Abstract
Background
Migraine with aura (MwA) is a debilitating disorder characterized by paroxysmal attacks of pain preceded or accompanied by reversible neurological symptoms. While the pathophysiology remains unclear, trigeminovascular system activation and cortical spreading depression have been implicated. This study aims to comprehensively investigate and characterize the diverse clinical features and manifestations of aura, as well as the types of acute medications self-administered for aura management.
Methods
A multicenter, cross-sectional study was conducted using data from the Italian Headache Registry (RICe). Aura characteristics, frequency, duration and associated migraine premonitory symptoms were collected. Acute medication use and timing (headache or aura phase) were assessed.
Results
The study included 272 patients with a diagnosis of MwA. Most patients (99.3%) experienced typical aura symptoms, with visual aura (96.3%) being the most prevalent, followed by sensory (33.0%) and speech and/or language aura (25.6%). Brainstem aura (8.5%) and motor aura (1.8%) were less common. Notably, 13.0% of patients reported aura relapses within 24 hours. Triptans (39.7%), non-steroidal anti-inflammatory drugs (47.8%) and nutraceuticals (59.9%) were commonly used for acute aura management.
Conclusions
This study reports several different aura manifestations, highlighting atypical features, aura relapse rates and treatment approaches for aura. These findings could contribute to a deeper understanding of aura and its management in clinical settings.
This is a visual representation of the abstract.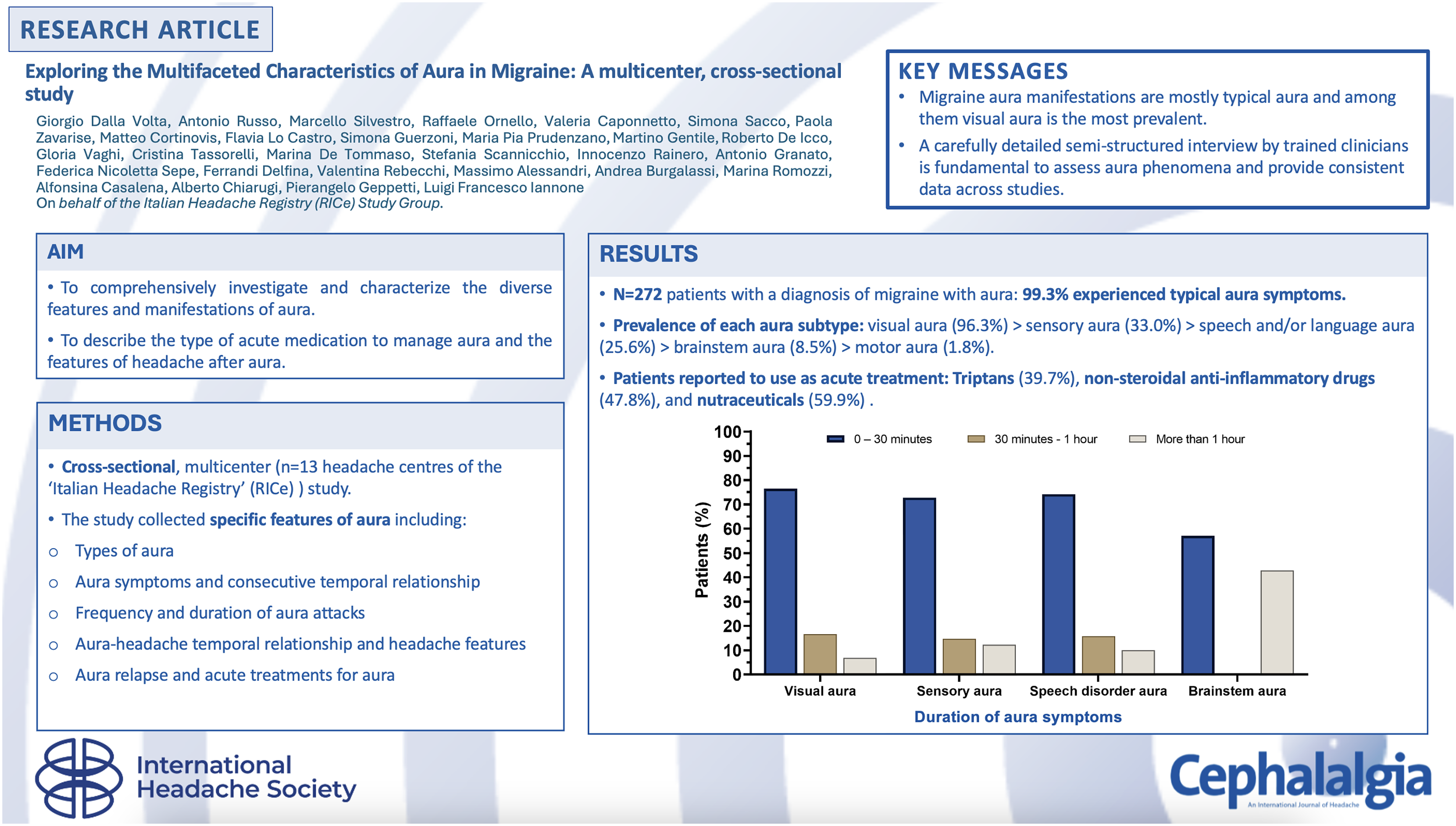
Introduction
Migraine is a common neurological disorder that ranks as one of the most debilitating medical conditions, characterized by recurrent, paroxysmal attacks of varying intensity and frequency (1,2). Close to one-third of individuals with migraine experience aura, a reversible neurological phenomenon that manifests with visual, sensory, speech and motor focal neurologic symptoms, usually lasting 5–60 minutes, either preceding or accompanying headache pain (3,4). Although the underlying mechanism is poorly understood, migraine appears to be caused by the activation and sensitization of the trigeminovascular system, which eventually results in the release of neurogenic inflammatory neuropeptides, including the calcitonin gene-related peptide (CGRP) (5,6). Cortical spreading depression, a self-propagating wave of depolarization spreading across the cerebral cortex, has been proposed as the pathophysiological mechanism that underlies migraine aura (3,7).
