Abstract
Background
Migraine is associated with obesity. These analyses evaluated weight change with atogepant used as a preventive migraine treatment.
Methods
Five atogepant clinical trials in adults with migraine (one phase 2b/3; four phase 3) were included: Three 12-week, randomized, placebo-controlled trials (episodic migraine: two; chronic migraine: one); one 40-week, open-label extension trial and one 52-week, standard care, randomized, long-term safety trial in episodic migraine. Change from baseline in body weight was measured.
Results
Mean baseline body mass indexes were 30.0–30.7 kg/m2 (pooled episodic migraine [United States only]) and 25.0–25.5 kg/m2 (chronic migraine [East Asia, Europe, and North America]). More participants treated with atogepant 60 mg once-daily compared to placebo experienced ≥7% weight loss at any time in the pooled episodic migraine placebo-controlled trials (4.9% vs. 2.8%), chronic migraine placebo-controlled trial (5.8% vs. 2.0%), and pooled open-label extension and long-term safety trials (24.0% vs.14.7% in standard care [long-term safety only]). In the placebo-controlled trials, weight loss with atogepant 60 mg once-daily was observed at week 2 (pooled episodic migraine: −0.32%; chronic migraine: −0.39%), increasing at week 12 (pooled episodic migraine: −1.02%; chronic migraine: −1.50%); compared to weight gain with placebo at week 12 (pooled episodic migraine: +0.49%; chronic migraine: +0.10%). In the long-term episodic migraine studies, weight loss with atogepant 60 mg once-daily was observed at week 4 (long-term safety: −0.42%; open-label extension: −0.76%), increasing at week 40 (long-term safety: −2.38%; open-label extension: −2.09%).
Conclusion
Atogepant was associated with modest dose- and duration-dependent weight loss.
Trial registration
ClinicalTrials.gov identifiers: NCT02848326 (CGP-MD-01); NCT03777059 (3101-301-002); NCT03700320 (long-term safety trial); NCT03939312 (open-label extension trial); NCT03855137 (3101-303-002).
Introduction
Migraine is a common neurologic disease that is associated with a substantial burden (1,–3). The risk of migraine is greater in those with obesity (body mass index [BMI] ≥ 30 kg/m2) than those with a BMI between 18.5–24.9 kg/m2 (4,–7). Additionally, those with migraine and obesity experiencing weight loss from surgical or nonsurgical interventions may also experience decreased attack frequency and severity (8,9). Epidemiologic studies have shown that the risk of migraine is increased by 27% in those with obesity (6). Compounding their negative impact, migraine and obesity are also associated with higher risks of other chronic medical conditions impacted by weight gain, including cardiovascular disease (10,–12). Therefore, optimizing migraine treatment outcomes includes understanding the impact of migraine treatments on body weight.
Basic research has suggested that calcitonin gene–related peptide (CGRP) is an important cofactor in the pathophysiology of both migraine and obesity (13). CGRP is involved in processes related to feeding behavior and metabolic functions, and elevated CGRP levels have been observed in obesity as well as migraine (14). When administered exogenously to mice, CGRP has been shown to decrease food intake and limit energy expenditure (15). However, α-CGRP knockout mice also experience improved metabolic function and possibly weight loss, suggesting the need for additional research to clarify this relationship (16). The hormone amylin is closely related to CGRP and also associated with migraine and obesity (17). Future research should explore the effects of CGRP on the central and peripheral nervous systems related to maintaining body weight and understanding the metabolic effects of CGRP in the pancreas, adipose tissue, and muscle (13).
Atogepant is an orally administered, small-molecule CGRP receptor antagonist indicated for the preventive treatment of migraine based on results of placebo-controlled trials in episodic migraine (EM) and chronic migraine (CM), and long-term EM studies (Online Supplemental Figure 1) (18,–22). In these studies, weight loss was consistently observed with atogepant, an observation that has not been reported with CGRP-targeted monoclonal antibodies. The current analyses evaluated the magnitude and time course of weight loss associated with using atogepant as a preventive migraine treatment.
Methods
Study design
These analyses assessed weight loss associated with atogepant using data from five clinical trials: a phase 2b/3 randomized, placebo-controlled trial (Study CGP-MD-01) (18), a phase 3 randomized, placebo-controlled trial (3101-301-002 [ADVANCE]) (19), a long-term safety (LTS) trial (3101-302-002) (20), and an open-label extension (OLE) trial (3101-309-002) (21) in people with EM, and a phase 3 randomized, placebo-controlled trial in people with CM (3101-303-002 [PROGRESS]) (22). Participants in the safety population with available baseline weight data and ≥1 post-baseline assessment during the study were included.
Study CGP-MD-01 evaluated atogepant 10, 30, and 60 mg once-daily and 30 and 60 mg twice-daily versus placebo. In the other EM placebo-controlled trial (3101-301-002), participants received atogepant 10, 30, and 60 mg once-daily versus placebo. Participants who completed CGP-MD-01 and 3101-301-002 were eligible for the 52-week LTS and 40-week OLE trials, respectively. The LTS trial also enrolled individuals who had not participated in the lead-in study (de novo participants). The LTS and OLE trials assessed the safety and tolerability of atogepant 60 mg once-daily. In the 52-week LTS trial only, an oral standard care (SC) arm was included to provide comparative data. Participants randomized to SC were permitted to use safe and effective preventive migraine treatments, which could be changed or discontinued, while allowing the participant to continue in the study. In the CM placebo-controlled trial (3101-303-002), the efficacy and safety of atogepant 30 mg twice-daily and 60 mg once-daily versus placebo were evaluated. The EM studies were conducted in the United States (US), while the CM study enrolled participants from North America, Europe, and East Asia (30–35% each). The current weight loss analyses focus on the following doses of atogepant: 10, 30, and 60 mg once-daily.
