Abstract
Background
Individuals with autism spectrum disorder (ASD) experience a wide array of neurological, psychiatric and medical comorbidities, yet little attention has been given to the potential link between ASD and migraine, one of the most prevalent neurological disorders worldwide. This study aimed to investigate whether a genetic predisposition for ASD is linked to migraine and its major subtypes, with and without aura. Additionally, potential moderator and mediators of the association between ASD and migraine were explored.
Methods
Polygenic scores (PGS) for ASD were constructed based on the genome-wide association study by the Psychiatric Genomics Consortium, on the UK Biobank cohort dataset comprising 337,386 participants using PRSice-2. Regression analyses were performed to investigate the association of ASD PGS with migraine and its major subtypes, with and without aura. Sex was explored as a potential moderating factor. The mediation analyses took into consideration variables such as education, personality trait neuroticism, body mass index (BMI) and four categories of comorbidities (psychiatric, vascular, neurologic and others).
Results
ASD PGS were significantly and positively associated with migraine (odds ratio (OR) = 1.04, 95% confidence interval (CI) = 1.02–1.05, p < 0.002), migraine without aura (OR = 1.05, 95% CI = 1.02–1.07, p < 0.002) and migraine with aura (OR = 1.05, 95% CI = 1.02–1.07, p < 0.002). No moderating effect of sex on the association between ASD PGS and migraine was observed. As for potential mediators, only the personality trait neuroticism significantly mediated the association between ASD PGS and migraine, with the proportion of effect mediated 8.75% (95% CI = 4–18%).
Conclusions
Our study suggests that individuals genetically predisposed to autism are at higher risk of experiencing migraine, including the two major subtypes, with and without aura. While emphasizing the complex shared genetic and pathophysiological interactions of these conditions, the role of personality trait neuroticism as a mediator of this relationship is highlighted.
This is a visual representation of the abstract.
Introduction
Autism spectrum disorder (ASD) is a group of complex neurodevelopmental disorders with lifelong persistence and is characterized by difficulties in social communication, and the presence of sensory alterations and/or patterns of restricted interests and repetitive behaviours (1). Approximately one in 100 children are diagnosed with ASD globally (2) and present with a broad range of co-morbidities or co-existing conditions (3). Migraine is the most common neurological condition and the most disabling disorder, affecting about one billion people globally (4). While these two conditions share common pathophysiological changes, such as neurotransmission dysregulation, particularly in the serotoninergic system; altered immune responses leading to neurogenic neuroinflammation; abnormalities in cortical minicolumn organization; and a dysfunctional gut-brain axis, along with shared susceptibility genes (5), population genetics-based investigation remains underexplored.
There are only a handful of studies that have explored the association between autism and migraine (6,–8) and these are limited in small sample sizes and provide inconclusive evidence. A comparatively large national level study conducted in Taiwan found that children and adolescents diagnosed with ASD have a significantly higher risk of developing migraine compared to those without ASD (9). A recent meta-analysis and systematic review by Pan et al. (10) investigating neurological disorders in ASD identified associations between migraine (including headaches) and ASD in nine studies. Among these, only three studies focused specifically on migraine diagnoses, one study on children (11) found a higher prevalence of migraine, another one on the transitional age of 14–25 years (12) reported a significantly less common prevalence of migraine, while one study on adults (13) reported no significant association with migraine.
Studies focusing on the relationship between ASD and migraine are mostly limited to children or adolescents (7,8,11). There are only a few studies in adults that have examined the association between ASD and migraine and yielded contradictory findings (6,13). In the meta-analyses by Pan et al. (10), no significant association of either headache or migraine with autism was observed when analyzed separately in adults. These findings underscore the need for further and more comprehensive investigation into the association between migraine and autism among adults. Furthermore, previous ASD migraine association studies focused mostly on clinical samples but not on a general population level (6,–8). A large population-based study among adults may assess the association between ASD and migraine with more statistical power.
While risk factors, personality traits and a list of psychiatric, vascular, neurologic and other comorbidities have been established and investigated as influential in migraine (14,–17), studies exploring the association between migraine and ASD currently fall short in investigating these connections. Moreover, migraine is two- to three-fold more prevalent among women than in men (18), while the prevalence ratio of ASD between men and women is 4:1 (19). Nevertheless, studies exploring sex-specific differences in migraine–ASD association are rare to find.
