Abstract
Purpose
Photophobia is a common and debilitating symptom associated with migraine. Women are disproportionately affected by migraines, with a higher prevalence and more severe symptoms compared to men. This study investigated the effects of cortical spreading depression on light-aversive and dark-seeking behaviors in a rat model, with an emphasis on sex differences.
Method
Experiments were conducted on seven to eight-week-old male and female Sprague Dawley rats. cortical spreading depression was modeled by injections of potassium chloride or artificial cerebrospinal fluid into the occipital cortex through a guide cannula. Key behavioral assessments included light-aversive and dark-seeking behaviors measured using a three-chamber box, pupil to iris ratio, periorbital tightening, periorbital allodynia and facial withdrawal thresholds.
Results
Our results demonstrated that cortical potassium chloride injections significantly increased photophobic behaviors, particularly in female rats. Specifically, potassium chloride-injected females demonstrated a significant reduction in the time spent in the light chamber (p = 0.001) and increased time in the dark chamber compared to control rats (p = 0.01), indicating heightened light-aversion. Females exhibited more substantial pupil constriction and eyelid closure at 180 min after potassium chloride injection compared to artificial cerebrospinal fluid injection, suggesting a stronger physiological response to light. Similarly, a greater percent of female rats displayed periorbital allodynia (withdrawal threshold <6 g) over a post injection time course compared to male rats. Among rats that exhibited periorbital allodynia at least two consecutive time points, females had significantly lower facial withdrawal thresholds than males at 60-, 90-, 120-, and 180-min post injection (p < 0.05), suggesting a difference in magnitude and duration. Furthermore, the area under the curve for the time course experiment indicated development of tactile allodynia in periorbital region among female rats (p < 0.0001).
Conclusion
Altogether these findings highlight the importance of considering sex-specific differences in developing therapeutic strategies for the treatment of migraine. We report for the first time a complete time course analysis of migraine-related responses, providing a comprehensive overview of the dynamics involved. The results suggest that potassium chloride-induced cortical spreading depression may offer a valuable model for studying the underlying mechanisms and sex differences of photophobia in migraine, aiding in the development of targeted treatments.
This is a visual representation of the abstract.
Introduction
Migraine is a complex, multiphasic, and multi-symptom neurological disorder characterized by unilateral, episodic headaches and sensory disturbances (1,2). In approximately 14% of cases, an aura involving neurologic symptoms occurs that is physiologically correlated to cortical spreading depression (CSD). CSD is a wave of neuronal and glial depolarization that slowly propagates across the cortex followed by a period of quiescence, or depression of cellular activity (3–6). This phenomenon may trigger the activation of trigeminal nerve pathways and pain processing centers within the brain, which are thought to contribute to the headache phase of the migraine (7,8).
Among the array of symptoms, individuals with migraine also experience photophobia, or sensitivity to light. Photophobia is classified into two main types: pain upon exposure to light and substantial discomfort due to glare (9). Previous studies have noted that migraineurs exhibit hypersensitivity to light even when not experiencing a headache, indicating an underlying heightened sensory response mechanism (9–13). Furthermore, functional neuroimaging studies report that migraineurs exhibit increased responsiveness to visual stimuli within the visual cortex and visual association areas, a phenomenon that may underlie the experience of photophobia (14). Given these findings, it becomes evident that increased excitability of the visual cortex may serve as a crucial link or even a potential precursor, to the phenomenon of photophobia.
Despite its common occurrence, the mechanisms underlying photophobia, especially in relation to CSD, are not fully understood. Emerging research underscores that the manifestation and intensity of migraine symptoms, including photophobia, exhibit sex differences, potentially due to variations in hormonal levels, pain perception, and neural response between sexes (15,16). Studies indicate that females, both in human and animal models, exhibit a greater predisposition to CSD and related migraine symptoms, such as photophobia and head pain, suggesting a significant role of hormonal differences in the pathophysiology of migraine (17–19).
This study used a three-chamber (light-transition-dark) box to investigate the sex-specific behavioral responses to CSD by assessing how often rats exhibit photophobia, the time courses of development of light sensitivity, and the magnitude of behaviors observed using a three-chamber, light-transition-dark, approach. Further experiments aimed to correlate the behavior to physiological changes by measuring orbital tightening, vertical palpebral aperture, and pupil:iris ratios. Understanding the sex-specific incidence and timing of photophobia in the CSD migraine model will advance our ability to develop targeted therapies that treat patients more effectively and improve their overall quality of life.
Methods
Drugs and reagents
Ketamine/xylazine was purchased from Sigma-Aldrich (St Louis, MO), and acepromazine, isoflurane from Vet One (IL, USA). All other chemicals, unless noted, were purchased from Sigma-Aldrich (St Louis, MO).
Animals
All studies were approved by the Institutional Animal Care and Use Committee of the University of Arizona (17–223) in accordance with the guidelines set forth by the National Institutes of Health Guide for the Care and Use of Laboratory Animals and the International Association for the Study of Pain. Male and female (7–8 weeks, 175–225 g) Sprague Dawley rats were purchased from Envigo. Upon arrival, three animals were housed per cage but were separated following dural cannulation. Justification for animal numbers was consistent with NIH policy (NOT-OD-15–102), and experiments were randomized to blinded treatment groups to give 80% power to detect a treatment effect size of 20% compared to a baseline response of 5% at a significance level of 0.05 (20). Numbers required to achieve statistical power were determined by G. Power3.1. The stage of estrus cycle was not determined in this study (21). All procedures were conducted in accordance with the policies and recommendations of the International Association for the Study of Pain, the NIH guidelines for laboratory animals, and were approved by the Institutional Animal Care and Use Committee (IACUC) of the University of Arizona (17–223). This study was conducted in accordance with the ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines to ensure rigorous and transparent reporting of our animal research methods and results (22).