Eligible participants were adults with ≥1-year history of either migraine with or without aura or CM, diagnosed according to the International Classification of Headache Disorders, 3rd edition (23). Eligible EM participants had 4–14 migraine days per month. Eligible CM participants had ≥15 headache days per month, including ≥8 migraine days. Additional inclusion and exclusion criteria are reported in the corresponding publications (18,–22).
All studies received approval from an institutional review board or independent ethics committee for each participating study center and were conducted in compliance with the Declaration of Helsinki and the International Council for Harmonisation Guidelines for Good Clinical Practice. All participants provided written informed consent before any study procedures were conducted.
Outcomes
The percent and kilogram changes from baseline in body weight across the study period were the main endpoints of interest and were assessed using data from the EM placebo-controlled trials (pooled), CM placebo-controlled trial, and LTS and OLE EM trials. For these outcomes, the long-term trials were assessed individually due to differences in baseline assessment points between rollover and de novo participants in the LTS trial. Because sex and race are associated with migraine and obesity (4,–6), subgroup analyses were conducted for percent and kilogram change from baseline in body weight to assess whether sex and race had an impact on these results.
Cumulative distribution function (CDF) plots of percent change in body weight from baseline to week 12 (EM [pooled] and CM placebo-controlled trials), week 40 (OLE study), and week 52 (LTS study) were calculated. CDF plots graphically represent the percent change from baseline in body weight at the end of treatment (horizontal axis) and the cumulative percent of participants experiencing up to that percent change (vertical axis). The last observation carried forward (LOCF) method was used to account for missing data.
The proportions of participants experiencing ≥7% weight loss at any time during the study period in the pooled EM placebo-controlled trials, CM placebo-controlled trial, and pooled LTS and OLE EM studies were calculated based on non-missing data. Point estimates and two-sided 95% exact CIs based on the Clopper-Pearson method are provided for incidence rates of ≥7% weight loss at any time with atogepant for baseline BMI, sex, and race. The 95% CIs were used to compare the incidence rates to those with <7% weight loss. If the 95% CIs cover the incidence rate in the reference group, no significant differences can be inferred.
A ≥ 5% threshold for weight loss at the end of treatment is a common efficacy endpoint in weight loss trials (24,25). In these five atogepant clinical trials, ≥ 7% weight loss and gain were used as a potentially clinically significant threshold for safety monitoring. For these analyses, ≥ 7% was also used as a more stringent threshold for assessing weight loss. To provide a more thorough assessment of the degree of weight loss observed with atogepant, ≥ 5% weight loss and gain were also evaluated.
Correlation analyses assessed whether certain patient characteristics (e.g., baseline BMI, efficacy endpoints) were related to weight change. Mean changes in body weight and incidence rates of ≥7% weight loss and gain were evaluated by BMI category: underweight: < 18.5 kg/m2; normal: ≥ 18.5 to <25 kg/m2; overweight: ≥ 25 to <30 kg/m2; obese: ≥ 30 kg/m2. Pearson correlation coefficients were calculated using the overall atogepant population to assess potential relationships between weight change and baseline BMI, and change from baseline in monthly migraine days (MMDs), patient-rated treatment satisfaction, and functional outcome measures. Assessments of change from baseline in monthly headache days (MHDs) and MMDs, and ≥50% reduction in three-month average of MMDs by BMI category for the pooled EM placebo-controlled trials and CM placebo-controlled trial were also conducted. To assess the potential relationship between potential weight loss-related adverse events (AEs) (decreased appetite, constipation, nausea) and ≥7% weight loss at any time, the Clopper-Pearson method was used.
Results
Participants
In the placebo-controlled trials, 1550 participants with EM and 516 participants with CM were analyzed (Table 1). Pooled analyses of 1424 participants from the long-term studies (LTS: n = 543; OLE: n = 685; SC [LTS only]: n = 196) were conducted for appropriate endpoints (e.g., CDF, ≥ 7% weight loss at any time). The mean (SD) baseline BMI for atogepant 60 mg once-daily was 30.0 (7.5) vs 30.6 (8.2) kg/m2 for placebo in the pooled EM placebo-controlled trials, 30.6 (7.7) kg/m2 in the pooled long-term EM trials vs 30.6 (8.0) kg/m2 for SC (LTS only), and 25.0 (5.5) kg/m2 vs 25.5 (6.0) kg/m2 for placebo in the CM placebo-controlled trial, indicating that the average BMI met the criteria for overweight or obese (26). The 10 mg and 30 mg once-daily atogepant groups had similar baseline BMI, and other demographic characteristics, compared with the 60 mg group (Table 1).
Baseline demographic characteristics.