Migraine has two major subtypes: with and without aura (20). There exist potential pathophysiological connections between ASD and migraine with aura (sensory disturbance), as well as shared characteristics such as onset in infancy, and suffering a lifelong imprint on cognition, socialization and language development (21,–23). However, very few studies that have explored the association between ASD and migraine with aura, are either based on small clinical samples (7) or focused only on women (24). The epidemiological context and the common genetic susceptibility of ASD and migraine have yet only been partly explored (5). A Recent Brainstorm Consortium report revealed shared genetic heritability between ASD and migraine including both types, with and without aura (25).
Polygenic scores (PGS) are composed of cumulative totals of susceptible genetic mutations (i.e. single nucleotide polymorphisms; SNPs) associated with specific target trait, with each SNP assigned a weight based on its effect size in association with the trait determined by genome-wide association studies (GWAS) (26). ASD PGS are useful predictors for autism diagnosis and associated with traits such as childhood trauma, lifetime self-harm, and suicidal behavior and ideation (27), childhood difficulties in social communication (28), autistic traits (29), cognitive aptitude (30) and everyday executive function (31). Previously, we have used UK Biobank cohort data (comprising approximately 500,000 participants) and successfully applied the PGS approach to investigate the association between the genetic risk for ASD and well-being spectrum traits (32). The present study aimed to investigate the potential association between genetic predisposition for ASD and diagnosed migraine, including its major subtypes (with and without aura), among adults in the UK Biobank cohort, using PSG analysis. Additionally, we aimed to explore the male–female distribution of genetic risk for ASD in relation to migraine. Specifically, in this study, we addressed the following research questions (Figure 1). (i) Is migraine and its major subtypes, with and without aura - associated with a common genetic background for autism? (ii) Does sex moderate the association between ASD PGS and migraine? (iii) Do education, personality trait and general health related variables mediate the effects of autism on migraine?
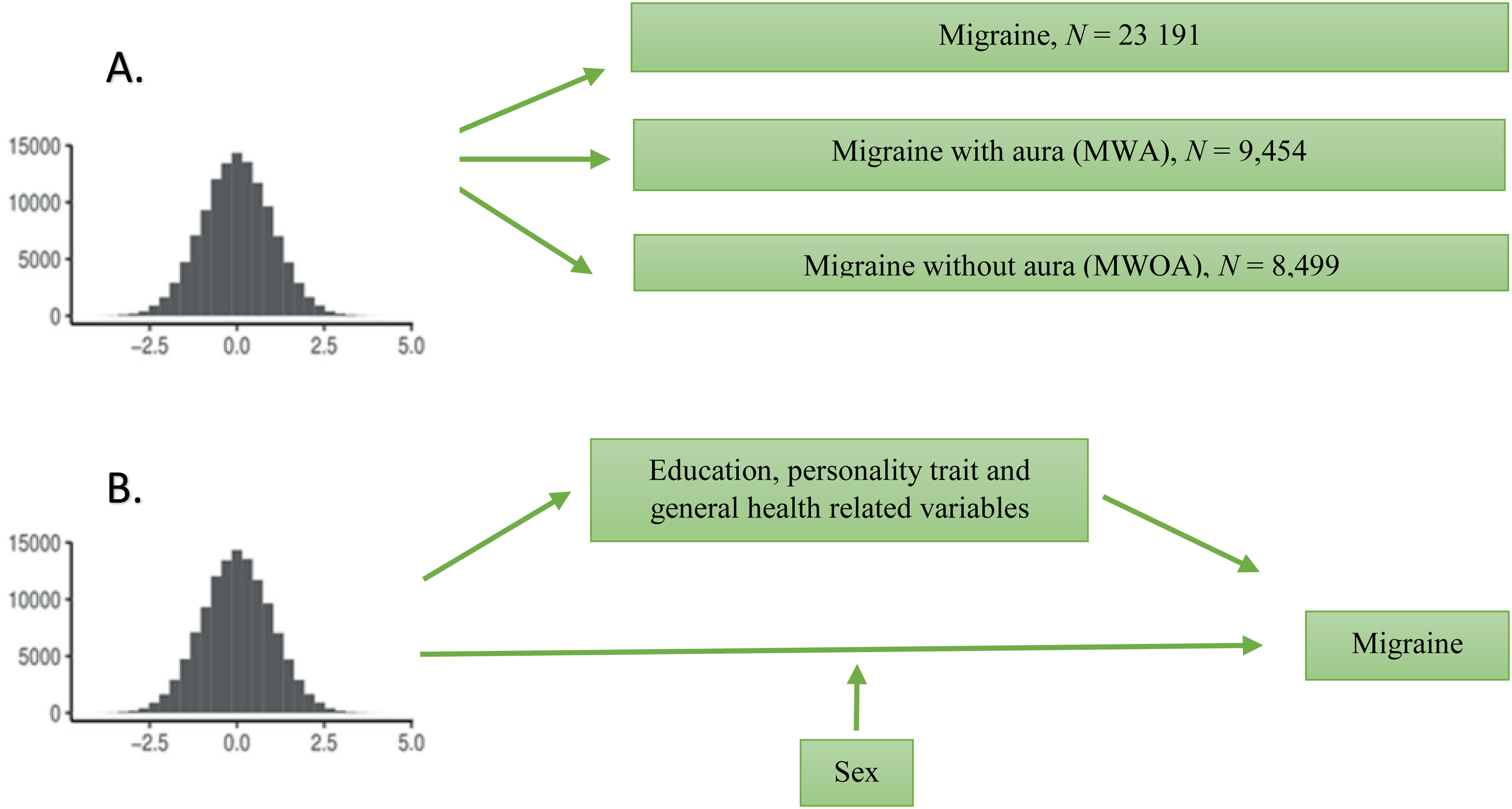
Schematic diagram of the study design. (A) Illustration of the polygenic score (PGS) association analyses. Polygenic scores (PGS) were calculated in the UK Biobank for autism spectrum disorder (ASD) and associations were tested between the PGS and migraine, including its two major subtypes, with and without aura. (B) Illustration of the mediation analyses and moderation analysis performed in the study. Mediation analyses were performed to investigate if education, personality, and general health-related variables (body mass index, psychiatric comorbidities, vascular comorbidities, neurologic comorbidities and other comorbidities) mediate the association between ASD PGS and migraine. Moderation analysis was performed to investigate whether sex influenced the strength of the relationship between ASD PGS and migraine.
