Abstract
Background
Several novel treatments targeting the calcitonin gene-related peptide pathway have been developed for migraine. We evaluated the efficacy of these medications, including atogepant, rimegepant, erenumab, eptinezumab, fremanezumab, and galcanezumab, for the prevention of migraine via network meta-analysis.
Methods
Databases, including MEDLINE via PubMed, EMBASE, and Cochrane central, were systematically reviewed, and all eligible phase 3 randomised controlled trials were included.
Results
Nineteen studies (n = 14,584 participants) were included. Studies included episodic (n = 11) and chronic (n = 4) migraine or both (n = 4). All interventions, except for eptinzumab 30
Conclusion
The results show that medications targeting calcitonin gene-related peptide were effective in preventing migraine compared to placebo. Considering limitations of single studies, different populations such as episodic and chronic migraine, and the absence of head-to-head trials, all novel treatments decreased mean monthly migraine and headache days, and showed higher 50%, 75% and 100% responder rates than placebo.
Introduction
Migraine is one of the leading causes of disability (1,2), with over one billion people worldwide affected. This highlights the importance of seeking proper management and therapeutic options for this neurological disease (2). Migraine treatment includes non-pharmacological and pharmacological acute drugs and preventive approaches (3,4). Preventive medications should be suggested for people with migraine with severe disabilities or more than four days of headache per month regardless of the disability degree (3,4).
In recent years, a new target-driven class of migraine preventive treatments that act by blocking calcitonin gene related peptide (CGRP) has been developed. CGRP is a neurotransmitter with an essential role in migraine pathophysiology. Currently, there are two types of medications targeting CGRP for migraine prevention: a) CGRP monoclonal antibodies, or CGRP-mAbs, including erenumab which blocks CGRP receptor, and eptinezumab, fremanezumab, and galcanezumab that are CGRP blockers, and b) small molecule CGRP receptor antagonists, or gepants, including atogepant and rimegepant. Rimegepant is also used for acute management of migraine. Gepants are administered orally, erenumab, fremanezumab and galcanezumab subcutaneously and eptinezumab via intravenous injection (5–7). Dose regimens also greatly differ among these drugs, as some are administered daily (i.e. atogepant), some every other day (i.e. rimegepant), some monthly (i.e. erenumab, galcanezumab and fremanezumab) and others every trimester (i.e. eptinezumab). Overall, anti-CGRP medications have shown promising results in managing and preventing migraine (8,9).
A meta-analysis allows the comparison of available data with a high level of evidence, even though the differences described above can only fully be accounted for with direct head-to-head trials. The current study is a systematic review of the available phase 3 randomised controlled trials for atogepant, rimegepant, erenumab, eptinezumab, fremanezumab and galcanezumab in the prevention of episodic and chronic migraine and network meta-analysis to evaluate their efficacy compared to placebo. Safety evaluation was also performed and is reported separately in another manuscript as part of this Special Collection.
Methods
The current systematic review and network meta-analysis was conducted based on Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA). The protocol was registered on the International Prospective Register of Systematic Reviews (PROSPERO) with registration number CRD42022310579 and can be accessed via the following link: https://www.crd.york.ac.uk/prospero/display_record.php?ID = CRD42022310579.
Search strategy
The following online databases were searched from inception until 11 February 2022 (with an updated search performed in May 2022): MEDLINE via PubMed, EMBASE and Cochrane central register of controlled trials. The search strategy was prepared to obtain all published randomised controlled trials (RCT) on the effect of atogepant, rimegepant, erenumab, eptinezumab, fremanezumab, or galcanezumab on the preventive treatment of migraine. The detailed search strategy for all databases is reported in online Supplementary Table 1.
