Abstract
The aim of the study was to evaluate sleep of children with migraine during the interictal period and the modifications of sleep which precede, are concomitant with, or follow migraine attacks. Eighteen patients with migraine without aura were compared with a group of 17 healthy age-matched children. Sleep parameters were monitored for two full weeks by means of actigraphs and self-report diaries. Headache diaries were also filled out in order to evaluate the occurrence and the characteristics of migraine attacks. Fifty-seven attacks were recorded during the monitoring period. During the interictal period, sleep parameters of children suffering from migraine did not differ from those of controls; only sleep onset latency was slightly prolonged in the migraine group. Timing of the attack affected nocturnal motor activity which presented the lowest values on the night preceding the attack, indicating a decrease in cortical activation during sleep preceding migraine attacks. Further studies should clarify if the observed reduction in nocturnal motor activity close to the attack is related to neurotransmitter imbalance.
Keywords
Introduction
Several epidemiological studies have shown the presence of co-morbidity between various sleep disorders and different headache subtypes (1). A study on psychiatric co-morbidity showed that, beside other psychiatric problems, sleep disturbances are the most frequent complaint in children with headache (2).
However, it has not yet been established if headache is the major causative factor of sleep disruption or if sleep is the main trigger event for headache attacks. Headaches are known to occur during sleep and in relationship with various sleep stages. A lack or an excess of sleep, a poor quality or inadequate duration of sleep are usually reported as possible causes of headache. Many headache patients, whatever the type of headache they have, complain of insufficient sleep, lack of restoration in the morning, severe snoring and other sleep disturbances (1). Several studies on headache in children outlined the contemporary presence of headaches and parasomnias (3, 4) and also a relationship between sleep breathing disorders and migraine or tension-type headache (5). It has been reported that sleep respiratory problems can cause a migraine attack (6), which is only indirectly due to sleep apnea events but seems to depend mainly on the interruption of sleep continuity (7). This is indirectly confirmed by the causal link between poor sleep quality and headache, which is reported by migraine patients. Bruni et al. (5) highlighted that both young migraine and tension-type headache sufferers have a worse sleep quality (shorter sleep duration, longer sleep onset latencies, more awakenings, etc.) than controls.
Few studies have focused on the whole sleep-wake cycle and on the circadian aspects of headache. A study by Fox and Davis (8), which analysed the circadian distribution of 3582 attacks of 1689 adult migraine subjects, demonstrated that migraine attacks occurred most frequently in the early morning. However, this finding does not clarify the exact relationship between the occurrence of a headache attack and disturbed sleep. A recent polysomnographic study showed that, during the night preceding migraine attacks, there was a significant decrease in the number of arousals, rapid eye movement (REM) density, beta power during slow-wave sleep, and in alpha power during the first REM period. This suggests that there is a decrease in cortical activation during sleep preceding migraine attacks (9).
Based on this study, sleep recordings offer an interesting insight into the pathophysiology of alterations preceding migraine headache attacks. However, it would be helpful to evaluate this relationship in a high number of subjects and for a long period. Recent sleep studies used actigraphy in order to have an objective estimation of sleep parameters in naturalistic conditions and over a long period of time (10, 11). Actigraph monitoring has been found to have good face validity and its reliability has been documented in numerous studies that found a high rate of agreement (85–90%) between actigraphic and polysomnographic measures of sleep; further, actigraphs have been found to successfully distinguish between sleep-disturbed and control children (12, 13).
The aims of the present study were: (i) to evaluate sleep quality of children suffering from migraine without aura in the interictal period, in comparison with healthy age-matched controls by means of actigraphic monitoring and (ii) to evaluate whether modifications of sleep quality precede, are concomitant with or follow a migraine attack.
Method
Subjects
Eighteen patients with migraine without aura (10 male, eight female; mean age = 9.8 years; S.D. = 1.21; range 8–12) were recruited from those attending our Pediatric Headache University Centre.
