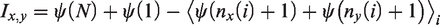Abstract
Objective
Growing evidence shows a critical role of network disturbances in the pathogenesis of migraine. Unilateral pattern of neurological symptoms of aura suggests disruption of interhemispheric interactions during the early phase of a migraine attack. Using local field potentials data from the visual and motor cortices, this study explored effects of unilateral cortical spreading depression, the likely pathophysiological mechanism of migraine aura, on interhemispheric functional connectivity in freely behaving rats.
Methods
Temporal evolution of the functional connectivity was evaluated using mutual information and phase synchronization measures applied to local field potentials recordings obtained in homotopic points of the motor and visual cortices of the two hemispheres in freely behaving rats after induction of a single unilateral cortical spreading depression in the somatosensory S1 cortex and sham cortical stimulation.
Results
Cortical spreading depression was followed by a dramatic broadband loss of interhemispheric functional connectivity in the visual and motor regions of the cortex. The hemispheric disconnection started after the end of the depolarization phase of cortical spreading depression, progressed gradually, and terminated by 5 min after initiation of cortical spreading depression. The network impairment had region- and frequency-specific characteristics and was more pronounced in the visual cortex than in the motor cortex. The period of impaired neural synchrony coincided with post-cortical spreading depression electrographic aberrant activation of the ipsilateral cortex and abnormal behavior.
Conclusion
The study provides the first evidence that unilateral cortical spreading depression induces a reversible loss of functional hemispheric connectivity in the cortex of awake animals. Given a critical role of long-distance cortical synchronization in sensory processing and sensorimotor integration, the post-cortical spreading depression breakdown of functional connectivity may contribute to neuropathological mechanisms of aura generation.
Keywords
Introduction
Migraine aura represents unilateral, fully reversible neurological symptoms, including sensory, motor or speech disturbances that show a slow, progressive onset and may be followed by headache (1,2). The symptoms are thought to reflect transient cerebral dysfunction caused by cortical spreading depression (CSD), a wave of intense cellular depolarization slowly propagating over the cortex at the rate of 3–5 mm/min (1,3). Electrophysiologically, CSD manifests as a large negative direct current (DC) shift and transient depression of cortical activity (3,4). The DC shift marks a period of strong neuroglial depolarization and breakdown of local ion homeostasis that lasts 1–2 minutes in healthy neuronal tissue. The depolarization phase of CSD is preceded by a brief burst of neuronal firing and is followed by depression of spontaneous electrographic activity of the cortex.
Propagation of CSD over the visual cortex is thought to underlie visual symptoms of aura, the most common in migraine patients (5). It is thought that a brief burst of neuronal firing before CSD onset produces positive visual symptoms (flashing lights, scintillations) and subsequent depression of neuronal activity underlies negative symptoms (scotoma, blurring of vision). Despite the widely accepted link between CSD and migraine aura, there are several gaps in translating CSD features to clinical aura symptoms and some patterns of aura described in migraine patients cannot be explained based on the known properties of CSD (6). This may be in part due to the fact that most information about CSD and its electrophysiological characteristics has been obtained in anesthetized animals and acute slice preparations. The conditions are associated with altered patterns of cerebral activity, widespread breakdown in network interactions and inter-areal connectivity that may significantly change characteristics and consequences of CSD. Much less is known about properties of CSD in non-anesthetized awake animals, better reproducing the brain state of migraine patients.
The unilateral pattern typical of aura symptoms suggests alterations in the interhemispheric balance of sensory processing during migraine aura. We hypothesized that a unilateral CSD, the electrographic correlate of migraine aura, alters interhemispheric interactions in the visual cortex. To test the hypothesis, we explored the effects of CSD on the functional connectivity between bilateral homologous cortical regions in awake, freely moving rats. Functional connectivity describes statistical association between activities of distant brain regions and is thought to reflect the strength of inter-regional communication. We recorded local field potential (LFP) in homotopic sites of the visual and motor cortices of the two hemispheres after initiation of a single unilateral CSD and tracked the evolution of functional connectivity in the immediate post-CSD period using two measures of functional connectivity – mutual information (7) and phase locking value or mean phase coherence (8). We found that unilateral CSD is followed by dramatic global loss of functional interactions between hemispheres for several minutes, simultaneously with abnormal electrographic activation of the ipsilateral cortex and development of atypical behavior.