Methods
Participants
We utilized baseline data sourced from the UK Biobank (UKB) project, which is among the largest prospective population-based cohorts in Europe. Participants aged 40–69 years were recruited between the year 2006 and 2010. A comprehensive description has been published elsewhere (33). Briefly, at baseline assessment, participants provided physical measurements and biological samples, completed computer-assisted self-administered questionnaires, and engaged in face-to-face interviews (34). Our analyses were restricted to participants of European ancestry (33) based on self-report and multidimensional scaling (UKB data field 22006), which fits the data of the autism GWAS conducted on participants of European ancestry (35). Participants were excluded if they had withdrawn consent (n = 12), had non-Caucasian origin (n = 92,845), were non-qualified for standard quality control (36) (n = 72,128), showed sex-mismatch between reported and genetic data (sex was incorporated as a covariate to effectively control its influence in the study analyses) or were outliers concerning heterozygosity and genetic relatedness (detailed below). Finally, 337,386 participants (46% male) were considered for the PGS model (Figure 2).

Flowchart of UK Biobank (UKB) participants for analysis. ICD-10, International Classification of Disease, version 10; MWA, migraine with aura; MWOA, migraine with aura.
Phenotypic information
The phenotypes of interest in the study were migraine and the major types of migraine – with and without aura. Baseline data of UKB were used for the phenotypic information retrieval and association analyses. Participants with either missing data or reporting ‘Prefer not to answer’ or ‘Do not know’ were excluded.
Migraine
Information regarding migraine diagnosis (n = 23,191) was extracted and merged from three sources: (i) migraine diagnosed by a doctor (UKB Data-field 120016); (ii) cases with migraine as primary or secondary diagnosis from hospital records, according to the International Classification of Disease, version 10 (ICD-10) (37) coding (UKB Data-field 41270); and (iii) medical condition reported by participants to a trained nurse, followed by placing in coding tree by the trained nurse (UKB Data-field 20002). However, we considered only the diagnosed migraine cases (either by doctor or by ICD-10 coding or both), and “self-reported” cases were only considered if those patient were either overlapped with the data from “diagnosis by doctor” or “ICD-10 diagnosis” from hospital records (Figure 2).