Dural cannulation
The dural cannulation procedure was performed as previously described (23–25). Animals were briefly anesthetized via intraperitoneal injection of either 80:10 mg/kg cocktail, ketamine:xylazine or 45:5:2 mg/kg cocktail of ketamine:xylazine:acepromazine. Throughout the procedure, rats were monitored for vital stability via heart rate, respiratory rate, and skin tone, as well as reflexiveness via toe pinch. Animals were placed into a stereotaxic frame (Stoelting Co.) and subsequently received a 1.5- to 2-cm incision to expose the skull; additional connective tissue was removed with a scalpel blade (Integra Life Sciences no. 10) as necessary. A hole (0.66- to 1-mm) was then made −6.0 mm A/P and −3.0 mm M/L from bregma using a surgical drill (Ram Productions MICROLAB 350/450) to expose, but not damage, the dura. A guide cannula (0.50 mm from the top of skull. 22 GA, #C313G; Plastics One) was inserted into the hole and secured with glue. Two additional 1.0-mm holes were made rostral and contralateral to the cannula to receive stainless-steel screws (#MPX-080–3F-1 M; Small Parts). Dental acrylic was poured over the surgical site to fix the cannula and the screws, and after solidifying, a dummy cap (#C313DC; Plastics One) was inserted into the guide cannula to ensure patency. The behavior assays began a minimum of five days post-surgery to avoid confounding acute post-surgical pain. After surgery, animals were followed up for three days to monitor their weight and activity. All animals recovered well without any complications.
Cortical injection
Prior to behavioral assessments, awake but lightly restrained rodents were cortically injected using a Hamilton injector (30 GA, #80308 701 SN, Hamilton Company) customized to project 1.0 mm beyond the dura into the occipital cortex. The injector was inserted through the guide cannula to deliver a brief 0.5 µL focal injection of 1 M KCl or artificial CSF (aCSF) into the cerebral cortex. aCSF was comprised of 145 mM NaCl, 2.6 mM KCl, 1 mM MgCl2, 1.2 mM CaCl2, and 2 mM Na2HPO4 (pH 7.4); both solutions were passed through a 0.2-µm syringe filter before injection. Cortical injection was considered t = 0 min across all behavioral experiments. Systematic randomization was used to allocate animals to aCSF and KCl treatment groups, with every other animal assigned alternately to each group.
Periorbital allodynia
Periorbital allodynia was evaluated before and after cortical injection (t = 30, 60, 90, 120, 180, 360 min, and 24 h). Rats were grouped based on their postsurgical baseline to ensure equivalent pre-injection thresholds (6–8 g). Rats were acclimated to the testing box 1 h prior to evaluation of periorbital mechanical allodynia with calibrated von Frey filaments (1 g, 1.4 g, 2.0 g, 4 g, 6 g, 8 g) as previously described by Edelmayer and colleagues (26,27). Behavioral responses were determined by applying calibrated von Frey filaments perpendicularly to the midline of the forehead at the level of the eyes with enough force to cause the filament to slightly bend while held for 5 s. A response was indicated by a sharp withdrawal of the head, vocalization, or severe batting at the filament with attempts to bite it. The withdrawal threshold was determined using a modified version of the Dixon up-down method (28). Animals with a periorbital withdrawal threshold at baseline or post-surgical baselines of < 6 g were excluded (n = 11) (Figure 1(A)).

Periorbital allodynia induced by cortical KCl injection presents differently in male and female rats. (A) Number of rats evaluated meeting inclusion criteria in total (B) Higher number of female rats exhibited periorbital allodynia (threshold <6 g) at individual timepoints post injection as compared to male rats n = 20/sex/timepoint. (C) Of rats having withdrawal threshold <6 g at two consecutive timepoints, females had significantly lower facial withdrawal thresholds than males at 60, 90, 120, and 180 min post-injection. Values are the mean ± SEM (n = 7/sex with sensitivity; * p < 0.05, **p < 0.01, ***p < 0.001 females versus males; ^ p < 0.05 from BL, post-Sx, ^^ p < 0.01 from BL, post-Sx).
Total time spent analysis in the light, transition, and dark zones
The dark-transition-light behavior apparatus consists of a box divided into three chambers (San Diego Instruments, CPP/CPA boxes for rats); each chamber is separated by a doorway that allows the animals to pass from one chamber to another. One of the end chambers was lit at ∼100 lux (maximal output of box), the center chamber acted as a dimly lit transition zone (∼30 lux, residual carry-over from the lit chamber), the other end chamber was un-lit, although there was still some carry-over (∼ 5 lux) (Figure 2(A)). As the animals move from one chamber to the next, they pass through a photobeam array which records their movement and calculates the number of entries to, and amount of time spent in each chamber. Animals were first acclimated to the behavioral apparatus over two days to limit novelty of the environment. Rats were placed into the boxes during acclimation and allowed to explore freely for 15 min; room lights (250 lux) remained on during this phase. Light sensitivity was measured before and after (t = 15, 60, 120, 180, 240, 300, 360 min, and 24 h) cortical injection. For baseline, room lights were turned off five minutes before animals were placed into the boxes to allow for retinal accommodation, and the light in the “light” box was set at the lowest setting to allow ∼10 lux across the apparatus. For all other timepoints, the light box was maintained at ∼100 lux and room lights were turned off. Animals were removed from the apparatus and returned to their home cage in between each testing interval. Exclusion criterium was spending ≥80% of the time in any chamber before cortical injection; no animals were excluded on this basis.

Naïve male, but not female rats develop dark preference in the absence of injury after repeated testing. (A) Diagram depicts the experimental setup for assessing photophobia in rats, with an emphasis on the light conditions at baseline and during testing. The upper part of the figure shows the baseline conditions with all chambers equalized at 10 lux. The middle section illustrates the calibrated light intensity levels for the test chambers: dark (5 lux), transition (30 lux), and light (100 lux), simulating varying environmental lighting conditions. The timeline at the bottom outlines the experimental procedure, including room habituation, the accommodation period with lights off, and the time points post cortical injection (15 min to 24 h) when rat behavior was observed. The right side provides a reference for light intensity levels (measured in lux) corresponding to different environmental and indoor scenarios for contextual understanding of the lux levels used in the experiment. This setup was intended to mimic conditions rats might encounter in natural settings and assess their light aversion and preference (B) The time spent by male and female rats in different chambers at various time points with repeated exposure to the testing apparatus. The dark chamber, transition zone, and light chamber are represented by black, grey, and yellow bars, respectively. Baseline (BL) measurements were taken before any injections were administered. Subsequent time points (15 min to 1440 min) represent the duration in each chamber following injections. Naïve male rats developed a preference for the dark chamber with repeated exposures whereas female rats did not. Data are expressed as mean ± SEM for both sexes, indicating the preference or aversion to light and dark environments over time.