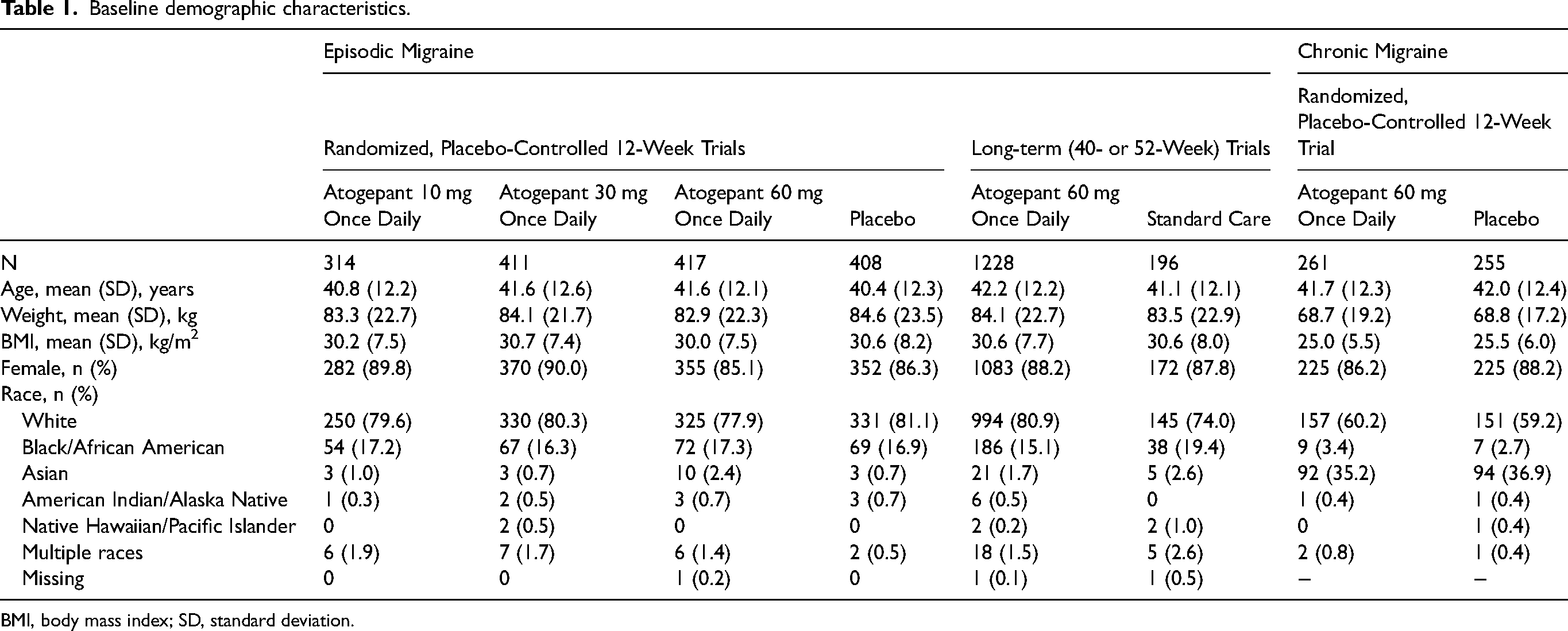
BMI, body mass index; SD, standard deviation.
Outcomes
Percent and time course of change in body weight
The magnitude and time course of weight loss with atogepant were similar for EM and CM. In the pooled EM placebo-controlled trials, atogepant was associated with dose-dependent weight loss beginning at week 2 (10 mg: + 0.09%; 30 mg: −0.29%; 60 mg: −0.32%) that increased in magnitude at week 12 (10 mg: + 0.12%; 30 mg: −0.51%; 60 mg: −1.02%) (Figure 1A). In comparison, the placebo group experienced weight gain (+0.49%) at week 12. In the CM placebo-controlled trial, atogepant 60 mg once-daily was also associated with weight loss at week 2 (−0.39%) that increased at week 12 (−1.50%) compared with +0.10% for placebo (Figure 1B).
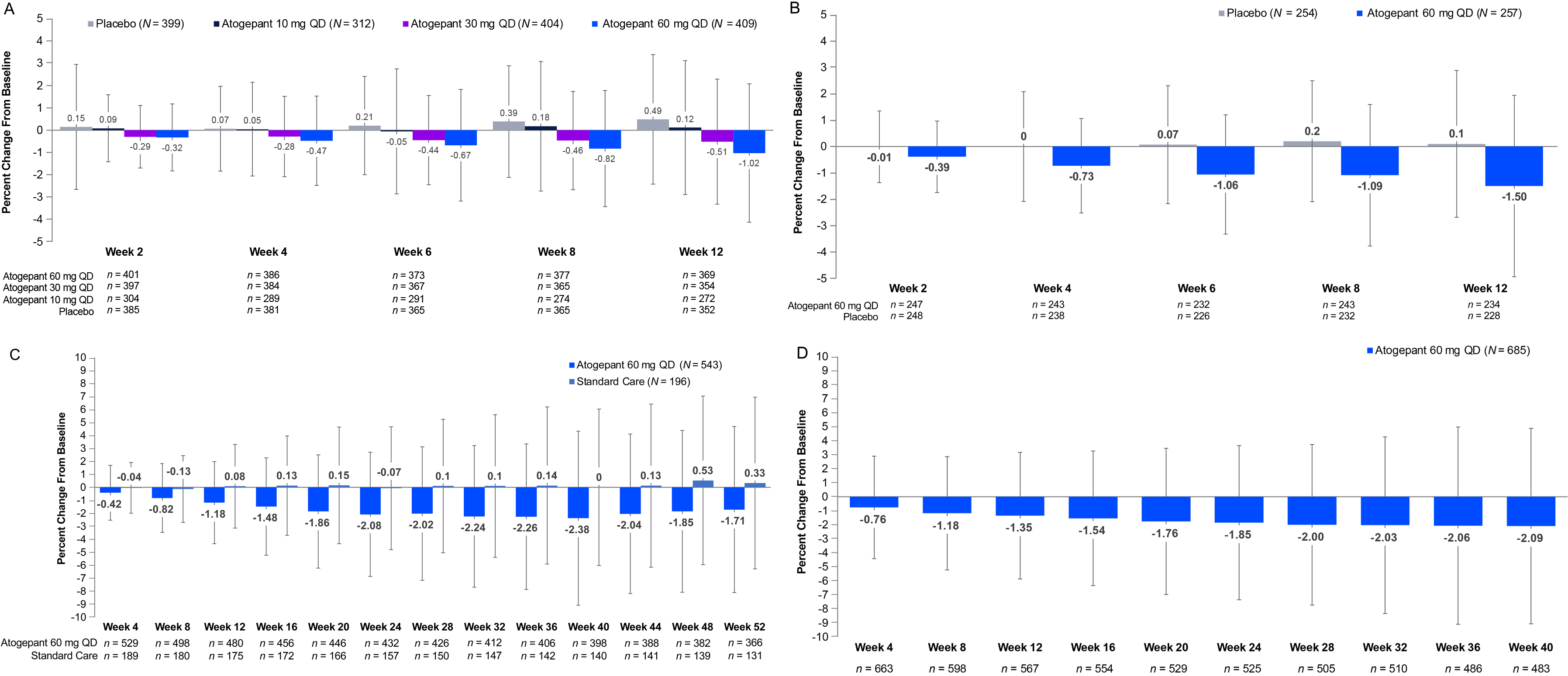
Percent change from baseline in body weight and time course of weight change with atogepant: (A) pooled EM placebo-controlled trials, (B) CM placebo-controlled trial, (C) EM long-term safety trial, and (D) EM open-label extension trial. N, number of patients in the safety population with available baseline and post-baseline weight data; n, number of patients with available data at each visit. CM, chronic migraine; EM, episodic migraine; LTS, long-term safety; OLE, open-label extension; QD, once daily.
