Abstract
Magnesium sulphate has been used in the acute treatment of migraines; some studies found it to be a highly effective medication in the acute control of migraine pain and associated symptoms. This randomized, double-blind, placebo-controlled study assesses the effect of magnesium sulphate on the pain and associated symptoms in patients with migraine without aura and migraine with aura. Sixty patients in each group were assigned at random to receive magnesium sulphate, 1000 mg intravenously, or 0.9% physiological saline, 10 ml. We used seven parameters of analgesic evaluation and an analogue scale to assess nausea, photophobia and phonophobia. In the migraine without aura group there was no statistically significant difference in the patients who received magnesium sulphate vs. placebo in pain relief. The analgesic therapeutic gain was 17% and number needed to treat was 5.98 at 1 h. There was also no statistical difference in relief of nausea. We did observe a significant lower intensity of photophobia and phonophobia in patients who received magnesium sulphate. In the migraine with aura group patients receiving magnesium sulphate presented a statistically significant improvement of pain and of all associated symptoms compared with controls. The analgesic therapeutic gain was 36.7% at 1 h. A smaller number of patients continued to have aura in the magnesium sulphate group compared with placebo 1 h after the administration of medication. Our data support the idea that magnesium sulphate can be used for the treatment of all symptoms in migraine with aura, or as an adjuvant therapy for associated symptoms in patients with migraine without aura.
Keywords
Introduction
Magnesium is the second most abundant cation in the intracellular fluids. It is essential for enzymatic activity as well as for neurochemical transmission and muscular excitability (1). Magnesium ion (Mg2+) influences vascular tone (2). Decrease of magnesium serum levels in experimental animals has been associated with triggering cortical spreading depression (3), with reuptake of cerebral neurotransmittors (4) and with an increase of platelet aggregation state (5). In addition, magnesium has been implicated in NMDA receptor modulation (6), in nitric oxide production (2), in substance P modulation and with the production and activation of catecholamines (7). As all of these actions may play a role in migraine pathophysiology, magnesium clinical effects are of great interest.
Magnesium, especially as magnesium sulphate, has been studied in the acute treatment of migraine and other headaches, with some studies finding it highly effective in the acute control of pain and associated symptoms (8). According to Mauskop et al. (9), refractory patients can sometimes benefit from intravenous infusions of magnesium sulphate.
We report a randomized, placebo-controlled, double-blind study evaluating the efficacy of magnesium sulphate in the acute treatment of pain and associated symptoms in patients with migraine without aura (MO) and migraine with aura (MA), as well as the possible effects of intravenous (IV) magnesium on the duration of aura.
Methods
We used the same methodological approach that we have utilized in other similar studies performed by our group (10).
Study duration and setting
The study was conducted at two public health units in the towns of Ribeira~o Preto and Sa~o Carlos, State of Sa~o Paulo, Brazil, from 1 April 1997 to 31 December 1999.
Inclusion criteria
Minimum age of 18 years.
Diagnosis of MO and MA according to the criteria of the International Headache Society (11). Only patients presenting aura during the attack under study were included in the group of migraine with aura.
Moderate to severe pain.
Agreement to participate in the study by signing an informed consent form, after approval by the Research Ethics Committee of the University Hospital, School of Medicine at Ribeira~o Preto, University of Sa~o Paulo.
Exclusion criteria
Known or reported intolerance or contraindication for magnesium sulphate.
Having taken any type of medication for that attack before presenting to the health unit for pain or any other accompanying symptoms.
Proven or assumed pregnancy.
Study design
The patients were randomized by drawing lots, 30 patients to each group (12). The sample size had been determined in another study (10), where we assumed a 25% response rate for placebo and 55% for the active drug, to work with 90% of power to detect the difference at a 5% significance level.
We first determined how long the patient had been in pain and defined this time as T0. We then applied scales for the evaluation of pain and of associated symptoms immediately before the administration of the study drug at T0 and at 30 min (Time 30 — T30) and 60 min after its administration (Time 60 — T60). The patients were questioned again, by telephone, 24 h after the administration of the study drugs.