They were selected according to the following criteria:
migraine without aura according to IHS criteria (1988);
more than one migraine attack per week (in order to increase the probability of recording during an attack);
absence of other disorders (as evaluated by clinical interviews as well as by neurophysiological recordings).
The migraine group was compared with a group of 17 healthy age-matched children (mean age = 9.6 years; S.D. = 0.99; range 8–12) recruited in a school in Rome.
Measures
Actigraphy
Actigraphs are used in sleep assessment to discriminate between sleep–wake states by means of computerized analysis of body movements. Basic Mini-Motionloggers from Ambulatory Monitoring, Inc. (Ardsley, NY, USA) were used in the current study to measure sleep parameters and nocturnal motor activity. These actigraphs employ a piezoelectric sensor, have a fixed sensitivity at 2–3 Hz and detect accelerations greater than 0.01 g force. The mechanism is housed in a metal, waterproof case and has a 32 K memory. The actigraphs were programmed to employ a zero-crossing mode using an auto actigraph interface.
Data were extracted using the ACT software and analysed by means of the ACTIONW2 program, according to the validated sleep estimation algorithm developed by Sadeh (14). Before analysing the data, recordings were visually inspected to reject any epochs where the actigraph had been removed from the subject's wrist.
Children indicated ‘lights out’ and ‘get out of bed’ events by pressing the event marker of the actigraph. Four sleep parameters (total sleep duration, sleep onset time, minutes of night awakenings, and sleep efficiency index) were computed.
Total sleep duration was defined as the number of minutes from sleep onset to wake onset.
Sleep onset was measured in minutes from the time the children indicated ‘lights out’ (by pressing the event marker to sleep onset) to the first period of sleep of more than 20 min.
Total time in bed was defined as the number of minutes from the ‘lights out’ event to ‘get out of bed’ event.
Sleep efficiency was calculated as the ratio between total sleep duration and total time spent in bed.
An index of motor activity (% epochs with > 0 activity score) in the period that subjects spent in bed was computed.
Self-report questionnaire
A self-report questionnaire was developed using previously validated paediatric symptom diary scales (15). This questionnaire was previously administered to a pilot sample of children in order to assess the ease of use and compliance with use of the diaries. Subsequently, a version of the questionnaire was prepared with graphics and language appropriate to the age of the patients.
The questionnaire contained sleep and headache diaries composed by simple questions about sleep patterns and about the characteristics of migraine attacks. The sleep diary collected information on sleep onset time, wake-up time; bedtime difficulties and nocturnal awakenings, with identical questions for each day.
The headache section collected information on the time of occurrence of the attacks, their duration and severity, and subjective evaluation of trigger factors. Additional questions regarded school activities, eating habits, tiredness and irritability. An Italian copy of the questionnaire can be obtained from the first author.
Procedure
The institutional review board approved the study. Informed consent was obtained from parents before enrolment in the study. Parents were provided with general information on how the actigraph worked and then it was placed on the wrist of the child's non-dominant hand. Furthermore, both parents and children were instructed that the diary was to be completed by each child just before he/she went to sleep and as soon as possible after he/she got up in the morning. The group of children with migraine wore the actigraph and completed the sleep diary for two full weeks (in order to increase the probability of recording a migraine attack), while the control subjects wore the actigraph and completed the sleep diary for one full week (in order to obtain a reliable recording of sleep parameters). At the end of the recording period, the parents returned the actigraph and the questionnaires to the Headache Centre. Children were also asked to complete the diary each morning and evening.
Data analyses
Statistica software (StatSoft Inc. 2001. STATISTICA– data analysis software system, version 6. http://www.statsoft.com) was used for all analyses. The results of the study are presented in two sections: (i) interictal phase and (ii) occurrence of migraine attack.