Migraine subtypes (with and without aura)
Information on migraine with (n = 9454) and without (n= 8499) aura was calculated as a probable score from participants’ completion of the ‘Experience of Pain’ questionnaire, an online survey hosted on the UKB platform, following a similar approach reported before (38). This questionnaire was designed to delve into the specifics of pain, including its nature, severity and duration. In cases where participants indicated experiencing headaches, they were presented with a more extensive set of inquiries. These additional questions sought to gather information on the duration, frequency, intensity and any accompanying symptoms of their headaches. The data collected through these responses were used in the creation of two key variables, namely “migraine with aura (MWA)” and “migraine without aura (MWOA)” representing probable migraine with aura and probable migraine without aura, respectively. To determine MWA and MWOA diagnoses, the criteria aligned with the standards outlined in the International Classification of Headache Disorders (ICHD-3) 20 were applied based on the information provided within the UKB ‘Experience of Pain’ Questionnaire. MWA diagnosis was characterized by the presence of visual or sensory aura symptoms gradually extending over several minutes or more either before or in close proximity to the onset of the headaches. Likewise, for MWOA, specific criteria included the unilateral location of headaches, a pulsating quality, moderate or severe pain intensity, aggravation triggered by routine physical activities and the presence of associated symptoms such as nausea/vomiting, as well as photophobia or phonophobia. Details of migraine subtype, with and without aura classification and characterization are mentioned in the supplementary material.
Mediators and moderator of migraine
Seven measures were considered as potential mediators of ASD PGS and migraine association based on the previous reports of association with migraine: education, personality trait, body mass index (BMI), psychiatric comorbidities, vascular comorbidities, neurologic comorbidities and other comorbidities. BMI is an important variable to consider in case of migraine, as obesity may lead to persistent activation of the trigemino-vascular system, leading to migraine progression (16). BMI (n = 336,293) information was extracted from UKB baseline assessment data (UKB Data-field 21001). Since previous studies reported that education acts as a protective factor for migraine (39), we coded information on education (n = 276,604) in UKB by distinguishing between with or without University/College degree (UKB Data-field 6138). Individuals with migraine have been reported to exhibit neurotic personality trait (14). We measured the neuroticism score (UKB Data-field 20127) in this study on the included participants’ (n = 337,386) responses to 12 questions from the Eysenck Personality Inventory Neuroticism – Revised (EPIN-R) scale (40). From previous report (15), we took into account the common comorbidities present among individuals with migraine. We considered the diagnosed cases from hospital records, coded according to ICD-10 classification (UKB Data-field 41270) and categorized them into four categories: (1) Psychiatric comorbidities (n = 31,586) including bipolar disorder, depressive disorder, anxiety disorders and post-traumatic stress disorder; (2) Vascular comorbidities (n = 19,937) including acute myocardial infarction, subsequent myocardial infarction, ongoing complications following acute myocardial infarction, old myocardial infarction, stroke, sequelae of stroke, brain stem stroke syndrome, cerebellar stroke syndrome and Raynaud's syndrome; (3) neurologic comorbidities (n = 14,265) including epilepsy, status epilepticus, multiple sclerosis, non-organic sleep disorders and sleep disorders; and (4) other comorbidities (n = 37,962) including fibromyalgia, temporomandibular joint disorder, vasomotor and allergic rhinitis, asthma, status asthmaticus, systemic lupus erythematosus, and other headache syndromes.
We considered sex as a moderator of the effect of ASD PGS on migraine, taking into account the nearly opposite male-female prevalence of migraine (two- to three-fold higher in females) (18) and ASD (four-fold higher in males) (19). We specifically considered moderators and mediators as outlined by Baron and Kenny (41). In this framework, a mediator is a variable that serves as a mechanism through which the independent variable influences the dependent variable, acting as an intermediary variable. Conversely, a moderator is a variable that impacts the strength of the association between the independent and dependent variables, essentially functioning as an effect modifier. This is akin to assessing an interaction effect.
Genotype quality control
In the study, we utilized imputed genotyping data from the UKB (33). Our approach to quality control (QC) aligns with the recommendations for constructing PGS using UKB data (33,42). The analyses were confined to SNPs that met specific criteria: a minor allele frequency > 0.5, an imputation information score (INFO) > 0.8 and genotype missingness < 0.02. The genetic data imputation process in UKB reported (43) to be carried out using the IMPUTE2 program, which provides INFO values on a scale from 0 to 1. A value closer to 1 signifies a higher level of certainty in the SNP imputation (44). To ensure the robustness of our dataset, participants for whom genetic data met QC standards within the UKB (data-field 22020) (42) were included. This allowed us to exclude individuals with a missing rate >0.02 on autosomes, displaying sex-discrepancies, outliers in terms of heterozygosity, genetic relatedness (up to third-degree relatives) or individuals with an ethnicity other than ‘White British’ based on genetic grouping (UKB data-field 22006) (42).
