Abstract
Objective
To describe longitudinal patterns of medication use throughout pregnancy in women with migraine.
Methods
We used the IBM MarketScan healthcare claims database in the US to create a cohort of pregnancies enrolled between 2011–2015 resulting in live or stillbirth. Migraine headache was identified based on ICD-9-CM diagnosis codes or procedure codes recorded in clinical encounters. Outcomes were patterns of prescriptions filled for medications that may be used to prevent migraine (antiepileptics, antihypertensives, antidepressants) or treat acute episodes (opioids, triptans, acetaminophen) and of other comorbid conditions (hypertension, psychiatric diagnoses, epilepsy). We used group-based multi-trajectory models to cluster women into similar longitudinal patterns of prescription fills.
Results
Of 859,501 pregnancies, 8168 had migraine. Within migraineurs, before pregnancy, the most commonly filled prescription was for a triptan (43.2%), followed by opioids (26.7%), acetaminophen (26.2%), antidepressants (24.9%), antiepileptics (18.6%) and antihypertensives (12.3%). Antiepileptics, antidepressants, and triptans were frequently discontinued early in pregnancy with few new users, while antihypertensives were discontinued by some users, but continued or initiated by a minority of users late in pregnancy. Opioids and acetaminophen were used intermittently throughout pregnancy. Comorbidities included hypertension (10.8%), epilepsy (4.7%), depression (14.0%), and anxiety (15.6%). Polypharmacy involving both preventive and acute medications was most common before pregnancy (31.4%) and declined in first trimester (14.7%). In all, 25.9% of women filled prescriptions for two or more acute medications before pregnancy.
Conclusions
Medication use patterns during pregnancy for women with migraine are complex. Patterns of polypharmacy and comorbidity during pregnancy highlight an under-studied area relevant for maternal and child health outcomes.
Introduction
Migraine is a debilitating neurological condition (1) that affects 25–30% of women of reproductive age (2). During pregnancy, migraine severity decreases after the first trimester in about two thirds of women, but persists unchanged or worsens for many others (3), and about 80% of women with migraine have at least some disease activity during pregnancy (4). The scope of pharmacologic treatment for migraine during pregnancy is not well documented, but because many of the most effective drugs for migraine treatment are either contraindicated in pregnancy or have uncertain safety profiles (5–12), uncertainty around medication safety during pregnancy may lead to discontinuation of effective medications, or switching to non-migraine-specific therapies that are associated with other risks (13).
Treatment decision making is further complicated by the lack of safety data when medications are combined. Polypharmacy in migraineurs may be specific to treatment of migraine itself or may be related to treatment of both migraine and other comorbidities, but the scope of polypharmacy during pregnancy is unknown. Nor do we know how starting, stopping, or switching migraine medications during pregnancy affects maternal and fetal outcomes. Until we can identify common patterns of concomitant medication use and understand their interaction with comorbidities, we will not be able to implement real world safety studies in pregnant women with migraine.
To evaluate pharmacotherapy in pregnant migraineurs, we i) describe the prevalence of multiple prescriptions for migraine medications throughout pregnancy; ii) use group-based multitrajectory modelling (14) to identify patterns of prescription filling for common classes of drugs used to treat migraine; and iii) descriptively examine relationships between comorbidities and multidrug trajectories.
Methods
Data source and cohort creation
Our study population came from the IBM MarketScan healthcare claims database in the US between 2011 and 2015. We identified a cohort of women aged 12–55 who were pregnant during our study period and had a live born or stillborn infant. Gestational age was ascertained using validated algorithms (15). Women were required to have continuous enrolment in their insurance plan from 90 days before estimated last menstrual period (LMP), through the end of pregnancy, and to have prescription drug coverage for the same period. Estimated LMP was determined by subtracting the gestational length from the end date of the pregnancy. Additional cohort details, including classifications of pregnancy type and estimation of gestational length, have been previously described (16).