Selection criteria
In the current systematic review and meta-analysis, we included phase 3 double-blind RCTs that fulfil the following PICOS criteria:
Population (P): Participants over the age of 18 that were diagnosed with migraine (episodic or chronic) based on the International Classification of Headache Disorders criteria, third edition (ICHD-III) (10) or ICHD-III beta edition (11). Intervention (I): Studies with at least one arm evaluating the effect of atogepant, rimegepant, erenumab, eptinezumab, fremanezumab or galcanezumab. All available doses of the drugs were included in this review. Atogepant 30 Each medication in each available dose was considered as a separate arm. Atogepant (10, 30, 60 and 120 Comparison I: Studies comparing one of the mentioned interventions with placebo. Outcome (O): Outcomes were selected based on the guidelines of the International Headache Society for controlled trials of preventive treatment of episodic (12) and chronic (13) migraine in adults. The following outcomes were collected if available as primary outcomes: change from baseline in monthly migraine days (MMD – where a migraine day is defined as a day with a migraine that lasts at least 30 minutes without intake of analgesics or a day with a headache that successfully responds to migraine-specific acute treatment), and ≥50% responder rate (defined as the percent change from baseline in the number of migraine days or moderate/severe headache days). In addition, changes from baseline in monthly headache days (MHD), acute medication days (AMD), ≥75% and 100% responder rate were collected as secondary outcomes. Study design (S): Phase 3 double-blind RCTs. Phase IIIb trials with difficult-to-treat populations who failed previous preventive treatments were not included.
Only studies in English language, RCTs, and studies on participants ≥18 years old were included. Open-label studies, post-hoc and secondary analysis reports, conference abstracts, letters or editorials were excluded.
Screening and data extraction
After removing duplications, all identified citations were imported into Rayyan, an online free web and mobile app, for screening (14). All citations were screened on different levels, including title and abstract screening and full-text screening according to the protocol by two independent reviewers, experienced in research on headache disorders (FH, DGA, FP, and RM) for each record. Conflicts were resolved by discussing with a third reviewer or senior author (PP-R).
Data extraction from included studies was performed using a predesigned excel spreadsheet. Independent reviewers (FH, DGA, FP, and RM) performed the data extraction in pairs, and conflicts were solved by discussing or involving a third reviewer or with a senior author. There were no missing data for primary outcomes. Authors were not contacted for further data and only published data reported in papers were used in the study.
In addition to mentioned primary and secondary outcomes, the following data were extracted: study title, year of publication, first author’s name, demographics of participants including age, sex and ethnicity, BMI, trial registration number, total number of included participants, migraine type (episodic or chronic), presence of aura, history of prior preventive treatments, study duration, intervention type, route of administration, intervention dose, and sample size in each group. In addition, data for assessing the risk of bias was also collected.
Risk of bias assessment
Risk of bias assessment was performed using version 2 of the Cochrane risk-of-bias tool for randomised trials (RoB 2) (15). The evaluation was done in five main domains, including ‘bias arising from the randomisation process’, ‘bias due to deviations from intended interventions’, ‘bias due to missing outcome data’, ‘bias in measurement of the outcome’ and ‘bias in selection of the reported result’. Judgment for each section could be ‘low risk of bias’, ‘some concerns’ and ‘high risk of bias’; finally, an ‘overall bias’ was reported for each study. The monthly migraine days outcome was used to assess the risk of bias in the current study. The assessment was done by two independent reviewers (FH and FP).