Orbital tightening/pupil constriction
Orbital tightening was measured by calculating vertical palpebral aperture relative to a reference point (nose tip to medial angle of the eye); pupil constriction was calculated using the ratio of pupil: iris diameter. Vertical palpebral aperture and pupil constriction were evaluated from static images captured immediately prior to, at 60-, and at 180-min after cortical injection. These timepoints were determined from peak photophobic behavior during the light-dark behavioral assay. Experiments were performed in naïve (n = 5), KCl-injected (n = 6), and aCSF-injected (n = 6) male and female rats. This behavioral assay was performed at room lux of 300, and between 09:00 and 13:00 h. Animals were returned to their home cage between timepoints. Images were captured using an iPhone 14 camera and analyzed using PowerPoint software. During image collection, rats were lightly restrained to prevent movement and promote photo clarity. Animals that had corneal damage or were unable to be successfully recorded were excluded from the experiment; nine rats met this exclusion criteria. Potential corneal damage was identified by the presence of a film covering the eye, which could have resulted from either dry eye or inadvertent damage during surgery. Vertical palpebral aperture opening, and pupil constriction ratios were compared relative to baseline levels.
Statistical analysis
Statistical analyses were conducted using GraphPad Prism 10.2.3 (GraphPad Software). Data were analyzed using two-way ANOVA with post-hoc tests for multiple comparisons. Time in chambers, periorbital allodynia, and vertical palpebral aperture measurements were assessed using repeated measures two-way ANOVA to analyze differences between treatment groups over time, with Tukey's test applied post hoc. Naïve data for pupil constriction and vertical palpebral aperture measurements were calculated using paired t-tests. Unless otherwise stated, the data were expressed as mean ± S.E.M. Differences were considered significant if p ≤ 0.05.
Results
Sex differences in periorbital allodynia after CSD
As a first step to establish CSD model in male and female rats, we measured postsurgical periorbital allodynia with calibrated von Frey filaments overtime. Tactile threshold was evaluated seven days after placement of cortical guide cannula and baseline measurements were taken before any injections were administered. Animals were considered sensitive when periorbital thresholds fell below 80% of the maximal threshold (<6 g); animals considered as insensitive did not have threshold below 6 g at two consecutive time points (Figure 1(A)). At individual time points, more female rats were identified as having periorbital allodynia than male rats at 60-, 90-, 120-, and 180-min post injection (Figure 1(B)). Notably, the percentage of rats (number allodynic/ total number of rats evaluated, n = 19/sex) exhibiting periorbital allodynia was substantially higher in females than males at each time point. For instance, at 30 min post injection, 80% of female rats exhibited periorbital allodynia compared to only 19% of male rats. This trend continued with 85% of females affected at 60 and 90 min, reached 100% prevalence at 120 and 180 min, and decreased to 75% at 240 min. In contrast, male rats showed much lower incidence of allodynia, never exceeding 29% at any time point (Figure 1(B)). The magnitude and duration of periorbital allodynia was next determined (Figure 1(C)). In rats showing sensitivity (n = 7/sex) the periorbital withdrawal threshold in females was significantly lower as compared to males starting at 90 min post injection, peaking at 120 min, and lasting until 180 min after cortical injections. As compared to baseline thresholds, female rats had an onset of significantly reduced thresholds by 60 min through 120 min, whereas male thresholds only reached statistical significance 90 min after injection. These results suggest a different time course and prevalence for the development of periorbital allodynia between sexes following cortical KCl injection, with females exhibiting a more pronounced response.
Sex differences in chamber preference in naive rats
Given the sex difference in periorbital allodynia, the next study analyzed the development of photophobia over time in male and female rats. A preliminary study was conducted to establish a baseline for light-aversion and dark-seeking behaviors in rats before proceeding with a more focused experiment to quantify the effects of cortical spreading depression (CSD) on photophobia. To have a control for comparison with post CSD behaviors, we begin with understanding the innate preferences and aversions of naive male and female rats to different light conditions. The first study evaluated the time course of light-aversion in male and female SD rats following surgical cannulation but receiving no injection. At baseline, when chambers were at an equalized lux level (10 lux), male and female rats spent similar amounts of time in each chamber (Figure 2(B)). When the lux of each chamber was calibrated to light (100 lux), transition (30 lux) and dark (5 lux), repeated exposure to the three-chamber set up over 24 h significantly decreased the amount of total time non-injected male rats spent in the light chamber as compared to the baseline condition (10 lux in all three chambers) (Figure 2(B), Male). This preference for the dark chamber intensified by 240 mins and peaked at 360 min time-point (p < 0.0001), indicating a robust trend towards dark-seeking behavior and light-aversion among males. Female rats, on the other hand, displayed a slightly different trend. While they also showed preference for the dark chamber, this behavior was less consistent and prevalent compared to males. Females had no significant differences in time spent in any chamber as compared to baseline or repeated exposure at 100 lux. Together, these data suggest that in the absence of cortical injection, males exhibit a preference for the dark chamber after five exposures to the chambers, and this does not occur to a significant extent in female SD rats.
Light-aversion in rats after cortical injections: Males
To determine how CSD impacts light sensitivity in male rats, they were cortically injected with either aCSF (0.5 uL) or KCl (1 M, 0.5 uL). To understand the complex nature of light-aversion and dark-seeking behaviors, we used three independent parameters: the time spent in the chamber, the number of entries into the chamber, and an index reflecting aversion to light or preference for darkness and transition, accounting for time spent in each chamber at baseline. These behaviors were quantified using analysis of variance (ANOVA) and area under the curve (AUC) analysis, providing a multifaceted view of the rats’ responses to light following CSD induction. ANOVA was used to evaluate the “effect of time,” showing how behaviors changed at different time points, and the “interaction between time and injection type,” indicating whether these changes varied based on the injection. The results for male and female rats showed significant changes in light sensitivity and related behaviors as depicted in Figure 3(A) to (H).