Migraine aura manifestations can be highly variable, including several neurological manifestations with different durations. These symptoms may be accompanied by headache, and patients may experience different aura subtypes throughout their lives, making this disorder a highly multifaceted and heterogeneous condition (8–10). Detailed characterization of the clinical features of aura is of paramount importance to improve diagnosis in clinical practice and increase disease knowledge.
Herein, we report a multicenter, cross-sectional study with the aim of comprehensively investigating and characterizing the diverse features and manifestations of aura and related symptoms experienced by patients diagnosed with migraine with aura (MwA), and the management of aura episodes.
Methods
Study design and ethics
In 2019, the “Italian Headache Registry” (Registro Italiano per le Cefalee, RICe) was established to evaluate primary headache disorder epidemiology in Italy; RICe currently involves 52 Italian headache centers (any level of care), and all patients attending these centers are proposed to be inserted in the registry.
Hence, we conducted a multicenter, cross-sectional study involving patients with a diagnosis of MwA, including 13 centers that were partners in the project. The study consisted of collecting specific features of aura, subsequent headache and acute treatments. All consecutive outpatients who had a diagnosis of MwA between January 2019 and December 2022 were included in the study (regardless of whether it was the first visit or follow-up visits) and provided informed consent to participate in the RICe study (Figure 1). Patients were then assessed by trained personnel with a semi-structured questionnaire that recalled detailed information about the nature, frequency, duration and relapses of aura. The study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
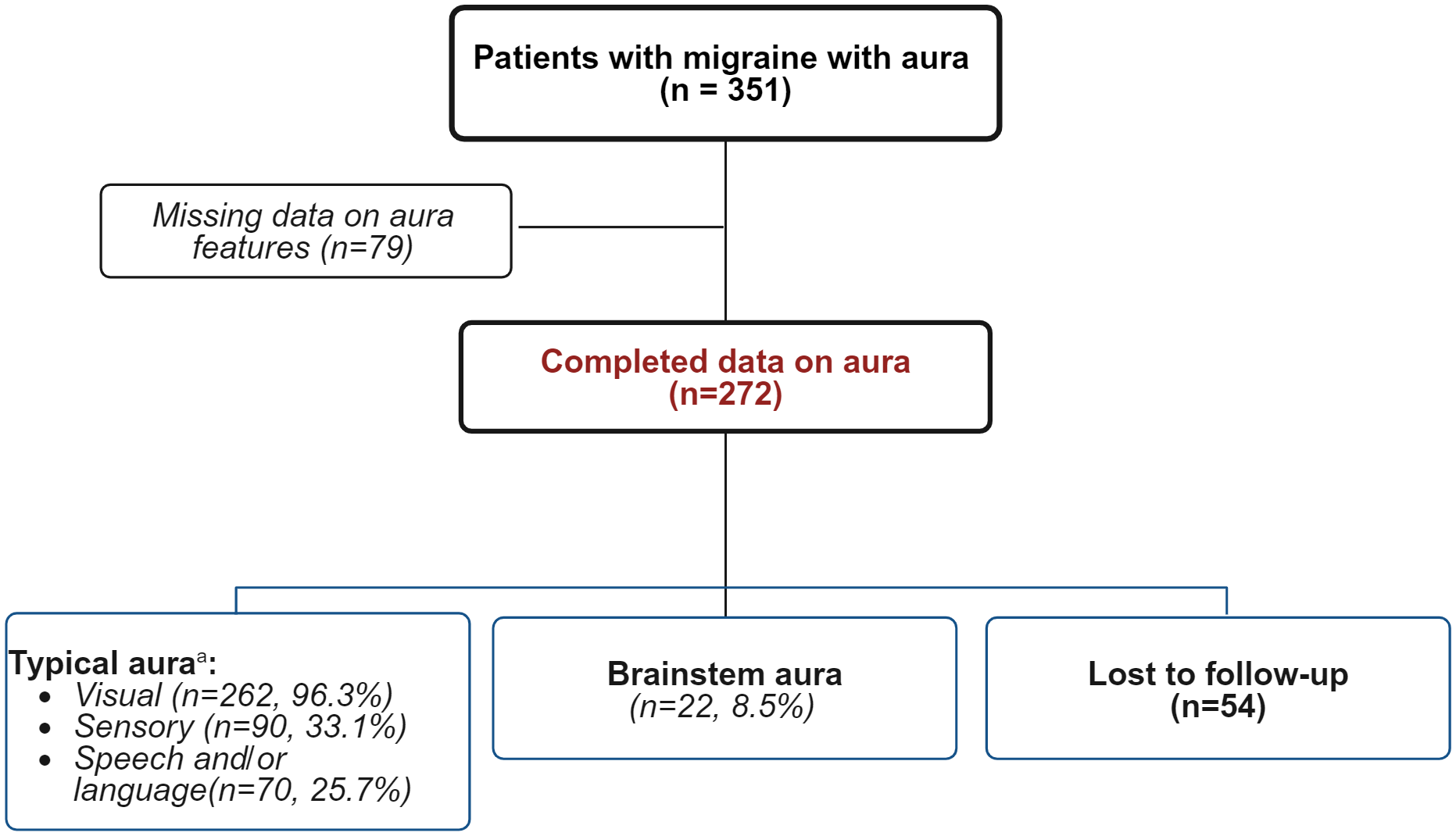
Flowchart of patients and type of aura.
Aims
The primary objective of this descriptive study was to comprehensively investigate and characterize the diverse features and manifestations of aura experienced by patients diagnosed with MwA. Specifically, the study aimed to provide an extensive account of the various types of aura symptoms encountered by patients, encompassing visual disturbances, sensory alterations and speech impairments. The secondary objectives were to describe the type of acute medication used by the patient to manage aura episodes with or without a specific indication or prescription, the timing of administration (aura or headache onset) and the features of headache after aura (if reported).
