. These settings are similar to those used in other studies at our lab, and comparable to other studies using a similar laser stimulator (26).
A diode laser beam marked the area of stimulation. A total of 50 stimuli were given during the main test with an interstimulus interval of 6–10 s. Before the main test, individual thresholds for pinprick pain were determined. The stimulus intensity was started at 2 J and increased by steps of 0.5 J every second stimuli until an NRS score of 8 or higher or 6.5 J was reached. When pain was reported, test subjects disclosed whether burning pain or pinprick pain was felt. The pain threshold (PT) was defined as an intensity yielding pinprick pain in at least one of the two stimuli. The stimulus intensity used in the main test was started at 4.5 J (9.0 J/cm2) on the first test day. If the test subject experienced pain lower than a NRS of 2 or higher than 8 with this stimulus intensity, the stimulus intensity was adjusted within the range of 3–6.5 J (≈ 6.0–10.9 J/cm2), before the 50 stimulations started. The mean intensity given was 5.62 J in patients and 5.77 J in controls (Table 1). Ten subjects were given 4.5 J, while the remaining subjects were given a higher intensity. Stimulus intensity in J, which were determined on the first test day, were identical on test day 1 and test day 2. The test subjects were instructed to score the pain of each stimulus verbally.
NSEP
High density electrical stimulation was given with an electrode made of platinum. The electrode had a diameter of 0.2 mm and protruded 0.2 mm from the surface of a polyoxymethylene frame. The electrode was custom made at the National Institute of Occupational Health, Oslo, Norway (11), based on the electrode used in a study by Inui et al (27) The electrode was attached to the volar forearm 1 cm medial to the halfway distance between the distal end of ulna and the insertion of the biceps brachii tendon. A Velcro strap (Alpine Biomed ApS, Skovlunde, Denmark) soaked in an isotonic NaCl solution, was used as the anode. It was placed 5 cm proximal to the ipsilateral cubital fossa. A constant current stimulator (DS7A and DG2A, Digitimer, Hertfordshire, England) delivered electrical stimuli made up of two unipolar pulses with 0.5 ms duration and 10 ms inter pulse interval. Subjects were lying in a supine position. Individual PT and stimulus intensity for nSEP were determined at both test days. PT were determined by three series of stimuli; for each series, stimuli started at 0 mA and increased in steps of 0.1 mA until the subject perceived the stimuli as painful. The mean of the two last values was set as the PT. If the stimulation gave a feeling of radiation or muscle contraction at any point during the experiment, the electrode placement was adjusted slightly.
For the main test three blocks were applied, each with 30 stimulations. Within each block, the stimuli were divided equally between two intensities: A (3×PT) and B (4×PT) and varied in pseudo-randomized order. Inter-stimulus interval was 10 to 15 s and inter-block interval was 2 min. Subjects were instructed to verbally rate pain intensity after each stimulus.
EEG processing
For data analysis, we used Brainvision Analyzer, version 2.1.0.327, Professional edition (Brain Products GmbH, Munich, Germany). First, the stimulus markers were investigated and edited in case of stimuli not being identified correctly. A filter was applied (0.3 Hz high pass, 100 Hz low pass and 50 Hz notch). Then data was segmented based on the stimulus markers with a range of 1000 ms pre stimulus and 1000 ms post stimulus and re-referenced to the nasion electrode. Later a semi-automatic artefact rejection was performed. All segments containing amplitudes greater than ± 65 microvolt underwent manual review, and segments with artefacts were removed.
After artifact rejection, the signal was baseline corrected from 100 ms pre stimulus. The signal was averaged across each block separately and for all blocks combined (3 blocks for nSEP and 2 blocks for LEP). Automatic peak detection was applied. The LEP N2-potential was defined as the most negative peak between 140–320 ms, and P2 as the most positive peak between 190–500 ms. For nSEP N2 was set at the most negative peak between 50–200 ms, and P2 at the most positive peak between 150–500 ms. Peak detections were manually controlled and corrected if needed. N2- and P2 peak latencies and amplitudes were extracted and N2P2 peak-to peak amplitude calculated.