The weight loss observed in the long-term studies overall was consistent with the placebo-controlled trials. Atogepant 60 mg once-daily was associated with duration-dependent weight loss beginning at week 4 in the LTS (−0.42%) and OLE (−0.76%) studies (Figure 1C-D). At week 40 of the OLE study, the percent change from baseline in weight for atogepant 60 mg once-daily was −2.09%. In the LTS study, the percent change from baseline was −2.38% at week 40 and remained approximately stable at week 52 (−1.71%).
Weight loss expressed as absolute changes in kilograms is presented in Online Supplemental Figure 2A-D. Weight loss seen with atogepant 30 mg twice-daily in CGP-MD-01 and the CM placebo-controlled trial was similar to that seen with 60 mg once-daily (data not shown). The subgroup analyses of change from baseline in body weight by race and sex found no noticeable differences (Online Supplemental Table 1).
Cumulative distribution function of change in body weight
The CDF plots for each atogepant dose are shown in Figure 2A-C. The pooled EM graph is shifted to the left with atogepant compared with placebo at every level of percent weight loss indicating a dose-dependent effect (Figure 2A).
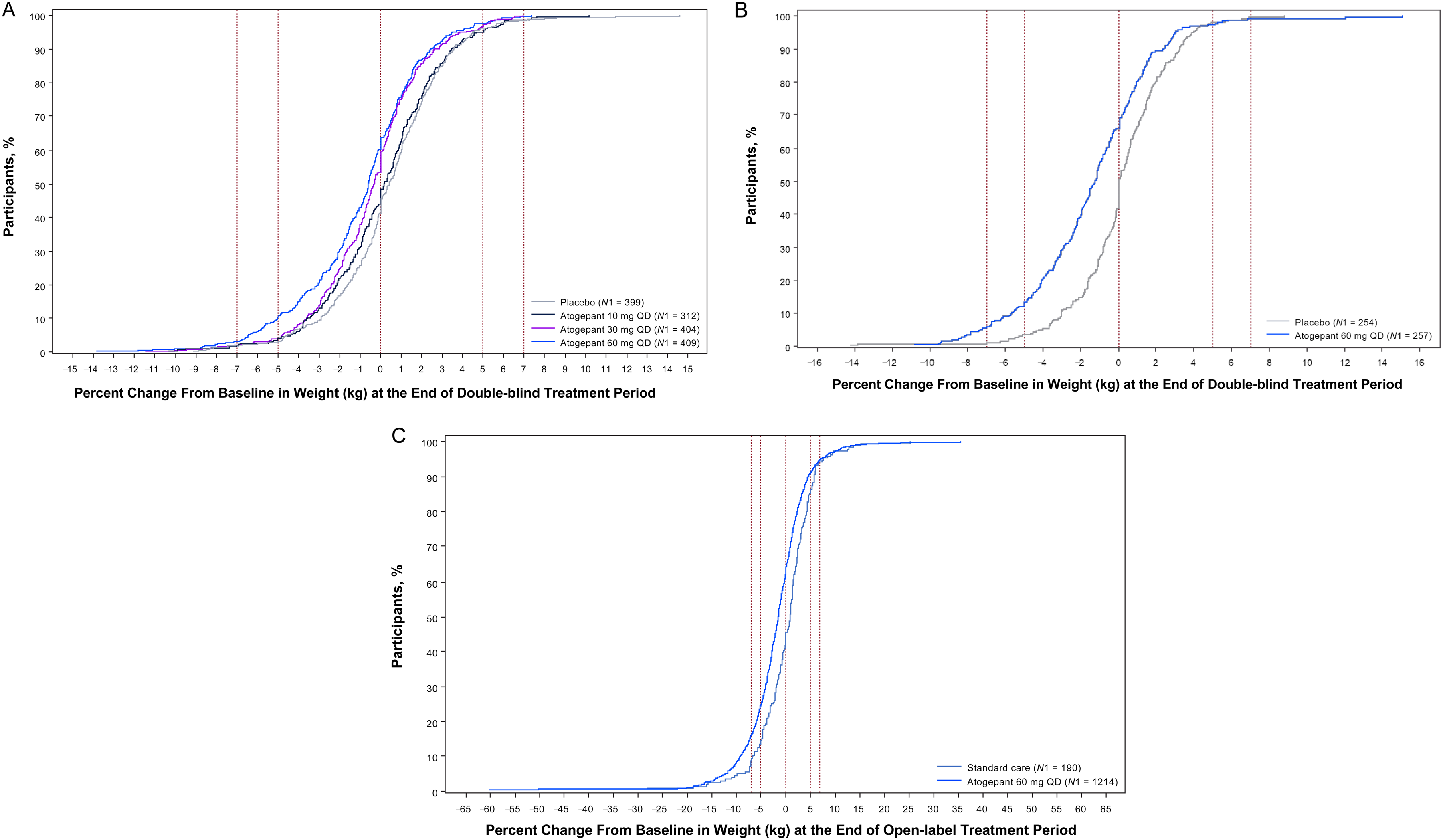
Cumulative distribution function plots for percent change from baseline in body weight (LOCF): (A) pooled EM placebo-controlled trials (week 12), (B) CM placebo-controlled trial (week 12), and (C) pooled EM long-term safety trials (weeks 40 or 52). N1 = Number of participants with available baseline values and ≥1 post-baseline assessment during the double-blind (A and B) and open-label (C) treatment periods in the safety population. Vertical lines represent 7%, 5%, and 0% weight gain and loss. CM, chronic migraine; EM, episodic migraine; LOCF, last observation carried forward; QD, once daily.