Patient features and variables collected
The study recruited adult patients diagnosed with MwA according to the International Classification of Headache Disorders 3rd Edition (ICHD-3) criteria (11), with or without other headache diagnoses. Patients who declined to provide informed consent, as well as those with incomplete clinical data, were excluded. Sub-diagnoses of MwA according to ICHD-3 have been reported if available. The variables collected for aura, as reported by the patients as the most common pattern in the last year, were:
Aura onset (age); Aura symptoms: visual, sensory, speech and/or language, motor, brainstem, retinal; Frequency of aura attacks categorized in the subsequent categories (in the last year): <5 per year, ≥5 per year and <1 per month, >1 per month and <1 per week, and ≥1 per week (without overlapping among groups); Duration (in minutes); Aura-headache temporal relationship (before and/or together with pain) Consecutive aura symptoms temporal relationship (if more than one symptom); Premonitory migraine symptoms; Aura relapse (defined as the recurrence of aura within the first 48 hours); Headache features after aura; Acute treatments for aura.
All variables were documented electronically and extracted using the RICe registry. The semi-structured questionnaire is reported in the supplementary material.
Statistical analysis and missing data
Considering the descriptive nature of the study, the sample size was not determined based on statistical considerations. All consecutive outpatients who had a diagnosis of MwA during the study period were included.
We reported the mean ± SD or median and interquartile range for continuous variables, and number (percentage) for categorical data. No imputation was performed for missing data, and the number of patients analyzed for a single variable is reported in the figure and table legends where appropriate. Few questions reported in the semi-structure questionnaire (see supplementary material; evidenced with an asterisk, (*)) were not consistently assessed by the centers and therefore not reported in the results. Multiple types of auras could be reported for the same patient, leading to a cumulative percentage exceeding 100% in some instances. The software SPSS, version 26.0 (IBM Corp., Armonk, NY, USA) was used for all data analyses.
Results
Cohort characteristics
Among the 351 patients with MwA diagnosis present in the database, we included a total cohort of 272 patients with reported data on aura. The detailed features of aura were not included for 79 patients, and therefore they were excluded from the analysis. The mean ± SD age of the patients was 43.3 ± 14.5 years. Among the cohort, 223/272 (82%) were female. The mean ± SD age of onset for aura was 23.9 ± 11.2 years. Regarding other concomitant headache diagnoses, the distribution was: 243 patients with migraine without aura (89.3%) and 35 (12.9%) with chronic migraine, other concomitant headache are reported in Table 1.
Patients demographic and clinical features.
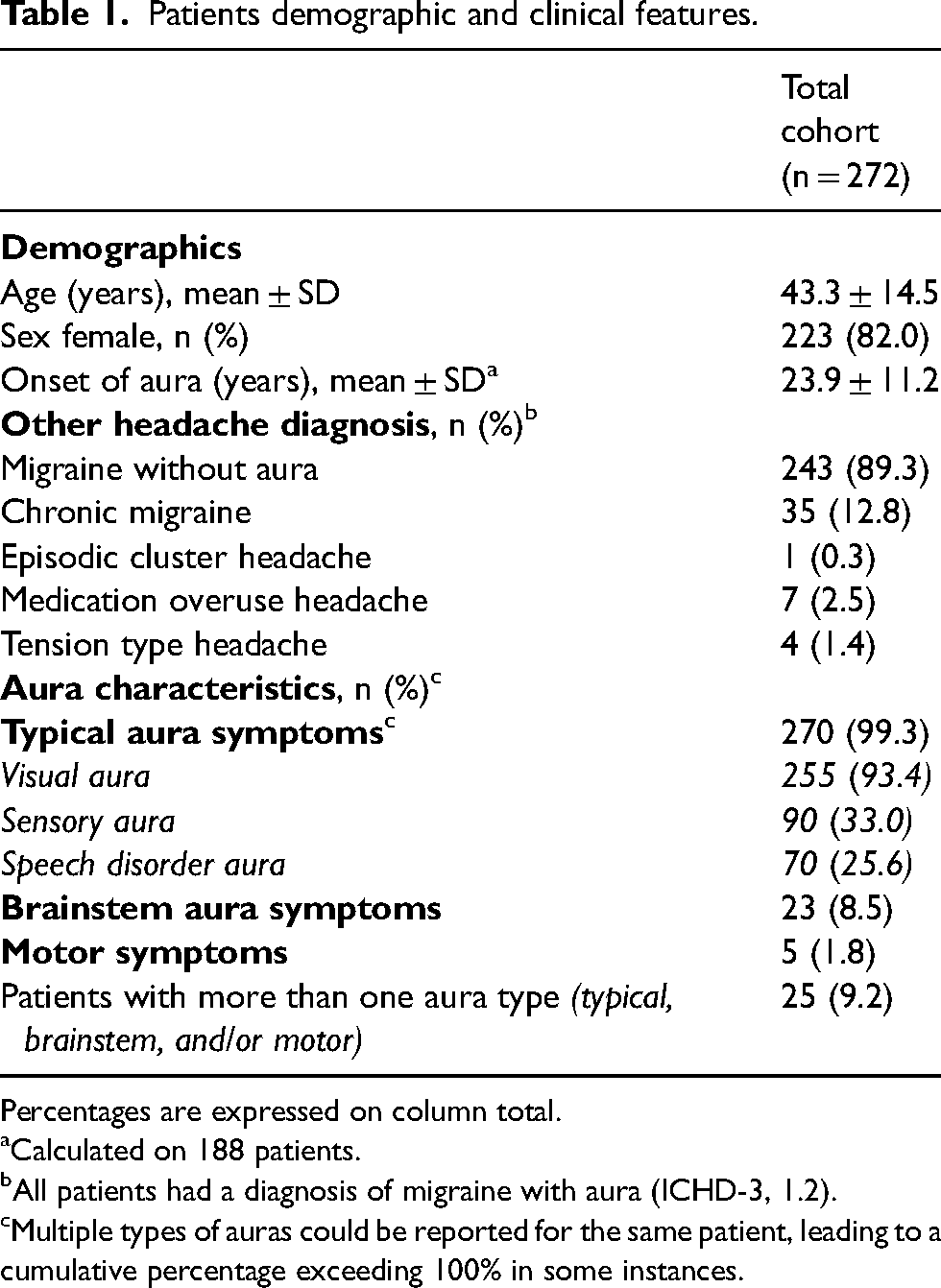
Percentages are expressed on column total.