Statistics
SPSS Statistics Version 26 and SYSTAT Version 13 were used for statistical analyses. As in a previous study by our group (12), Cz was selected as the primary sites for the analysis because the largest N2P2 amplitude is close to Cz (28). Fz was chosen as an additional recording site according to recommendations by Cruccu et al (29) LEP N2P2 amplitudes from the Cz-Nasion deviation were square root transformed, and LEP N2P2 amplitude for the Fz-Nasion deviation and nSEP N2P2 amplitudes were transformed using the natural logarithm; LN(x + 0.1). After these transformations, distribution of normality of latencies and transformed amplitude variables were assessed and found to be satisfactory with visual inspection of histograms and QQ-plots, and Shapiro-Wilk test.
Primary outcome variables were LEP and nSEP N2P2 amplitude (for all blocks and both intensities combined) and N2P2 amplitude habituation. Habituation was quantified by habituation slope for both LEP and nSEP (calculated from block amplitudes with least squares lines regression for each subject) including two blocks for LEP and three blocks for nSEP. Secondary outcome variables were LEP and nSEP N2 and P2 latencies.
Repeated measure ANOVA were used to compare the effect of sleep condition on primary variables in migraine subjects with interictal measurements after both sleep conditions (n = 21) and controls (n = 31). In the analysis of amplitudes and latencies, group (interictal migraine or control) were included as between-subject factors and sleep condition (habitual sleep and sleep restriction) as within-subject factors. In the analysis of N2P2 amplitude habituation analysis, block was added as an additional within-subject factor. Repeated measures ANOVA for nSEP habituation were corrected for non-sphericity with the Huynh-Feldt method. Significant factors in the repeated measure ANOVA regarding N2P2 amplitude, habituation, and latencies for the two sleep conditions were compared with a paired Student's t-test for each group separately. Variables for difference in nSEP amplitude habituation between habitual sleep and sleep restriction were correlated with migraine history duration, usual migraine headache intensity, usual migraine attack duration, migraine days in the last three months, migraine attack frequency, HADS depression and anxiety scores, and insomnia severity score.
To detect a medium sized effect (0.65 SD) with a power of 80% and significance level of 5% in a paired samples t-test, a group size of n = 21 is needed. Based on experience from prior studies, we estimated that 45 migraineurs would be needed to get enough migraine patients with two interictal recordings. P-values < 0.05 are reported as significant.
Ethics
LEP and nSEP stimulations are mild to moderately painful and without risk of injury. The methods in this project are widely used, and well known to be safe. The study was approved by the Norwegian Regional Committee for Medical Research Ethics (approval number 2014/2322). All subjects gave informed consent.
Results
Age, sex distribution, BMI, days since last menstruation and ESS score were similar in migraine subjects with interictal examinations after both sleep conditions and controls, while HADS scores were slightly higher in the migraine group as expected. All interictal migraine patients had 1–7 migraine days/month. and on average a headache history of approximately 20 years (Table 2). Mean PT for nSEP and LEP were similar in migraine patients and controls, as well as after the two sleep conditions. Total sleep time and time in bed after sleep restriction and habitual sleep measured by actigraphy were similar in the interictal migraine and control groups (Table 1). In both groups, 52% of the participants had a habitual sleep condition before restricted sleep.
Mean (SD) for pain thresholds (PT), total sleep time and time in bed measured by actigraphy, reaction time (PVT) and sleepiness (KSS) after habitual sleep and sleep restriction.

Mean (SD). No differences were seen between the migraine subgroup with interictal measurements after both sleep conditions and all included migraine patients. PT: Pain threshold, PVT: Psychomotor vigilance test measuring reaction time, KSS: Karolinska sleepiness scale score at beginning of laboratory visit, i.e., 08:00 or 9:30AM (scale from 1: ‘alert’ to 9: ‘very sleepy’). J: joule.