Methods
Animals
Male Wistar rats (3–4 months old) weighing 285–400 g (Stolbovaya Animal Breeding Center, Moscow, Russia) were used. Rats were housed in individual cages under controlled environmental conditions (a 12 h light-dark cycle, lights on at 7:00 AM, 20–23°C) with free access to food and water. Experiments were carried out in accordance with the EU Directive 2010/63/EU for animal experiments and the experimental protocol was approved by the Ethical Committee on Animal Experimentation of IHNA. The experimental protocol used in this study included measures to minimize animal suffering and the number of animals used. All animals were handled, treated and monitored in the same way.
Surgery
Guide cannula for sham microinjections and electrodes for CSD recording were stereotactically implanted under chloral hydrate (400 mg/kg ip, AppliChem, Germany) anesthesia 2 weeks before the onset of experiments. A stainless-steel guide cannula (OD, 0.6 mm; ID, 0.4 mm) was implanted bilaterally in the primary S1 somatosensory cortex (AP −2.8; ML 4.8; V 1.5). The same length stylet was inserted into the guide cannula to prevent its obstruction. For recording full-band LFP signals including DC potential shifts, glass electrodes with an inner carbon fiber (having a tip diameter of 50–100 µm; (9)) were bilaterally implanted in the primary M1 motor (AP +1.2; ML 2.3, V 1.5) and primary V1 visual (AP: −5.8, ML 3.5; V 1.5) cortex. The reference electrode was placed over the cerebellum and all electrodes were soldered to a pin connector. Cannula and electrodes were fixed on the skull with acrylic cement. After the surgery, rats were placed in individual cages and let to recover for 14 days. A week before the experiment, rats were handled for 3 consecutive days to minimize the stress caused by the experimental procedure and to ensure that dummy cannulas could be easily removed.
Induction of CSD
To avoid painful experience from damaging the dura mater, CSD was induced by local mechanical stimulation of cortical parenchyma via the preliminary implanted cannula. To induce the spreading depression, a rat was gently handled and a stainless-steel needle was inserted into the guide cannula, protruding 1 mm from its tip. In sham tests, a short needle (a dummy cannula) was inserted into the guide cannula without damaging the cortical tissue.
Experimental design
Experiments were performed in awake, freely moving rats with simultaneous video monitoring of behavior and recording of full-band LFP signals. Each rat was individually placed in a shielded experimental chamber (60 × 40 × 40 cm) and the implanted connector was attached to the recording cable. Electrical activity of the cortex was recorded with a four-channel, high-input impedance (1 GΩ) DC amplifier and A/D converter (E14-440, L-Card, Russia). The data were stored on a PC (1 kHz sampling rate) and analyzed off-line.
After a 5-min period of habituation in the chamber and a 10-min period of baseline LFP recording, a sham stimulation of the S1 cortex or local mechanical stimulation was performed. Cortical LFP signals were registered for 10 min post-stimulation. Spatiotemporal characteristics of CSD and its behavioral manifestations (based on analysis of video recordings) were estimated.
Out of eight rats implanted with a cannula and recording electrodes, one animal was excluded due to connector loss after surgery that prevented electrographic data from being obtained. A total of seven rats were therefore included in the analysis. In each rat, two unilateral CSD were induced with an interval of a week between repeated tests. Electrographic and behavioral manifestations of the first and the second CSDs did not differ significantly and the data were pooled (n = 14). Sham stimulations were used as negative controls (n = 14).
Data analysis
LFP recordings were filtered with a bandpass filter 0–100 Hz (DC) or 1–100 Hz (AC). For analysis of temporal dynamics of spectral power and functional connectivity, LFP recordings without significant artifacts and line noise during the 500-sec period after sham (n = 7) and CSD-inducing (n = 7) stimulations were used. The investigator (TM) analyzing the data was blinded to the group treatment. Active behavior (exploration, walking, turning and scanning movements of the head) and grooming were scored for each 20-sec interval after CSD induction.