The patients were medicated in the medication room of the health unit, where they lay down and were submitted to catheterization of a peripheral vein maintained with 0.9% physiological saline (0.9% PS), 0.5 ml (10 drops) per min. Then, according to the group to which they had been randomly assigned, they were medicated according to the following protocol:
Placebo: intravenous (i.v.) injection of 10 ml 0.9% PS.
Magnesium sulphate: i.v. injection of 1 g, diluted to 10 ml of 0.9% PS.
The study was double blind.
Parameters for the assessment of pain
Analgesia was assessed according to the parameters indicated below.
Parameter 1: pain intensity measured by a 10-point verbal-analogical scale (13).
Parameter 2: pain intensity measured by the traditional 4-point scale. 0=no pain, 1=mild pain, 2=moderate pain, 3=severe pain. Positive headache response was a patient's pain changing from 2 or 3 to 1 or 0 after study drug at particular endpoints.
Parameter 3: pain free. Pain intensity changing from 2 or 3 to 0 at predetermined time point.
Parameter 4: therapeutic gain (TG). TG was defined as the ‘active response rate – placebo response rate’. We used the endpoint efficacy (parameter 2) for calculations.
Parameter 5: number needed to treat (NNT). NNT was defined as the reciprocal of TG, i.e. 1/TG.
Parameter 6: recurrence of pain. Recurrence was considered to be present when the patient reported pain free at any time after administration of study drug with a headache returning afterwards.
Parameter 7: use of rescue medication. Use of rescue medication was noted if the patient used some type of analgesic medication before the re-evaluation performed 24 h after the administration of the study drug.
Parameters for the assessment of aura and of associated symptoms
Associated symptoms (nausea, photophobia and phonophobia) were also quantified by the patient as absent (tabulated as ‘0’), mild (‘1’), moderate (‘2’) or severe (‘3’).
The presence or absence of aura was determined at the first evaluation and at T30 and T60.
Criteria for discharge and re-evaluation
After 1 h patients who felt better were discharged even if they had received the placebo. Those whose clinical improvement was not satisfactory, with the need for further treatment, were treated with the rescue medication. Patients were re-evaluated, by phone 24 h after the administration of the substance under study. This re-evaluation consisted of the application of the same scales as applied previously.
Statistical analysis
Data were analysed statistically by descriptive statistics for all variables studied. The demographic variables were analysed by the chi-square test and by contingency tables, and the efficacy and adverse events (AEs) of the medications were compared by the non-parametric Mann–Whitney test. We considered P<0.05 as statistically significant.
The statistician responsible for the analysis worked in a blinded fashion.
The study was approved by the Research Ethics Committee of the University Hospital, School of Medicine at Ribeira~o Preto, USP.
Results
Thirty patients with MO were randomly assigned to receiving placebo. Forty per cent of them were male. Mean age was 27 years. The magnesium sulphate group consisted of 30 patients, 30.5% of them male. Mean age was 31.4 years. The female : male ratio was 1.5 for the placebo group and 2.0 for the magnesium sulphate group. The mean for T0 was 4.4 and 4.5 h. There were no significant differences between these demographics.
Similarly, 30 patients with MA were randomly assigned to receiving either placebo or magnesium sulphate. In the placebo group, 23.3% were male and mean patient age was 28.2 years. In the magnesium sulphate group, 20.0% were male and mean patient age was 27.1 years. The female : male ratio was, respectively, 3.3 and 4.0. The mean for T0 was 3.7 and 2.8 h, respectively. There was no significant difference between these variables.
Assessment of analgesia
The comparison of pain intensity evaluated by the pain scale (parameter 1) is presented in Fig. 1. Statistical difference between magnesium sulphate and placebo was only observed in those patients with MA at 60 min (T60).
