Abstract
The last three decades have produced several novel and efficient medications to treat migraine attacks and reduce attack frequency. Additionally, promising approaches for the development of acute therapy and migraine prophylaxis continue to be pursued. At the same time as we witness the development of better and more efficient medications with continuously fewer side effects, we also realise that the high cost of such therapies means that only a minority of migraine patients who could benefit from these medications can afford them. Furthermore, information on cost-effectiveness is still lacking. Here, we compare availiable data, highlight open questions and suggest trials to close knowledge gaps. With good reason, our medicine is evidence-based. However, if this evidence is not collected, our decisions will continue to be based on marketing and assumptions. At the moment, we are not doing justice to our patients.
Keywords
Introduction
Recent years have seen the emergence of a number of new and effective drugs for the treatment of migraine and very promising approaches to the development of acute therapy and migraine prophylaxis continue to be pursued. This unprecedented success story of the last 30 years obscures our neglect that, as a result of the high costs, these new medications are only available to a very small fraction of migraine patients worldwide. At the same time, a large part of all scientific publications and main symposia of scientific congresses (and thus all the money spent) revolve around the innovations, and not around medications which have been on the market for more than 20 years. International professional societies such as the International Headache Society (IHS) have as their declared main objective “the benefit of people affected by headache disorders”. We will only come closer to this goal if new and effective medications that have been investigated in valid RCTs are available to all, or at least the majority of all migraine patients.
A critical appraisal: therapy of the acute migraine attack
Until recently, acute migraine attacks were treated with analgesics and non-steroidal anti-inflammatory drugs or triptans. Triptans are very effective and have a good side effect and safety profile. In a meta-analysis, sumatriptan had a pain free rate of 29%. Compared with sumatriptan, 10 mg of rizatriptan showed better efficacy and consistency, and similar tolerability; 80 mg of eletriptan showed better efficacy, similar consistency, but lower tolerability; 12.5 mg of almotriptan showed a similar efficacy at two hours (but better than other results); 2.5 mg of naratriptan and 20 mg of eletriptan showed lower efficacy and (the first two) better tolerability; 2.5 mg and 5 mg of zolmitriptan, 40 mg of eletriptan and 5 mg of rizatriptan showed very similar results (1). A recent real world study including 4,777,524 medication-outcome pairs from 3,119,517 migraine attacks among 278,006 users showed a mean odds ratio of 4.8 for triptans compared to ibuprofen and superiority of triptan over opioids, ergots and non-steroidal anti-inflammatory drugs (2). The data are sufficiently good to conclude that triptans are the drugs of choice in the treatment of acute migraine attacks, both in terms of benefit and side-effect profile. However, some patients are non-responders to triptans and, in many cases, the non-response is a result of application errors such as administering the triptan too late during a migraine attack when the headache has already reached its peak level. Triptans act via serotonin 5-HT1B and 5-HT1D receptors. Initially, it was assumed that the effect of triptans was mediated by a constriction of dilated dural arteries during the migraine attack (3,4). This assumption led to the contraindications in the original marketing authorisation, namely serious vascular diseases. Subsequently, it was shown, however, that the effect of triptans is mediated predominantly via 5-HT1D receptors in terminal nerve endings of the trigeminal nerve, in the trigeminal ganglion, in the trigeminal nucleus area and in the thalamus (5). Consequently, further studies showed that the vasoconstrictive properties of triptans in humans are minimal (6). In addition, the mechanism of stroke or myocardial infarction is in most cases plaque rupture and local thrombosis or embolism and not vasoconstriction (7). Vasoconstriction mainly plays a role in Prinzmetal angina and vasospasm after subarachnoidal haemorrhage. Despite these new findings, the package inserts (summary of product characteristics or SmPC) for triptans were never changed, such that contraindications such as uncontrolled hypertension, stroke, myocardial infarction, angina pectoris and other serious vascular diseases are still mentioned. However, large observational studies have not shown an increased risk of acute coronary syndromes and ischaemic strokes in patients who treat their migraine attacks with triptans (8). A query of the database of the Drug Commission of the German Medical Association did not reveal a single report of a stroke or myocardial infarction after the use of a triptan in the last three years in Germany (personal communication, German Medical Association). Because the patent protection of the triptans has expired, there will be no initiatives to change the SmPCs of the triptans regarding contraindications.