Interictal phase
Data for both groups were considered. The results of one-way ANOVA designs are shown, in which Group (Between Factor; 2 levels: Migraine vs Controls) is the independent variable and sleep parameters and activity index are the dependent variables. For each dependent variable, in order to avoid the influence of attack occurrence, only sleep during the nights which were distant at least 48 h from an attack is considered and averaged (NIGHTS WITHOUT ATTACK). Considering that no control subjects experienced a headache attack, all recording nights were averaged for these subjects.
Occurrence of migraine attack
Only data collected on the migraine group were considered. Considering the time of occurrence of the attacks, the nights of children with migraine were classified into four classes: (i) NIGHTS WITHOUT ATTACKS (see above); (ii) NIGHTS PRECEDING THE ATTACK (nights in which sleep started less than 24 h before the attack); (iii) NIGHTS OF THE ATTACK (namely nights in which sleep started less than 24 h after the attack); (iv) NIGHTS AFTER THE ATTACK (namely nights in which sleep started in the period of 24–48 h after the attack). Nights belonging to the same class were averaged. This classification allowed us to define a 4-level within factor (RECORDING PERIOD).
For each sleep parameter and for the activity index, a repeated-measures ANOVA was conducted in which RECORDING PERIOD (within factor; 4 levels) was the independent variable.
Results
The children with migraine were monitored for a total of 252 days in which 57 attacks were observed (23% of monitoring days), with a mean of 3.2 attacks per patient (S.D. 2.8). Twenty-two attacks were short in duration, lasting 1 hour or less, while 14 lasted between 2 and 3 h and 21 lasted for four or more hours. Thirty-two attacks were of mild-moderate intensity, while 25 were of high or very high intensity.
There were 28 attacks in the morning (08.00–12.00 h), 22 attacks in the afternoon (13.00–20.00 h), while 17 occurred in the late afternoon and evening. None of these attacks occurred after sleep onset. The tiredness in the morning after the attack was absent or low after 39 attacks, of medium intensity after 14 attacks and only after four attacks subjects reported a substantial increase in the level of tiredness. No modification of the usual time of sleep onset was observed in relation to the occurrence of migraine attacks. No between-groups difference emerged for sleep onset time and wake-up time.
Interictal phase: differences between groups
The mean values of actigraphic sleep parameters recorded in both groups are reported in Table 1. No significant differences between children suffering from migraine and controls were observed in sleep parameters. A tendency towards statistically significant difference was observed for sleep onset latency: migraine patients showed a time to fall asleep significantly longer than that of control subjects.
Actigraphic parameters in migraine (interictal period) patients and normal controls

n.s., non significant.
Occurrence of migraine attacks
Only one subject presented migraine attacks on two consecutive days and therefore the corresponding nights could not be classified univocally as specified in the Method section. For this reason, these actigraphic data were not included in the analysis.
The repeated measures ANOVAs showed a significant effect of the RECORDING PERIOD for the nocturnal motor activity index (F = 3.11; P = 0.034). Averages of nocturnal motor activity in the four moments of recording are displayed in Fig. 1. Sheffé post-hoc comparison showed that this significant effect is due to the level of motor activity in the NIGHTS WITHOUT THE ATTACK (mean = 14.0; S.D. = 5.85) which is higher than the level of motor activity during the NIGHTS PRECEDING THE ATTACK (mean = 10.96; S.D. = 5.23; P = 0.005), during NIGHTS OF THE ATTACK (mean = 11.88; S.D. = 5.40; P = 0.045) and during the NIGHTS AFTER THE ATTACK (mean = 11.90; S.D. = 5.93; P = 0.047).
