Abstract
A population-based longitudinal study suggests that obesity is a strong risk factor for the development of headaches on 15 or more days per month. Little is know about the influence of weight on the response to headache preventive treatment. Herein we prospectively assessed the influence of the baseline body mass index (BMI) on the response to headache preventive treatment. We included adults with episodic or chronic migraine (ICHD-2), or transformed migraine (Silberstein and Lipton criteria) that sought care in a headache clinic. BMI was assessed in the first visit. Baseline information included headache frequency, number of days with severe headache (prospectively obtained over 1 month), and headache-related disability (HIT-6). The same information was obtained after 3 months of preventive treatment. Subjects were categorized based on BMI in: normal weight (≤24.9), overweight (25-29.9), or obese (≥30). We contrasted the headache end-points using ANOVA with post-test and Kruskal-Wallis with post-test. We used logistic regression to model BMI and headache parameters adjusting for covariates. Our sample consisted of 176 subjects (79.5% women, mean of 44.4 years). At baseline 40.9% had normal weight, 29.5% were overweight and 27.3% were obese. No significant differences were observed in the number of headache days at baseline. After treatment, frequency declined in the entire population, but no significant differences were found by BMI group. Regarding the number of days with severe pain per month, there were also no significant differences at baseline (normal = 6.1, overweight = 6.5, obese = 6.7), and improvement overall (P = 0.01). However, changes were greater in the obese (reduction in 2.7 days with treatment) and overweight (3.9) vs. normal (1.5, P < 0.01). Finally, HIT scores at baseline did not differ by BMI group (normal weight = 63.8, overweight = 64.1, obese = 63.6). However, compared with the normal weighted group, change in HIT scores (followup baseline) were greater in the obese (6.4 vs. 3.5, P < 0.05) and overweight groups (6.8 vs. 3.5, P < 0.05). In the logistic regression model, BMI did not account for changes in disability, headache frequency, or in the number of days with severe headache per month, after adjusting for covariates. Contrary to what we hypothesized, obesity at baseline does not seem to be related to follow-up refractoriness to preventive treatment.
Introduction
Recent evidence suggests that a subgroup of migraine sufferers may have a clinically progressive disorder (1). In a longitudinal epidemiological study, Scher et al. showed that, over the course of 1 year, 3% of individuals with episodic headache (headache frequency 2–104 days per year) progressed to a group defined as chronic daily headaches (CDH, attack frequency >180 days per year) (2). Similarly, a clinic-based study found that 14% of individuals with episodic headache developed daily or almost daily headaches within 1 year (3).
Although limited evidence exists about the risk factors for migraine progression, obesity was the strongest variable associated with chronification within subjects with episodic headache, as assessed in a population study (4). In that study, obese headache subjects developed CDH at a rate 5.53 times higher when compared with non-obese headache subjects over a 1-year period.
Both migraine and obesity are very prevalent disorders in the population. The American Migraine Study II, conducted in 1999, estimated that nearly one in four US households had a member with migraine, and that 28 million US residents had severe migraine headaches in that year (5). Results of the National Health and Nutrition Examination Survey (NHANES, 1999–2000) indicate that an estimated 64% of US adults are either overweight or obese, defined as having a body mass index (BMI) of ≥25 (6). Furthermore, during the past 20 years there has been a dramatic increase in the prevalence of obesity in the USA. In 1991, just four states participating in the Center for Disease Control Behavioural Risk Factor Surveillance System (BRFSS) had obesity prevalence rates of as high as 15–19% and none had rates at or above 20%. In 2003, 15 states had obesity prevalence rates of 15–19%; astonishingly, 31 states had rates of 20–24%; and four states had rates of >25% (6).
Obesity is comorbid to a number of chronic pain syndromes. In fibromyalgia, obesity is associated with increased disease severity and refractoriness to treatment, and weight loss is associated with improved physical functioning (7). Additionally, obesity is an important independent predictor of back and neck pain in the population (8). Although limited evidence suggests that obesity is a risk factor for migraine progression, and that a higher proportion of subjects with frequent headaches are obese in comparison with those with episodic headaches (2, 4), little is know about the influence of baseline weight status on the severity and disability of the headache syndrome, as well as on the response to headache preventive treatment. Therefore, we aimed to assess the influence of BMI on response to preventive treatment in subjects with refractory headaches who sought care in a headache clinic.