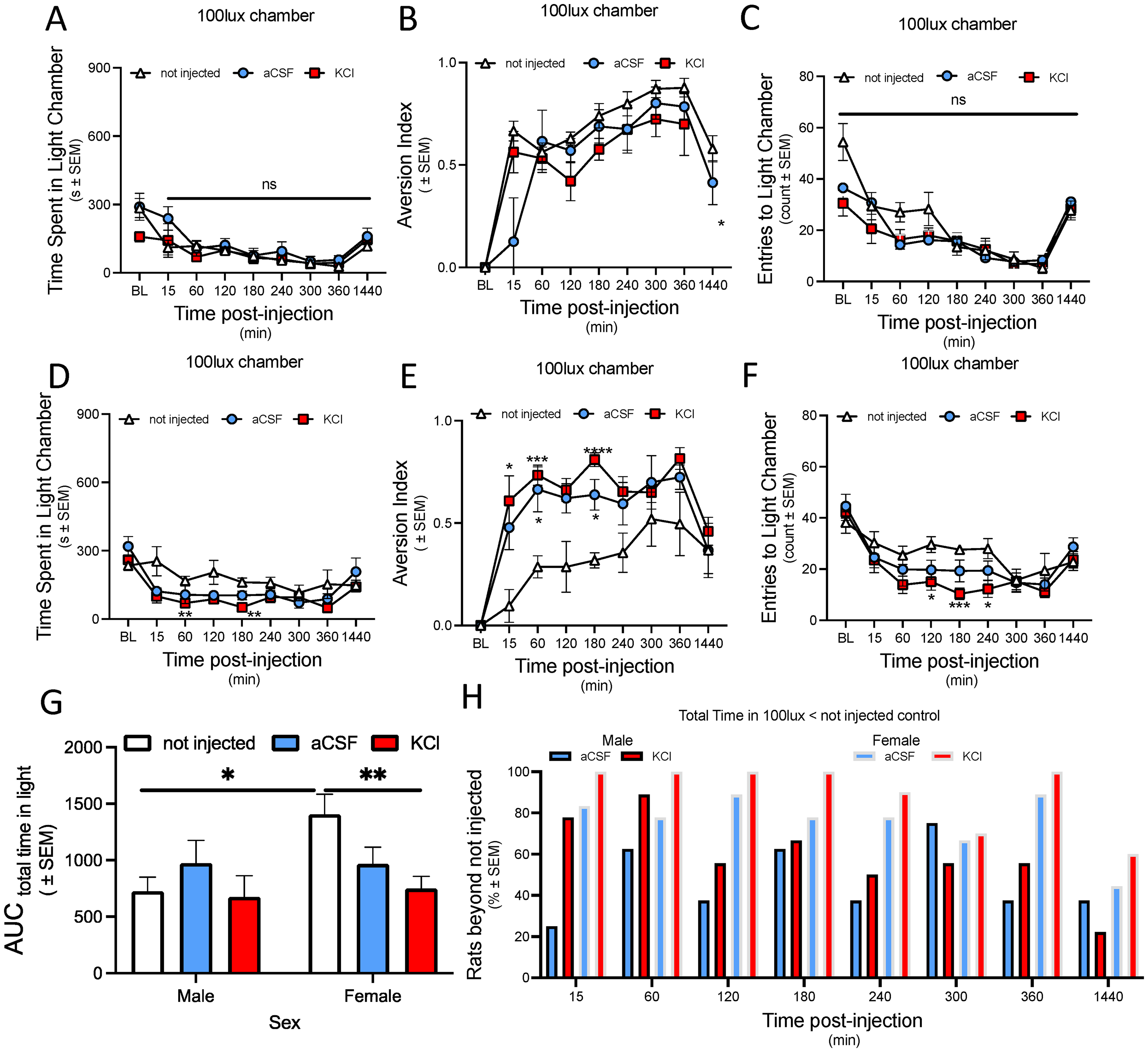
Cortical injection of either aCSF or KCl induces light aversion in female but not male rats.
Male rats spent significantly less time in the light (100 lux) chamber regardless of treatment group (F (2.937, 60.94) = 16.34, p < 0.0001), with no significant interaction between the observation time and injection type (F (16, 166) = 1.278, p = 0.2168) (Figure 3(A)).
Next, we looked at the aversion index, which accounts for the proportional reduction in time spent in the light chamber compared to baseline (Figure 3(B)). The total time spent in the light chamber varied significantly over time (F (4.378, 86.46) = 22.68, p < 0.0001) but KCl and aCSF injections were not different from controls. Interestingly, the ‘non-injected’ control group maintained a high overall aversion to light, particularly evident at 24 h post injection as compared to the KCl group (F (16,158) = 2.196, p = 0.0157) (Figure 3(B)).
The number of entries into the light chamber (Figure 3(C)) were analyzed as an additional behavioral indicator, complementing our measures of the aversion index (time spent in the light chamber). There was a significant time-dependent trend in the frequency of chamber entries (F (5.228, 109.1) = 34.79, p < 0.0001), with no difference between treatment types, signifying that the tendency to enter the light chamber varied over time irrespective of the treatment (Figure 3(C)).
Although the differences in time spent in the light chamber among the treatment groups did not reach statistical significance, it was notable that rats injected with KCl exhibited the shortest duration in the light chamber, as reflected by their lower area under the curve (AUC) value (675.7 ± 186.2, N = 9). The ‘non-injected’ group also maintained a high overall aversion to the light (AUC = 726 ± 124, N = 7). While the aCSF group displayed an intermediate behavior with the highest AUC for time spent in the light chamber (AUC = 972.3 ± 201.1, N = 8) (Figure 3(G)), suggesting they exhibited the lowest overall photophobic response among the groups.
These findings indicate that the photophobic response demonstrated by male rats over time was independent of the treatment type, suggesting that in males the aversion to light is a dynamic process influenced more by external factors independent of cortical injection.
Light-aversion in rats after cortical injections: Females
In analyzing the female rat's data following cortical injections, we found significant time and treatment dependent changes in light-aversion behaviors, as reflected in both the aversion index and time spent in the light chamber (Figure 3(D) to (F)). The mixed-effects model analysis showed a significant effect of time (F (4.943, 85.27) = 15.87, p < 0.0001), and an interaction between time and cortical injection (F (16, 138) = 2.135, p = 0.0099). Indicating that light-aversion varied over time depending on the type of injection (KCl or aCSF). At both 60- and 180-min post injection, the KCl group spent significantly less time in the light compared to the non-injected group (p value of 0.0078 and 0.0036, respectively (Figure 3(D)).