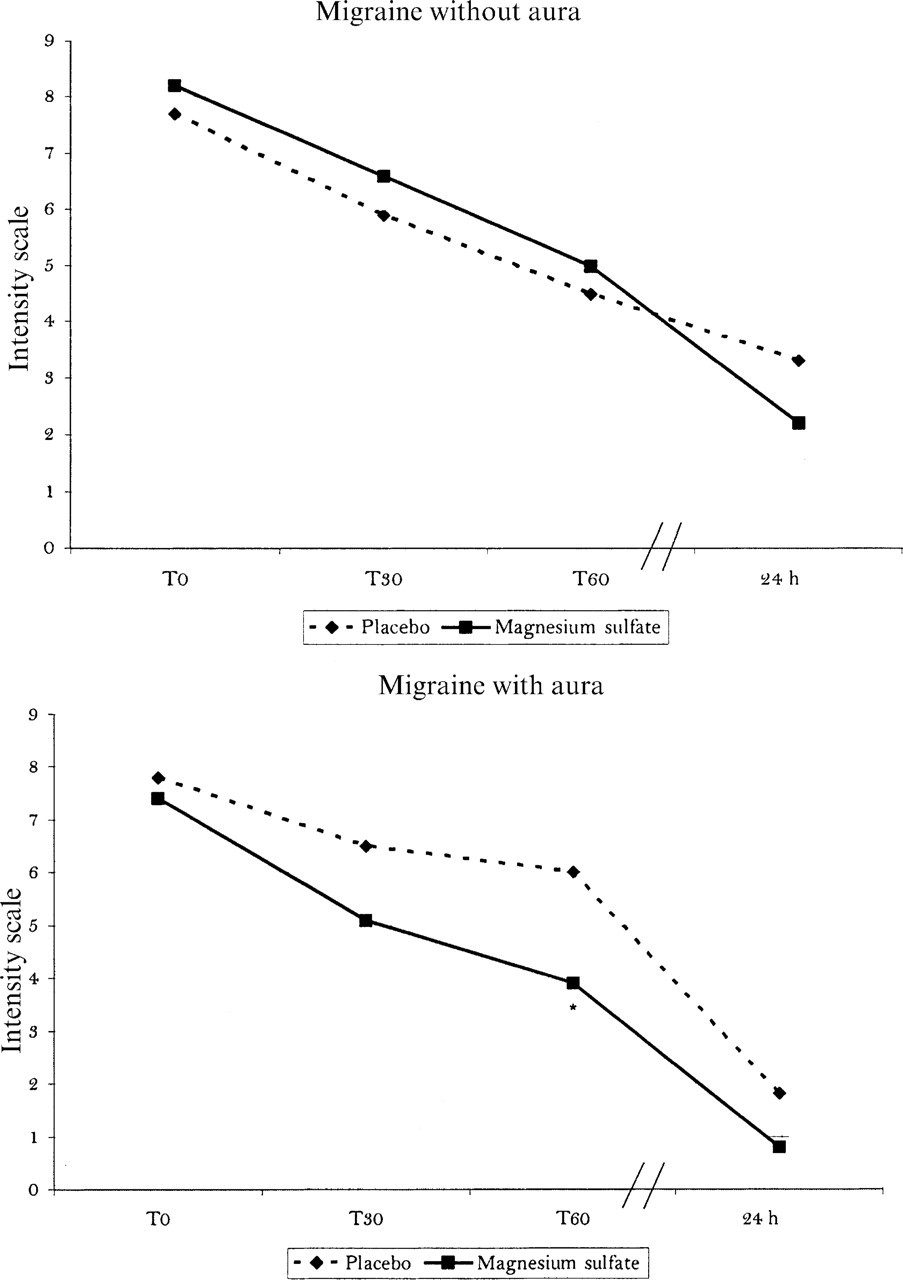
Evolution of pain intensity measured by a 10-point scale (parameter 1). Comparison between magnesium sulphate and placebo.