Methods
Participants of this study were subjects from 18 to 65 years old, with episodic or chronic migraine according to the criteria defined by the Second Edition of the International Classification of Headache Disorders (ICHD-2 (9), or transformed migraine (according to the criteria proposed by Silberstein and Lipton) (10), that sought medical care in a headache clinic during the years 2003 and 2004. According to the ICHD-2, all individuals with transformed migraine had a combination of migraine and tension-type headaches.
After agreeing to participate, and before receiving any intervention, subjects had their weight and height assessed. The BMI was calculated as the ratio between weight in kg and the square of the height in meters (BMI = kg/m2).
Subjects received headache calendars and collected headache information prospectively for 1 month, before any intervention was conducted. Information on frequency and severity of the headache was abstracted from the headache calendars. We also collected (from the headache calendars and standardized questionnaires given at the time of the first visit) information on location of pain, quality of pain, duration of headache, premonitory features, aura, associated symptoms, and relationship between pain and physical activity. Finally, baseline information included the assessment of headache-related disability, using the Headache Impact Test (HIT-6).
All subjects included in this study had assessment of their psychological comorbidity during their first visit by taking the Minnesota Multiphasic Personality Inventory-2 (MMPI-2). The MMPI-2 is a 567-item, true–false questionnaire that evaluates the personality of the subject on three validity and 10 clinical scales (11). For this study we included information on scale 1 (hypochondriasis), scale 2 (depression), scale 3 (hysterias) and scale 7 (psychasthenia).
After the 1-month baseline assessment, subjects received one of several preventive medications. Subjects that were prescribed medications that can cause weight loss (i.e. topiramate, zonisamide) were not included in this study.
The same information obtained at baseline was again assessed after 3 months of preventive treatment.
Data were summarized using frequency counts and descriptive statistics. We categorized individuals as underweight (BMI < 18.5), normal weight (18.5–24.9), overweight (25–29.9) or obese (≥30), and contrasted the headache endpoints using
This study was approved by the IRB. All subjects signed approved consent forms as well as an IRB-approved HIPAA form (American Health Insurance Portability and Accountability Act of 1996) before enrolment.
Results
Our sample consists of 176 subjects, of whom 139 (78.9%) were women (mean age 44.4 years), and 37 (21.1%) were men (mean age 36.2 years). Respondents were mostly caucasians (90.3%) and highly educated (75% had at least finished high school). A total of 38.1% of the patients was referred to the clinic by other doctors, while 61.9% sought care spontaneously.
Nearly half (48.9%) of the participants had migraine, while 51.2% had transformed migraine. When enrolled in this study, four (2.3%) were underweight, 72 (40.9%) had normal weight, 52 (29.5%) were overweight, and 48 (27.3%) were obese. Underweight subjects were excluded from our analyses due to our small sample.
When enrolled in the study, subjects were using, alone or in combination, β-blockers (31.8%), calcium channel blockers (25%), antiepileptic drugs other than topiramate or zonisamide (35.7%), vitamins and minerals (28.9%), antidepressant medications (35%) and other medications (10.2%). After 3 months of treatment, subjects gained an average of 0.6 kg (±1.1), a non-statistically significant difference.
There were no significant differences between the proportions of men and women that were overweight or obese at the baseline visit. There were no significant differences in regard to the proportion of subjects with abnormal scores in the MMPI-2 scales by weight group (data not shown). A total of 54.7% of the women and 62.2% of the men were overweight or obese at the time of the first visit (Fig. 1).