Calculated on 188 patients.
All patients had a diagnosis of migraine with aura (ICHD-3, 1.2).
Multiple types of auras could be reported for the same patient, leading to a cumulative percentage exceeding 100% in some instances.
Frequency of aura in the last year was available for 216 patients and categorized according to predefined cut off: < 5 attacks per year (n = 81; 37.5%), ≥ 5 attacks per year and <1 per month (n = 31; 14.4%), ≥ 1 per month and <1 per week (n = 71; 32.9%), ≥ 1 attacks per week (n = 33; 15.3%).
According to the ICHD3 classification criteria, 270/272 (99.3%) patients experienced typical aura symptoms, 23 (8.5%) patients had brainstem aura and five (1.8%) had motor aura (Figure 1). Notably, 25 patients (9.2%) reported experiencing more than one type of aura in the same attack, including typical, brainstem and/or motor symptoms. No cases of retinal aura fulfilling the ICHD-3 criteria were reported in our cohort. All details are reported in Table 1.
Aura features
Typical aura
Among the group of patients with typical aura, visual aura was the most prevalent (n = 262; 96.3%), followed by sensory aura (n = 90; 33.1%) and speech and/or language aura (n = 70; 25.7%).
Regarding visual aura, 96/272 patients (35.3%) reported more than one subtype. Positive scotomas (colored, bright, white dot or dots) were reported by 224 patients (82.4%), while 39 (14.3%) described negative scotomas. A smaller subset of patients (n = 10; 3.7%) reported visual snow-like disturbances and 37 (13.6%) experienced blurred vision or dyschromia. The duration of visual aura varied, with 17 patients (6.9%) reporting more than one hour, 41 (16.6%) from 30 minutes to one hour, and 189 (76.5%) between 5 and 30 minutes (Figure 2).
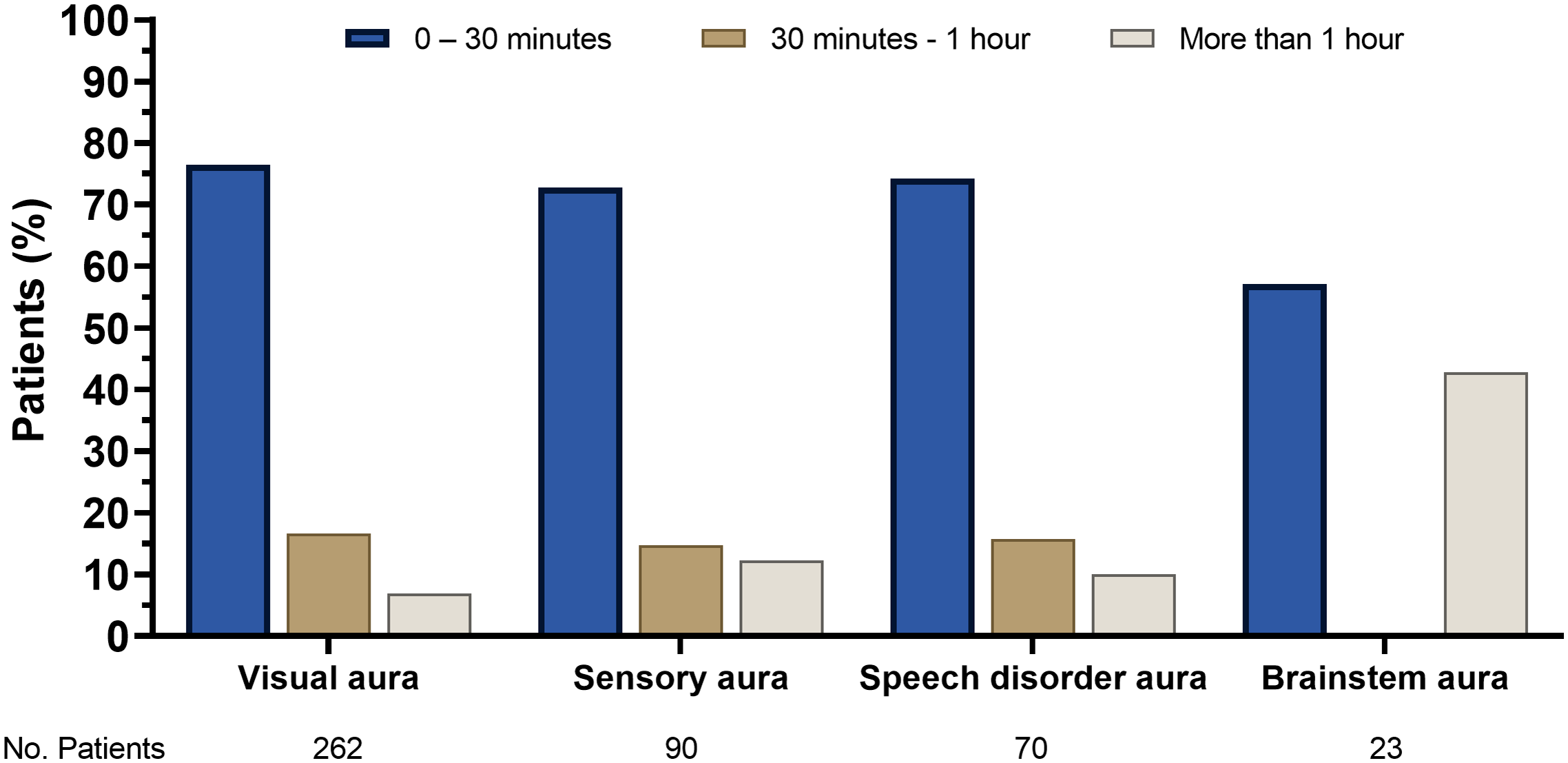
Usual duration of aura subdivided by subtype.