Subsequent analysis for the aversion index (Figure 3(E)) revealed a significant effect of time (F (5.039, 86.92) = 22.56, p < 0.0001), with no significant interaction between time and treatment (F (16, 138) = 1.596, p 0.0774). The KCl-injected females had the highest aversion to light, which started at 15 min (p = 0.0165), remained elevated at 60 min (p = 0.0003), and peaked at 180 min (p < 0.0001) post injection as compared to the non-injected group (Figure 3(E)). The aCSF-injected group also displayed an intermediate aversion index, with a significant difference from the non-injected control at 60 min (p = 0.0332) and 180 min (p = 0.0109) post injection, which may suggest some level of stress or discomfort related to the injection procedure (Figure 3(E)).
This sustained aversion is further supported by the AUC data, where the KCl group had the least time spent in the light with a total area of 749.5 ± 107.7 (N = 10) compared to other groups, indicating a robust aversion to light across all time points (Figure 3(G)). The overall AUC for the aCSF group was 966.2 ± 147.6 (N = 7), higher than that of the KCl group, which suggests that while the initial aversion was less intense than that induced by KCl, it did not decrease as much over time (Figure 3(G)).
The control group (non-injected) exhibited the least light-aversion overall, with the highest AUC value of 1405 ± 179 (N = 5) for time spent in the light chamber, signifying the least photophobic response across the time course (Figure 3(G)). This suggests that the physical act of injection or the induction of CSD, rather than nonspecific effects of the procedure, primarily drives the photophobic response in female rates.
The analysis of the number of entries into the light chamber underscored significant behavioral trends (Figure 3(F)). The mixed-effects model analysis confirmed a significant effect of time (F (4.632, 79.91) = 17.32, p < 0.0001) on the number of entries, but no significant interaction was found between these factors (F (16, 138) = 1.467, p = 0.1205). The KCl-induced photophobia in female rats was evident, as they entered the light chamber significantly less frequently at 120 min (p = 0.0209), 180 min (p < 0.0001), and 240 min (p = 0.0311) post injection relative to non-injected controls (Figure 3(F)). This pattern highlights the pronounced impact of KCl on photophobic behavior, as demonstrated by the reduced frequency of light chamber entries over time.
Incidence of light sensitivity in male and female rats over time
To further understand the incidence of light sensitivity over time, we assessed the percentage of rats in each group that spent less time in the light chamber compared to the average of the non-injected control group at each time points (Figure 3(H)). This analysis helps to highlight the immediate effects of the treatments and the prevalence of light sensitivity over time. Both male and female rats showed increased photophobic behavior following KCl injection, particularly within the first hour compared to non-injected controls. In female rats, KCl injection induced a strong and immediate photophobic response, with all females avoiding the light chamber at 15, 60,120, 180 and, 240 min. The aCSF-injected females displayed a more variable response, indicating less consistent light sensitivity compared to the KCl group.
Male rats also demonstrated increased light sensitivity following KCl injection, but the response was less pronounced and more variable than in females. While both male and female rats injected with aCSF showed some degree of light-aversion, the effect was notably stronger and more consistent in the KCl-injected females. These findings emphasize the significant impact of KCl on inducing sustained photophobic behavior, particularly in female rats (Figure 3(H)).
Dark-seeking behavior in rats: Males
In addition to light avoidance, we examined dark-seeking behaviors in male and female rats. We assessed time spent in the dark chamber, a dark-seeking index, and the number of entries into the dark chamber (Figure 4(A) to (H)).

Cortical injection of KCl, but not aCSF induces dark seeking in female but not male rats.
In male rats, cortical injection of aCSF or KCl did not statistically change the time spent in the dark chamber as compared to non-injected rats (F (16, 158) = 0.9972, p = 0.4626) (Figure 4(A)). When considering the total time spent in the dark chamber, the KCl-injected males displayed a greater duration within the dark environment across most time points compared to aCSF-injected and non-injected controls, inferred from their higher AUC value of 5387 ± 275.7 (N = 9), compared to 4967 + 434.9 (N = 8) for aCSF and 4878 + 343 (N = 7) for non-injected groups (Figure 3(G)).
The dark-seeking index was significantly affected by time (F (2.092, 41.58) = 7.891, p = 0.0011), but not by the interaction of time with cortical injection (F (16, 159) = 1.274, p = 0.2198) (Figure 4(B)). This indicates that the overall preference for the dark chamber was influenced primarily by the passage of time rather than the specific treatment received. At 15-min post cortical injection, the dark-seeking index was statistically higher in the KCl-injected group compared to rats non-injected (Figure 4(B)), suggesting a more rapid initiation in behavior (p = 0.0040). However, there were no differences between treatment groups at any later time points.
The number of entries into the dark chamber reflects the frequency of dark-seeking behavior (Figure 4(C)). The non-injected group made more entries compared to KCl and aCSF group over time (F (5.575, 115.7) = 33.33, p < 0.0001). This was particularly evident between aCSF and non-injected group (F (16, 166) = 2.150, p = 0.0085) at baseline, 15-, and 60-min post injection. (Figure 4(C)).
Dark-seeking behavior in rats: Females
After receiving KCl injections, female rats spent significantly more time in the dark chamber then non-injected controls (F (4.821, 83.17) = 7.982, p < 0.0001). With regards to comparison between the non-injected and KCl groups, this phenomenon was most significant at 120 and 180 min (F (16, 138) = 1.997, p = 0.0172, (p = 0.0093, 0.0017) (Figure 4(D)).
Similarly, the KCl group showed increased preference for dark relative to the aCSF-injected group that persisted at 180 (p = 0.0033), 240 (p = 0.0055), and 360 min (p = 0.0435), suggesting a sustained reaction to KCl injection. These results were further validated by the AUC values (Figure 4(G)), which show that the KCl group had the biggest AUC for time spent in the dark chamber (AUC = 5428 + 242.6, N = 10), indicative of a robust and persistent dark-seeking behavior. In comparison, the aCSF group had an AUC of 3852 ± 325.7 (N = 7) and the non-injected group had an AUC of 4323 ± 237.3 (N = 5), indicating less time spent in the dark chamber compared to the KCl group (Figure 4(G)).
