Abstract
This study compares the results of brushing allodynia (BA) during migraine attacks to those of self-reported cutaneous allodynia. We recruited 100 patients (20 with chronic migraine, 80 with episodic migraine) and performed a gauze-brushing test to detect BA. A previous experience of cutaneous allodynia was queried. Seventeen patients reported having allodynia before, whereas 22 patients exhibited BA. The frequency of self-reported allodynia (12.5% vs. 35.0%, p = .040) but not BA (21.3% vs. 25.0%, p = .765) differed between patients with episodic migraine and those chronic migraine. Patients with aura (N = 8) had a higher frequency of both self-reported allodynia (62.5% vs. 12.2%, p = .003) and BA (87.5% vs. 17.6%, p < .001) than those without aura (N = 92). Some allodynia symptoms, including avoidance of washing hair, touching the head, combing hair and lying on the site of head pain, were associated with BA (all p < .05). Our study showed that BA during migraine attacks correlated well with prior allodynia symptoms. The frequencies of BA and self-reported allodynia depend on the composition of different migraine subtypes and the timing of the gauze-brushing test.
Introduction
Cutaneous allodynia is common during migraine attacks (1). There are four types of cutaneous allodynia (2–4): (1) static mechanical (pressure and punctate), (2) dynamic mechanical (such as BA), (3) heat and (4) cold. They have different neural processes, involving both peripheral and central mechanisms (3–5). During the progression of an acute migraine attack, when the peripheral trigeminal nerve fibers are sensitized, the patients begin to feel throbbing pain. As the attack progresses, the second-order sensory trigeminal nucleus caudalis neurons are sensitized. The patients feel unpleasant if they comb or wash their hair, wear glasses or do any other activities that involve their face or scalp (1). Later, when the third-order neurons in the thalamus are sensitized, the patients may experience extracephalic allodynia (1,6).
Quantitative sensory testing (QST) can be used to determine static and thermal allodynia (1,7,8), whereas BA can be determined by the gauze-brushing test (9–11). Burstein et al. showed that nearly 80% of patients developed allodynia, as detected by QST, during migraine attacks (1) and that allodynia predicted a poor response to triptans in patients with migraine attacks (12). By contrast, using the gauze-brushing test, only 14% to 42.7% patients were found to develop BA during migraine attacks (2,13,14). Intriguingly, a recent Belgian study showed that headache intensity rather than BA is the best predictor of treatment efficacy (15).
A questionnaire designed to identify patient symptoms of cutaneous allodynia was validated by QST and successfully used in clinics and in field studies (8,16,17). Compared with QST, the gauze-brushing test is more convenient to perform in clinical practice. However, its relationship to patient self-reported cutaneous allodynia is not well described. In addition, most previous studies measured BA in patients with migraine during interictal periods (2). The degree of discrepancy between ictal and interictal periods is also not known. This study aimed to determine the frequency of BA and correlate it with self-reported cutaneous allodynia in Taiwanese patients during acute migraine attacks.
Methods
Patients visiting the Neurology Outpatient Clinic at the Yuan Shan Veterans Hospital or the Yi-Lan General Hospital for acute migraine or probable migraine attacks were recruited. The Institutional Review Board of the National Yang-Ming University approved this study protocol. Prior to entering the study, all patients provided written informed consent.
Patients were diagnosed according to the criteria of the International Classification of Headache Disorders, second edition (ICHD-2), 2004 (18). Only patients with migraine with (code 1.2) or without aura (1.1) or with probable migraine (code 1.6) were recruited. In addition, they were classified into episodic migraine (EM) and chronic migraine groups (CM, ≥ 15 headache days/month for ≥ three months, with ≥ eight days of migraine attacks per month) (19). We also recorded demographics and headache profiles on each of the participating patients. The headache intensity was recorded with a 0–10 verbal numeric scale.