We defined migraine using an algorithm that prioritized specificity. Migraine cases met one of four criteria: i) Two or more International Classification of Disease 9 Current Modification (ICD-9-CM) codes 346.xx (migraine) in the pre-pregnancy period; ii) one ICD-9-CM code for migraine before pregnancy AND one triptan prescription during the study period; iii) one ICD-9-CM code for migraine before pregnancy AND one neurology encounter; or iv) two or more ICD-9-CM codes for migraine at any time AND one neurology encounter. Details and sample size for inclusion criteria are outlined in Table 1. Alternate cohort definitions considered as sensitivity analyses included a) not considering triptan prescription as an inclusion criterion, b) requiring at least one code for migraine be assigned during a neurology visit, and c) including all women with any ICD-9-CM code for migraine present before pregnancy; alternate definitions are included in Supplemental Table 1.
Eligibility criteria for migraine pregnancy cohort.

Medication use
We searched outpatient claims for the generic names of drugs used in the treatment of migraine, including preventive medications (antiepileptic, antihypertensive, and antidepressant medications) and acute medications (triptans, opioids, and acetaminophen); acetaminophen and opioids were present in both single- and multi-drug formulations. Supplemental Table 2 has a full list of included medications. We captured dispensing during four intervals: Pre-pregnancy baseline ([LMP-90 days]–[LMP-1 day]), first trimester (LMP–[LMP+90]), second trimester ([LMP + 91 days]–[LMP +180 days]), and third trimester ([LMP + 181 days]–[end of pregnancy]). We defined polypharmacy as filling two or more prescriptions during the same interval for distinct classes of medications that may be used for migraine (e.g. a triptan and an antihypertensive during first trimester; see Supplemental Table 3). We evaluated polypharmacy for preventive medications, acute medications, and preventive/acute combinations.
Maternal characteristics
Maternal characteristics were coded using ICD-9-CM and Current Procedural Terminology (CPT) codes. Comorbid conditions including hypertension, epilepsy, depression, anxiety, diabetes, obesity, smoking and other pain conditions (i.e. arthritis, musculoskeletal pain) were coded from inpatient and outpatient claims, during the pre-pregnancy baseline and the full study period. We also captured information on healthcare utilization, including prenatal care, contact with a neurologist, and emergency medicine encounters where migraine was the main presenting condition (Supplemental Table 4).
Statistical analysis
We used group-based trajectory modelling (GBTM) and group-based multi-trajectory modelling (GBMTM), an unsupervised clustering method based on finite mixture models, to identify groups of women with similar patterns of prescription fills from the pre-pregnancy baseline period throughout the course of pregnancy. The goal of our analysis was to identify a) single drug trajectory models using GBTM for characterizing longitudinal patterns of use for a single medication, and b) multiple drug trajectory models using GBMTM for characterizing longitudinal patterns of prescription fills for up to six medications.
For single- and multi-drug models, analysis followed the same series of steps to select the model or models with i) the optimal number of groups and ii) adequate fit based on shape of group trajectories. Model building began with fitting models with group numbers ranging from 2–5 (single drug GBTM) and 4–6 (multiple drug GBMTM) with all linear trajectory shapes, and examining model fit, trajectory plots and mean posterior probability of group membership; group numbers were adjusted and model fitting proceeded iteratively. Model selection was based on the Bayesian information criterion (BIC), in which the model with the highest (least negative) BIC value is preferred. The Bayes factor, which is the odds of one model (i) being the correct model compared to another model (j), was used to determine meaningful differences in BIC values. The Bayes factor is approximated by eBICi – BICj, and a large Bayes factor was considered stronger evidence for a difference between models.
To select between models with different group trajectory shapes (0 = constant, 1 = linear, 2 = quadratic, 3 = cubic), we assessed model adequacy based on a) estimated and assigned group proportions, b) average posterior probability of group membership, and c) odds of correct classification. An adequate model is one in which the proportion assigned to each trajectory group (based on the maximum posterior probability rule) is similar to the estimated group proportion; the average posterior probability of group membership is at least 0.7 for all groups, and the odds of correct classification (the ratio of the odds of correct classification based on the average posterior probability of group membership and the odds of correct classification based on random assignment) should be at least 5 for all groups.
Because of the large number of possible models, it is not possible to exhaustively test all possible combinations of group number and trajectory shape. From a set of models that were essentially statistically equivalent, we selected final models based on their qualitative clinical relevance and/or interpretability of results. The final model choice was made in consultation with a neurologist specializing in migraine (RB). Statistical details of GBTM (14) and GBMTM (17), including guidelines for selecting a final model (18), have been previously published.