The rationale behind the development of new drugs for the treatment of acute migraine attacks was the patient population for whom triptans are not or not sufficiently effective and patients with actual or perceived contra-indications to triptans. Three calcitonin gene-related peptide (CGRP) receptor antagonists, the so-called gepants, were developed: for the treatment of acute migraine attacks, rimegepant 75 mg, ubrogepant 50 and 100 mg and zavegepant nasal spray 10 mg. There are no significant differences in the efficacy of the three gepants, and the 95% confidence intervals of the odds ratios overlap (Table 1) (9). Unfortunately, there are no direct comparative studies between triptans, analgesics, non-steroidal anti-inflammatory drugs and the approved gepants. It is rather likely that sumatriptan 100 mg and rizatriptan 10 mg are more effective than the three gepants.
Two hour pain free rates and corresponding odds ratios (OR) and 95% confidence intervals (CI).
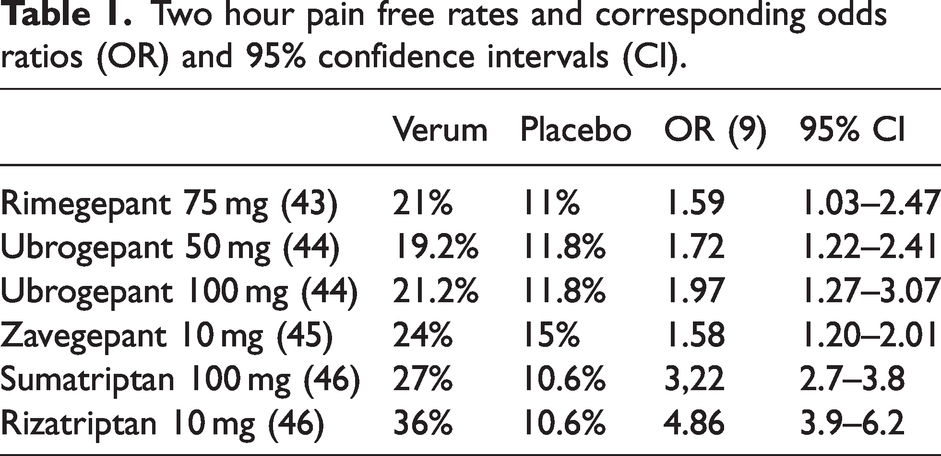
The target group for treatment of acute migraine attacks with a gepant are, as described above, patients with proven ineffectiveness of triptans. It is surprising that there are no randomised trials with patients in whom the lack of efficacy or ineffectiveness of a triptan has been prospectively tested. In such a study, patients would first have to be treated with a triptan in the first phase and only non-responders (i.e. patients who do not respond to the triptan in three out of three migraine attacks) would then receive a gepant or a triptan in a randomised study. Such studies are undoubtedly expensive and the pharmaceutical industry should take its responsibility and make such studies available.
Another class of new migraine triptans are the ditans. Lasmiditan has shown efficacy in the treatment of migraine attacks but has central side effects (10–12). Lasmiditan has no vasoconsstrictive properties and therefore would be an alternative for patients with clear contraindications for triptans (13)
Because gepants block the CGRP receptor and thus probably prevent the dilation of collaterals in cases of ischaemia, they should also not be used in patients with severe vascular disease (14). However, it would be extremely unlikely that a migraine patient would coincidentally suffer an ischemic stroke in temporal relation to taking a gepant.
Acute medication: cost–benefit analysis
Three gepants are approved and commercially available in the USA. Table 2 shows the cost of one tablet of each of the gepants compared with sumatriptan and rizatriptan in the USA. For comparison, the cost of rimegepant and sumatriptan and rizatriptan in Europe is also shown. These figures show the most important problem for the use of gepants: at current prices, there are few migraine patients outside the USA who can afford to treat migraine attacks with a gepant. This deprives millions of people with migraine who do not respond adequately to triptans of a potentially effective therapy. It also raises the question of the unique selling point: what is the added benefit of a medication that costs 31 times (USA) or six times (Europe) as much as the triptans?