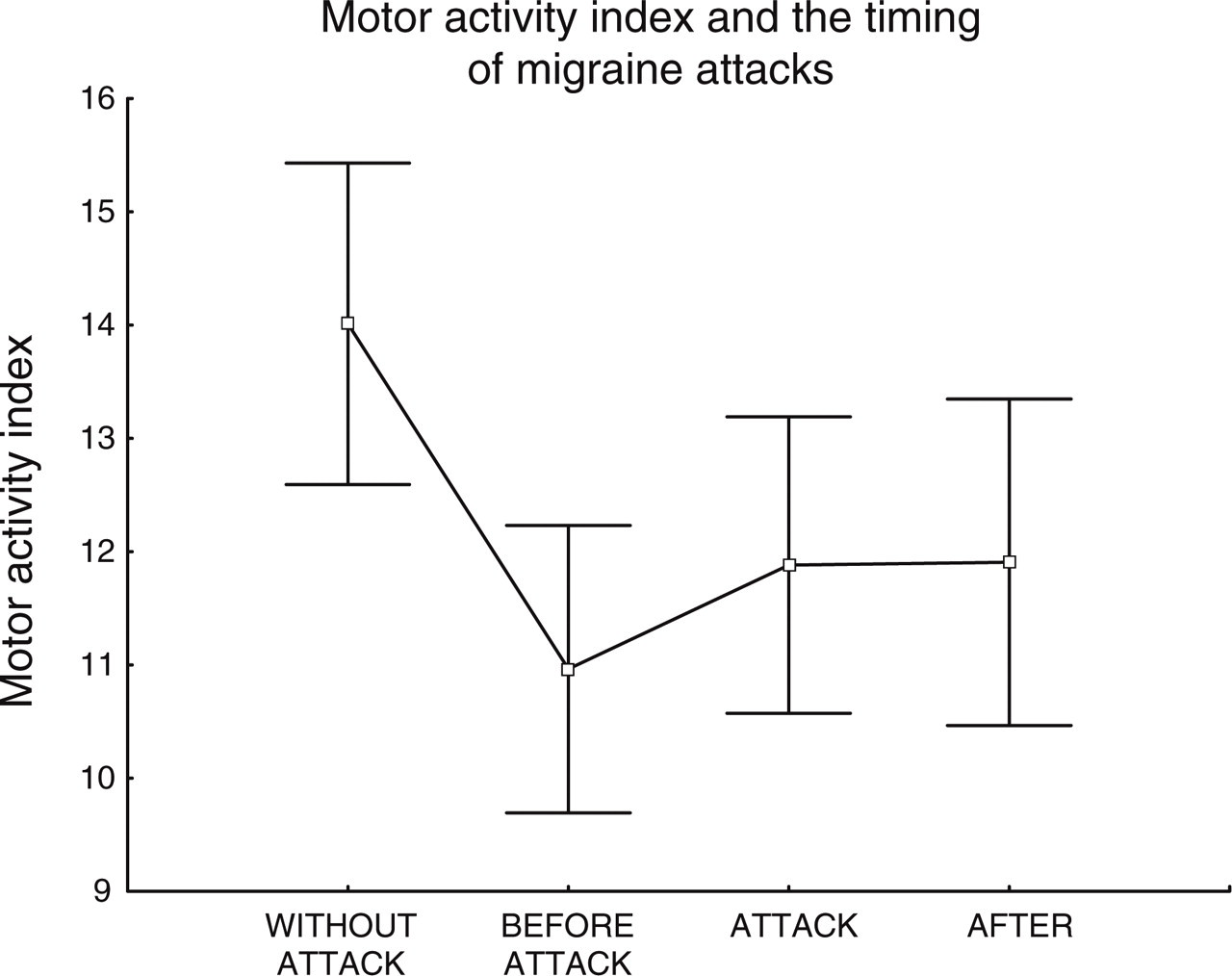
Nocturnal Activity Index was averaged across nights according to the following classification: WITHOUT ATTACK = nights that were distant at least 48 h from an attack; BEFORE ATTACK = nights in which sleep started less than 24 h before the attack; ATTACK = nights in which sleep started less than 24 h after the attack; AFTER = nights in which sleep started in the period of 24–48 h after the attack.