We used alluvial plots to visualize the frequency of prescription fills over time and examined the clinical characteristics and healthcare utilization associated with membership in each group identified by the final multi-drug GBMTM model. Sensitivity analyses examined clinical characteristics, prescription fills, and performance of the best GBTM and GBMTM models under alternate cohort definitions, and characteristics of groups identified from alternate models. GBTM and GBMTM was carried out using PROC TRAJ in SAS (14). Figures were constructed using the ggplot2 and ggalluvial packages in R/RStudio.
Standard protocol approvals, registrations, and patient consents
This study received ethics board approval from the Harvard T.H. Chan School of Public Health (Boston, Massachusetts).
Data availability
Data from the IBM MarketScan healthcare claims database are publicly available and can be purchased from IBM via data use agreements. Information on algorithms used to create the cohort is available from the authors upon request.
Results
Sample characteristics
Of the 859,501 pregnancies that met inclusion criteria, 8168 met criteria for migraine and were included in further analyses. Common comorbidities included hypertension (10.8%), obesity (10.7%), smoking (5.8%), epilepsy (4.7%), depression (14.0%), anxiety (15.6%), and other pain conditions (54.6% with musculoskeletal pain). In all, 17% had at least one emergency room visit for migraine. Characteristics of women with and without migraine (main definition) are found in Supplemental Table 5. Alternate cohort definitions resulted in a migraine cohort with broadly similar characteristics (Supplemental Table 6).
Preventive and acute migraine therapy
Before pregnancy, the most common prescription among the medications we examined was for a triptan (43.2%), followed by opioids (26.7%), acetaminophen (26.2%), antidepressants (24.9%), antiepileptic drugs (18.6%) and antihypertensives (12.3%). The percent of women filling a prescription for each medication at each time point, and the changes in percentages over time are shown in Figure 1 (see Supplemental Table 7 for prescription fills under alternate migraine definitions). Longitudinal patterns in the alluvial diagrams suggest these drugs can be conceptually divided into three main categories. Antiepileptics, antidepressants, and triptans (Figure 1(b), (c), and (d)) are rapidly discontinued at pregnancy onset and acquire few new users during pregnancy. Antihypertensives (Figure 1(a)) are discontinued by some users, but use persists for a subset of women throughout pregnancy, and some new users begin treatment late in pregnancy. Opioids and acetaminophen (Figure 1(e) and (f)) appear to be used intermittently before and during pregnancy, with no apparent rapid discontinuation.

Longitudinal patterns of prescription fills, among ever-users of a particular medication class. Alluvial diagrams for filling a prescription for (a) antihypertensives, (b) antiepileptics, (c) antidepressants, (d) triptans, (e) opioids, and (f) acetaminophen during the 90 days before the last menstrual period (pre-LMP), and the first, second, and third trimesters. Proportions are for the full migraine cohort, but figures exclude women who never filled a prescription for the medication of interest during the study period.
The single drug GBTMs shown in Figure 2 provide similar information to the alluvial diagrams. For single trajectory models, we were unable to find a five-group solution or result for antidepressants, and although five-group solutions converged for other drugs, these models generally fit poorly and had at least one group with very few members, while three- and four-group solutions had adequate fit for all drug classes (Supplemental Table 8). Based on the best models, antihypertensive users (Figure 2(a)) are grouped into never-users, persistent users, discontinuers, and new users, while antidepressant and antiepileptic users are characterized as never users, persistent users, and discontinuers (Figure 2(b) and (c)). Triptan groups are differentiated by how quickly they discontinue at pregnancy onset (Figure 2(d)). However, models for both opioid and acetaminophen prescription fills show a small group of high probability persistent users (Figure 2(e) and (f)); the acetaminophen model additionally shows a group with increasing probability over the course of pregnancy (Figure 2(f)).