Demographical and clinical data on migraine and control subjects.

Mean (SD) or number of participants. aHospital Anxiety and Depression scale. bUsual intensity of migraine attacks, 1 = light – can keep doing a task, 2 = moderate – can do light tasks, 3 = strong – have to lie down, 4 = extremely strong – cannot lay still. cAverage number of headache days per month for the last three months. dFrequency + Intensity score for photophobia during migraine attacks: eFrequency + Intensity score for phonophobia during migraine attacks: Frequency: 1: Rarely (less than 1/4 of attacks), 2 = Less than 1/2 attacks, 3 = up to 3/4 attacks, 4: almost every attack). Intensity: 0 = none, 1 = light, 2 = moderate, 3 = severe. NA: Not applicable. f excluding women with menopause and amenorrhea > 50 days. HS: Habitual sleep. SR Sleep restriction.
Amplitudes and latencies
For LEP and nSEP amplitude there were no significant effects, neither for diagnosis, sleep condition, nor for the interaction between sleep condition and diagnosis (Table 3). We observed significantly longer N2 nSEP latency at Cz in the interictal migraine group compared to controls (Table 3; significant group factor, independent of sleep condition). For migraine subjects and controls combined, we observed longer LEP N2 latency at Fz after sleep restriction compared to habitual sleep (Table 3, significant sleep condition factor, independent of group).
N2-P2 amplitudes and N2 and P2 latencies of laser evoked potentials (LEP) and nociceptive somatosensory evoked potentials (nSEP) (mean and SD).

Bold: Significant findings.
NSEP and LEP amplitude habituation
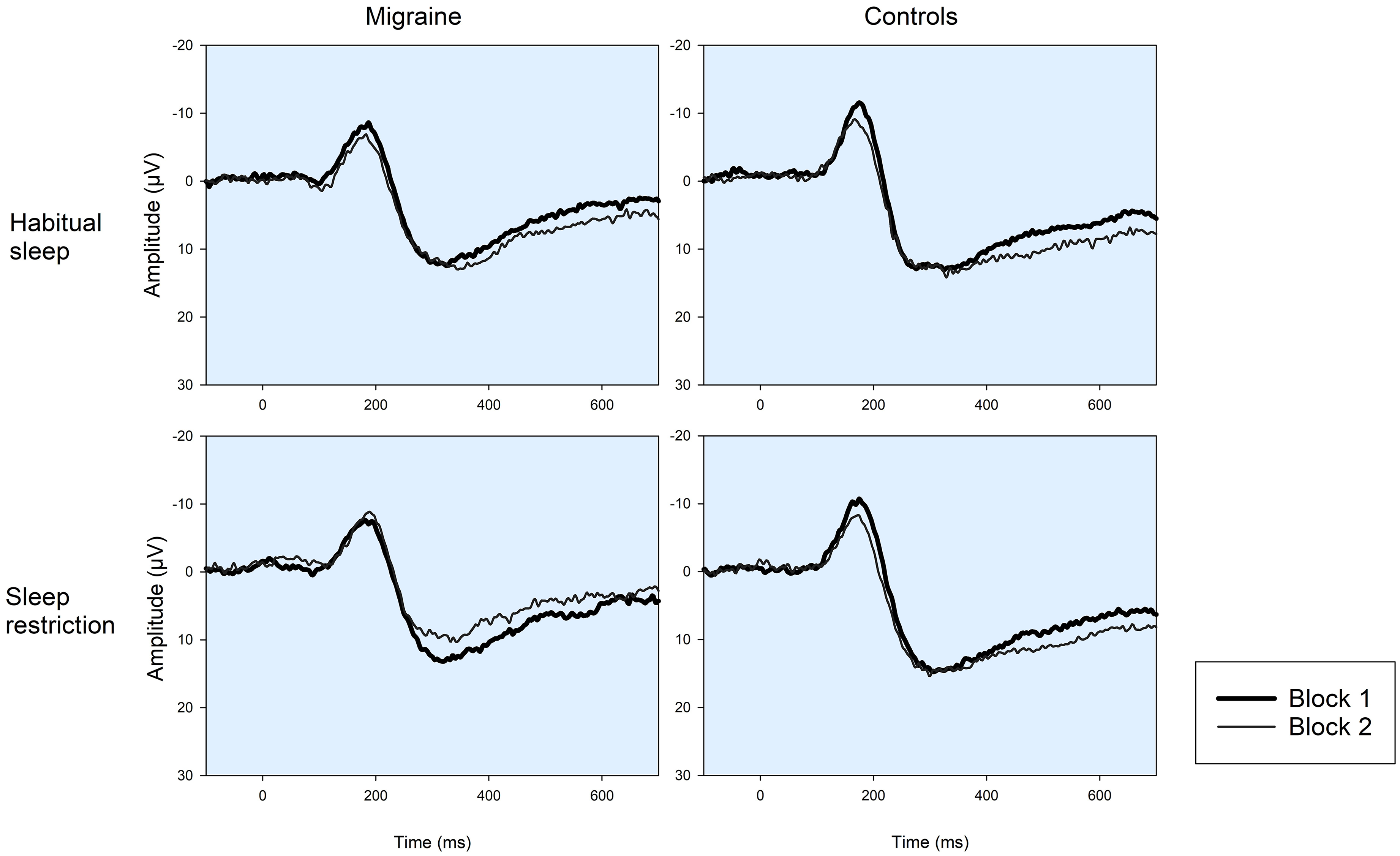
The nSEP sleep × block × group interaction was significant at both Cz and Fz; p ≤ 0.012 (Table 4). Contrast analysis shows that migraine subjects had reduced nSEP habituation after habitual sleep compared to controls (p < 0.035, Table 5).