In assessing the dark-seeking index (Figure 4(E)), we found that dark-seeking behavior by KCl-treated female rats is time-dependent (F (4.742, 81.80) = 18.56, p < 0.0001) and there is an interaction between time and injection type (F (16, 138) = 2.211, p = 0.0073). This effect was especially pronounced at 60 min post-KCl injection, with a statistically significant difference with respective to the non-injected group (p = 0.0075) (Figure 4(E)). The preference towards darkness became more pronounced over time, with a peak at 180 min post injection (p < 0.0001).
Post KCl injection, female rats entered the dark chamber less frequently with regards to time (F (4.544, 79.52) = 12.75, p < 0.0001) than their non-injected or aCSF-injected counterparts, reaching statistical significance at 240 min post injection (p = 0.0192) relative to the non-injected group (Figure 4(F)). This behavior suggests that the KCl-induced CSD might have reduced their overall mobility, manifesting as a stronger preference for remaining within the dark chamber rather than remaining mobile.
Together, these findings indicate that female rats have a more pronounced, treatment-specific response to CSD induction. Notably, the KCl-treated females exhibited significant photophobia and an increased preference for dark environments, suggesting heightened sensitivity to light and altered sensory processing following CSD. Furthermore, there is a consistent pattern of behavior across various measures of light-aversion and dark-seeking post-CSD. This pattern indicates that the CSD not only affects immediate sensory responses but also leads to sustained changes in behavior in female rats.
Incidence of dark-seeking in male and female rats over time
To further assess the impact of CSD on dark-seeking behaviors, we evaluated the percentage of animals in each injection group (KCl and aCSF) that spent more time in the 5-lux chamber compared to the average time spent by the non-injected control group at each time point (Figure 4(H)). In male rats, the percentage of KCl-injected animals spending more time in the 5-lux chamber did not consistently exceed than of the control groups (aCSF and non-injected), indicating no strong or lasting preference for the dark chamber over the course of the experiment. However, KCl-injected males generally spent more time in the dark chamber compared to aCSF-injected males at most time points evaluated.
In contrast, female rats injected with KCl demonstrated a consistent and significant occupancy in the 5-lux chamber, with the percentage of time spent in the dark chamber notably surpassing the control averages at 60-, 180-, 240-, and 360-min post injection. These CSD induction data delineate a clear sex difference in the susceptibility to, and time variance, in photophobic behaviors after a single cortical KCl injection to model CSD induction (Figure 4(H)).
Transition chamber: Males
In addition to dark and light chambers, we included a transition chamber (30 lux) to better understand the propensity for dark-seeking and light-aversion in rats following cortical injections (Figure 5). Our analysis of male rat behavior revealed significant differences in occupancy in the transition chamber with respect to time (F (4.281,88.84) = 8.231, p < 0.0001), but there was no effect of injection type on the time spent when comparing KCl and aCSF injections to the non-injected controls (F (16,166) = 0.9020, p = 0.56) (Figure 5(A)).
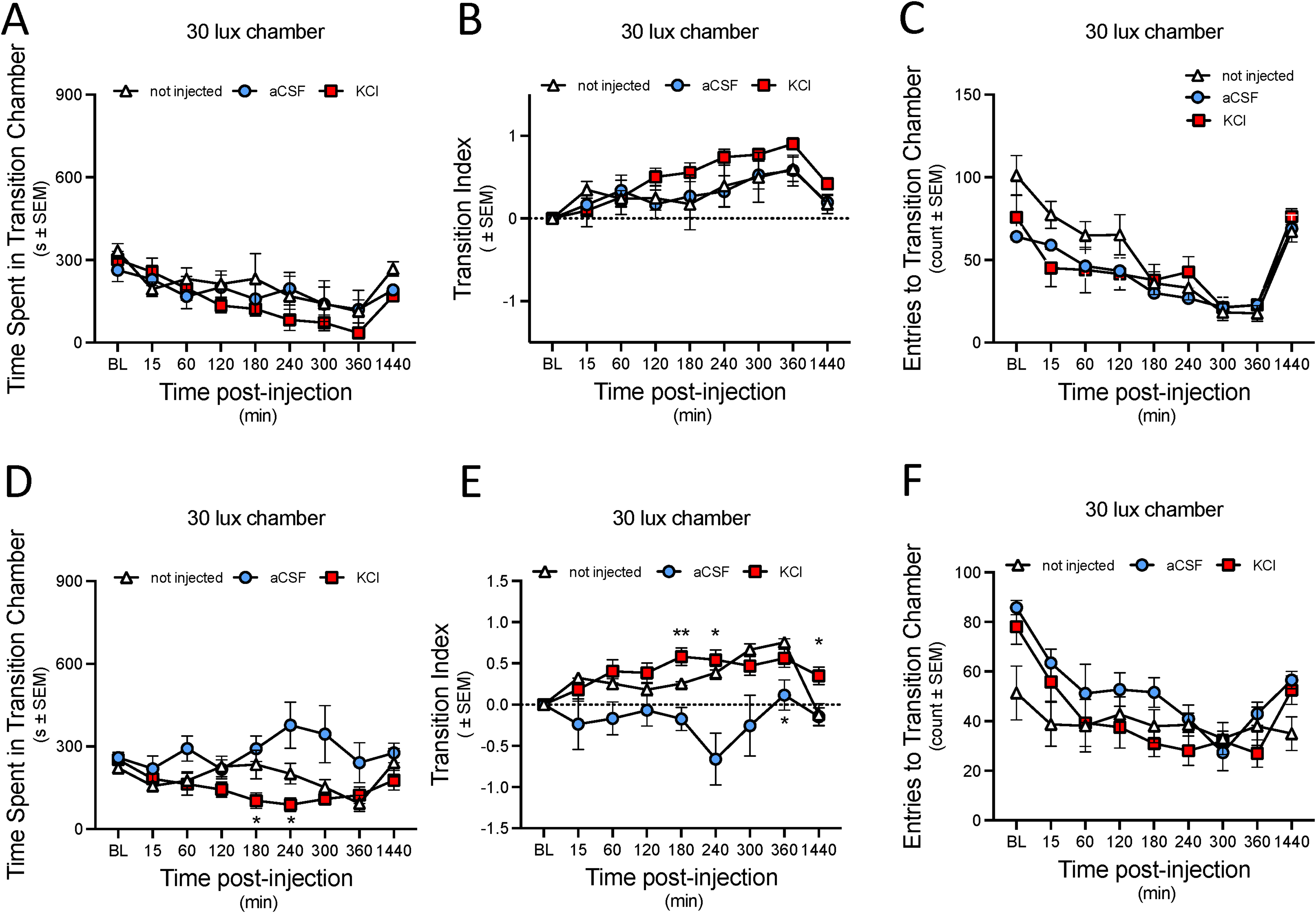
Cortical KCl reduced the time spent in the transition chamber in females, not males, without changing the total exploratory entries to the chamber. (A) Total time spent in the transition chamber by male SD rats. (B) Calculated transition index considering time spent in the transition chamber across time points. (C) Total number of entries into the transition chamber made by male rats. (D) Total time spent in the transition chamber by female SD rats. (E) Calculated transition index considering time spent in the transition chamber across time points in female rats. (F) Total number of entries into the transition chamber made by female rats. Data are expressed as mean ± SEM.