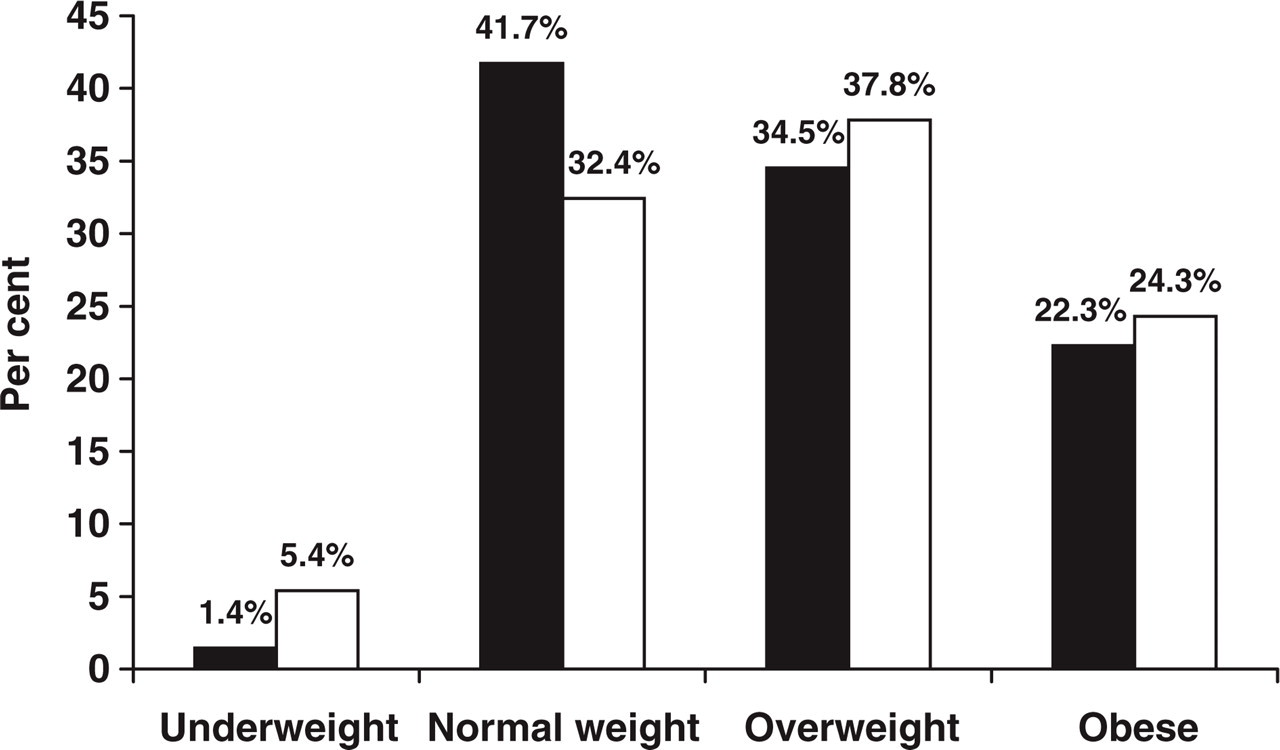
Participants in our study by body mass index and gender. ▪, Female; □, male.
We did not observe significant differences with regard to headache frequency among the three weight groups, comparing the number of days with headache per month at baseline (normal = 19.3, overweight = 22.2 and obese = 21.2), or at follow-up (14 vs. 14.5 vs. 16.2). In the three groups, preventive treatment was associated with reduction in headache frequency. However, the mean reduction in frequency following treatment was not significantly different by weight group either (Fig. 2).

Number of days with headache per month, comparing the baseline month and the third month of preventive treatment, by body mass index group. ▪, Baseline; □, 3 months; hatched, difference.
We found no significant differences regarding the number of days with severe pain per month at baseline (normal weight = 6.1, overweight = 6.5, obese = 7.3), or at follow-up (normal = 4.5, overweight = 2.5, obese = 4.6). In all groups there was a statistically significant reduction in the number of days with severe pain after 3 months vs. baseline. Interestingly, the reduction in the number of severe headache days (follow-up–baseline) was higher in the overweight group compared with the normal weight group (4.0 vs. 1.6, P = 0.0024). No significant differences were found comparing the overweight vs. obese groups, nor the normal weight vs. obese (Fig. 3).