Cutaneous allodynia questionnaire
The questionnaire was designed to identify the symptoms of cutaneous allodynia. Patients were asked to report, based on their prior experiences during migraine attacks, whether they had to avoid the following activities: combing hair, shaving, wearing glasses, washing hair, touching the head, touching the forearms, lying on the site of head pain, wearing bracelets (or watches) or washing hands. These activities were related to dynamic, pressure-related or thermal allodynia. The patients answered ‘yes’ or ‘no’ to these questions. If the patients answered that they had to avoid at least one of the above activities during migraine attacks, they were classified into the self-reported allodynia group; otherwise, they were placed in the non-allodynia group.
Brushing Allodynia
The gauze-brushing test was done in the clinic. We applied a 4-by-4-inch gauze diagonally folded twice to brush the bilateral chin, forehead and forearms of the patient with the frequency about 1–2 Hz 10 times (2). The patient was asked if he or she felt any discomfort during the brushing. If the answer was yes (over either forehead, chin or forearms), the response to BA was defined as ‘positive’; otherwise, the response was defined as ‘negative’.
Statistics
Descriptive statistics were presented as means ± SD or percentages. Means were compared using the unpaired t-test, and proportions were compared using the Chi-square or Fisher’s exact tests when appropriate. All calculated p values were two-tailed and statistical significance was defined as a p value of less than. 05.
Results
Headache profile of all study patients a
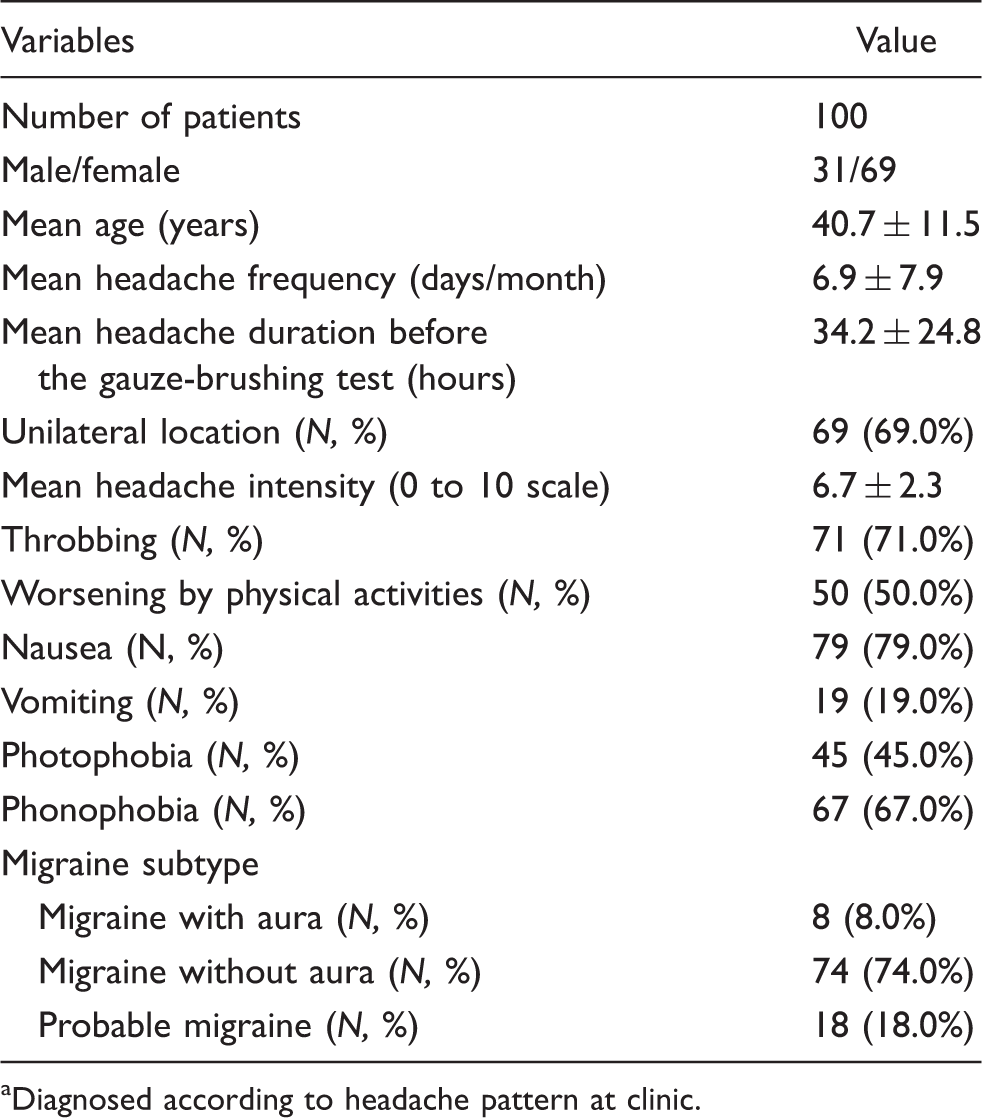
Diagnosed according to headache pattern at clinic.