With respect to sleep parameters, repeated measure ANOVAs did not show any statistical differences due to the recording period for sleep duration (F = 1.01; P = 0.39), sleep efficiency (F = 0.91; P = 0.43), minutes of wake after sleep onset (F = 0.95; P = 0.42), and the difference in the mean sleep onset latency did not reach statistical significance (F = 2.17; P = 0.10).
Discussion
Our study represents the first attempt to evaluate direct relationships between sleep and migraine using an objective method over a long period of time; in contrast, other studies investigated this topic using parental ratings and self-reports measures (5, 16). Actigraphy has been widely used to evaluate sleep in children of different ages (10, 17) and has proved to be a reliable tool for assessing sleep parameters (11, 12, 14).
As children with migraine are reported as potentially sleep disordered subjects, an objective evaluation of sleep could help to clarify the temporal relationships between the occurrence of migraine attacks and sleep disruption (1).
In the present study, 57 attacks were recorded during the 252 days of sleep monitoring of 18 children with migraine. Comparison between interictal sleep parameters of children with migraine and those of age-matched controls did not show any substantial modification of actigraphic sleep parameters in migraine patients during the pain-free intervals. A difference was found in sleep latency which did not reach the usual statistical significance, but it is consistent with previous findings, which showed a longer time to fall asleep in children suffering from migraine (5, 18).
The intragroup analysis of migraine subjects showed that sleep parameters were not influenced by the occurrence of the attack, as no differences were found the night before, after and during migraine attacks. This is probably due to the fact that, in our group, no attacks occurred during the night, directly causing sleep disruption. Therefore, the hypothesis of a causal link between disturbed sleep and occurrence of migraine attack is not supported by the present findings.
The only actigraphic parameter affected by the timing of the attack was the nocturnal motor activity index which showed a typical trend with the lowest values on the night preceding the attack that increased slowly the following nights. It is worth noting that the greatest reduction of nocturnal motor activity was observed during the night preceding the attack. This finding is indirectly supported by recent studies that have evaluated the relationship between migraine attacks and sleep using neurophysiological methods (9, 19).
Using non-linear EEG analysis Strenge et al. (19) found a loss of dimensional complexity in the first two non-rapid eye movement sleep states in the migraine attack night, providing evidence of a global dimension decrease that is related to cortical network changes during a migraine attack. Also, they found attack-related neuronal complexity reductions already at the very beginning of the migraine night before the occurrence of pain. The same authors stated that this phenomenon could reflect changing activity in the noradrenergic system (20) which could also be involved in the reduction of motor activity observed in the present study.
Furthermore, a general decrease in cortical activation during sleep preceding migraine attacks has been found in a recent polysomnographic study (9). The authors, analysing sleep alterations preceding migraine attacks, found no alterations in the occurrence of specific sleep stages, but a significant decrease in the number of arousals, in rapid eye movement (REM) density, in beta power in slow-wave sleep and in alpha power during the first REM period. These results suggest a decrease in cortical activation during sleep preceding migraine attacks.
It has been reported that typical premonitory signs, such as irritability, withdrawal, depression and drowsiness, occurred during the 24 h preceding a migraine attack (21, 22) and that tension, irritability and tiredness, were significantly increased in the 2 days before a migraine headache (23). All these subjective and neurophysiological findings might reflect a decrease in cortical activation occurring some hours or even days before the migraine attack. Our study, showing a reduction in nocturnal motor activity during the night preceding the migraine attack, might indirectly support the hypothesis of a decreased cortical activation that is most evident during the night preceding migraine attack and slowly progresses to normality in the following nights.
Further studies should clarify if the observed reduction of motor activity during the nights close to the attack can be directly related to a fluctuation of levels of neurotransmitters; also the identification of the pathophysiology of alterations occurring during the temporal transitions from interictal to ictal cortical states could be an interesting topic for further research.