PGS construction
PGS computation was done using the clumping and thresholding algorithms for each UKB participant in PRSice-2, version 2.3.5 (45,46). This algorithm generates PGS taking into account the weighted averages of common risk polymorphisms that reflect an individual's genetic predisposition to a specific condition. The weights assigned to each allele are determined by the regression β value obtained from the reference GWAS conducted on the base dataset, and individuals are then scored based on the number of trait-associated alleles they possess (0, 1 or 2). The latest iPSYCH-PGC autism GWAS summary statistics (released in November 2017) consisting 18,381 individuals with autism and 27,969 individuals from the general population, was used as the base dataset in this study (47). The PGS generated from this GWAS of ASD, explained 2.45% of the total variance of ASD and 11% SNP-based heritability (h2SNP) (47).
SNPs in linkage disequilibrium (LD) were grouped. We clumped SNPs using an LD-based r2 ≥ 0.1 and a 250-kb genomic distance following current guideline (48). We generated PGS for seven p-value thresholds (p = 1, 0.75, 0.5, 0.25, 0.1, 0.01 and 0.001), as shown in the supplementary material (Figure S1). These thresholds were selected to achieve a balanced signal-to-noise ratio due to the highly polygenic nature of autism (47). The number of SNPs at each threshold is listed in the supplementary material (Table S2).
Statistical analysis
Association analysis
Logistic regression analyses were conducted using standardized PGS as the independent variable. The phenotypes of interest migraine and its major subtypes, with aura (MWA) and without aura (MWOA) were investigated as primary outcome variables. Age, sex, genotyping batch and the first 20 genetic principal components (UKB data field 22009) were included as covariates in the regression model, all of which were standardized. Seven p-value thresholds were investigated for each of the three variables, migraine, MWA and MWOA. Thus, we identified significant results at a Bonferroni corrected α = 2 × 10−03 for the regression analyses. Given that outcome variables were binary traits, the variance (R2) explained for these traits were measured using Nagelkerke's pseudo R2. Statistical analysis were performed using R, version 4.0.3 (R Foundation).
Mediation and moderation analysis
Mediation and moderation analyses were performed to investigate the effect of other variables in the association of ASD PGS and migraine. We examined the interaction between sex and ASD PGS. The significance for the moderation analysis output was α = 5 × 10−02 (one ‘sex × PGS interaction’ test conducted). We performed a series of mediation analyses aimed at identifying potential factors that could mediate the relationship between ASD PGS and migraine. First, we investigated whether the ASD PGS were associated with the mediators, and included only those variables that were associated with ASD PGS. Then, we investigated each mediator separately, without considering them in parallel or serially taking into account: (i) the inability to establish a causal relationship between the mediators; (2) temporal ordering not being able to be determined in this cross-sectional dataset; and (3) some of these variables exhibiting moderate correlations with each other. Seven variables were considered in the mediation analyses. Thus, we identified significant results at a Bonferroni corrected α = 7 × 10−03. In the moderation analysis with migraine as the dependent variable, and in all mediation analyses, we limited our investigations to a PGS p-value threshold of 0.01, as this explained the maximum variance in migraine. Package ‘mediation’, version 4.5.0, in R using 1000 simulations was used to perform the mediation analyses.
Results
We investigated whether ASD PGS were associated with migraine and its major subtypes, with and without aura. PGS at all seven p-value thresholds were significantly and positively associated with migraine and the major types, with and without aura (Table 1).
Autism spectrum disorder polygenic score (ASD PGS) association with migraine and its major subtypes.

The result of the PSG analyses for the three phenotypes at seven different p-value thresholds is provided. For each analysis, we report the odds ratio (OR), confidence interval (CI) and the accompanying standard errors (SE), Z-score (Z) and p-value of the Z-score (p). For the regression, we included age, sex and the first twenty genetic principal components as covariates, all of which were standardized. Variance explained (R2) is Nagelkerke's pseudo R2, and reported in percentages. MWOA, migraine without aura; MWA, migraine with aura. Bonferroni corrected α of p = 2.4 × 10−3.
For migraine, the highest variance was observed at p = 0.01(7509 SNPs, R2 = 0.030%, p < 0.002) (Table 1; see also supplementary material, Table S2). Dividing the cohort into centiles based on ASD PGS (p = 0.01), the top 10% had, on average, a 12% increase in migraine compared to the bottom 10% (Figure 3).