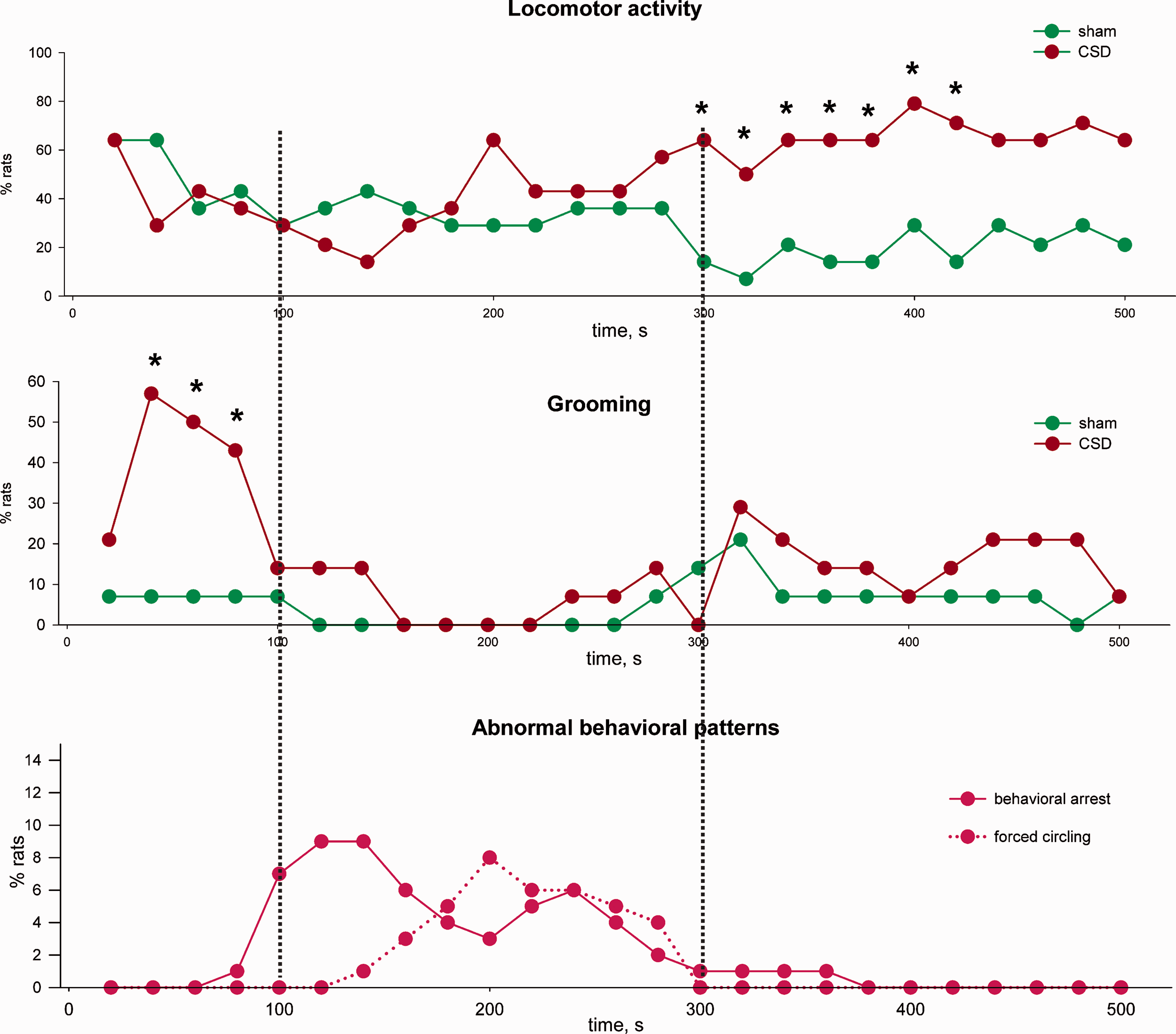
Power spectra were computed using a Fast Fourier Transform (FFT) routine for five frequency bands: delta (1–4 Hz), theta (4–10 Hz), beta (10–25 Hz), low gamma (25–50 Hz) and high gamma (50–100 Hz). Epochs containing artifacts were excluded from analysis. 500-sec sections of LFP recordings obtained immediately after sham and CSD-inducing stimulations were selected. Further, these sections were divided into 20-sec intervals and the power spectrum and measures of coupling were evaluated for each section. To analyze the coupling dynamics between the left and right hemispheres, the mutual information function (7) and the phase locking value (8) were used.