Similarly, the transition index did not reveal any statistically significant differences on time spent in the transition chamber between KCl, aCSF, and non-injected groups (F (16,159) = 0.8693, p = 0.61). However, KCl males with respect to KCl baseline spent less time in the transition chamber (F (4.111, 81.70) = 7.667, p < 0.0001), suggesting a reduced preference to remain in the transition chamber compared to the aCSF and non-injected groups (Figure 5(B)).
This trend was mirrored in the number of entries into the transition chamber (Figure 5(C)), with all injected and non-injected group making the fewest entries on average over time (F (5.567, 77.25) = 29.19, p < 0.0001), suggesting a diminished transitional activity (Figure 5(C)).
Transition chamber: Females
In female rats, following cortical injections, the time spent in the transition chamber was significantly dependent on the injection type (F (16, 129) = 2.240, p = 0.0068). The KCl-treated group spent significantly less time in the transition chamber at 180- and 240-min post injection when compared to the aCSF-treated group, with p-values of 0.0145 and 0.0299, respectively (Figure 5(D)).
When examining the transition index (Figure 5(E)), a notable decrease in the preference to remain in the transition chamber was evident over time (F (4.068, 90.01) = 3.718, p = 0.0073) with reference to injection type (F (16, 177) = 2.807, p = 0.0005). A marked difference was observed at the 180-min time point, where the transition index of the KCl injected group was statistically higher than that of the aCSF injected (p = 0.0017) and non-injected (p = 0.0339) groups. The aCSF group also showed significant reductions in the transition index compared to non-injected controls at this time point (p = 0.0367). This pattern persisted, with significant differences between the KCl and non-injected groups at 240 (p = 0.02) and 1440 (p = 0.04) minutes post injection, as well as between the KCl and aCSF groups at 1440 (p = 0.01) minutes. Additionally, the aCSF group continued to show significant reduction in the transition index from the non-injected group at 240 and 360 min (Figure 5(E)). Notably, the frequency of entries into the transition chamber was not statistically significant different among the KCl, aCSF, and non-injected groups, indicating that the number of entries was influenced by time rather than treatment (F (3.716, 56.68) = 11.47, p < 0.0001) (Figure 5(F)). These transition data suggest that moving between the light (100 lux) and dark (5 lux) through the 30-lux chamber occurred in all groups, but that the aCSF-injected rats preferred this light level as compared to KCl or not-injected controls
Pupil constriction induced by cortical aCSF or KCl
To determine whether eye physiology is altered by treatments that change behaviors, we measured the pupil diameter and vertical palpebral aperture. In naive animals and at baseline after cannula implantation, pupil diameter relative to iris outer diameter does not differ between the sexes at room lighting of 300 lux (Figure 6(A)).

Pupil constriction occurs during light aversion whereas lid closure aligns with dark seeking independent of sex. (A) Ratio of pupil to outer iris diameter in naïve male and female rats in room lighting. (B) Pupil constriction in male rats after dural cannulation (BL), 60 and 180 min after aCSF or KCl injected into the cortex. (C) Pupil constriction in female rats after dural cannulation (BL), 60 and 180 min after aCSF or KCl injected into the cortex aligning with peak aversion but not dark seeking in Figures 3 and 4. (D) Ratio of Vertical Palpebral aperture to tip of nose to inner corner in naïve male and female rats in room lighting. (E) Vertical Palpebral aperture relative to the distance in male rats after dural cannulation (BL), 60 and 180 min after aCSF or KCl injected into the cortex. (F) Vertical Palpebral aperture relative to the distance in female rats after dural cannulation (BL), 60 and 180 min after aCSF or KCl injected into the cortex aligning with peak dark seeking but not light aversion in Figures 3 and 4. Data are expressed as mean ± SEM.
Pupil diameter is reduced 60 min after cortical aCSF and KCl injection in both sexes (Figure 6(B) and (C)), suggesting constriction in both sexes. However, this constriction did not differ between aCSF and KCl injections at either time point evaluated. In male rats, the reduction in pupil diameter was significant only for aCSF injection (p = 0.0278) (Figure 6(B)). For female rats, there was a significant reduction in pupil diameter for both KCl (p = 0.049) and aCSF injection (p = 0.059) (Figure 6(C)). These data suggest that pupil constriction occurs at times of light-aversion, with females showing a more pronounced response across treatments compared to males.
Sex differences in vertical palpebral aperture responses
The degree of eyelid closure, measured using vertical palpebral aperture (see Methods section), did not differ between male and female rats in the uninjured state or after cannula implantation (Figure 6(D)). Males displayed a significant difference in eye closure at baseline between KCl- and aCSF-treated rats (p = 0.0056) (Figure 6(E)).
In female rats following cortical injection of aCSF or KCl, no significant differences in eyelid closure were observed at baseline or 60 min. By 180 min post injection, animals injected with cortical KCl showed a significant reduction in vertical palpebral aperture as compared to aCSF injected animals (p = 0.0172) suggesting that KCl injection causes lid closure at times of light aversive behavior. Together, these results suggest light sensitivity during CSD-like conditions manifests in eye physiology, with pupil constriction occurring during light-aversion and lid closure aligning with dark-seeking, independent of sex (Figure 6(F)).
