Abstract

We read with great interest International Headache Society (IHS) global practice recommendations for the acute pharmacological treatment of migraine. 1 Based on available guidelines and expert consensus, the IHS has developed an optimal recommendation and an essential one for each of the seventeen questions outlined from the treatment of acute migraine, aiming to improve the quality of acute migraine treatment worldwide. However, we would like to raise several issues concerning these recommendations and provide our consideration on them.
According to the Guidelines of the International Headache Society for controlled trials of acute treatment of migraine attacks in adults: Fourth edition, 2 and several national and regional guidelines (Table 1), the definition of Effective Treatment of a migraine attack involves achieving pain free, headache relief (from severe/moderate to mild or no headache) or freedom from the most bothersome associated symptom (MBS) two hours after the intake of the drug,3–6 although the IHS accepted that “headache relief” should be used only as a secondary endpoint to facilitate comparison of the result of new clinical trials with those of previous programs. 2 Treatment failure is indicated if the above definitions are not matched.
Summary of statements on effective treatment of a migraine attack in several national and regional guidelines and other guidance documents

Headache is subjective, and for the assessment of headache intensity, guidelines of the IHS recommended “Headache intensity should be measured on a four-point scale where 0 = no headache; 1 = mild headache; 2 = moderate headache; and 3 = severe headache. Alternatively, a 100-mm visual analogue scale (VAS) or an 11-point numerical rating scale (NRS) can be used”. 2 The 100-mm VAS and 11-point NRS were also commonly used to calculate the percentage reduction of pain intensity to evaluate the efficacy of pain treatment.7,8 Similarly, on a four-point scale, “headache relief” can be a reduction in headache intensity from 3 points (severe headache) or 2 points (moderate headache) to 1 point (mild headache) or 0 points (no headache), which is a reduction in headache intensity of 67%, 100% or 50%, 100% (expressed as percentages) (Figure 1(a)). Therefore, a reduction in headache intensity of ≥50% (headache relief) could be considered as one of the Effective Treatment criteria for a migraine attack.
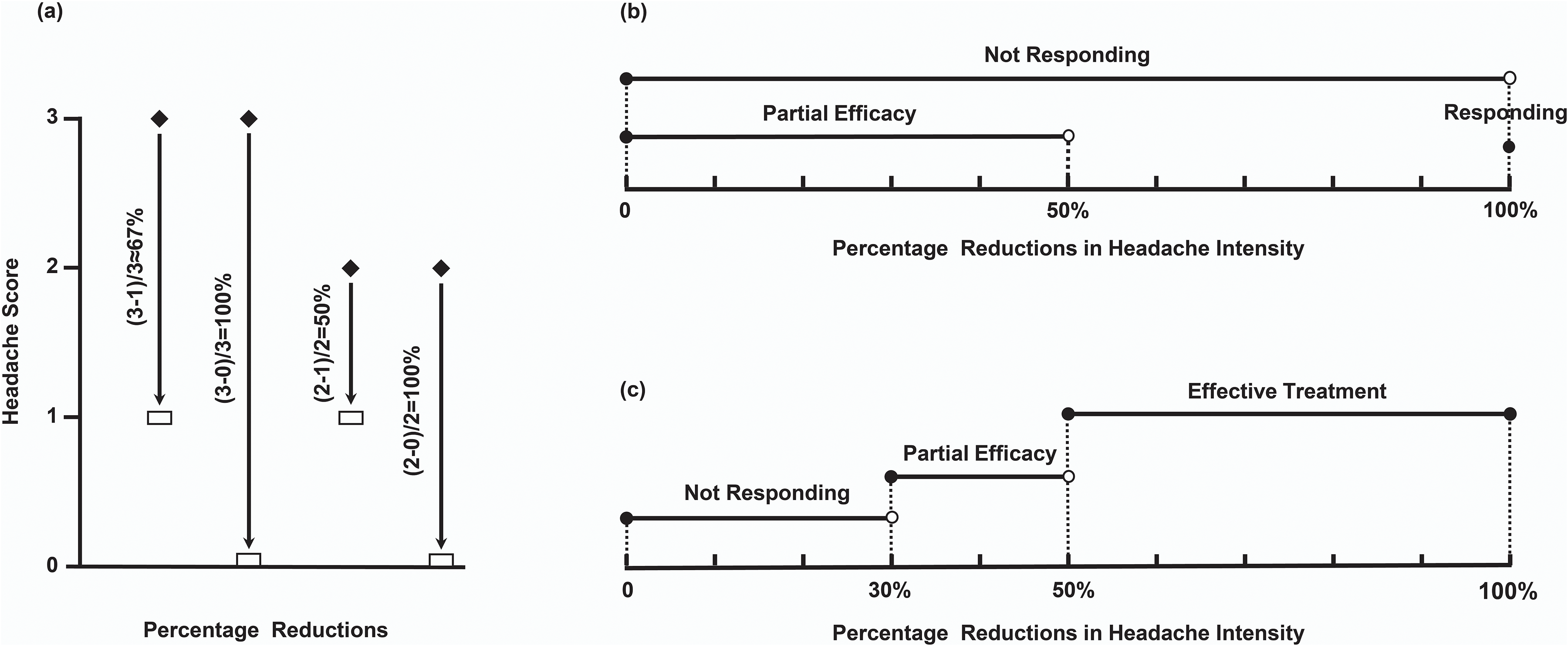
Schematic representations of the percentage reductions in headache intensity. (a) Percentage reductions in headache intensity on the four-point scale for “headache relief” as defined in the guidelines of the International Headache Society (IHS) for controlled trials of acute treatment of migraine attacks. (b) Percentage reductions in headache intensity for “Not Responding” and “Partial Efficacy” as defined in the IHS practical recommendations. (c) Percentage reductions in headache intensity for widely accepted “Effective Treatment”, and “Not Responding” and “Partial Efficacy” of our recommendation.