Repeated measures ANOVA F-statistics show how pain-SEP amplitude habituation (block) is affected by sleep restriction and group

Repeated measures ANOVA with group (interictal migraine or control) as between-subject factor and block (B1, B2, B3 for nSEP and B1, B2 for LEP) and sleep-condition (habitual sleep and sleep restriction) as within-subject factors. Bold: Significant findings. ×: interaction. ns: non-significant.
Amplitude-habituation slopes (mean and SD) for LEP block 1–2 and nSEP block 1, 2 and 3.

Amplitude habitation was analyzed by ANOVA, showing significant three-way group × sleep × block interaction for nSEP (p ≤ 0.012, Table 4). Contrasts were compared by the Habituation Slope (least square regression slope for N2P2 block amplitude for block 1, 2 and 3 for nSEP and block 1 and 2 for LEP.
Paired Student's t-test shows increased nSEP amplitude habituation following sleep restriction in the interictal migraine group but not in the control group (p < 0.025, Table 4, Figure 3 and 4). For LEP N2P2 amplitude, we observed no differences in amplitude habituation between groups; Table 4), and no effect of sleep condition on amplitude habituation (Table 5, Figure 5 and 6).

Grand average nociceptive somatosensory evoked potentials (nSEP) from the Cz-nose derivation for migraine patients in the interictal phase and healthy controls following habitual sleep and sleep restriction. To measure amplitude habituation responses were averaged in blocks. Waveforms for the first and last block are indicated by thick and thin lines respectively. Migraine patients had significantly reduced nSEP amplitude habituation compared to controls, and nSEP amplitude habituation was increased in the migraine group following sleep restriction.