Mean scores distribution of migraine and its major subtypes based on centiles of autism spectrum disorder (ASD) polygenic scores (PGS). An illustration is provided for the mean scores of the three phenotypes of interest in relation to the percentiles of the PGS at the p-threshold explaining maximum variance respectively (p = 0.01 for migraine, p = 0.001 for MWOA, p = 0.1 for MWA). The cohort was segmented into 100 groups based on their PGS. Each dot on the plot corresponds to the average phenotypic score for a specific group. Colors indicate the gradient of percentile, with yellow (lighter color) representing the highest percentile and red (darker color) representing the lowest percentile. MWA, migraine with aura; MWOA, migraine without aura.
The highest variance for MWOA, was observed at p = 0.001 (1378 SNPs, R2 = 0.034%, p < 0.002) (Table 1; see also supplementary material, Table S2). Centiles based analysis of the cohort on ASD PGS (p = 0.001) showed that the top 10% had, on average, a 21% increase in MWOA compared to the bottom 10% (Figure 3).
For MWA, the highest variance was observed at p = 0.1 (40,798 SNPs, R2 = 0.028%, p < 0.002) (Table 1; see also supplementary material, Table S2). Dividing the cohort into centiles based on ASD PGS (p = 0.1), the top 10% had, on average, a 14% increase in MWA compared to the bottom 10% (Figure 3). We repeated the association analyses between ASD PGS and the three phenotypes of interest, excluding individuals with self-reported physician-posed ASD diagnosis (UKB Data-field 20544) in UKB. The results remained consistent and statistically significant, as detailed in the supplementary material (Table S3).
We investigated whether sex moderates the effect of ASD PGS on migraine. Sex did not show significant (p = 0.688) interaction with PGS to predict migraine (Table 2A). The main effects of ASD PGS and Sex were significant. Female sex was significantly and positively associated with migraine. Additionally, we extended our investigation to assess whether the nature of the moderation effect was consistent across the subtypes of migraine, MWOA, and MWA. The findings were in line with the observations from the moderation analysis of migraine (see supplementary material, Table S4).
Mediation and moderation analyses for migraine.

The summary results are shown for the mediation and moderation analyses for migraine with respect to autism spectrum disorder polygenic score (ASD PGS) (p = 0.01). A. Interaction effects: we tested the interaction between PGS and sex, and report the two main effects and the interaction effect. From the analysis, we report the regression coefficient (beta) and the accompanying SE, Z-score (Z) and p-value of the Z-score (p). B. Mediation effects: A summary is provided of the proportion mediated by the mediators that were found to be potential for the relationship between ASD PGS and migraine (see Methods). Detailed results of the mediation analysis series are reported in the supplemental materiial (Table S3–S5).
We investigated if seven different variables (see supplementary material, Table S1), BMI, education, neuroticism, psychiatric comorbidities, vascular comorbidities, neurologic comorbidities and other comorbidities mediate the relationship between ASD PGS and migraine. ASD PGS were significantly associated with all except vascular comorbidities (see supplementary material, Table S5). With the exception of education, all mediating variables were significantly associated with migraine (see supplementary material, Table S6). Thus, five variables were taken forward for mediation analyses. Only neuroticism among them showed a significant mediating effect on the relationship between ASD PGS and migraine (Table 2B). The proportion of mediated effect for neuroticism was 8.75% (95% confidence interval (CI) = 4–18%), average causal mediated effect or ACME = 4.94 × 10−04 (95% CI = 2.8 × 10−04-0). Details are enlisted in the supplementary material (Table S7).
Discussion
To our knowledge, the present study is the first to investigate the genetic propensity of ASD to associate with migraine and its major subtypes, with and without aura. Furthermore, the study is the first to investigate the direct association of genetic predisposition for ASD with the migraine major subtypes, with and without aura at a population level. We show that individuals with a higher genetic predisposition for ASD are more likely to experience migraine, including both major subtypes, with and without aura. Given the intriguing known sex differences in the prevalence of ASD and migraine and the scarcity of studies focusing on the sex effect in the relationship, we aimed to address this gap. We observed that sex does not modify the association between genetically predisposed ASD and migraine. Unlike previous studies that only reported an association between ASD and migraine, we additionally investigated the potential mediating effect of education, personality trait and general health-related variables and found that neuroticism significantly mediates the association between ASD and migraine.