Participants with ≥7% weight loss
In the pooled EM placebo-controlled trials, the proportions of individuals experiencing ≥7% weight loss at any time also suggests dose-dependency (Figure 3A). The proportions of participants in the pooled EM placebo-controlled trials with ≥7% weight loss at any time who received atogepant 10, 30, and 60 mg once-daily were 3.8% (12/312), 3.2% (13/404), and 4.9% (20/409), respectively, compared with 2.8% (11/399) for placebo. Similar proportions of individuals experienced ≥7% weight loss at any time in the CM placebo-controlled trial (Figure 3B). In the pooled LTS and OLE trials, the proportions of participants with ≥7% weight loss at any time was 24.0% (291/1214) for atogepant 60 mg once-daily compared with 14.7% (28/190) for SC in the LTS trial. The proportions of individuals experiencing ≥5% weight loss with atogepant at study end are presented in Online Supplemental Figure 3.
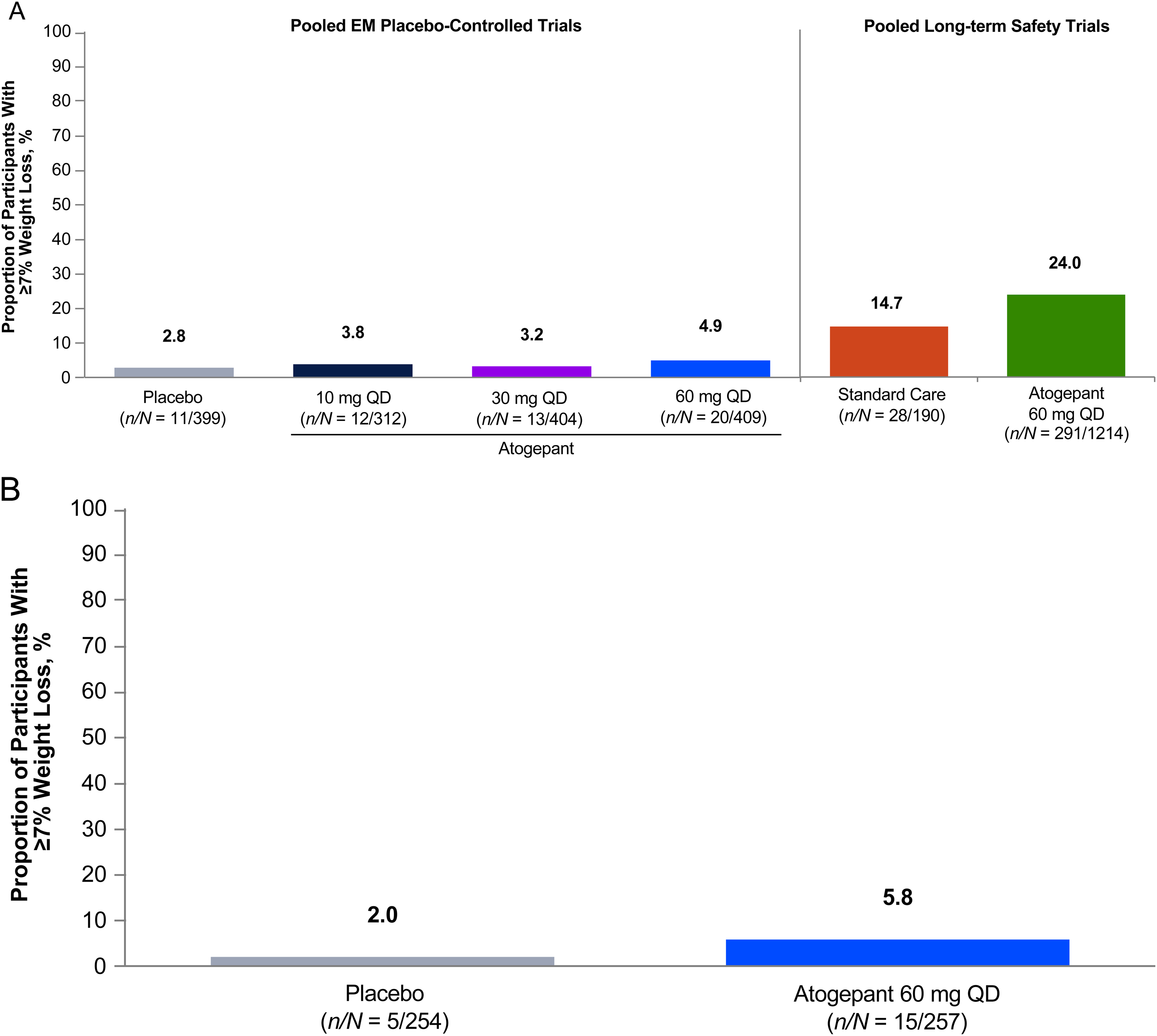
Proportions of participants with ≥7% weight loss at any time during the treatment period: (A) pooled EM placebo-controlled and long-term safety trials and (B) CM placebo-controlled trial. Percentages are calculated based on number of participants with non-PCS baseline values and ≥1 postbaseline assessment. CM, chronic migraine; EM, episodic migraine; PCS, potentially clinically significant; QD, once daily.