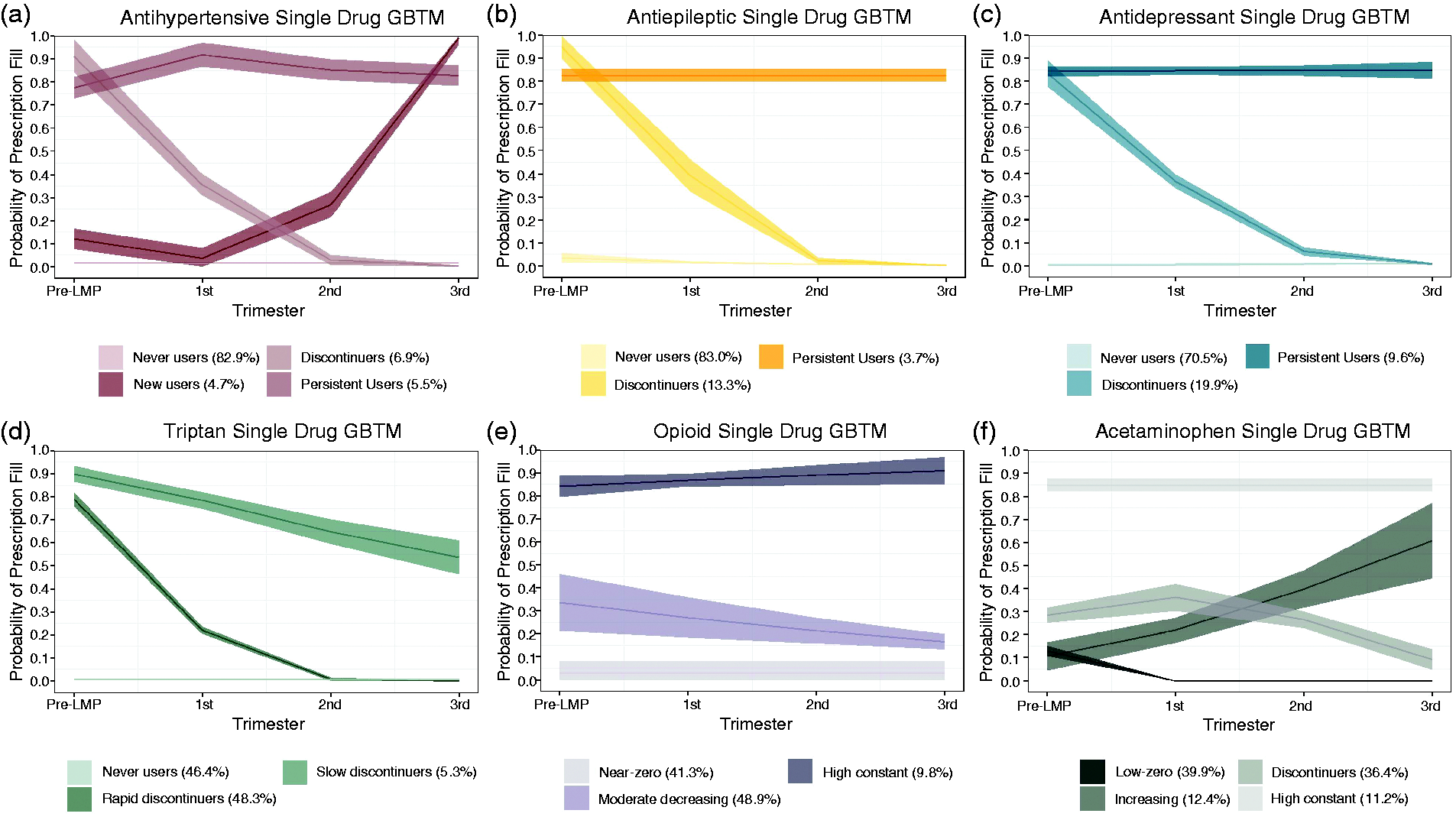
Groups identified from best single drug group-based trajectory models. Predicted probabilities with 95% confidence intervals filling a prescription for (a) antihypertensives, (b) antiepileptics, (c) antidepressants, (d) triptans, (e) opioids, and (f) acetaminophen during the 90 days before the last menstrual period (pre-LMP), and the first, second, and third trimesters. Probabilities with confidence intervals are based on results from single-drug group-based trajectory models (GBTM).