Data analysis
Demographic and clinical characteristics of patients randomised in the included studies were reported as mean and standard deviations or as frequencies. Frequentist random effects network meta-analyses were applied to the primary and secondary outcomes following Rücker (16) and Rücker and Schwarzer (17). Since one has to be cautious in comparing the results of the different routes of administration, as there may be an administration effect (18), separate network meta-analyses were performed for each route of administration, i.e. subcutaneous, intravenous and oral. In addition, pairwise meta-analyses were performed using the inverse variance method with restricted maximum likelihood (REML) estimation (19). Mean differences with 95% confidence intervals were estimated for continuous variables, and odds ratios with 95% confidence intervals for binary outcomes. Subgroup analyses were performed by repeating each analysis among studies only on participants with a) episodic migraine (EM) and b) chronic migraine (CM). In addition, analysis on the effect of medications on MMD was repeated after removing the studies in which MMD was not evaluated as a primary outcome. In studies with zero events arms, the treatment arm continuity correction with weights summing up to one by Sweeting et al. (20) was applied. This method considers different sample sizes and shall outperform fixed continuity corrections. Even if it is difficult to detect and to explore heterogeneity with such few studies for the single drugs, heterogeneity is here analysed in a graphical way for the pairwise comparisons. Additionally, the
Results
Included studies
The search yielded a total of 2314 citations from PubMed (n = 256), Embase (n = 1091) and Cochrane central (n = 967). After removing duplicates (n = 1181), 1133 citations were screened in two stages based on 1) title and abstract and 2) full text. Finally, 19 studies were eligible and included in the study. Figure 1 shows the flowchart of the study selection process in detail.

Flowchart of study identification, screening and inclusion.
Out of 19 studies, five (23–27) evaluated erenumab (n = 2939), five (28–32) fremanezumab (n = 3771), four (33–36) galcanezumab (n = 3364), two (37,38) eptinezumab (n = 2019), two (39,40) atogepant (n = 1744) and one (41) rimegepant (n = 747). All studies were published after 2017. A summary of baseline characteristics of all included studies is reported in Table 1, while online Supplementary Table 2 reports overall features for each medication.
Summary of baseline characteristics of all included studies.
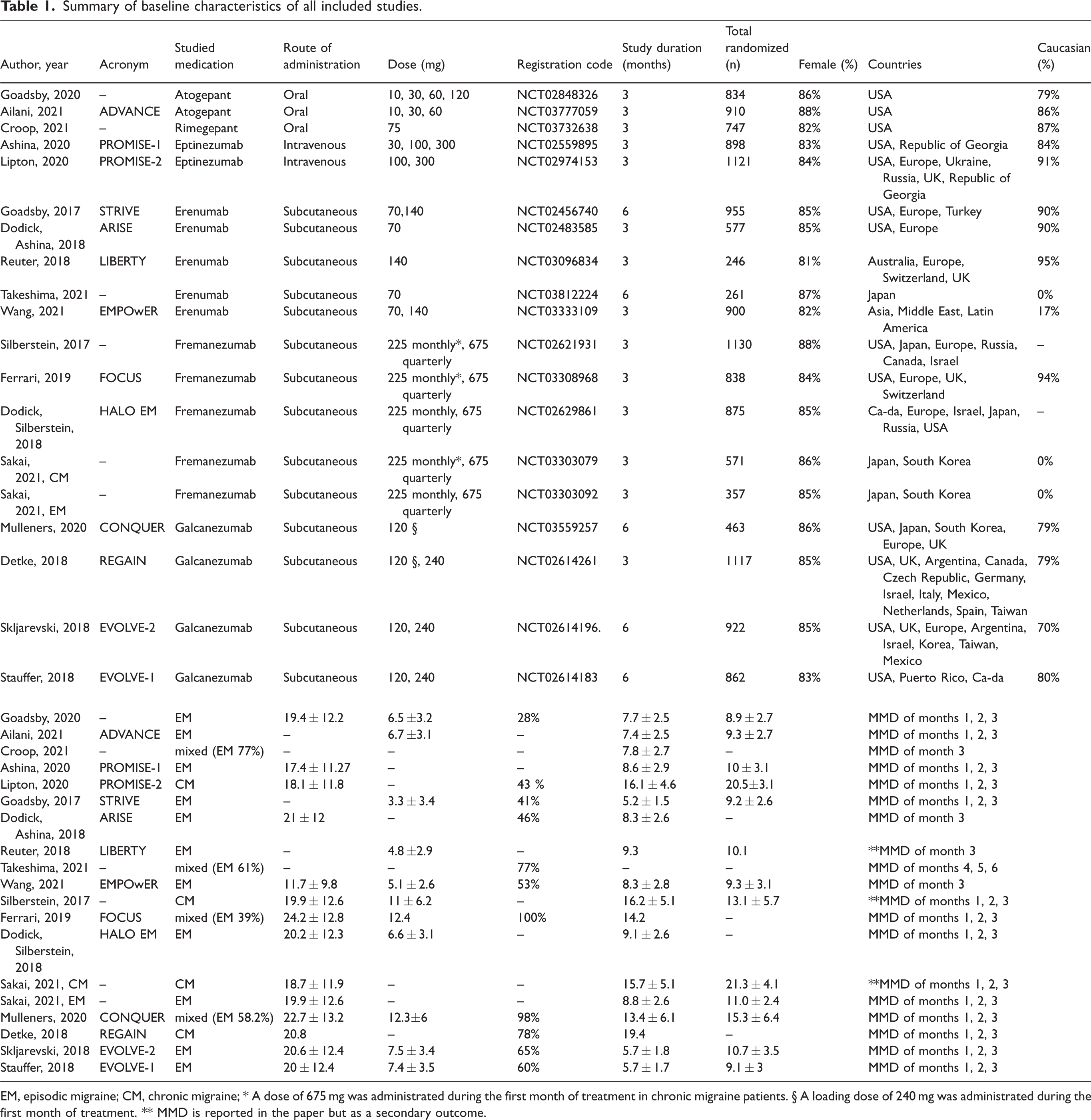
EM, episodic migraine; CM, chronic migraine; * A dose of 675