Participants with ≥7% weight gain
Atogepant was not generally associated with potentially clinically significant weight gain, and the proportions were lower than in the applicable control groups. The percentages of participants in the EM and CM placebo-controlled trials experiencing ≥7% weight gain at any time were 0–2% for atogepant 10, 30, 60 mg once-daily compared with 1–3% for placebo (Online Supplemental Table 2). In the LTS and OLE trials, 7.3% and 9.2% of participants receiving atogepant 60 mg once-daily experienced ≥7% weight gain at any time, respectively, compared with 12.6% for SC (LTS only).
Correlation analyses of ≥7% weight loss and baseline BMI subgroups
Across all studies, there was a trend toward higher proportions of individuals with healthy weight, overweight, and obesity experiencing ≥7% weight loss at any time compared with those who were underweight (Online Supplemental Table 3). In the EM placebo-controlled trials, participants receiving atogepant 60 mg once-daily who were underweight (<18.5 kg/m2; n = 4) experienced an increase in body weight (+4.41%) at week 12. However, those with BMIs in the normal range (≥18.5 to <25 kg/m2; n = 107) experienced −0.54% weight loss at week 12, while those with a BMI indicating overweight (≥25 to <30 kg/m2; n = 103) or obesity (≥30 kg/m2; n = 155) experienced higher degrees of weight loss (−1.64% and −1.09%, respectively). In the long-term studies, a similar trend was observed: underweight participants tended to gain weight and those who were normal weight, overweight, and obese lost weight. In the CM placebo-controlled trial, underweight participants lost weight (atogepant 60 mg once-daily: −2.62% [n = 8]; placebo: −1.31% [n = 7]) along with the other BMI subgroups. However, the small sample size led to wide variance, impeding drawing conclusions.
No underweight participants experienced ≥7% weight loss at any time in the EM and CM placebo-controlled trials, and one participant who was underweight experienced ≥7% weight loss at any time in the LTS and OLE trials. Furthermore, the percentage of participants with ≥7% weight gain at any time in the overweight and obese populations was ≤1% in the EM and CM placebo-controlled trials (Online Supplemental Table 3).
Correlation between ≥7% weight loss and AEs
For the pooled EM placebo-controlled trials, the rates of AEs for those with ≥7% or <7% weight loss at any time were similar (Online Supplemental Table 4A). However, in the CM placebo-controlled trial and pooled LTS and OLE EM studies, the incidence of decreased appetite was slightly higher in participants with ≥7% weight loss at any time (Online Supplemental Table 4B-C).
Correlation between change from baseline in weight and efficacy and functional outcomes
In the pooled EM and CM placebo-controlled trials, there was no evidence of a correlation between baseline BMI and change in body weight with atogepant 60 mg once-daily (Online Supplemental Figure 4). In the pooled long-term studies, there was a weak negative correlation between baseline BMI and percent (Pearson r = −0.072; P = 0.013) and mean (Pearson r = −0.129; P < 0.001) change in body weight in the long-term studies.
In the pooled EM placebo-controlled trials only, there was a weak positive correlation between change from baseline in MMDs (Pearson r = 0.067; P = 0.009) and body weight (Online Supplemental Table 5). However, as the magnitude of correlation coefficient is very small, we are unable to draw clear conclusions. In the pooled EM placebo-controlled trials and CM placebo-controlled trial, participants receiving atogepant across all BMI groups experienced similar degrees of change from baseline in MMDs and MHDs, and proportions experiencing ≥50% reductions in three-month average MMDs (data not shown). Weak positive correlations were also observed between change from baseline in body weight and patient satisfaction in the pooled EM placebo-controlled trials, CM placebo-controlled trial, and LTS trial.
Correlation analyses did not observe consistent relationships among change in body weight and Activity Impairment in Migraine-Diary (AIM-D) Physical Impairment and Performance of Daily Activities scales, Headache Impact Test-6 total score, and Role Function–Restrictive (RFR) score of the Migraine-Specific Quality of Life questionnaire (MSQ) v2.1. However, in the LTS and CM placebo-controlled trials, there were weak correlations between change from baseline in MSQ v2.1 RFR and body weight.
Discussion
These analyses showed consistent, modest, dose- and duration-dependent weight loss with atogepant when used as a preventive migraine treatment during 12-week EM and CM placebo-controlled trials and 40- and 52-week long-term studies. Atogepant 60 mg once-daily was associated with the greatest degree of weight loss, which was observed as early as week 2 in the placebo-controlled trials (pooled EM: −0.32%; CM: −0.39%) and week 4 of the LTS (−0.42%) and OLE (−0.76%) trials. At week 12, the degree of weight loss increased in the placebo-controlled trials (pooled EM: −1.02%; CM: −1.50%). Weight loss was −2.09% at week 40 (OLE) and −1.71% at week 52 (LTS). In all five trials, the proportions of participants experiencing ≥7% weight loss at any time were greater for atogepant 60 mg once-daily (pooled EM: 4.9%; CM: 5.8%; pooled long-term: 24.0%) compared with placebo and SC (LTS only). Although at the population level, the mean weight loss may be modest, for some individuals the degree of weight loss may be clinically significant.