Polypharmacy before and during pregnancy
We examined the proportion of women with migraine with prescriptions for multiple medication classes during the same interval. Polypharmacy involving both preventive and acute medications was most common before pregnancy (31.4%) and declined at pregnancy onset (14.7% in the first trimester) with subsequent decreases to 8.9% and 7.8% in the second and third trimesters, respectively. In all, 25.9% of women filled prescriptions for two or more acute medications before LMP, with a slight decrease over the course of pregnancy (21.0% in the first trimester, 18.9% in the second, 16.3% in the third), while use of multiple preventives decreased sharply at pregnancy onset (12.2% pre-LMP to 6.3% in the first trimester) but remained stable during the second and third trimesters (2.7 and 3.0%).
Before pregnancy, antidepressants were more commonly combined with triptans (12.5%); during the first trimester, the most commonly combined preventive and acute classes were antidepressants and acetaminophen (6.3%). As pregnancy progressed, antidepressants continued to decline, and by the third trimester, antihypertensives in combination with acetaminophen (3.7%) or opioids (3.3%) were marginally more common than antidepressants or antiepileptics in combination with any acute medications. Combinations of antihypertensives with antiepileptics were most common before and early in pregnancy (6.2 and 2.9%) before decreasing later (<1%), while combinations of antihypertensives with antidepressants were more common during the second (1.3%) and third (1.6%) trimesters. Opioids and acetaminophen were the most commonly combined acute medications for all intervals and increased from before pregnancy (12.2%) to first and second trimesters (13.9% and 16.5%). Additional details on the frequency of various types of polypharmacy, including specific combinations of medications, is included in Table 2.
Polypharmacy in medications used to treat migraine: number filling prescriptions for multiple medication classes before and during pregnancy

1Numbers are n (%); cell counts less than 5 are reported as “< 5” to preserve anonymity.
2Includes individuals with zero prescription fills, fills for only one class of medication, and combinations of only acute or only preventive medications.
3Combinations included at least one acute and at least one preventive medication.
4Combinations of acute + preventive drugs may add to > 100% (groups not mutually exclusive).
Multitrajectory models
We used the results of the final six group multi-drug GBMTM (model fit statistics included in Supplemental Table 9), along with the associated proportions of prescription fills (Figure 3) and clinical characteristics (Table 3) to characterize each trajectory group. Group 3, the largest group (40.1%), overwhelmingly used triptans before pregnancy with minimal use of preventives or prescription analgesics; they also had lower levels of other comorbidities and less intensive use of healthcare, suggesting this group can be referred to as “low severity/comorbidity”. Group 1, comprising 16.6% of the sample, had high levels of acute treatments rapidly discontinued at pregnancy onset, and high initial antiepileptic use with most but not all discontinuing, as well as the highest recorded epilepsy prevalence; we labelled this the “moderate severity” group. Group 2 (5.2%), dubbed the “cardiovascular comorbidity” group, had high persistent use of antihypertensives throughout pregnancy and also moderate persistent analgesic use, along with the highest levels of hypertension, diabetes, and obesity. Group 4 (18.9%) had similar prescription fill patterns to the low severity/comorbidity group, except that they increasingly filled prescriptions for analgesics as pregnancy progressed, and had more recorded pain diagnoses in addition to migraine, as well as more emergency room visits for migraine-related reasons; we termed them the “pain comorbidity” group. Group 5 (9.5%), the “psychiatric comorbidity” group, had higher persistent antidepressant use, and higher prevalence of comorbid depression and anxiety. Group 6 (9.7%) had the highest persistent use of analgesics, and high levels of preventive therapies and triptans prior to pregnancy, with slower discontinuation than in other groups; they also had higher levels of many comorbidities and more intense use of healthcare, suggesting a heterogeneous “medically complex” group.