Treating an acute migraine attack: cost per tablet in the USA and Europe, lowest prices by internet pharmacies.
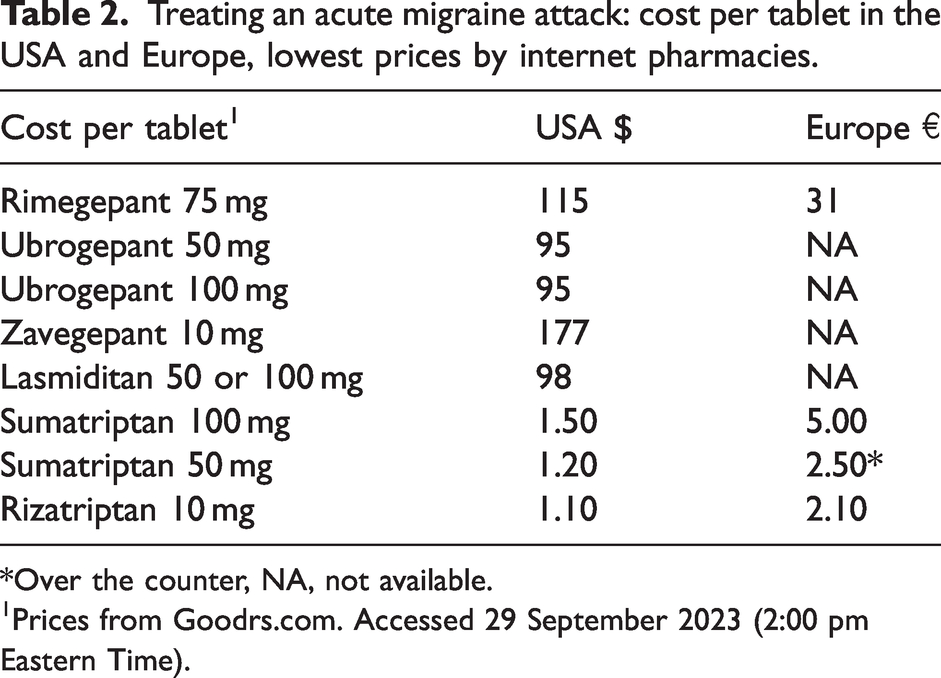
*Over the counter, NA, not available.
Prices from Goodrs.com. Accessed 29 September 2023 (2:00 pm Eastern Time).
Acute medication: what data and information are still missing?
Clinical neurologists and headache specialists lack information to make evidence-based, valid decisions about the best treatment of migraimne attacks for their patients.
There are no randomised controlled trials that have directly compared a triptan such as 50 or 100 mg of sumatriptan with an approved gepant. There are no randomised trials to date to demonstrate the efficacy of gepants in patients who have prospectively shown that they do not benefit from triptans. There have been no randomised trials in patients who cannot take triptans because of real or assumed contraindications. There are no data on the safety of gepants in patients with severe vascular diseases. There are no valid cost–benefit analyses for the gepants so far. The proof that gepants do not lead to medication overuse headache is still lacking
Clinical implications acute migraine medication
There is no doubt that the development of the gepants is an important step in the treatment of acute migraine attacks. The main advantage of gepants over triptans is that thery are well tolerated. It is very likely that gepants can also be used in patients with vascular diseases. Gepants might not lead to medication overuse and medication overuse headache (15). The main obstacle for the use of gepants is the very high price.