Our investigation reveals that migraine, including both major subtypes with and without aura, are significantly more likely to occur in adults genetically predisposed to ASD, with the risk increasing as ASD PGS rise. While previous epidemiological studies support this link, they are limited by small sample sizes (n = 18–105), a focus on children or adolescents, and clinical settings (6–8,11). Population-based studies on migraine in individuals with ASD are limited and show contrasting findings across age groups, ethnicities, geographical region and are further limited by not considering other comorbidities or not specifically examining migraine–ASD link (9,12,13). Our findings, based on genetic predisposition in a European non-clinical cohort of approximately 350,000 adults using high-quality ASD-related SNPs, minimize confounding factors and allow for broader population-level conclusions with robust statistical power.
The association between migraine and ASD may be explainable by shared pathophysiological and genetic elements (5,49). Both conditions involve calcium channel mutations, neurotransmission dysregulation (especially in the serotoninergic system), neuroinflammation, cortical minicolumn abnormalities and gut–brain axis dysfunction (5). Shared susceptibility genes further suggest a link. While our findings show a genetic predisposition to ASD is associated with migraine, further research is needed to fully understand the molecular mechanisms driven by shared heritability (5,50).
We found that both migraine subtypes, with and without aura, are significantly associated with ASD. While limited studies, often with small samples, have shown a link between migraine with aura and ASD (7,24), our study is the first to demonstrate this connection at a population level through genetic risk patterns. These findings support earlier small-scale epidemiological studies (8,11). While the associations remain consistent for both migraine subtypes in the analyses, it is important to interpret the results with caution. Although the findings are empowered and robust with large numbers, the diagnoses are approximations and subject to potential variation. Previous clinical observations, limited in sample size are thus strengthened by high statistical power, which allows drawing conclusions for a general population. Future studies may aim to validate and strengthen these findings by using broadly-phenotyped clinical samples from larger, more representative population cohorts. Further research may also explore to what extent certain lifestyle factors trigger migraine, specifically in individuals with ASD (8,51).
We demonstrate that ASD PGS is significantly associated with migraine including both types, with and without aura, explaining approximately 0.03% of the variance. While these proportions may seem to be quite small, the magnitude is not unexpected to be found in a general population, as we investigate in the study. Our presented centile analyses further demonstrates the strength and nature of the associations, highlighting the potential to identify individuals at increased risk for ASD and heightened susceptibility to migraine (52). While our PGS is based on the latest and largest autism GWAS available, the percentage of “explained variance” could potentially be increased with an even larger discovery sample size and greater inclusion of significant markers in the GWAS (53). In light of these aspects the findings made in the study can be considered robust. However, despite the potential of PGS as a powerful tool for clinical utility, even with small effect sizes, the results should be interpreted and communicated with an emphasis on a deep understanding of genetics and genomics (54–56).
Sex does not impact the positive relationship between genetic predisposition for ASD and migraine according to our results. The population level analysis incorporating genetics shows, regardless of sex difference, the imbalance in prevalence of ASD and migraine (18,19), likely does not stem from, or even balances out, in the association between common genetic background of ASD and migraine, at the same time as confirming the higher prevalence of migraine in females. Aligned to our findings, Sullivan et al. (8) found no gender difference, while focused on sensory hyperreactivity and anxiety in 81 children with ASD. By contrast, and limited to specific geographical regions, a Taiwanese study reported higher migraine risk in males with ASD (9), while Northern Californian studies reported higher prevalence among females with ASD (12,13). Further sex-stratified studies across different regional populations, and genetic correlation analyses could shed more light on this aspect. Additionally, the relationships between sex hormones and migraine, with findings such as exogenous estrogen exacerbating migraine (57) and dihydrotestosterone being linked to de novo migraine (58), are intriguing. Investigating their role in the ASD–migraine association, which was beyond the scope of the present study, may also offer further explanation regarding the finding of no effect of sex in ASD–migraine relationship.
We show that the personality trait neuroticism significantly mediates a portion of the association between ASD PGS and migraine. While this provides a framework for additional exploration, we may hypothesize a mechanism to explain the mediating effect of neuroticism. As broadly discussed in a review by Sanchez et al. (59), neuroticism may be associated with problems in the ability to endure migraine pain. Personality traits can affect an individual's vulnerability and non-adaptive coping strategy usage while facing disease conditions such as pain (60) and neurotic personality is reported for pain perception alteration (61). Further investigations using longitudinal models are needed to explore how personality traits develop with age and impact migraine in individuals with ASD. However, gathering large-scale longitudinal data from childhood to adulthood is challenging. As a result, cross-sectional mediation analysis is a valuable alternative, justifying its use in this dataset.