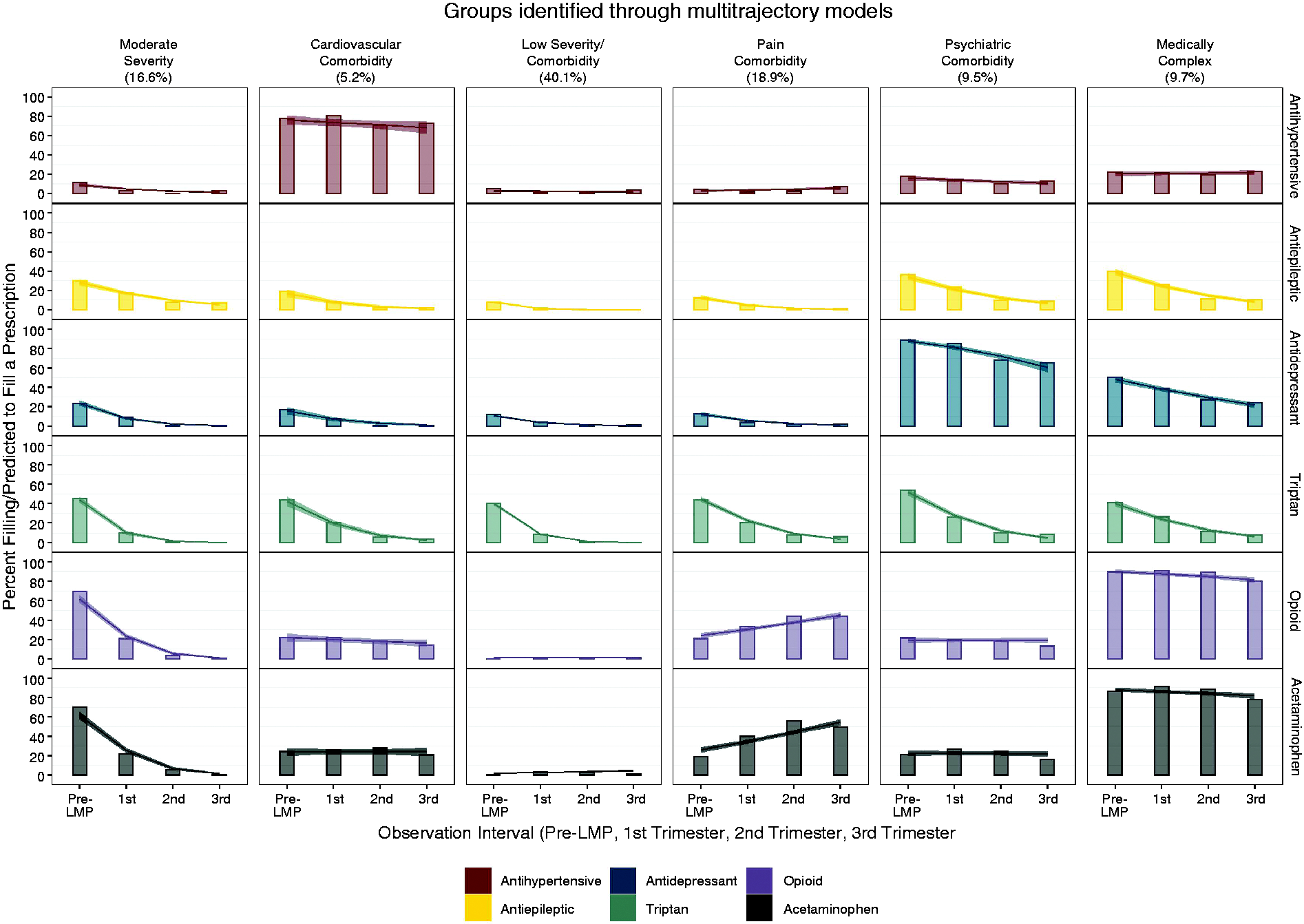
Observed and predicted prescription fills for groups identified from the best multi-drug group-based multi-trajectory model. For the final six drug group-based multi-trajectory model, bar plots show the observed percent of prescription fills for each medication, while the lines with 95% confidence intervals show the predicted probability of filling a prescription, ordered by trimester, within trajectory group (vertical columns) and medication class (horizontal rows)
Maternal characteristics, comorbidities, and healthcare utilization for trajectory groups from group-based multi-trajectory model.

LMP: last menstrual period.
1Numbers are n (%); cell counts less than 5 are reported as “< 5” to preserve anonymity.
Sensitivity analyses
We compared the characteristics of the groups identified by the six-group model to those of the five-group model (Supplemental Table 10). Using the six-group model as the reference standard, the low severity, pain comorbidity, psychiatric comorbidity, and medically complex groups all retained more than 93% of their constituents. The cardiovascular comorbidity group retained only 84% and the moderate severity group was more diffuse, with 52% of constituents persisting together and the remaining 48% distributed across psychiatric, cardiovascular, and low severity groups. Examining prescription fills and clinical characteristics suggests selecting the five-group rather than the six-group solution would have resulted in a low severity group that included a number of observations previously categorized as moderate severity, and a much more heterogeneous group comprising elements of the cardiovascular and pain groups (Supplemental Table 10). Additional sensitivity analyses in which the best-performing single drug GBTM and multiple-drug GBMTM were re-run under alternate cohort definitions produced results similar to those reported in the main analyses (Supplemental Tables 11–16).