A critical appraisal: migraine prevention
Until five years ago, migraine prophylaxis was carried out with substances whose efficacy for the prevention of migraine was discovered by chance. Only methysergide and pizotifen were developed as specific migraine preventive drugs but are either no longer available or no longer used. Beta-receptor blockers such as propranolol (16), flunarizine (17), topiramate (18), valproic acid (19), amitriptyline (20) and candesartan (21) have been shown to be effective in placebo-controlled trials. For chronic migraine, there is evidence of efficacy for onabotulinumtoxinA (22). The problem with oral migraine prophylactics was not efficacy but tolerability. Unfortunately, many patients discontinued migraine prophylaxis because of adverse drug reactions (23). In addition, most of the randomised controlled trials addressing oral drugs for migraine prevention would nowadays considered inadequate from a methodological point to establish if the study drug was superior to placebo. Therefore, there was a clear clinical need to develop new migraine medications with proven efficacy and a better side effect profile.
Three monoclonal antibodies against CGRP (eptinezumab, fremanezumab and galcanezumab) and a monoclonal antibody against the CGRP receptor (erenumab) were developed as specific migraine prophylactics. In randomised placebo-controlled trials, all monoclonal antibodies were more effective than placebo in reducing the mean number of migraine days per month and 50% responder rates. This was true for both episodic and chronic migraine patients. In a meta-analysis, the efficacy of monoclonal antibodies against CGRP or the CGRP receptor was comparable (24).
The tolerability and safety of monoclonal antibodies in migraine prophylaxis is very good (25). A dose of 240 mg of galcanezumab had the highest rates of adverse drug reactions. Otherwise, there were no significant differences between the individual monoclonal antibodies. Erenumab has a slightly higher risk of constipation.
Initially, health care systems in Europe restricted the prescription of monoclonal antibodies for patients who did not respond to two to five previous oral therapies or onabotulinumtoxinA, valproic acid or could not tolerate them or had contraindications (e.g. topiramate in women) (26).
Migraine prophylaxis with gepants
Two of the gepants namely rimegepant 75 mg every other day (27) and atogepant once daily (28–30) were studied for the prophylaxis of migraine. The study of rimegepant was based on the observation from open-label, long-term studies that patients who treated their migraine attacks with rimegepant also experienced a reduction in the frequency of migraine days (31). Table 3 shows efficacy data for CGRP antagonists, CGRP antibodies, and topiramat and propranolol. Atogepant is approved for the prophylaxis of episodic and chronic migraine. Rimegepant is only approved for the treatment of episodic migraine.
Which patients should be treated with monoclonal antibodies to CGRP or the CGRP receptor or gepants?
Patients with chronic migraine and medication overuse are likely to benefit most from monoclonal antibodies and gepants. Patients in whom oral migraine prophylactics or onabotulinumtoxinA for chronic migraine are ineffective, not tolerated or contraindicated should be treated with a monoclonal antibodies. Data from randomised trials for this patient population are not yet available for gepants.
However, most health systems severely limit reimbursement for the new migraine prophylactics because of the high annual treatment costs.
Migraine prophylaxis: cost–benefit analysis
The treatment costs for the monoclonal antibodies are currently still very high (Table 4). This is in partly because of the high costs for the production of humanised monoclonal antibodies. The costs for the gepants are even higher. A systematic review of the economic evaluation of erenumab, fremanezumab and galcanezumab showed that they are not cost-effective compared to OnabotulinumtoxinA (32). As a result of the high cost of treatment, very few patients are currently treated with monoclonal antibodies outside the USA. In Germany, with a population of 84 million people, there were an estimated 35,000 patients with migraine in 2022 who were treated with a monoclonal antibody (personal communication,). In addition, many patients and physicians are not aware that new options for the prevention of migraine exist.