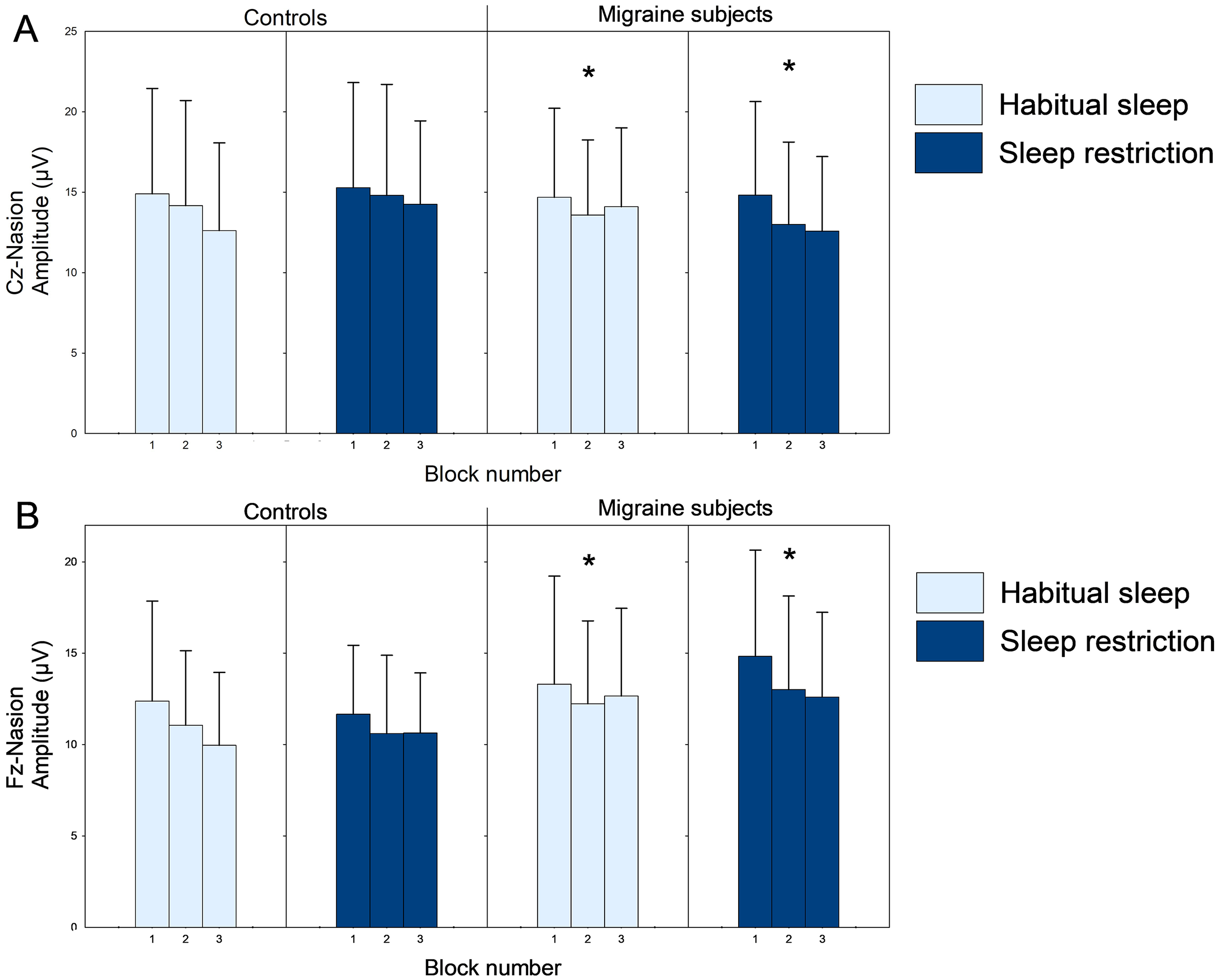
Block amplitudes (mean ± SD) for nociceptive evoked potentials to high-density electrical stimulation (nociceptive somatosensory evoked potentials, nSEP) at the Cz-Nasion (A) and Fz-Nasion (B) bipolar deviations for controls and interictal migraine subjects after habitual sleep and sleep restriction. *NSEP amplitude habituation was significantly lower in migraine patients after habitual sleep compared to controls (Student's t-test, p < 0.035) and increased significantly following sleep restriction in migraine subjects (paired Student's t-test, p < 0.025).