Table 1 presents the comparison of headache response (parameter 2) and pain free (parameter 3). We observed two different patterns: patients with MO that received magnesium sulphate showed no benefit from magnesium sulphate compared with placebo. On the other hand, there was statistical benefit for both headache response and pain-free endpoints for patients with MA at both T30 and T60 min.
Comparison of headache response (parameter 2) and pain free (parameter 3) in patients randomly assigned to receiving placebo or magnesium sulphate

∗ P<0.05.
†Patients were allowed to use rescue medication between T60 and 24 h. MgS, magnesium sulphate.
TG (parameter 4) and NNT (parameter 5) results are shown in Fig. 2. Despite the low therapeutic gain and the high number needed to treat observed in the MO group, and emphasizing that comparing the differences between patients with MA and MO was not the aim of this study, it is interesting to note a statistically significant difference in both TG and NNT between these groups both at T30 and T60.

Evolution of therapeutic gain and number needed, 30 min, 60 min and 24 h after magnesium sulphate administration.
Recurrence (parameter 6) and the need for rescue medication (parameter 7) are presented in Fig. 3. Patients that received magnesium sulphate had lower levels of recurrence than patients that received placebo in both groups. We noted lower levels of rescue medication utilization in the MA group only, which might reflect the higher efficacy of the magnesium sulphate observed in this group.
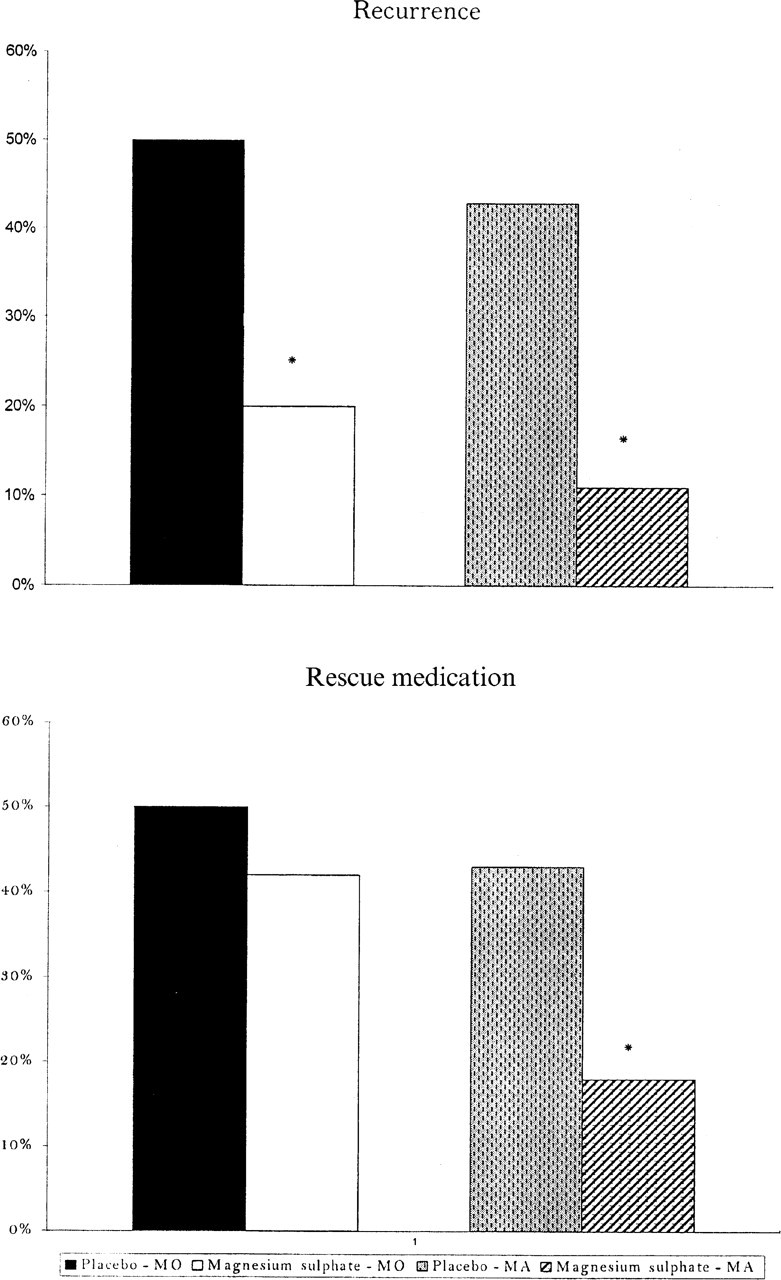
Pain recurrence and rescue medication utilization. Comparison between magnesium sulphate and placebo in the migraine with aura and migraine without aura groups.