Cost for one year of treatment in the USA and Europe
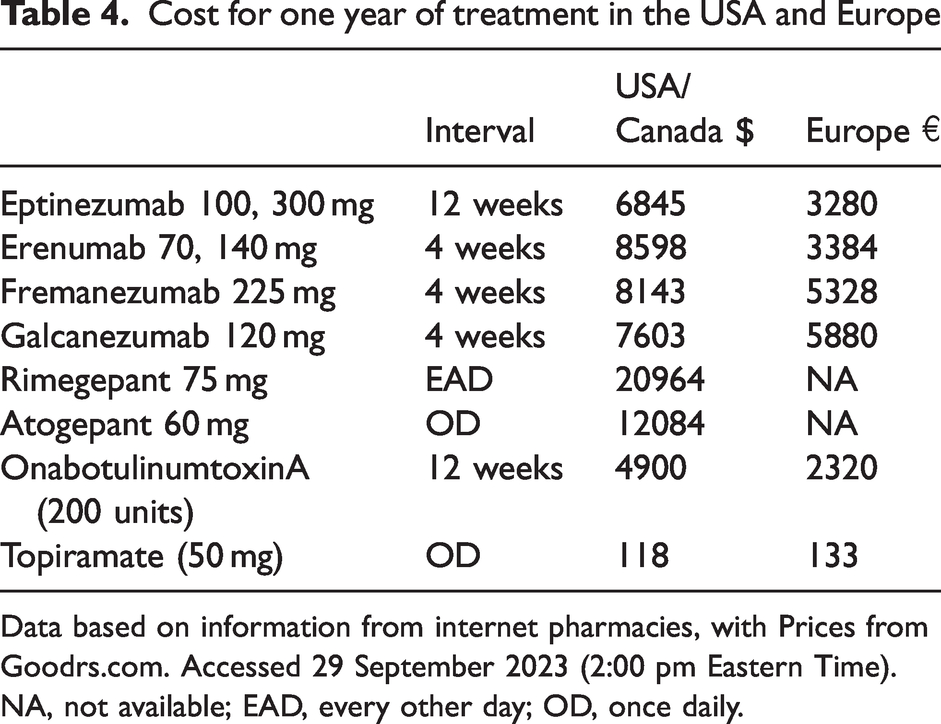
Data based on information from internet pharmacies, with Prices from Goodrs.com. Accessed 29 September 2023 (2:00 pm Eastern Time).
NA, not available; EAD, every other day; OD, once daily.
The high cost of the monoclonal antibodies and gepants means that only a minimal fraction of all migraine patients who could benefit from these drugs receive modern migraine prophylaxis.
Prophylaxis: which data and information are still missing?
Clinical neurologists and headache specialists lack the information they need to make evidence-based, valid decisions about the best treatment for their patients.
Head–to-head comparative studies of monoclonal antibodies are completely lacking. Comparative studies of monoclonal antibodies and gepants with oral migraine prophylactics and in chronic migraine with onabotulinumtoxinA are also lacking. To date, there is only one direct comparison study between erenumab and topiramate with the question of the side effect rate, in which, unsurprisingly, erenumab was not only better tolerated than topiramate, but also caused a significantly higher reduction in monthly migraine days in the secondary objectives (33). Topiramate is known to be poorly tolerated. Comparative studies should be conducted with propranolol, which is significantly better tolerated than topiramate. Prospective study at the end of therapy: most patients experience a rapid increase in migraine frequency as soon as successful anti-CGRP therapy is discontinued (34,35). In future studies, successful therapy with monoclonal antibodies should be terminated after six months in randomised and placebo-controlled trials, either abruptly (as is currently common practice) or gradually (over longer intervals between new injections). Pharmaceutical companies, however, are probably not interested in conducting studies on how their drugs are best discontinued. Prospective studies on the duration of therapy. In these studies, patients should be treated with a monoclonal antibody or a gepant for nine or 12 months. Then they should be randomised to continuation of drug prophylaxis or placebo. In this way, it will be possible to prospectively assess the percentage of patients who need to be retreated with the new migraine prophylactics after a treatment pause. Prospective randomised studies on switching therapy when efficacy is poor or insufficient. Patients in whom a monoclonal antibody was not effective after three months should be randomised and treated for a further 3 months with the previously used monoclonal antibody or an alternative antibody or a gepant. Prospective randomised trials of the combination of traditional oral migraine prophylactics or in chronic migraine with onabotulinumtoxinA in patients who have had an inadequate response to a monoclonal antibody or gepant (responder rate >30–50%).
Which patients should currently receive limited or no treatment with monoclonal antibodies against CGRP or the CGRP receptor or gepants?