Seventeen (17%) patients reported having previously experienced at least one occurrence of cutaneous allodynia during migraine attacks. Patients with CM (N = 7, 35%) had a higher frequency than those with EM (N = 10, 12.5%, p = .04). The proportions of different allodynia symptoms did not differ between the CM and EM patients. Patients with migraine with aura had a higher proportion of self-reported cutaneous allodynia in comparison with those with migraine without aura or those with probable migraine (62.5% vs. 12.2% vs. 16.7%, p = .001).
Twenty-two (22%) patients exhibited BA with the gauze-brushing test. The frequencies of BA were similar between patients with EM and those with CM (21.3% vs. 25.0%, p = .765). The patients with migraine with aura were more likely to have BA than those with migraine without aura or those with probable migraine (87.5% vs. 17.6% vs. 11.1%, p < .001). Analysis of the other variables in demographics or headache profile did not demonstrate differences between patients with and without BA (data not shown).
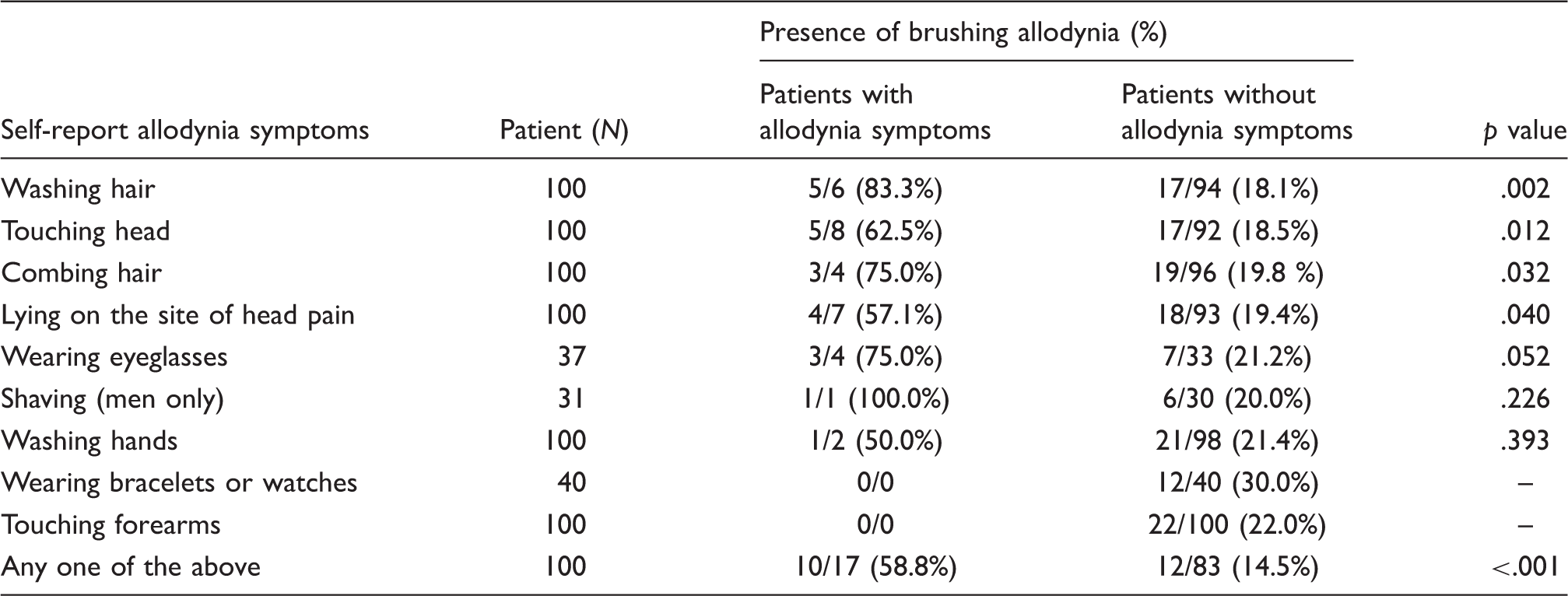
Presence of brushing allodynia in patients with or without self-reported allodynia symptoms
Discussion
Our study showed 22% of migraine patients developed BA during a migraine attack. The frequency of BA was higher in patients with migraine with aura (87.5%) but did not differ between patients with EM and those with CM. Of note, we found that patients with migraine with aura or with CM were at higher risk to experience and report allodynia. The frequency of BA in this study was similar to that reported in previous studies (14%–42.7%) (2,13,14). However, the frequencies of BA were lower than those of allodynia detected by QST (75.3%–79%) in prior studies (1,8). The frequency of self-reported allodynia for our questionnaire was low (17.0%). This may be in part related to the questionnaire design (i.e., we asked if the patients had ever ‘avoided’ rather than ‘felt pain or unpleasant’ (8,16) from certain activities during migraine attacks).
Our study showed an association between BA and self-reported allodynia. About 59% patients with self-reported allodynia demonstrated BA, whereas about 15% of patients without self-reported allodynia did. These relevant activities, ‘washing hair’, ’touching head’ and ‘combing hair’, not only represent dynamic allodynia but also are located in the cranial region, where allodynia occurs more frequently. In contrast, ‘lying on the site of head pain’, which is considered a