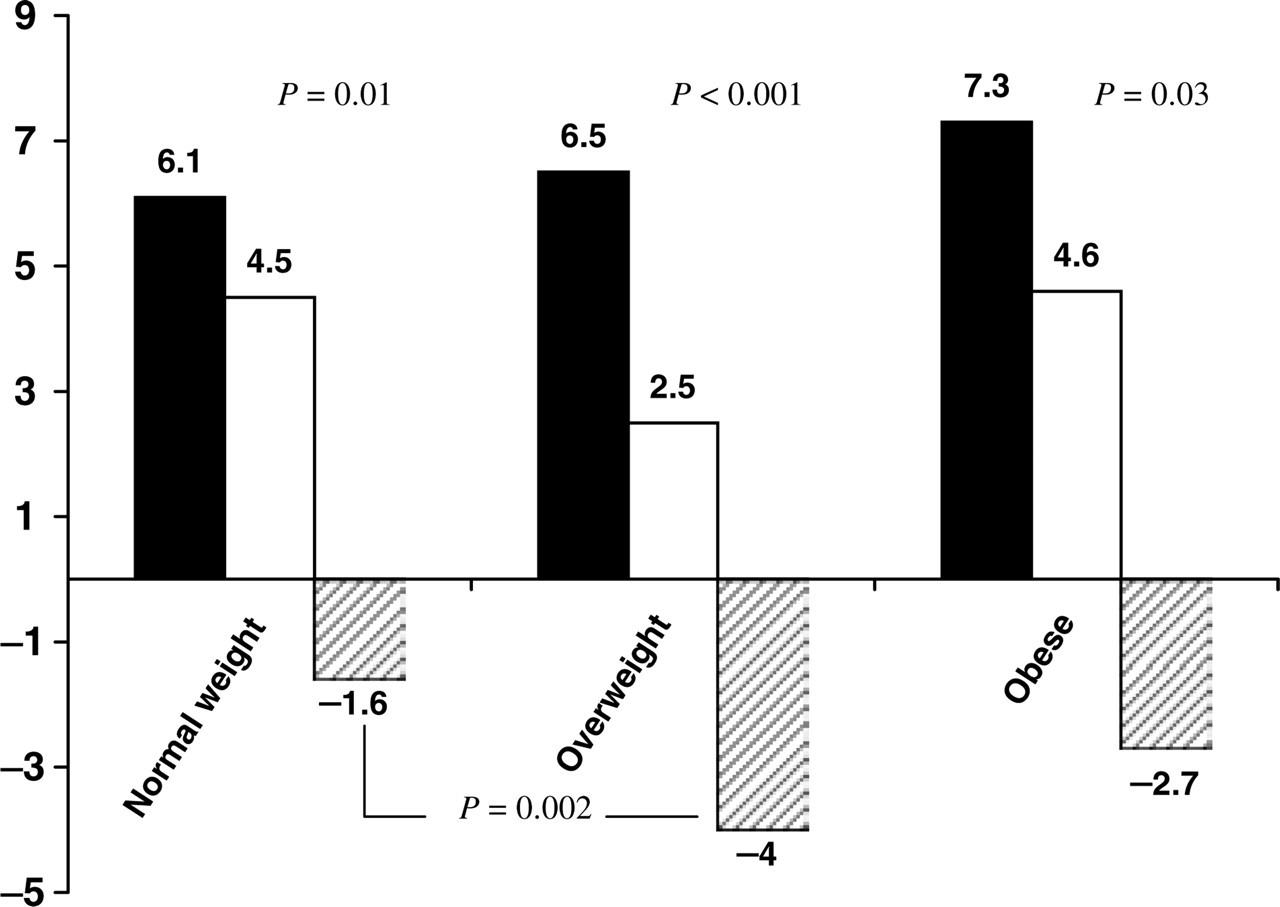
Number of days with severe headache per month, comparing the baseline month and the third month of preventive treatment, by body mass index group. ▪, Baseline; □, 3 months; hatched, difference.
Although the HIT scores at baseline were not significantly different (normal weight = 63.8, overweight = 64.1, obese = 63.6), and in the three classes there were significant reductions in these scores with treatment (pre vs. post), the differences between follow-up and baseline scores were significantly superior in obese vs. normal (P = 0.01) and in overweight vs. normal (P < 0.05) (Fig. 4).

Disability scores, as asessed by the headache impact test (HIT-6), comparing the baseline month and the third month of preventive treatment, by body mass index group. ▪, Baseline; □, 3 months; hatched, difference.
We did not find differences in the proportions of attacks with nausea during the study (normal weight = 39.6%, overweight = 33.4%, obese = 41.3%), or in the proportion of attacks with photophobia (normal weight = 70.2%, overweight = 67.3%, obese = 69.4%) or phonophobia (normal weight = 52.1% overweight = 47.0%, obese = 46.1%). Finally, there were no differences in the duration of the treated attack or in the utilization of rescue medication.
In the logistic regression model, BMI did not account for changes in disability, headache frequency, or in the number of days with severe headache per month, after adjusting for covariates (number of preventive medications used, MMPI-2 scales, gender, age, marital status, aura, location of pain), although our sample size limited many of the logistic analyses.
Discussion
Recent evidence supports the concept that some patients with migraine have a progressive form of the disease (1–3, 12). In 75% of migraine sufferers, sensitization of the second-order trigeminal neuron in the brain stem occurs during the course of a migraine attack (central sensitization, CS) (13, 14). It can be hypothesized that repetitive CS at the level of the trigeminal neuron correlates with neuronal dysfunction and neuronal death, as suggested by the observation that iron deposition occurs in the midbrain periaqueductal grey area in subjects with chronic headaches (15).