CGRP has numerous biological functions and is a particularly potent vasodilator in reduced perfusion and ischaemia. In addition, CGRP receptors are also found, for example, in the intestine, in the endothelium of the bronchi and in the skin. For purely pathophysiological reasons, there are therefore a number of diseases in which both the monoclonal antibodies against CGRP and the gepante should only be used with great restraint or caution (36,37) (Table 5). The new migraine drugs are currently contraindicated in pregnant women, children and adolescents.
Patients in whom monoclonal antibodies or gepants should be used with care

COPD, chronic obstructive pulmonary disease.
Clinical implications of new migraine preventive drugs
Gepants are an important new therapeutic option for the treatment of acute migraine attacks, particularly in patients in whom triptans are ineffective, not tolerated or contraindicated. Monoclonal antibodies against CGRP or the CGRP receptor and gepants offer an effective and well-tolerated option for migraine prophylaxis, particularly in patients in whom previous oral migraine prophylactics or onabotulinumtoxinA have not been effective, are not tolerated or are contraindicated. Patients with the highest benefit from monoclonal antibodies are those with chronic migraine and medication overuse. All monoclonal antibodies showed efficacy in these patients. In the past, this had only been shown for onabotulinumtoxinA. The particular advantage of monoclonal antibodies and gepants is that they are well tolerated. The subcutaneous or intravenous administration of the monoclonal antibodies ensures high adherence. So far, there are no serious safety concerns. A final conclusion on the saftey of monoclonal antibodies requires the treatment of large numbers of patients including those with a hypothetical risk such as ischaemic stroke, acute coronary syndrome, inflammatory bowel disease or severe chronic obstructive pulmonary disease (38)
Conclusions and call for action
At the current prices for monoclonal antibodies and the gepants, only extremely few patients can benefit from this therapy. Professional societies and pharmaceutical companies should take their responsibility and set the goal that at least 50% of all migraine patients can be treated with modern migraine medication, instead of the currently small minority. In some studies reporting on monoclonal antibodies (39,40), the impression was also given that triptans are dangerous in the treatment of acute migraine attacks. This is simply wrong (41). Moreover, given the good tolerability of CGRP antagonists and antibodies, devaluing the gold standard for marketing reasons is unnecessary. So far, there has been one published study comparing a monoclonal antibody with an oral migraine prophylactic. (33). The much more important question of therapeutic benefit, which can be clearly answered in a direct comparison, is being bypassed. The elephant in the room here is the fact that most of the medical prophylactics used so far can be discontinued after about six to nine months because the migraine frequency remains permanently low. The conventional prophylactics therefore have a positive effect on the migraine biology. CGRP antibodies usually only work as long as they are given and, even after a year of treatment, the frequency of attacks rises again as soon as the therapy is discontinued (42). This is not even taken into account when calculating the costs (Table 4). We also simply do not know how a therapy with CGRP antibodies, which may be necessary for many years, will be tolerated long-term. Studies are urgently needed here. So far, there have been no prospective studies in which non-responders to triptans or patients with contraindications to triptans were examined. There have also been no randomised trials of the new migraine prophylactics regarding the duration of treatment, the consequences of drug interruption, switching in case of ineffectiveness, and the efficacy, tolerability and safety of combination therapy.
Our therapy decisions should be based on comprehensible data. With good reason, our medicine is evidence-based. However, if this evidence is not collected, our decisions will continue to be based on marketing and assumptions. We are not doing justice to our patients.
Clinical implications
Gepants are an important new therapeutic option for the treatment of acute migraine attacks, particularly in patients in whom triptans are ineffective, not tolerated or contraindicated. Monoclonal antibodies against CGRP or the CGRP receptor and gepants offer an effective and well-tolerated option for migraine prophylaxis, particularly in patients in whom previous oral migraine prophylactics or onabotulinumtoxinA have not been effective, are not tolerated or are contraindicated. The patient group with the highest benefit from monoclonal antibodies is patients with chronic migraine and medication overuse. The particular advantage of monoclonal antibodies and gepants is that they are well tolerated. The subcutaneous or intravenous administration of the monoclonal antibodies ensures high adherence. So far, there are no serious safety concerns. The high costs of the new medications impede the use of these medications for the majority of migraine patients.