Grand average laser evoked potentials (LEP) from the Cz-nose derivation for migraine patients in the interictal phase and healthy controls following habitual sleep and sleep restriction. To measure amplitude habituation responses were averaged in blocks. Waveforms for the first and last block are indicated by thick and thin lines respectively. For LEP amplitude habituation there were neither significant differences between groups nor sleep conditions.

Block amplitudes (mean ± SD) for laser evoked potentials (LEP) at the Cz-Nasion (A) and Fz-Nasion (B) bipolar deviations for controls and migraine subjects after habitual sleep and sleep restriction. LEP amplitude habituation did not differ between groups or sleep conditions.
The nSEP amplitude habituation difference between sleep restriction and habitual sleep correlated with insomnia severity (r = 0.51, p = 0.014) in migraine (Figure 7) but not in controls (r = −0.11, p = 0.56). Other clinical correlations were not significant (p > 0.07).

Difference in nSEP habituation slope following sleep restriction and habitual sleep correlated with insomnia severity in interictal migraine patients (Cz-nasion recording. Pearson r = 0.5, p = 0.014, n = 21).
Discussion
The main finding of this study was increased nSEP amplitude habituation in the interictal migraine group, but not the control group, following sleep restriction. For nSEP but not LEP, the interictal migraine also had lower habituation than controls after habitual sleep. In addition, there was a significantly higher nSEP N2 latency in interictal migraine compared to controls in both sleep conditions. LEP N2 latency were increased in both controls and migraine patients after sleep restriction.
Our findings of increased habituation following sleep restriction indicate that insufficient sleep causes increased inhibitory responses to repeated nociceptive stimuli in migraine. We also found that the difference in nSEP amplitude habituation between sleep restriction and habitual conditions in migraine, increased with the level of subjective insomnia symptom load. This finding suggests a larger effect of insufficient sleep in migraine patients with insomnia symptoms.
Previous studies have reported both increased and decreased cortical responses in migraine. It has been suggested that these varying findings are caused by altered thresholds for homeostatic inhibitory mechanisms (4). Increased habituation to repeated nSEP stimulation in migraine after sleep restriction may suggest a decreased inhibitory homeostatic threshold following reduced sleep. Alternatively, increased inhibitory effects in migraine may be caused by increased cortical activation reaching the inhibitory threshold (4). Insufficient sleep is known to downregulate dopamine receptors in striatum, increasing thalamic activity, which in turn may cause increased activation to the level of an inhibitory threshold (30). Interestingly, high frequency TMS may restore or increase VEP and conventional somatosensory evoked potential (SEP) habituation in interictal migraine (31,32) and topiramate may restore normal LEP amplitude habituation in migraine (33). Furthermore, increased or even normalized habituation has been reported in the ictal compared to the interictal period in migraine subjects (34). Thus, our findings suggest similar effects on homeostatic inhibitory threshold effects from interictal sleep restriction as the ictal phase, high-frequency rTMS and topiramate may have in migraine.
NSEP has not been investigated in migraine subjects previously, and this study is the first to report reduced nSEP amplitude habituation in interictal migraine following habitual sleep. A previous study of healthy subjects also found habituation of nSEP amplitude, but no effect of sleep restriction on amplitude habituation (12). Reduced amplitude habituation has been reported previously for other types of evoked potentials including conventional SEP (35), indicating that reduced amplitude habituation in migraine may apply to sensory processing in general, and not just pain-related activity.
We did not find altered interictal LEP amplitude habituation in interictal migraine in contrast to some (14,15) but not all (36) previous studies. Compared to LEP, the electrical stimulation in nSEP may activate more subtypes of Aδ-fibres, and likely also results in a more synchronous activation of these fibers (37). The trigeminovascular system has a relatively higher amount of Aδ-fibers than extracephalic regions and is also the system primarily involved in migraine pain (38). Concordantly, it is possible that alterations in nociceptive processing in migraine can be more easily revealed by nSEP than LEP. Evoked potential amplitudes and amplitude habituation in general show a large interindividual variability and habituation to various types of evoked potentials have been studied in migraine with discrepant results (4,32,39). Our results indicate that insufficient sleep may increase amplitude habituation in migraine, at least for nSEP, and that this effect is stronger in patients with more insomnia symptoms. It is possible that this effect can have contributed to the previous discrepant findings regarding amplitude habituation in migraine. Future studies investigating the neurophysiology of migraine should therefore include an evaluation of sleep patterns and insomnia symptoms of the included subjects. Nevertheless, the increased nSEP habituation in migraine patients following sleep restriction is the more robust finding in the present study, as this is based on repeated measurements in the same subjects.