Assessment of associated symptoms
There was no significant difference in the number of patients that received placebo or magnesium sulphate who had associated symptoms. These values were, for the MO group: nausea (76.7% for the placebo group and 72.2% in the magnesium sulphate group), photophobia (90.0% and 91.7%) and phonophobia (93.3% and 86.1%) at T0. For the MA group they were, respectively: nausea 93.3% and 90%; photophobia 83.3% and 90%; phonophobia 80% and 86.7%.
The evolution of the intensities of associated symptoms after administration of the study drug is presented in Figs 4 and 5. We observed a decrease in the mean of the intensity of photophobia and phonophobia, at T30 and T60, in patients that received magnesium sulphate, in both MO and MA groups.

Evolution of the intensity of nausea, photophobia and phonophobia in the migraine without aura group. Comparison between magnesium sulphate and placebo.

Evolution of the intensity of nausea, photophobia and phonophobia in the migraine without aura group. Comparison between magnesium sulphate and placebo.
Aura
Twenty-one patients (70%) who received placebo and 20 patients (67.7%) who received magnesium sulphate continued to have aura at T0. All patients had visual aura fulfilling the criteria of the International Headache Society (11). The percentage of patients who continued to have aura after administration of either placebo or magnesium sulphate at the time periods evaluated is presented in Fig. 6. Those patients who received magnesium sulphate had a lower likelihood to continue to have aura at 30 and 60 min compared with those who received placebo. Five patients that were randomized to receive placebo still had aura at T60, fulfilling the criteria for migraine with prolonged aura. We did not observe prolonged auras in the magnesium sulphate group.
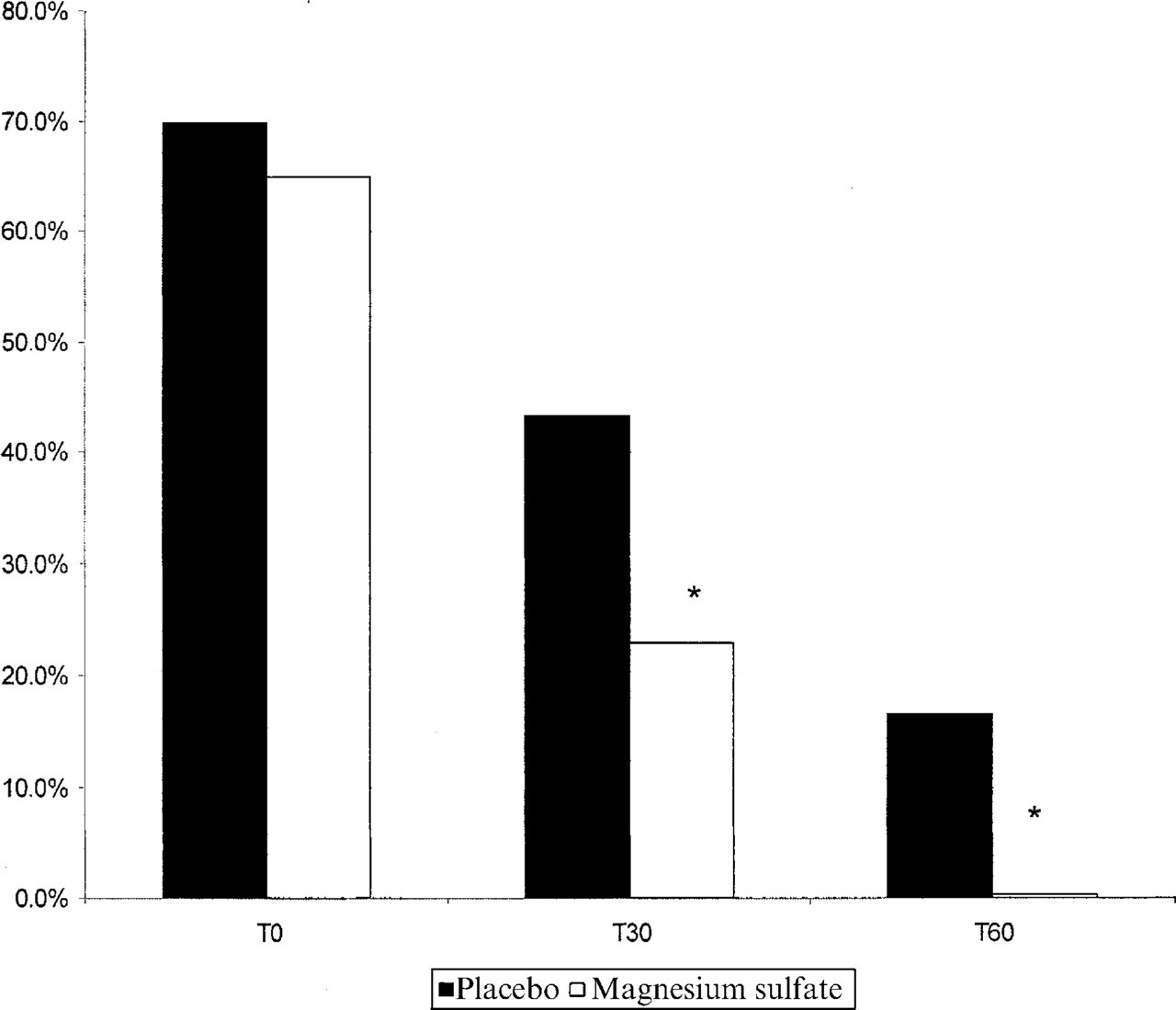