Discussion
In this cohort of women with migraine, one in every five showed evidence of first trimester polypharmacy: Use of multiple acute medications was most common, followed by combined acute-preventive therapy, with preventive polytherapy occurring less often. The most frequent combinations were antidepressants with antiepileptics, triptans, or non-migraine-specific analgesics. We were able to identify groups with distinct longitudinal patterns of medication use during pregnancy, largely distinguished by patterns of preventive medication use and corresponding comorbidities. While many medications are reduced or discontinued at pregnancy onset, we observed groups that persistently used antiepileptics, antihypertensives, or antidepressants, or had an increased reliance on analgesics. Although the largest single group was characterized by a lack of significant comorbidities and low longitudinal medication use, most of our sample comprised women with a more complicated clinical presentation.
Identification of longitudinal patterns of medication use during pregnancy is important for several reasons. The precautionary principle has resulted in the widespread practice of recommending discontinuation of most medications during pregnancy, including certain classes of preventive medications (e.g. antidepressants, antihypertensives) that could be associated with improved maternal and fetal outcomes. For example, despite an extensive literature on migraine during pregnancy and cardiovascular disease (19–21), serious consideration of the role of treatment is lacking. Likewise, comparative safety studies are needed on different strategies for acute migraine treatments during pregnancy, such as the practice of discontinuing triptans in favor of opioids.
As expected, use of medications before and during pregnancy was considerably higher in our migraine study sample than the general population of pregnant women. During the first trimester, preventive medication use ranged from 10–16%; other studies in similar populations not limited to specific diagnoses noted much lower prevalence (22–24), as did a study among pregnant women with migraine (25). However, the overall pattern of pharmacotherapy discontinuation in our study, with greater discontinuation for preventive drugs and less for acute treatments other than triptans, is consistent with many drug utilization studies in pregnancy in both the US (26,27) and Europe (28).
Our results suggest that recommendations for discontinuing many migraine-specific medications, including triptans and preventive drugs that are effective against migraine, may drive increases in other drugs perceived to be safer. More than one group in our study showed evidence of continued or increasing reliance on non-specific analgesics rather than migraine-specific therapies during pregnancy. While acetaminophen continues to be the preferred acute therapy for migraine in pregnancy (29), switching from a migraine-specific treatment to a non-specific treatment such as acetaminophen has been associated with increased self-reported pain (13). This issue deserves additional scrutiny, given the increased risk for neonatal abstinence syndrome associated with prenatal opioid exposure (30) and recent evidence suggesting an increased risk of behavioral problems in children with prolonged prenatal exposure to acetaminophen (31,32).
It is important to interpret the groups identified through group-based trajectory modelling with caution. Methods for identifying latent or unobserved groups are prone to over-extraction, meaning we risk classifying our sample into distinct groups when the underlying distribution is truly homogenous (33): We would, in some sense, invent subclasses of migraineurs. While several of the groups were retained through multiple sensitivity analyses, others were not, and the group we labelled “moderate severity” was particularly unstable. Given the complexity of migraine and its clinical heterogeneity (34), we believe the groups identified in our main analysis provide valuable additional insight; however, we caution against treating the groups as “real”.
This study has several limitations. Because our study relies on administrative claims, the trajectories we have identified are for prescription filling, not necessarily for using the medications in question. Studies comparing self-reported medication use in pregnancy to prescription records have noted wide variability in the validity of these data, with chronic medications such as antidepressants being very reliably recorded, but acute/occasional use drugs like acetaminophen being substantially less valid (35,36). In addition, our claims-based study was unable to link prescriptions to specific diagnoses. With the exception of triptans, medications used to treat migraine are also used to treat other disorders, including epilepsy, cardiovascular disease, psychiatric diagnoses, and a range of pain conditions, and it is unclear whether many of the medications included in our study were prescribed specifically for migraine, for other comorbid conditions, or both. Differences in comorbidity profiles between groups indicate that some degree of preventive medication use is likely due to treatment of conditions such as hypertension or depression. Regardless of indication, however, these prescription-fill trajectories likely represent exposure in our population of interest.