If migraine is a progressive disease, it is clear, however, that it is not progressive in everyone. The prevalence of migraine decreases after peaking at intermediate ages (5, 16), and although the prevalence of frequent headaches increases with age (suggesting that some individuals with migraine may be evolving to CDH) (2, 17), the incidence of CDH in individuals older than 40 years is lower than the remission rates for migraine in the same age range. Migraine may be progressive therefore in a minority of individuals, who appear to be at a higher risk.
As migraine is at times a progressive disease, a better understanding on its natural history (from episodic to chronic) is of great interest and may lead to the elucidation of effective preventive factors. Additionally, if there are risk factors associated with disease progression, subjects that have risk factors may require more aggressive treatment. Research in this field is still in its infancy and very limited, and hypotheses are at this point largely speculative. Nonetheless, a clinic-based study found that medication overuse and frequency of headache at baselines are important risk factors (3), and an epidemiological study found that obesity was the most important risk factor associated with the development of frequent headaches in the population (4).
We were interested in studying the relationship between obesity and refractory headaches. It is known that elevated plasma calcitonin gene-related peptide (CGRP) levels are present in obese individuals, particularly women, and that fat intake may be associated with increased CGRP secretion (18). CGRP is an important postsynaptic mediator of trigemino-vascular inflammation (19), and experimental CGRP inhibitors are effective in the acute treatment of migraine (20). Additionally, obesity itself is a proinflammatory (adipocytes secrete a variety of cytokines, including interleukin-6 and tumour necrosis factor-α, that promote inflammation) (21) and prothrombotic state (22), and migraine is associated with neurovascular inflammation (23). Migraine is also comorbid with several cardiovascular disorders, as well as a risk factor for stroke, especially in women who have migraine with aura (24–26).
We hypothesized that obese migraine sufferers would be more refractory to preventive treatment than would non-obese migraine sufferers. This hypothesis was not confirmed by our data, which can be summarized as follows: (i) a large proportion of individuals with refractory headaches that seek care in a headache clinic are overweight or obese, although the proportion does not seem to differ substantially from that found in the population; (ii) obesity does not contribute to the headache-related disability, nor is it associated with an increased prevalence of depression; (iii) being overweight and obese at baseline is not associated with treatment refractoriness. Indeed, disability was significantly more reduced in the obese and overweight subjects after preventive treatment, compared with the normal-weight group; (iv) in the logistic regression model, BMI did not account for changes in disability, headache frequency, or in the number of days with severe headache per month, after adjusting for covariates.
Our study partially disagrees with another clinic-based study (27). In that study, although headache severity and the frequency of pain were similar among the weight groups (similar to our findings), increased BMI was associated with more days per week with both reduced activity and complete disability (different from what we found).
Some caution is required in interpreting our data. First, we assessed BMI at baseline and correlated it with clinical features at baseline, as well as outcomes at follow-up. We did not model changes in BMI during treatment with treatment outcomes, due to our limited power to do so, as well as due to the fact that we excluded medications that may cause weight loss. Most preventive medications used in clinical practice are not weight neutral. It would be very interesting to compare the effect of weight gain vs. weight loss during the treatment with response to therapy. This was not done in our study but is planned for the future. Second, we include patients with migraine and transformed migraine in the same proportions. Although the preventive treatment strategies used in our clinic for both diseases are basically the same, the response to treatment may be very different. Due to this, it would be very important to separate both groups for analysis, and we did not do so due to power limitations in detecting a difference. However, we did run interim analyses in both groups to detect a trend that could suggest different patterns of responses, and found none. Future studies should assess a pure population. Third, we assessed a refractory headache population, and our findings may not reflect those patients with less severe migraine seen in settings other than a headache clinic. Finally, we were not able to model several covariates (race, or use of different analgesics, for example) due to sample size limitation. Also, we did not assess response to acute therapy as a function of BMI.
Understanding the natural history of a disease is a necessary step in the search for better treatment and satisfactory outcomes. As pointed out by Mercker, ‘you can’t protect what you don’t know’ (28). Risk factor identification, as well as assessment of predictors of treatment success or treatment failure, are fundamental in this process. In this study we aimed to assess the influence of BMI on preventive treatment outcomes. We conclude that: (i) the majority of refractory headache subjects that need preventive treatment in a tertiary care setting are overweight or obese, although the proportions do not seem to be significantly different from those in the population; (ii) although obesity seems to be a risk factor for migraine progression, obesity at baseline does not seem to be related to post-therapy refractoriness to preventive treatment.