The association between atogepant treatment and weight loss is a novel finding, as weight loss has not been reported with other CGRP-targeted preventive migraine treatments. Notably, the observed weight loss did not show any clear correlations with related AEs (e.g., nausea, constipation), efficacy of treatment (e.g., reduction in MMDs), or change in physical activity (e.g., AIM-D and MSQ subscales); however, participants treated with atogepant 30 or 60 mg once-daily experienced higher degrees of weight loss than the atogepant 10 mg once-daily group. While these dose-dependent effects on weight loss suggest a pharmacologic mechanism producing this effect, the underlying mechanism of weight loss is unknown. It is unclear whether the weight loss observed in our clinical trial data is a direct result of CGRP antagonism or a function of downstream modulation of obesity-related proteins. Pharmacological characterization of atogepant antagonism suggests that human adipocytes express a functional CGRP receptor and atogepant exhibits amylin-1 receptor antagonism (27,28). Further research is needed to more fully characterize the mechanism underlying the weight loss observed with atogepant.
The relationship between CGRP and migraine and obesity is not fully understood, and thoroughly reviewing that research is beyond the scope of this publication. However, this link has been suggested to involve various factors, including feeding behavior, insulin resistance, and low-grade systemic inflammation associated with obesity (5,12,14,29). For example, CGRP modulates neuronal circuits in the parabrachial nucleus and hypothalamus that are involved with appetite control, which affects food intake and meal size (29). Glucose metabolism and insulin sensitivity are also affected by CGRP activity and, as a result, CGRP has been shown to reduce diet-induced obesity (16). Lastly, obesity-related chronic inflammation can increase CGRP levels and lower the activation threshold for trigeminal neurons and increase migraine frequency in at-risk individuals (14).
Commonly used preventive migraine treatments are often associated with treatment-emergent weight gain, which may reduce compliance and lead to discontinuation. Specifically, amitriptyline and valproate have been shown to cause weight gain of up to 7 kg when used as preventive migraine treatments (30). A meta-analysis reported that the relative risk of weight gain with tricyclic antidepressants for treating headache was 1.69 (95% CI: 0.74, 3.88) compared with placebo (31). Weight gain is among the most common AEs with the calcium channel blocker flunarizine (number needed to harm: 6) (32). Weight gain has been reported with some CGRP-targeted treatments (e.g., erenumab, galcanezumab) but was not considered clinically relevant by the authors due to the similar rates observed with placebo and low incidence (33,34). Obesity has also been shown to be a negative predictor of response to some CGRP mAbs, which has been suggested to involve the relationship between CGRP and migraine, particularly CM, and the increased CGRP levels observed in obesity (14,35). Our findings demonstrated that the EM and CM placebo-controlled trial populations treated with atogepant had similar efficacy across BMI categories.
In contrast to other preventive migraine treatments, topiramate, an anticonvulsant indicated for preventive migraine treatment in people ≥12 years, is associated with weight loss (−2.3% to −3.8% from baseline) (36–38). Among individuals receiving topiramate as a preventive migraine treatment, anorexia is among the most common AEs, which could contribute to weight loss. Although our findings demonstrate weight loss with atogepant, no underweight participants in the EM and CM placebo-controlled trials and only one underweight participant in the LTS trial experienced ≥7% weight loss.
There are some notable limitations to these analyses, including the fact that these clinical trials were not primarily designed to assess the impact of treatment on weight. Due to this, assessments of overall physical activity and caloric intake were not included; however, performance of daily activities and degree of functional restriction were captured. Safety assessments, including weight measurements, were also conducted at each visit and analyses of change from baseline in weight were prespecified. Similarly, the proportion of participants who experienced ≥7% weight loss or gain at any time post-baseline was prespecified as an analysis of potentially clinically significant weight change for safety monitoring. This cutoff has also been previously used in a controlled trial of a lifestyle-based weight-loss intervention and a galcanezumab migraine clinical trial (34,39). Weight-loss interventions use ≥5% to define clinically meaningful weight loss (24,25); however, in the current analyses, 7% was thought to provide a more stringent threshold for assessing weight loss. Finally, the BMI subgroup analyses are limited by the small populations that were analyzed, particularly in the underweight group.
Despite these limitations, these analyses have numerous strengths, including the consistent observation of weight loss across five studies with multiple atogepant doses and a large and diverse migraine population. These characteristics allowed for assessing various scenarios (e.g., short- and long-term treatment, different doses), making these findings relevant to clinicians. Future analyses should further explore the disease course of individuals who do and do not experience weight loss during preventive migraine treatment, particularly since weight loss has been associated with improvements in migraine with obesity (40). Determining the role of CGRP antagonism in this relationship could provide additional important information to clinicians.
Conclusion
These analyses of five large trials demonstrated that atogepant was associated with consistent and modest dose- and duration-dependent weight loss during EM and CM placebo-controlled trials and long-term EM studies in relation to the applicable comparators.
Data from five large clinical trials demonstrated that atogepant was associated with consistent and modest dose- and duration-dependent weight loss when used as a preventive treatment in people with episodic or chronic migraine. Given the high rate of comorbid obesity in people with migraine, the impact of atogepant on weight is an important factor to inform clinical decision making.
Supplemental Material
sj-docx-1-cep-10.1177_03331024241299753 - Supplemental material for Weight loss with atogepant during the preventive treatment of migraine: A pooled analysis
Supplemental material, sj-docx-1-cep-10.1177_03331024241299753 for Weight loss with atogepant during the preventive treatment of migraine: A pooled analysis by B. Lee Peterlin, Dale S. Bond, Jessica Ailani, David W. Dodick, Yingyi Liu, Rosa De Abreu Ferreira, Jonathan H. Smith, Brett Dabruzzo, Peter J. Goadsby, and Joel M. Trugman in Cephalalgia
Footnotes
Acknowledgments
AbbVie funded this study and contributed to the study design, the collection, analysis, and interpretation of data, and the review and approval of the final manuscript for publication. All authors had access to relevant data and participated in the drafting, review, and approval of this publication. No honoraria or payments were made for authorship. Medical writing support was provided by Dennis Stancavish, MA, of Peloton Advantage, LLC, an OPEN Health company, and was funded by AbbVie.