The strengths of this study include employing the robust PGS tool, which leverages data from the latest and largest ASD GWAS (iPSYCH-PGC), along with one of the most extensive cohorts available that possess high-quality genetic and comprehensive epidemiological data (UKB). This approach ensures the study's execution with substantial statistical power, which enabled us to also stratify according to migraine subtypes, with and without aura. Furthermore, while large population-based cohorts often exhibit underreported cases of ASD (27,32) and cohorts specifically focused on adult ASD are rare to find, they often come with enrichment of high-quality genetic data, comprehensive living-related outcomes and demographic information. The utilization of the PGS method in this context enabled the investigation of genetic data associated with underreported diagnoses and facilitated the assessment of a direct connection with extensive population-based migraine data. Nonetheless, this study needs to be also evaluated in light of its limitations. The UKB cohort is reported to have healthy volunteer bias (62) and the reported ASD prevalence (0.1%) in UKB (32) is lower than the actual (1–2%) ASD prevalence in the UK (47), limiting generalizability. However, our focus on genetic predisposition, fixed at birth, reduces the likelihood of confounding factors. Despite using the largest ASD GWAS, our ASD PGS explains only 2.5% of the variance, compared to the SNP-based heritability of 11% (47). Future research may refine the PGS by incorporating rare variants and copy number variations, while we exclusively focused on common variants known to account for the major part of ASD liability (63). The findings of this study should be interpreted with caution. Both MWA and MWOA were classified as probable diagnoses based on participants’ responses to the “Experience of Pain” questionnaire. Although the diagnoses relied on diagnosed migraine patients’ self-reported headache experiences, the questionnaire covered most, but not all, of the ICHD-3 criteria, resulting in “probable” rather than definitive diagnoses. The number of identified MWA cases were found to be higher than MWOA cases, which contrasts with the epidemiological understanding (64). This discrepancy may be partly explained by the fact that more participants met the criteria for MWA than MWOA, probablyt as a result of the limitations in the UK Biobank's ‘Experience of pain’ questionnaire, lacking certain ICHD-3 diagnostic criteria for migraine subtypes, such as specific questions about the number of attacks or headache duration. Additionally, the MWA diagnosis focused on aura criteria, such as visual disturbances and tingling/numbness, which likely captured most MWA cases. Because visual auras are common, and sensory auras are the second most frequent type (64), it is understandable that these symptoms were prioritized in the questionnaire. While PGS predictions have limitations in establishing causality (65), our study enhances the epidemiological understanding of the ASD–migraine link and contributes toward unraveling causality (66).
This research shows that individuals genetically predisposed to ASD are at higher risk for experiencing migraine, including its two major subtypes, with and without aura. The relationship between ASD risk and migraine is not affected by sex and the personality trait neuroticism plays a significant role in mediating the relationship. This highlights the hypothesis of shared genetic and pathophysiological interactions between autism and migraine, which need to be further explored.
Clinical implications
People genetically predisposed to autism are more likely to experience migraine, including migraine with and without aura. The findings are based on genetic data from over 300,000 individuals Sex does not affect the association Personality trait neuroticism partly explains this relationship The findings highlight the complex genetic and psychological connections between autism and migraine
Supplemental Material
sj-docx-1-cep-10.1177_03331024241312666 - Supplemental material for Migraine and its major subtypes – with and without aura are associated with polygenic scores for autism
Supplemental material, sj-docx-1-cep-10.1177_03331024241312666 for Migraine and its major subtypes – with and without aura are associated with polygenic scores for autism by Salahuddin Mohammad, Giorgia Bussu, Gull Rukh, Helgi B. Schiöth and Jessica Mwinyi in Cephalalgia
Footnotes
Author contributions
SM wrote the paper with support from GB, GR, HBS and JM. SM designed the work and performed the analyses. GB, GR, HBS and JM provided critical revision of the article. All authors have read and agreed to the published version of the manuscript submitted for publication.
Data availability
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
SM is supported by the Prime Minister's Fellowship, People's Republic Government of Bangladesh. GR is supported by the Svenska Sällskapet för Medicinsk Forskning.
Ethical statement
Ethical approval for the collection of UK Biobank data was granted by the North-West Multicentre Research Ethics Committee (11/NW/0382). Subsequent approval for the use of this data within our department (UKB application 30172), was granted by the Regional Ethics Committee of Uppsala, Sweden. All participants included in the study provided informed consent (![]() ).
).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