Participants were on average 41 ± 12 years old and 85% of them were female. Most participants were Caucasian (68%), with a mean BMI between 25.4 ± 4.9 and 30.4 ± 7.6 kg/m2. Eleven studies (23–25,27,29,31,35–37,39,40) enrolled only participants with episodic migraine and four studies (30,32,34,38) only those with chronic migraine, one for galcanezumab, one for eptinezumab and two for fremanezumab. The remaining studies (26,28,33,41) enrolled both episodic and chronic migraine participants. Of all the erenumab studies, only one included a subgroup of CM patients (26). Of note, in the fremanezumab studies with both EM and CM participants, the monthly fremanezumab treatment was given with a higher ‘loading’ dose of 675
As shown in Table 1, the double-blind treatment period was of three months in most studies, although for some (26,27,33,35,36) this lasted a total of six months. Further, not every trial evaluated the primary efficacy outcome of change in MMD in the same way. Most studies considered the mean MMD during the double-blind treatment period with respect to baseline, whereas three studies evaluated the mean MMD of the third month of the double-blind treatment period only (weeks 9–12) and one measured months 4, 5, 6. Moreover, two of the fremanezumab (30,32) studies and one erenumab (23) study did not have MMD as primary outcome, however, as this measure was still reported as a secondary outcome, we included it in our analysis.
Figure 2 reports the network of all included arms in the study separated based on the route of administration. Online Supplementary Table 3 reports the detail of baseline characteristics of each included arm. All included studies had a low risk of bias (with 100% agreement between the reviewers), as reported in online Supplementary Figure 1.

Network of included arms in the meta-analysis based on the monthly migraine days outcome. The lines between the interventions show the direct comparison in different studies. There are three separate nodes based on the drug route of administration.
Primary outcomes
Monthly migraine days (MMD)
Based on the network meta-analysis, all medications in all different doses reduced MMD compared to placebo (Figure 3). Out of medications with subcutaneous administration, fremanezumab 675

Comparison of different medications in available doses with placebo for primary outcomes. Results from three separate network meta-analysis based on the route of administration. Subgroup analysis based on migraine type (episodic vs. chronic) is reported on the right side of the figures. Only studies with 100% episodic or 100% chronic migraine participants were included in the subgroup analysis. n = number, MD = mean difference, OR = odds ratio, 95% CI = 95% confidence interval.