Evolution of the percentage of patients with aura, 30 min and 60 min after administration of magnesium sulphate or placebo.
Discussion
Multiple threads of research over the last 15 years have led to the concept that migraine is generated from a hyperexcitable brain (14–16) and, despite the fact that we do not know where migraine is generated, in the cortex or brainstem, one possible scenario would include a cascade of events beginning with cortical activation, followed by brainstem activation, leading to activation of ascending and descending pathways, with initiation of a perimeningeal vasodilatation and neurogenic inflammation (14, 15, 17, 18). A variety of causes for hyperexcitability of the migrainous brain have been suggested. These include low magnesium levels, mitochondrial abnormalities, dysfunctions related to nitric oxid, or a channelopathy (19, 20).
Magnesium binds to NMDA receptors to maintain calcium homeostasis (7). Low magnesium can result in opening of calcium channels, which in turn could result in increased intracellular calcium, aspartate and glutamate release and secondary alterations of extracellular potassium. This ionic change and amino acid release could initiate cortical spreading depression, eliciting the cascade of events described above (20). In addition, low magnesium levels are also associated with increased platelet aggregation, increased serotonin levels and vasoconstriction (21).
A number of studies have shown that magnesium sulphate may be effective as an acute care medication in patients with migraine attacks. One study performed by Mauskop et al. (22) showed that intravenous magnesium aborted severe migraine in those patients with low ionized magnesium, but not in those patients with normal ionized magnesium. The same group also found that magnesium sulphate could be effective for the acute treatment of cluster and other kinds of headaches (23).