Sensory aura, specifically paresthesia, was reported by 90 patients (33.1%). These sensations were localized in various body regions, including one upper limb (n = 87; 32.0%), one lower limb (n = 7; 2.7%), half of the face (n = 39; 14.3%) and half of the tongue (n = 15; 5.5%). The duration of a single episode of sensory aura in 10 patients (12.3%) lasted more than one hour, in 12 (14.8%) ranged from 30 minutes to one houe and, in the majority (n = 59; 72.8%), subsided within five to 30 minutes.
Speech and/or language aura was experienced by 70 patients (25.7%), with 51 (18.8%) presenting anomia and 23 (8.5%) exhibiting dysarthria. In terms of duration, in seven patients (10.0%), it lasted more than one hour, in 11 patients (15.7%) from 30 minutes to one hour and, in 52 patients, (74.3%) between five and 30 minutes. In Table 2, a comprehensive description of the typical aura features, their duration, and localization is reported.
Typical aura features, duration and localization. a

Percentages are expressed on column total if not otherwise specified.
Multiple types of auras could be reported for the same patient, leading to a cumulative percentage exceeding 100% in some instances.
Calculated on 247 patients.
Calculated on 81 patients.
Calculated on 70 patients.
Brainstem aura
Overall, 23/272 patients (8.5%) reported brainstem aura symptoms. Only five (5/23; 21.7%) patients reported isolated brainstem aura. Among the brainstem aura features, a decreased level of consciousness was reported in four patients (1.5%). Vertigo was experienced by 14 patients (5.2%), while diplopia and tinnitus were reported by seven patients (2.6%), respectively. Hypoacusis and ataxia were reported by four (1.5%) and two (0.7%) patients, respectively. The duration of brainstem aura symptoms was highly variable (data were available for seven patients): three patients reported a duration of more than one hour, while four patients (57.1%) reported a duration of five to 30 minutes. Notably, no instances of brainstem aura lasting between 30 minutes and one hour were reported in this subset of patients. From this group, nine patients (9/23; 39.1%) presented with more than one subtype of brainstem aura. The features and duration of brainstem aura are reported in Table 3.
Brainstem aura features and duration. a
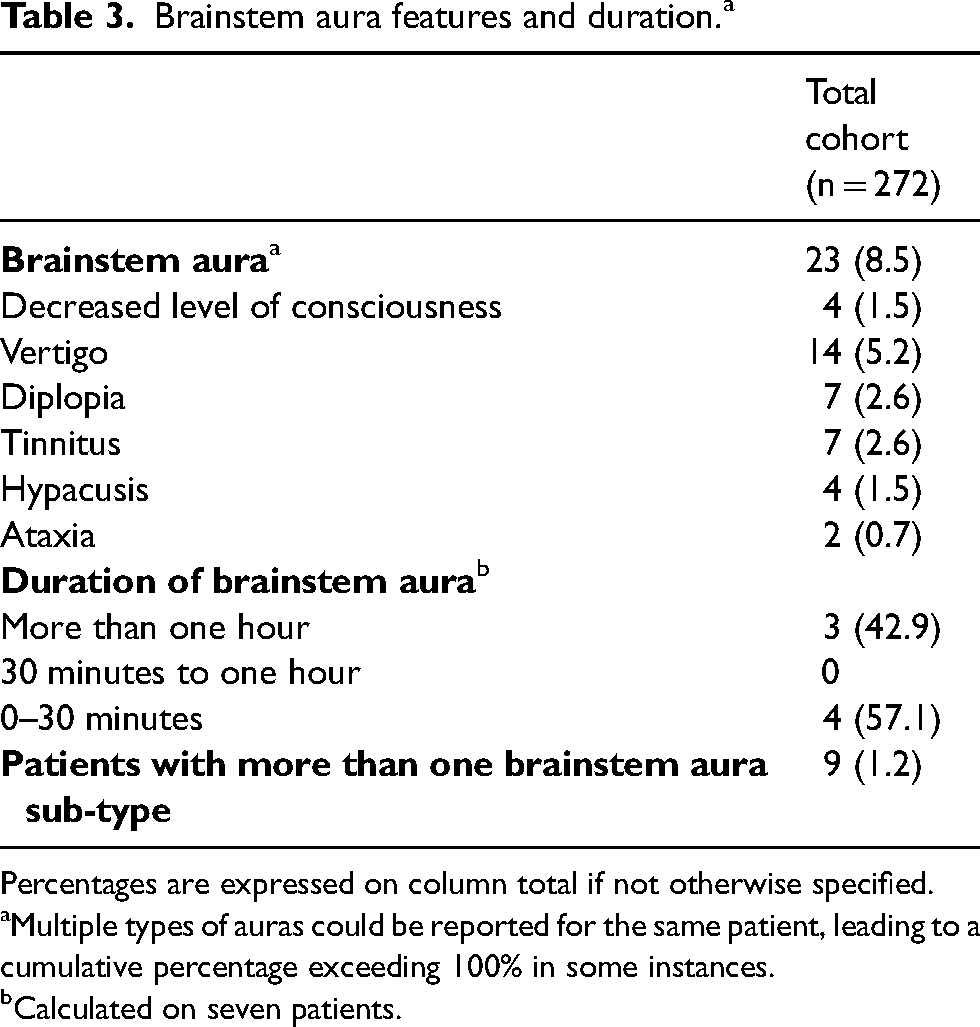
Percentages are expressed on column total if not otherwise specified.
Multiple types of auras could be reported for the same patient, leading to a cumulative percentage exceeding 100% in some instances.
Calculated on seven patients.
Motor aura
A small subset of patients (5/272; 1.8%) reported motor symptoms. Motor aura localized in both upper limbs was reported by one patient (0.4%), whereas motor aura limited to one upper limb was reported by fuur patients each (1.5%). No genetic data were available. Motor aura features and duration are reported in Table 4.
Motor aura features and duration.a

Percentages are expressed on column total if not otherwise specified.
Aura relapse and migraine premonitory symptoms
In the total cohort of 272 patients, 38 patients (14.1%) reported aura relapses. In 34 patients (12.6%), aura relapse occurred in the first 24 hours, while, in four patients (1.5%), it occurred the day after. The mean ± SD duration of aura relapse was 85.2 ± 84.4 minutes (on 22 patients with available data).