All oral medications in different doses reduced MMD compared to placebo, with atogepant 120
When considering studies only involving episodic migraine participants (middle columns in Figure 3) fremanezumab and galcanezumab still showed the highest efficacy in both available formulations followed by erenumab 140
When repeating this analysis by including only studies that reported MMD as a primary outcome (and thus excluding three studies) results were found to be nearly identical (see online Supplementary Figure 2).
≥50% responder rate
Data for this outcome was available for all studies and interventions. The network meta-analysis on medications with subcutaneous and intravenous route of administration showed significantly higher ≥50% responder rate for all medications compared to placebo (Figure 3) with fremanezumab 675
A similar pattern was observed by repeating the analysis in studies only including participants with episodic migraine. The analysis on chronic migraine studies showed that all available medications (eptinezumab 100 and 300
Secondary outcomes
Figure 4 reports the result of the network meta-analysis of the comparisons of all available medications with placebo for the secondary outcomes, including MHD, AMD, ≥75% responder rate and 100% responder rate.

Comparison of different medications in available doses with placebo for secondary outcomes. Results from three separate network meta-analysis based on the route of administration. Subgroup analysis based on migraine type (episodic vs. chronic) is reported on the right side of the figures. Only studies with 100% episodic or 100% chronic migraine participants were included in the subgroup analysis. n = number, MD = mean difference, OR = odds ratio, 95% CI = 95% confidence interval. Studies on eptinezumab did not report monthly acute medication day and 100% responder rate.
All arms with available data (atogepant, fremanezumab and galcanezumab all doses, eptinezumab 100 and 300
Out of the arms with data on ≥75% responder rate, all arms of subcutaneous and intravenous medications had a significantly higher rate compared to placebo. There was no data on 100% responder rate for intravenous arms, but subcutaneous medications showed a higher rate compared to placebo which was statistically significant for all arms except for erenumab 70
Pairwise analysis, network meta-regression and further analyses
Results of pairwise analyses for all outcomes are reported in online Supplementary Figures 3–8. In general, pairwise analyses supported the results found in the network meta-analysis. All arms with available data reduced MMD, MHD and AMD compared to placebo and had a higher rate of ≥50%, ≥75% and 100% responder rate. When looking at the heterogeneity within each treatment dose combination, some heterogeneity can be observed graphically. Naturally, heterogeneity can be better detected when more studies are available. Here, two fremanezumab studies in particular (28,29) seem to have larger treatment effects compared to the other three fremanezumab studies, as they showed higher reduction in MMD and higher ≥50% responder rate. To explore heterogeneity, network meta-regression was conducted. A significant effect can be detected for age (effect estimate: −0.19 [95% CI: −0.33; −0.06]). Using the SIDE approach for separating indirect from direct evidence, no significant local inconsistencies could be detected.
Discussion
The current study aimed to evaluate the efficacy of atogepant, rimegepant, erenumab, eptinezumab, fremanezumab, and galcanezumab on patients with migraine through network meta-analysis, in 19 phase 3 randomised controlled trials published from 2017 to May 2022 (23–41). It should be mentioned that all comparisons between different treatments are indirect and there are no head-to-head studies in the literature for comparison of CGRP mAbs and gepants with each other.