Our data can be summarized as follows:
Relief of pain with IV magnesium sulphate was not different from treatment with placebo in the MO group, and was better than placebo at T60 in the MA group.
Relief of nausea followed the same pattern as pain relief.
Magnesium sulphate was highly effective in relieving photophobia and phonophobia in both MO and MA groups.
A smaller number of patients that received magnesium sulphate had aura, 30 and 60 min after its administration, when compared with placebo.
We observed a greater response, in all symptoms, in the MA group than in the MO group.
Our methodology involved the assessment of pain and associated symptom intensity at T0 and at 30 min, 60 min and 24 h after drug administration. The fact that we limited our evaluation of efficacy to only 1 h may be criticized. However, this option was dictated by the needs of the health service where the study was conducted. As this was a public health unit in a developing country, with overcrowding, excess demand and all the other problems of these clinics, we thought it would be inappropriate to maintain a patient under observation for more than 1 h unless his clinical needs required it. We tried to minimize this limitation by re-contacting the patients 24 h after drug administration, but we know these data must be interpreted with caution, as some patients may have used rescue medications prior to our call. Although the guidelines of the International Headache Society recommend an evaluation at 2 h (24), we consider our procedure to be justified, both because of the anticipated speed of i.v. treatment and for the above stated sociological reasons.
There were significant differences in the MO and MA groups in terms of response to magnesium sulphate, which may have important pathophysiological implications. A recent study performed by our group showed that after placebo administration, the amount of reduction of symptom intensity in MO patients was significantly greater than that observed in MA patients (25). Although comparison was not the primary aim of the study, it is clear that patients with MA had a better response after magnesium sulphate than patients with MO. We did not measure the ionized serum magnesium levels but if only patients with low levels respond to this therapy (23), we think that at least two possible explanations might be possible for the differences observed between the MO and MA groups: (i) we had an allocation bias, with more patients with low ionized serum magnesium levels randomized to the MA group; (ii) the ionized serum magnesium levels are in fact lower in patients with MA than in those with MO. Further studies with ionized levels drawn will be necessary in order to clarify this critical question.
The analgesic TG in MO and MA groups was, respectively: 1—T30, 6.7% and 13.4%; 2—T60, 16.7% and 36.7%; 3—24 h, 10% in both groups. TG observed in MO was very low, while the TG observed in the MA group was correspondingly quite high, comparable with the TG of sumatriptan 50 mg, which, according to several studies, is between 24% and 37% (26).
The intensity of nausea, in both groups, followed the intensity of pain. One hypothesis would be that treatment which lowered brain hyperexcitability in the MA group was therefore able to lower the intensity of both pain and nausea.
The genesis of photophobia seems to involve an interaction between the trigeminal and visual pathways (27). This hyperexcitability may be the manifestation of a derangement of the central mechanisms of neurosensory control (28). There appear to be both peripheral and central components acting on photophobia. The action of magnesium sulphate on photophobia is easily explained in primarily central terms, reducing brain hyperexcitability. In an analogous way, phonophobia may involve changes in the auditory system-trigeminal connections. A reduced cochlear threshold is observed in migraneurs (28). The action of magnesium sulphate on this symptom is similar to that proposed for photophobia.
As noted, both decreased brain magnesium and a channel disorder may be relevant in starting cortical spreading depression, the increasingly accepted basis of aura. Also as previously stated, aura is associated with a state of neuronal hyperexcitability, with increased CSF excitatory amino acids such as glutamate and aspartate, and with low levels of magnesium in CSF (14). This pathophysiological model is compatible with the action of magnesium sulphate in aura that we observed.
In conclusion, intravenous magnesium sulphate might be a reasonable option for the acute treatment of migraine with aura. It is effective for the treatment of associated symptoms in both migraine with and without aura patients, as well as of the aura itself. Our data support the idea that magnesium sulphate can be used alone, treating patients with migraine with aura, or as an adjuvant therapy for the associated symptoms in patients with migraine with and without aura.