Author contributions
Study design: Y.L., J.H.S., B.D., J.M.T.; Study investigator: J.A.; Enrolled patients: J.A.; Collection and assembly of data: Y.L., J.H.S., B.D., J.M.T.; Data analysis: Y.L.; Data interpretation: All authors; Manuscript preparation: Y.L., R.D.A.F, J.H.S., B.D., J.M.T.; Manuscript review and revisions: All authors; Final approval of manuscript: All authors.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: B.L.P. has served as a consultant and/or on the speaker bureaus for AbbVie, Amgen, Biohaven, Eli Lilly, GlaxoSmithKline, Impel, and Lundbeck Pharmaceuticals and has received research support from GlaxoSmithKline Pharmaceuticals, the Landsberger Foundation, Luitpold, and the National Institutes of Health. D.S.B. has no conflicts to report. J.A. has served as a consultant for AbbVie, Aeon, Dr. Reddy, Eli Lilly and Company, GlaxoSmithKline, Gore, Ipsen, Linpharma, Lundbeck, Merz, Neurolief, Pfizer, Satsuma, Scilex, Theranica, and Tonix; provided editorial services to Current Pain and Headache Reports, SELF, and Medscape; and received clinical trial support from Ipsen, Parema, and Satsuma. D.W.D. reports the following conflicts: (5-years): Consulting: Amgen, Atria, CapiThera Ltd., Cerecin, Ceruvia Lifesciences LLC, CoolTech, Ctrl M, Allergan, AbbVie, Biohaven, Escient, GlaxoSmithKline, Halion, Lundbeck, Eli Lilly, Novartis, Impel, Satsuma, Theranica, WL Gore, Genentech, Nocira, Perfood, Praxis, AYYA Biosciences, Revance, Pfizer. Honoraria: American Academy of Neurology, Headache Cooperative of the Pacific, Headache Cooperative of New England, Canadian Headache Society, MF Med Ed Research, Biopharm Communications, CEA Group Holding Company (Clinical Education Alliance LLC), Teva (speaking), Amgen Japan (speaking), Eli Lilly Canada (speaking), Lundbeck (speaking), Pfizer (speaking), Vector Psychometric Group, Clinical Care Solutions, CME Outfitters, Curry Rockefeller Group, DeepBench, Global Access Meetings, KLJ Associates, Academy for Continued Healthcare Learning, Majallin LLC, Medlogix Communications, Medica Communications LLC, MJH Lifesciences, Miller Medical Communications, WebMD Health/Medscape, Wolters Kluwer, Oxford University Press, Cambridge University Press. Non-profit board membership: American Brain Foundation, American Migraine Foundation, ONE Neurology, Precon Health Foundation, Global Patient Advocacy Coalition, Atria Health Collaborative, Atria Academy of Science and Medicine, Arizona Brain Injury Alliance, Domestic Violence HOPE Foundation/Panfila, CSF Leak Foundation. Research support: Department of Defense, National Institutes of Health, Henry Jackson Foundation, Sperling Foundation, American Migraine Foundation, Henry Jackson Foundation, Patient Centered Outcomes Research Institute (PCORI). Stock options/shareholder/patents/board of directors: Ctrl M (options), Aural analytics (options), Axon Therapeutics (board/options), ExSano (options), Palion (options), Keimon Medical (options), Man and Science, Healint (options), Theranica (options), Second Opinion/Mobile Health (options), Epien (options), Nocira (options), Matterhorn (shares), Ontologics (shares), King-Devick Technologies (options/board), Precon Health (options/board), ScotiaLyfe (board), EigenLyfe (options/board), AYYA Biosciences (options), Axon Therapeutics (options/board), Cephalgia Group (options/board), Atria Health (options/employee). Patent 17189376.1-1466:vTitle: Onabotulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis (Non-royalty bearing). Patent application submitted: Synaquell® (Precon Health). P.J.G. reports support for the present study and personal fees during the conduct of the study from AbbVie; a grant from Celgene; personal fees from Aeon Biopharma, Amgen, BioDelivery Sciences International, Inc., CoolTech LLC, Dr Reddy's, Eli Lilly, Epalex, Impel NeuroPharma, Lundbeck, Novartis, Praxis, Sanofi, Satsuma, ShiraTronics, Teva Pharmaceuticals, and Tremeau; personal fees for advice through Gerson Lehrman Group, Guidepoint, SAI Med Partners, and Vector Metric; fees for educational materials from CME Outfitters, Omnia Education, and WebMD; fees for publishing from Massachusetts Medical Society and Oxford University Press; and fees for medicolegal advice in headache. He has a patent for Magnetic stimulation for headache, which is licensed to eNeura without fee. Y.L., R.D.A.F., J.H.S., B.D., and J.M.T. are employees of AbbVie and may hold AbbVie stock.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This study was sponsored and funded by Allergan (now AbbVie). Medical writing support was provided to the authors by Dennis Stancavish, MA, of Peloton Advantage, LLC, an OPEN Health company, and was funded by AbbVie.
Data sharing statement
AbbVie is committed to responsible data sharing regarding the clinical trials we sponsor. This includes access to anonymized, individual, and trial-level data (analysis data sets), as well as other information (e.g., protocols, clinical study reports, or analysis plans), as long as the trials are not part of an ongoing or planned regulatory submission. This includes requests for clinical trial data for unlicensed products and indications.
These clinical trial data can be requested by any qualified researchers who engage in rigorous, independent, scientific research, and will be provided following review and approval of a research proposal, Statistical Analysis Plan (SAP), and execution of a Data Sharing Agreement (DSA). Data requests can be submitted at any time after approval in the US and Europe and after acceptance of this manuscript for publication. The data will be accessible for 12 months, with possible extensions considered. For more information on the process or to submit a request, visit the following link: ![]() then select “Home.”
then select “Home.”
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