Only 25/272 patients (9.2%) reported premonitory symptoms of migraine with aura attacks, with 10 (3.7%) reporting more than one symptom. These symptoms included mood changes (n = 6; 2.2%), anxiety (n = 5; 1.8%), yawns (n = 4; 1.5%), and paleness and confusion (n = 3 each; 1.1%).
Acute symptomatic treatment use
Out of 272 patients, 108 patients (39.7%) reported triptan use. Notably, 29 patients (26.9%) employed triptans at the onset of aura, while 22 (20.8%) utilized them at the onset of headache. The remaining 57 patients (52.3%) did not specify the timing of triptan use. The use of non-steroidal anti-inflammatory drugs (NSAIDs) was reported by 130 patients (47.8%). Among them, 33 patients (25.3%) used NSAIDs at aura onset, while 54 patients (41.5%) used them when the headache started. Forty-three patients (33.2%) did not specify the timing of use. One hundred sixty-three patients (59.9%) reported the use of dietary supplements (including magnesium, nisin, magnesium plus tanacetum-hydroxytriptofan, coenzyme Q) for the acute management of aura, mainly at aura onset. All data are reported in Table 5.
Acute analgesics use.
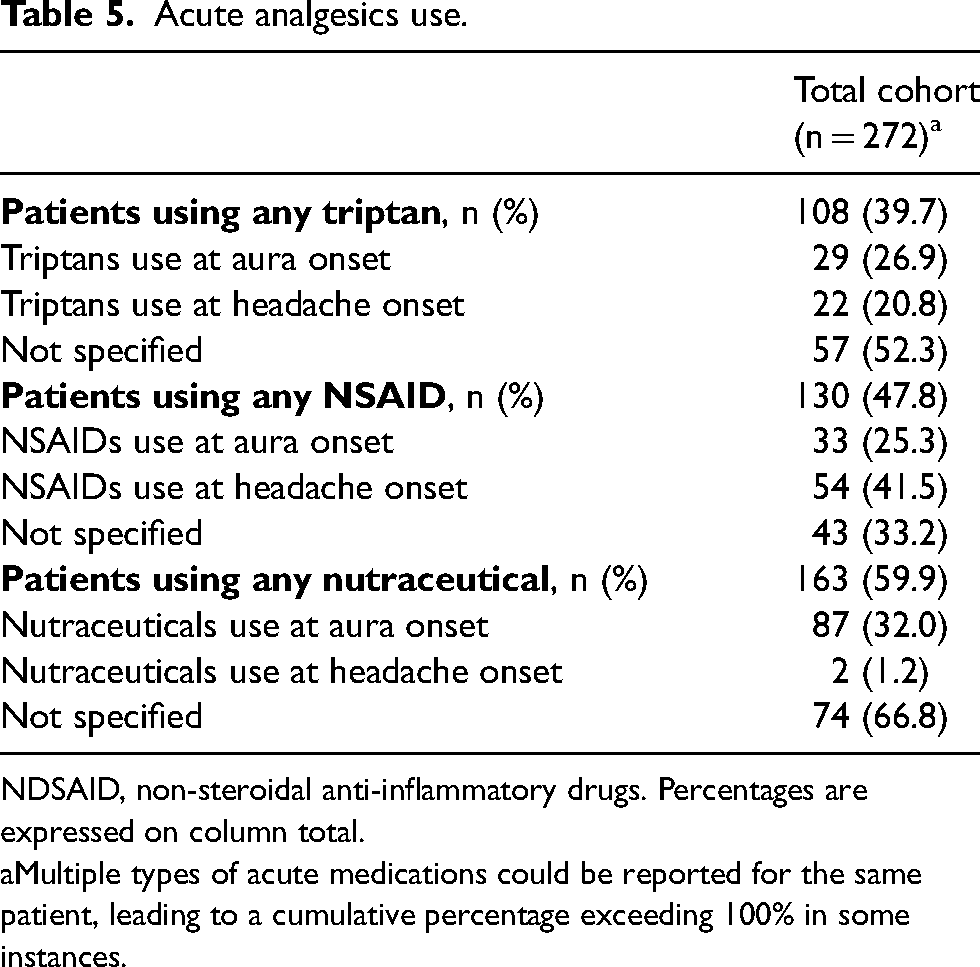
NDSAID, non-steroidal anti-inflammatory drugs. Percentages are expressed on column total.
Multiple types of acute medications could be reported for the same patient, leading to a cumulative percentage exceeding 100% in some instances.
Headache features and accompanying symptoms in migraine with aura attacks
These data are available only for patients with a diagnosis of migraine with aura and without aura (n = 243). Among them, 197 patients (81.0%) reported the detailed features of the migraine attack and were included in the analysis. When compared to headache attacks without aura, 95 patients (48.2%) found the headache to be more painful after aura (pain intensity), 14 (7.1%) experienced lower pain intensity and 88 (44.7%) reported no meaningful difference. According to a Numeric Rating Scale (NRS) rating the perceived pain level, 15 patients (8.2%) reported mild pain (NRS: 1–4), 109 patients (59.6%) moderate pain (NRS: 5–8) and 59 patients (32.2%) severe pain (NRS: 9–10).
Accompanying symptoms during headache episodes following aura were diverse, with the most frequent being photophobia/phonophobia (55.1%), nausea/vomiting (55.9%) and vertigo (12.5%). The other accompanying symptoms are reported in Table 6. Among 88 patients with available data, aura symptoms occurred along with headache in 39 patients (44.3%). Considering asthenia regardless of headache occurrence, 116 of 195 patients (59.5%) reported it; among these, 55 patients (28.2%) reported asthenia lasting less than 24 hours, while 61 patients (31.3%) reported more than 24 hours. All data are reported in Table 6.
Headache following aura in patients experiencing episodes with aura and without aura.