Discussion
This set of studies investigated the effects of cortical spreading depression (CSD) on the timelines of induction of periorbital allodynia, light-aversion and dark-seeking behaviors, pupil constriction, vertical palpebral aperture, and periorbital allodynia in male and female rats. Current results indicate that cortical injection of KCl, but not aCSF induces periorbital allodynia in male and female rats with differences in incidence, magnitude and duration. Yet, both aCSF and KCl induced significant photophobic behavior in female rats as compared to males. We have previously validated and published that 1 M KCl successfully induces CSD in both male and female rats, leading to consistent behavioral and physiological outcomes across the sexes (23,24). Additionally, we observed significant pupil constriction in both sexes following aCSF and KCl injections, with a greater effect in female rats. Furthermore, we found that KCl-injected females had significant eyelid closure at 180 min post injection, indicating an association between light-aversion and eyelid closure. Although previous studies have reported photophobic behaviors, ours is the first to provide a complete time course of these behaviors, offering new insights into how they develop and change over a 24-h period. Importantly, the observed temporal dynamics and sex differences of periorbital allodynia and light sensitivity after CSD modeling support a distinct underlying mechanism may be responsible for these effects in male and females.
Our study differs significantly from previous research, particularly in terms of species used, sex inclusion and the timing of intervention following CSD. Tang et al. (29) conducted their study exclusively on male mice, inducing CSD 24 h after surgery and then examining photophobic behavior 24 h post-CSD. They found that male mice in the KCl group developed significant photophobia following CSD. In contrast, we used both male and female rats, inducing CSD after a one-week recovery period following surgery and observing photophobic behaviors over a 24-h period immediately following CSD induction. Notably, male rats in our study did not develop photophobia within the 24-h observation period post-CSD induction, suggesting species-specific and model selective differences in response to CSD induction. This extended recovery period in our study helps to reduce the confounding effects of surgical trauma, providing a clearer picture of CSD-induced behaviors. Moreover, by using a full 24-h observation period post-CSD, our study captures the temporal dynamics of photophobia more comprehensively, which might be overlooked in studies with shorter observation windows.
The species difference between the studies also has important implications. The choice for using rats in our study was driven by practical and experimental advantages they offer in the neuroscience and behavioral research. Because of their larger brain size compared to mice, the cannula causes less damage to surrounding tissue and a smaller region is affected. Additionally, rats are easier to handle and experience less stress from human contact, which is a significant consideration in behavioral experiments (30). This similarity suggests that our findings may be more directly translatable to human conditions, particularly in the context of migraine research. Additionally, our study extends these findings by demonstrating a clear sex difference in the response to KCl-induced CSD, with female rats showing a more pronounced and prolonged photophobic response. These data are consistent with clinical populations, as females exhibit increased prevalence and severity of migraine symptoms (16,31–33). Across all assays, KCl-injected female rats exhibited greatest pain, photophobic behavior, and orbital tightening responses relative to other groups. When taken together, these experiments describe a comprehensive time course of migraine-like symptomology of light and touch sensitivity.
Our results correspond with our initial hypothesis that CSD induces more severe photophobia and migraine-like behavior in females compared to male rats. These sex differences in symptom frequency observed in female rats suggest an underlying biological mechanism influenced by both hormonal factors and differences in neural circuitry. Previous research has shown that estrogen affects cortical excitability and contributes to the development of CSD. The amplitude of the spreading depression depends on the estrogen level (34), suggesting that estrogen influences migraine pathogenesis by more than one mechanism. Previous studies with R192Q mutant mice, a model of familial hemiplegic migraine type 1 have shown that estrogen makes female mice more susceptible to CSD, while testosterone has a protective effect in males by reducing cortical excitability. These findings highlight the important role of sex hormones not only in genetic models of migraine but also in baseline sex differences as seen in our study (35,36).
In addition to hormonal influences, differences in neural circuits between males and females may also contribute to the observed variations in photophobic behavior. Research indicates that males and females can have distinct neural pathways and brain region activation patterns in response to pain stimuli. Recent imaging studies have demonstrated that female migraineurs exhibit significant structural and functional differences in brain regions associated with pain and sensory processing compared to male migraineurs and healthy controls. For example, female migraineurs have been found to have a thicker posterior insula and precuneus cortex, which are areas involved in pain perception and emotional processing. These structural differences are accompanied by functional disparities, with females showing a unique pattern of functional connectivity in response to noxious stimuli, particularly involving brain regions associated with emotional circuitry more than sensory processing. This suggests that the brains of female migraineurs are differentially affected by the disease compared to males, potentially leading to more pronounced pain and photophobic responses (37).
The present study has few limitations. First, is the lack of direct hormonal measurements in our study, which prevents us from conclusively determining the role of hormones in the observed sex differences. Second, sex differences in periorbital allodynia after KCl cortical injection were evident; however, aCSF sex differences were not assessed currently. A previous study has shown that dural pinprick alone is sufficient to induce CSD in a majority (73.9%) of rodents but is insufficient to produce sustained activation and sensitization of trigeminal afferents believed to be necessary for migraine-induced cutaneous allodynia (23). In females, this observation was similar. It is possible that the acute pain response from the dural pinprick resulted in conditioned behavior in aCSF-injected rats that manifests as photophobia. Therefore, caution should be exercised when extrapolating these findings to human conditions.
In summary, the present study provided quantitative evidence that CSD induces allodynia and photophobic behavior, with significant sex and temporal manifestations observed in male and female rats. Moreover, we demonstrated that female rats exhibited a more pronounced and prolonged photophobic response compared to males, highlighting the importance of considering sex differences in migraine research. From a clinical viewpoint, the present study supports the view that CSD can differentially impact males and female prevalence, symptom presentation, and severity of migraine like behaviors observed in women. These differences have been documented at a behavioral level with ongoing studies exploring physiological and molecular mechanisms of sexually dimorphic responses to light and periorbital touch.
Article highlights
Female rats exhibited a more intense and prolonged photophobic response to KCl injections compared to males. KCl injections caused significant light-aversion in females, while males showed no significant change in light-aversion across treatment groups. No sex differences were observed in pupil-to-iris ratios following KCl or aCSF injections. Females demonstrated greater eye closure and periorbital allodynia post-KCl injection compared to males, indicating stronger pain-related responses in females.
Footnotes
Availability of data and materials
The data generated and analyzed during this study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Mechanisms of Pain and Photophobia in Migraine and Dry Eye NIH/NEI 1UO1EY034680-01 and with support from the Comprehensive Center for Pain and Addiction at the University of Arizona.