Pain-evoked N2 latency was affected more by migraine diagnosis and sleep restriction than amplitudes. Event related potentials (ERP) amplitudes have greater variability than latency, and ERP latencies are considered more reliable and clinically relevant than amplitudes (40). Altered latencies in nociceptive ERP following sleep restriction in healthy volunteers may be caused by increased synaptic instability in pain-processing networks (41). Our results indicate synaptic instability in pain-processing networks following sleep restriction also in migraine subjects. Interestingly, it has recently been suggested that synaptic dysfunction in brainstem-thalamus-cortex axis is of importance in migraine pathophysiology (42).
Strengths and limitations
A major strength of this study is the strict blinded study design, which included blinding for diagnosis and sleep condition both during recordings and data processing. Stimulus intensity was not analyzed in the present study since NSEP amplitude is almost identical after 3 × PT and 4 × PT stimuli and was not affected by sleep restriction in healthy subjects (12). A possible limitation is the number of migraine subjects with interictal recording. However, 21 migraine subjects had examinations in the interictal period after both sleep conditions, so the power to detect medium effect sizes was acceptable.
Both migraine subjects and controls had some insomnia symptoms. As expected, the controls subjects had less insomnia symptoms than the migraine group (1), and the migraine subjects were evaluated for insomnia by a neurologist. It is therefore unlikely that the participants in this study met the diagnostic criteria for insomnia, although we cannot rule this out completely.
Variation in inter-stimulus interval for nSEP and LEP may reduce amplitude habituation. However, a previous study investigating LEP amplitude habituation in migraine with a similar method reported amplitude habituation in both migraine and controls (36). Therefore, the method applied in the present study should be adequate for investigating amplitude habituation.
Conclusion
We found increased amplitude habituation to electrical nociceptive stimulation in migraine following sleep restriction and decreased habituation after habitual sleep. These findings may indicate that the migraine brain may have an increased vulnerability for insufficient sleep and suggest that insufficient sleep may result in increased cortical inhibitory responses in migraine. This effect may be due to a generally increased cortical responsivity and reduced thresholds for homeostatic inhibitory mechanisms in migraine. However, the findings should be interpreted with caution as they were only seen with electrical and not laser nociceptive stimulation.
Article highlights
Migraine patients showed signs of increased cortical inhibitory responses to nociceptive stimulation after insufficient sleep. Migraine patients may have an increased vulnerability for insufficient sleep.
Footnotes
Acknowledgements
We are grateful for the contributions from all subjects who participated in the study, and from our collaborators Gøril Bruvik Gravdahl, Lars Jacob Stovner, Erling Tronvik, Knut Hagen, Mattias Linde, and Stine Schei and Marit Stjern. The examinations were performed at the core facility NeXt Move, Norwegian University of Science and Technology (NTNU).
Authors’ contributions
All the authors have read and approved the final version of the manuscript. JOH, MU, TS, DM and PMO designed and planned the study. JOH conducted the examinations, TS performed data analyses and statistical analyses, and PMO and JOH drafted the first version of the manuscript. PMO, MU, DM, MM and TS and contributed to the process of data analysis and made important contributions to the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics
The work has been carried out in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki). The study was approved by the Regional Committee for Medical Research Ethics Central Norway (approval number 2014/2322). Written, informed consent was obtained from all participants.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding from NTNU, Norwegian University of Science and Technology.