With regards to primary outcomes, all drugs and doses reduced MMD compared to placebo, with a range from about 2.3 days for fremanezumab 675
All arms with available data on mean monthly headache day outcome, including atogepant 10, 30, 60 and 120
Arms including atogepant 10, 30 and 120
The current meta-analysis shows the beneficial effects of anti-CGRP monoclonal antibodies in preventive migraine treatment compared to placebo. Our results support a previously published meta-analysis (42) on the impact of CGRP monoclonal antibodies on episodic migraine. Authors reported that medications improved preventive migraine outcomes such as MMD, AMD, and ≥50% responder rate. According to their results, eptinezumab showed the lowest amount of reduction in MMD compared to placebo among all medications (mean difference: –1 [95% CI: –2.20; 0.20]) in participants with episodic migraine. Our results presented a similar finding, as all doses of eptinezumab had lower MMD reduction compared to placebo among all arms in participants with episodic migraine. However, our sub-analysis looking into drug effects of both EM and CM groups allowed us to show that eptinezumab 100 and 300
Masoud et al. (43) showed that fremanezumab reduced MMD to a higher degree than other monoclonal CGRP antibodies. Our results reflect this outcome, but further show that galcanezumab at all doses had very similar efficacy rates. A network meta-analysis by Wang et al. (44) showed that all anti-CGRP monoclonal antibodies were effective in the reduction of MMD compared to placebo and that all medications were statistically similar compared to each other in ≥50% responder rates except for fremanezumab, which was significantly superior to eptinezumab. Our results showed similar pattern but as we had three nodes and performed separate network meta-analysis based on route of administration, we could not compare fremanezumab with eptinezumab. Further, their comparisons across drugs did not include different doses and the literature search, which included phase 2 trials, was until October 2020. Soni et al. (45) reported similar results to our review by evaluating the effect of different CGRP mAbs at different doses on participants with chronic migraine; they included phase 2 and 3 trials.
Overall, when considering methodological differences in the trials such as inclusion of chronic vs episodic migraine, duration of the randomisation period (three or six months) and different use of primary efficacy outcomes, the differences in efficacy profiles of the four antibodies are quite minor and all show clear superiority to placebo. A recent review comes to a similar conclusion (46), and for this same reason, the updated European Headache Federation guidelines on using CGRP mAbs for migraine prevention state these medications should be considered as first-line treatment (47). However, this might not be an option in many countries, as reimbursement and insurance coverage often require the proven failure of other prophylactic medications, such as onabotulinumtoxinA. These guidelines also state that efficacy should be checked after at least three months of treatment and, if there is no satisfactory responder, changing from one medication to another might be adequate (47).
Our results further showed that atogepant in all available doses improved efficacy outcomes after three months, including MMD, ≥50% responder rate, MHD, and AMD compared to placebo. Based on available data, rimegepant also improves MMD and AMD compared to placebo. For reasons that include oral administration and a shorter half-life (particularly important for women who plan a pregnancy), these gepants, particularly atogepant at the 120
Study strength and limitations
The current study is limited to papers in English and to published data. In addition, we did not include post-hoc analyses or open-label follow up studies, however, these have been covered in other papers within this Special Collection. Further, a parallel meta-analysis investigating the tolerability and efficacy of these same treatments is included in the Collection.
This study shows some limitations which are inherent with the available evidence on the topic, such as caucasian over-representation and episodic migraine predominance, as well as with the types of included studies. A direct example is that of studies using different timings for randomisations periods (three vs six months) and different primary outcomes (MHD instead of MMD reduction), showing heterogeneity even within the same intervention.
Our results should thus be regarded with caution, as they cannot substitute direct comparative studies and have limitations which are within the nature of a meta-analysis. To ensure that treatments with different routes of administration are comparable, direct comparison studies employing a double dummy technique are needed. The here included studies do not only differ in terms of route of administration, but also in terms of other characteristics. Overall, studies on fremanezumab and galcanezumab had older participants and with longer duration of the disease. These treatments showed also the largest effects compared to placebo regarding the primary outcome. This is another finding that highlights the importance of head-to-head studies to compare different anti-CGRP medications with each other in order to diminish the risk of confounders. It is also important to mention that our search was finalised in May 2022 and thus might have excluded important studies published after this date.
Although including just phase 3 trials resulted in missing some data, the current network meta-analysis is a report and summary of high-quality evidence. The result of RoB showed that all the studies had a low risk of bias. In addition, in the current study, all available migraine medications targeting CGRP are included at all doses as separate arms. Another strength of our study is represented by the analogous results of the pairwise comparisons that were conducted in parallel to the network meta-analysis.