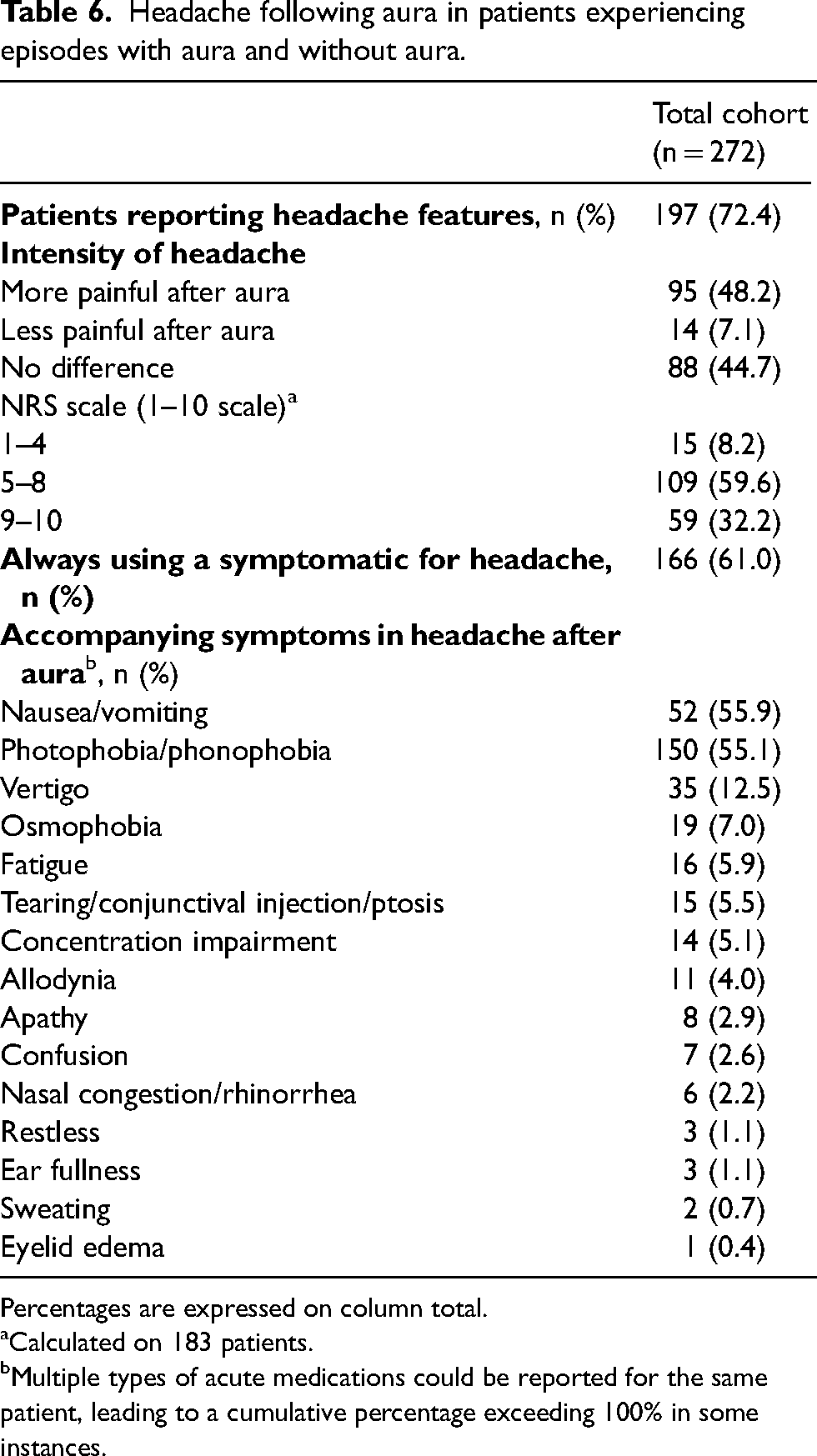
Percentages are expressed on column total.
Calculated on 183 patients.
Multiple types of acute medications could be reported for the same patient, leading to a cumulative percentage exceeding 100% in some instances.
Discussion
In this study, we included and detailed the aura features of numerous patients experiencing the aura phenomenon. In line with the literature, our data confirm that most patients manifest visual symptoms (96.0% of patients), followed by somatosensory aura (33.0% of patients) and language/speech aura (25.0% of patients). Furthermore, the features of less common types of aura, such as brainstem and motor aura, have been characterized. These findings are in line with previous observational studies and the recent paper from the REFORM study (12,13). Interestingly, in the REFORM study (13), the percentage of aura features reported is very similar to our data (visual aura: 94.7%; somatosensory: 35.7%; language/speech: 13.6%). These results suggest that semi-structured interviews performed by trained specialists could be very useful in correctly diagnosing and characterizing less common aura phenomena. These data are also in line with previous observational studies collected in a systematic review (10).
Regarding visual aura manifestations, more than 80.0% of patients reported positive symptoms, while only 14.0% reported negative symptoms. These percentages are comparable or slightly higher (67.0%) when compared to previously published data (8,9,13,14). Other reported visual phenomena included visual snow-like disturbances (3.7%) and dyschromia/blurred vision (13.0%). Somatosensory aura was predominantly reported as a gradually spreading paresthesia with different body localizations, in line with other studies (14); speech- and/or language aura was experienced by 25.7% of patients, with anomia or dysarthria as reported manifestations. Notably, 9.2% of patients experienced more than one type of aura, including typical, brainstem, and/or motor symptoms.
In relation to aura duration (including all types of aura), only 10.0% of patients reported aura lasting more than 60 minutes. Similarly, in the REFORM study (13), it was found that visual aura persisted for less than 60 minutes in 89.8% of patients, and for speech/language, this duration exceeded 60 minutes in 16.1% of cases. However, somatosensory aura lasted for over 60 minutes in approximately one-third of the participants. The majority of patients in our cohort reported aura duration of less than 30 minutes. It is worth noting that this finding may be biased by the presence of different aura symptoms in several patients, with the consequence that patients experiencing only one symptom would tend to report a shorter aura duration than those patients who experience different types of aura symptoms in succession.