The IHS practice recommendations 1 used “not responding” instead of “failure”, defining it as “not achieving pain-free two hours after the intake of the drug”, which denied that a reduction in headache intensity of ≥50% is effective for migraine attacks. Different from the previous binary classification of “effective” and “failure”, this IHS practice recommendations 1 proposed the term “Partial Efficacy” and defined it as “not achieving pain relief two hours after the intake of the drug”, which we strongly agree with. But the definitions of Not Responding and Partial Efficacy maybe overlap (Figure 1(b)).
Thus, to distinguish Effective Treatment, Partial Efficacy and Not Responding, more precise definitions are needed. We recommend defining Effective Treatment as a ≥ 50% reduction in headache intensity or satisfactory patient-reported response, Partial Efficacy as not achieving a 50% reduction and Not Responding as not achieving a 30% reduction or unsatisfactory patient-reported response two hours after the intake of the drug (Figure 1(c)).
These cutoffs of our proposal are arbitrary. According to the consensus statement from the Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials (IMMPACT), a reduction of 10–20% in the percentage of pain intensity difference (PercentPID) was considered to be the minimally important change, with a 30% reduction corresponding to moderate relief, and a 50% reduction indicating substantial relief. 9 Although pain intensity difference (PID) and PercentPID are rarely used in trials of acute migraine treatment, they were considered secondary endpoints and yielded results that were similar to those on a four-point scale used in trials, particularly when assessing acute treatments for mild pain. 2 Recently, IHS recommended “a ≥ 50% decrease in monthly migraine days or moderate-to-severe headache days” as one of the success criteria and “Less than 30% reduction in monthly migraine days or moderate-to-severe headache days” as an ineffective criterion for pharmacological preventive treatment of migraine. 10 Drawing on the above, the cutoff values of 30% and 50% that we suggest may be set to 50% and 75% or others in the future, which needs to be confirmed by more clinical trials.
We are in full agreement with “In people with severe migraine attacks, triptans may be more effective than non-steroidal anti-inflammatory drugs and therefore triptans can be used as the first line treatment”. 1 But, for certain patients, particularly those showing a partial response to non-steroidal anti-inflammatory drugs (NSAIDs) or analgesics, the IHS practice recommendations provided no specific recommendation. 1 Diener et al. 11 have found that combination analgesics were more effective than either of these drugs given as monotherapy through drug trials. Also, a “strong for” recommendation for aspirin-acetaminophen-caffeine was newly added for the short-term treatment of migraine by the US Department of Veterans Affairs and US Department of Defense Clinical Practice Guideline. 12 When NSAIDs or individual analgesics show only Partial Efficacy, we recommend the use of a combination analgesic such as a fixed combination of acetylsalicylic acid, acetaminophen and caffeine. This suggestion may be listed as an essential one, given that it provides therapeutic alternatives for certain countries or regions where pharmacological options remain limited.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