symptom of pressure allodynia, was also related to BA. It is known that pressure allodynia is mediated by nociceptors with C-fiber, whereas, BA is governed by A β-mechanoreceptive fibers (5,20–22). In line with prior studies, different forms of allodynia, such as pressure allodynia and BA, can occur together after occurrence of central sensitization (2–4). Our results support this finding and suggest that different types of allodynia may have different underlying mechanisms but may share some common pathways (2). Of note, a component of thermal allodynia should also be considered in migraine patients who avoid washing their hair or hands during migraine attacks.
Several studies have shown that migraine aura was associated with cutaneous allodynia (2,13,17). Our study also showed patients with migraine with aura had a higher frequency of BA than those without aura. However, patients with migraine with aura accounted for 8% of our participants, which may in part explain the lower frequency of BA in this study. According to LoPinto et al., (2) the frequency of BA was higher in transformed migraine patients than in EM patients (46.7% vs. 16.0%, p = .02) during interictal periods, but the presence of BA correlated well with self-reported allodynic symptoms in their study. However, another study reported that when patients with EM were tested during an acute attack, the frequency of BA increased significantly (60% vs. 9.9%) (9). Because our study was done during migraine attacks, the frequency of BA was similar in the CM and EM groups. Therefore, the frequency of BA and self-reported allodynia symptoms depend on the sample composition of different migraine subtypes and the timing of the tests.
Our study had limitations. First, the setting of this study was a primary care clinic; therefore, extrapolation to the general population or another clinical setting should be made with caution. Second, our allodynia questionnaire might have thus been restrictive and have reduced the sensitivity in the assessment of cutaneous allodynia. Third, this study was cross-sectional in design. The mean duration from headache onset to performance of the gauze-brushing test was around 30 hours; it is possible that development of BA diminished during the interim period before the gauze-brushing test.
Footnotes
Acknowledgements
This study was supported in part by grants from the National Science Council of Taiwan (97-2628-B-010-007-MY3), Taipei Veterans General Hospital, and Yuan-Shan Veterans Hospital.