For patients suffering from migraine both with and without aura, headache pain intensity is often higher in attacks preceded by aura compared to those not preceded by aura symptoms. In our cohort, 16.5% of the patients reported the following as accompanying symptoms of migraine with aura attacks (during or after aura): pronounced asthenia, difficulty in concentration, apathy, or confusion. These symptoms could last more than 24 hours after the end of the headache attack. These findings are often not reported by clinicians, highlighting the importance of structured semi-interviews to investigate all migraine phases, including the premonitory phase, accompanying symptoms and aura symptomatology, in clinical practice.
Finally, we investigated treatment strategies for aura episodes and related headache: 40.0% of patients used oral triptans, equally at aura onset and at headache onset. Although some previous studies reported that the use of triptans (specifically sumatriptan, eletriptan and zolmitriptan) during the aura phase was ineffective (15,16), other later studies have shown that treatment with triptans (including subcutaneous sumatriptan) during the aura phase prevented the development of the headache (17). To note, the administration of triptans during the migraine aura phase seems not to affect aura duration or features (15), but no strong evidence supports the use of triptans during the aura phase, and they are contraindicated in hemiplegic migraine and migraine with brainstem aura (18). Almost half of the patients used NSAIDs not only often at headache onset, but also at aura onset (25.0%). Finally, 60.0% of patients used dietary supplements suggested for the acute treatment of the aura phenomena (generally at aura onset).
Several lines of evidence support the role of CGRP in migraine with aura: (i) anti-CGRP monoclonal antibodies resulted effective in both migraine days with or without aura (19,20); (ii) anti-CGRP monoclonal antibodies administration modified the clinical features of aura (21,22); and (iii) CGRP infusion is able to induce aura in migraine with aura patients (5). Starting from these observations, it would be interesting to investigate the effects of the acute use of gepants during the aura phase.
Several strengths characterize our study, including the use of a semi-structured interview performed by trained specialists and the large sample size. There are, however, some limitations. The study's descriptive nature (cross-sectional) limits the ability to capture precisely the duration of aura, the presence of some symptoms (prodromal/premonitory) or the within attack variability. In addition, the use of convenience sampling may introduce selection bias, and reliance on self-report data might be susceptible to recall bias. Finally, as expected, some variables have missing data, as reported appropriately within this paper.
Conclusions
Visual symptoms are the predominant manifestation of aura, followed by somatosensory and speech/language symptoms. The accompanying symptomatology and the investigation of less common aura manifestations are of paramount importance to better characterize migraine with aura. A carefully detailed semi-structured interview by trained clinicians, is fundamental to better assess aura phenomena and provide consistent data across studies.
Article highlights
Migraine aura manifestations can be highly variable in clinical features and duration. Visual symptoms are the predominant manifestation of aura, followed by somatosensory and speech/language symptoms. A carefully detailed semi-structured interview by trained clinicians, is fundamental to assess aura phenomena better and provide consistent data across studies.
Supplemental Material
sj-doc-1-cep-10.1177_03331024241299453 - Supplemental material for Exploring the multifaceted characteristics of aura in migraine: A multicenter, cross-sectional study
Supplemental material, sj-doc-1-cep-10.1177_03331024241299453 for Exploring the multifaceted characteristics of aura in migraine: A multicenter, cross-sectional study by Giorgio Dalla Volta, Antonio Russo, Marcello Silvestro, Raffaele Ornello, Valeria Caponnetto, Simona Sacco, Paola Zavarise, Matteo Cortinovis, Flavia Lo Castro, Simona Guerzoni, Maria Pia Prudenzano, Martino Gentile, Roberto De Icco, Gloria Vaghi, Cristina Tassorelli, Marina De Tommaso, Stefania Scannicchio, Innocenzo Rainero, Antonio Granato, Federica Nicoletta Sepe, Delfina Ferrandi, Valentina Rebecchi, Massimo Alessandri, Andrea Burgalassi, Marina Romozzi, Alfonsina Casalena, Alberto Chiarugi, Pierangelo Geppetti and Luigi Francesco Iannone in Cephalalgia
Footnotes
Acknowledgments
Author contributions
All authors critically reviewed the manuscript, agreed to be fully accountable for ensuring the integrity and accuracy of the work, and read and approved the final version of the manuscript submitted for publication.
Data availability
Data supporting the findings in the present study are reported in the article. The data collected and analyzed for the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: PG received personal fees from Allergan, Eli Lilly, Novartis, Amgen and TEVA; grants from Amgen, TEVA, Eli-Lilly, Allergan and Chiesi; is on the Scientific Advisory Board for Endosome Therapeutics; and is a founding scientist of FloNext srl, Spinoff of the University of Florence. FDC received personal fees from TEVA, Eli Lilly and Novartis. LFI received personal fees from Eli-Lilly and TEVA, and a travel grant from Lundbeck. SS reports personal fees as a speaker or advisor from Abbott, Allergan-AbbVie, AstraZeneca, Eli Lilly, Lundbeck, Novartis, NovoNordisk, Pfizer and Teva, and research grants from Novartis. RO reports personal fees from Novartis, Eli Lilly and Teva, and non-financial support from Novartis, Allergan, Eli Lilly and Teva. SG received personal fees from Allergan/AbbVie, Eli Lilly, Novartis, Teva and Lundbeck. Grazia Sances received personal fees as a speaker or was on the Advisory Board for Eli-Lilly, Novartis, TEVA, Lundbeck and Pfizer. RDI received speaker honoraria and advisory board participation: Eli-Lilly, TEVA, Lundbeck, Pfizer, AbbVie; MPP received speaker honoraria and advisory board participation from Abbvie, Idorsia, Eli-Lilly, Novartis, Pfizer, TEVA. The other authors have no conflicting interests.
Ethical statement
This study is part of the Registro Italiano Cefalee (RICe) study approved by the local Ethics committee (Comitato Etico Area Vasta Toscana Centro; CEAVC Studio RICe, 14591_oss and subsequent amendments 2022-609).
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
ORCID iDs
RICe study group
Francesco De Cesaris14, Giulia Vigani15, Agnese Onofri3, Adriana Fallacara5, Maria Elena Roca5, Grazia Sances7, Michele Corrado6,7, Elena Mazzotta6,7, Gabriele Garascia10, Andrea Marcinnò9, Daniele Michelis11 and Fabrizio Valleggi13.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