Future directions
There is a lack of studies on comparing CGRP mAbs and gepants with each other and other medications. As an example, erenumab showed less adverse events and more efficacy based on ≥50% reduction in MMD, when compared to topiramate in a phase 4 clinical trial (48). Direct comparisons with a double dummy technique between different anti-CGRP medications in clinical trials, as well as comparisons with other available medications, are needed in the future.
Conclusion
The current systematic review and meta-analysis showed that the monoclonal antibodies against CGRP or its receptor, namely erenumab, eptinezumab, fremanezumab and galcanezumab, as well as small molecule CGRP receptor antagonists atogepant and rimegepant, are effective in the prevention of migraine compared to placebo. All medications reduced MMD and had higher ≥50% responder rate compared to placebo, even though MMD for eptinezumab 30
Article Highlights
A systematic review and network meta-analysis of phase 3 clinical trials on the efficacy of the calcitonin gene-related peptide mAbs and gepants on migraine prevention was conducted.
This study showed that novel treatments targeting the CGRP pathway including, atogepant, rimegepant, erenumab, eptinezumab, fremanezumab, and galcanezumab, are effective in the prevention of migraine compared to placebo.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231159366 - Supplemental material for Evaluating the efficacy of CGRP mAbs and gepants for the preventive treatment of migraine: A systematic review and network meta-analysis of phase 3 randomised controlled trials
Supplemental material, sj-pdf-1-cep-10.1177_03331024231159366 for Evaluating the efficacy of CGRP mAbs and gepants for the preventive treatment of migraine: A systematic review and network meta-analysis of phase 3 randomised controlled trials by Faraidoon Haghdoost, Francesca Puledda, David Garcia-Azorin, Eva-Maria Huessler, Roberta Messina and Patricia Pozo-Rosich in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024231159366 - Supplemental material for Evaluating the efficacy of CGRP mAbs and gepants for the preventive treatment of migraine: A systematic review and network meta-analysis of phase 3 randomised controlled trials
Supplemental material, sj-pdf-2-cep-10.1177_03331024231159366 for Evaluating the efficacy of CGRP mAbs and gepants for the preventive treatment of migraine: A systematic review and network meta-analysis of phase 3 randomised controlled trials by Faraidoon Haghdoost, Francesca Puledda, David Garcia-Azorin, Eva-Maria Huessler, Roberta Messina and Patricia Pozo-Rosich in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FH, EMH and FP report no conflict of interest.
RM reports personal fees from Eli-Lilly, Lunbeck and Bromatech for participating in advisory boards and speaker activities over the last 36 months.
DGA in the last 24 months has received personal compensation for consulting/advising from the World Health Organization. Non-profit board membership in the Spanish Society of Neurology, and the European Union of Medical Specialist section of Neurology. Research funding from the Regional Health Administration (Gerencia Regional de Salud SACYL) in Castilla y Leon, Spain. Speaker/travel grants/clinical trials from Teva, Allergan, Amgen, Eli Lilly, Lundbeck, Novartis, and Biohaven. There are no conflicts of interest in regard to this manuscript.
PP-R in the last 36 months has received honoraria as a consultant and speaker for: AbbVie, Biohaven, Chiesi, Eli Lilly, Medscape, Lundbeck, Novartis, Pfizer and Teva. Her research group has received research grants from AbbVie, Novartis and Teva; has received funding for clinical trials from Alder, Amgen, Biohaven, Electrocore, Eli Lilly, Lundbeck, Novartis, Teva. She is the Honorary Secretary of the International Headache Society. She is in the editorial board of Revista de Neurologia. She is an editor for Cephalalgia, Headache, Neurologia, Frontiers of Neurology and advisor for The Journal of Headache and Pain. She is a member of the Clinical Trials Guidelines Committee of the International Headache Society. She has edited the Guidelines for the Diagnosis and Treatment of Headache of the Spanish Neurological Society. She is the founder of ![]() . PP-R does not own stocks from any pharmaceutical company.
. PP-R does not own stocks from any pharmaceutical company.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