Our study population was a sample of commercially insured women in the US, meaning that either the woman or her spouse or partner obtained insurance through their employers, and remained enrolled throughout the study period. Although this sample is likely representative of women with migraine actively seeking care, there are differences in sociodemographic and economic characteristics between commercially insured, publicly insured, and uninsured women, and it is possible we would have observed different trajectory groups if our study included all pregnant women in the US. It is possible we would see different results in samples outside the US: While the ICD-9/10 coding system is used internationally, formularies and prescribing preferences differ before and during pregnancy, and replications in other samples would be desirable. We deliberately defined migraine in our sample to prioritize specificity rather than sensitivity and have therefore likely missed true cases of migraine in the larger sample. Because this definition of migraine relies on characteristics resulting from intensive contact with the healthcare system, our sample likely over-represents the most active and/or severe cases of migraine. In a sensitivity analysis with a less strictly defined migraine sample, we observed lower prevalence of medication use and different relative proportions of comorbidities, although the results were broadly comparable to the main analysis. As such, the patterns identified in the main analysis may be most relevant for clinicians treating a more severe and chronic migraine patient population. Despite these limitations, our study has important strengths. Using group-based multi-trajectory modelling, we highlighted clinically heterogeneous groups of pregnant women within a strict a priori definition of migraine. Selection of the final trajectories in our study was done in consultation with a clinician with expertise in the treatment of migraine in pregnancy. While we do not suggest that these trajectories are objective exposure groups or migraine subtypes, this method allowed us to distil high-dimensional data into important clusters of exposure patterns, which may be used to define dynamic or time-varying treatment regimes (37–39) in future studies. Our study also underlines the importance of evaluating the safety of different types of medications in combination, which is a clinically relevant but poorly studied research area. By demonstrating how the use and discontinuation of multiple medications interact, our study informs future directions for research into migraine treatment during pregnancy. Lastly, most drug utilization studies in pregnancy focus on the use of a particular drug or drug class in the general pregnant patient population; by contrast, our study examines the more clinically relevant question of polypharmacy in pregnant women with migraine.
Conclusion
Observational studies are critical for studying the safety of medication use during pregnancy (40,41). Using a large, administrative database, we showed that polypharmacy is present in one out of every three women with migraine who become pregnant and is a highly understudied condition. At the same time, concern over the safety of medications during pregnancy may result in avoidance of effective migraine-specific treatments and reliance on less effective medications. This study identifies a critical need for an increased focus on migraine in pregnancy, including its clinical comorbidities and pharmacological management, as an opportunity for improving maternal and child health.
Clinical implications
Pregnant migraineurs are a clinically heterogeneous group, with one in five characterized by polypharmacy and many more by comorbid conditions. Two out of every three pregnant women with migraine were characterized by persistent longitudinal patterns of preventive or acute medication use, increased prevalence of psychiatric, hypertensive, or pain comorbidities, or a combination. This study highlights an under-studied area of maternal health with important implications for future research priorities and clinical care.
Supplemental Material
sj-pdf-1-cep-10.1177_0333102420975394 - Supplemental material for Polypharmacy and comorbidities during pregnancy in a cohort of women with migraine
Supplemental material, sj-pdf-1-cep-10.1177_0333102420975394 for Polypharmacy and comorbidities during pregnancy in a cohort of women with migraine by Mollie E Wood, Rebecca C Burch and Sonia Hernandez-Diaz in Cephalalgia
Footnotes
Acknowledgements
The authors wish to thank Donald Halstead for his comments on an earlier version of this manuscript.
Author note
Statistical analyses were carried out by Mollie Wood.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MW was funded by a grant from the National Heart Lung and Blood Institute (T32 HL098048). She has no financial or other conflicts of interest.
SH-D reports being an investigator in research grants to her institution from GSK, Eli Lilly, and Takeda, and has consulted for Roche and UCB.
RB receives compensation for her work as an associate editor for Neurology. She has no other financial conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Mollie Wood was funded by NHLBI (T32 HL098048)
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