Mutual information is a statistical function of two random variables that describes the amount of information contained in one random variable relative to another. Mutual information is determined through the entropy and conditional entropy of two random variables as
The phase locking value is a numerical indicator quantitatively characterizing the phase synchronization of two oscillators and reflecting the stability of the difference of these phases. With the exact fitness of the phases, the coefficient is 1; in the absence of synchronization it is 0. The phase locking value was defined as
Statistical analysis
Data were expressed as mean ± SEM. Differences in mutual information and phase synchronization values between CSD and sham groups as well as differences in spectral power values between cortical regions ipsilateral and contralateral to CSD were compared using analysis of variance (ANOVA) for repeated measures followed by post-hoc Newman-Keuls test and one-way ANOVA. Regional difference in temporal characteristics of CSD and hemispheric decoupling were evaluated using Wilcoxon signed rank test. To estimate effects of CSD on the incidence of various behavioral patterns, Fisher’s exact test was used. Level of significance was set to p < 0.05 for all statistical tests.
Results
Electrographic changes in cortical activity of awake rats induced by unilateral CSD
A local mechanical stimulation of the S1 somatosensory cortex induced a single unilateral CSD propagated to the ipsilateral V1 visual and M1 motor cortices in all tests but one (13/14). Figure 1 shows representative recordings of DC shifts and respective changes in spontaneous cortical activity (AC recordings) in the ipsilateral M1 and V1 regions of the cortex in an awake rat. Negative DC-potential shifts, which indicated the appearance of CSD at the recording sites, were registered during the first 100 sec, initially invading the visual V1 cortex and later the motor M1 cortex (Figure 1(a), Table 1). Cortical AC-LFP amplitude was transiently depressed after CSD but the depression was rapidly replaced by enhanced cortical activity (Figure 1(b)). Extended AC recordings showed that the late activity consisted of isolated negative spikes, recurrent sharp waves and slow sharp waves superimposed on depressed cortical activity and masking the electrographic depression (Figure 1(d),(e)). No spike-wave discharges typical of absence epilepsy were recorded after CSD. The epileptiform patterns began to occur at 100–120 sec after CSD initiation, gradually intensified and faded out by about 260–300 sec (Figure 1(d),(e), Table 1). The aberrant electrographic activation was detected after all recorded CSDs (n = 13), although its intensity varied. No such abnormal activity was observed in the unaffected contralateral cortex (Figure 2). Time-frequency plots of the LFP recordings showed mild transient depression of high-frequency oscillations and late low-frequency activation of the cortex ipsilateral to CSD (Figure 2(c)).
Electrographic correlates of a single CSD in awake freely behaving rats. (a) Representative recordings of CSD-associated DC potential shifts (Fullband DC/AC-LFP, 0–100 Hz frequency band) obtained in the primary motor (M1) and visual (V1) regions of the cortex before and after induction of a single unilateral CSD by mechanical stimulation of the S1 somatosensory cortex (marked by arrow); (b) AC recordings obtained after filtering (bandpass 1–100 Hz) the recordings shown in (a); ((c)–(e)) expanded fragments from the AC recordings shown in (b). Calibration bars: 2 mV, 50 sec (a), 0.2 mV, 50 sec (b), 0.2 mV, 1 sec ((c)–(e)).
Temporal characteristics of CSD and CSD-associated interhemispheric functional disconnection in awake rats.

*Onset/termination after initiation of CSD in the somatosensory cortex.
#Significant difference between the visual and motor cortices (Wilcoxon test).

Lateralized pattern of CSD-induced changes in cortical activity in awake rats. (a) Representative fullband LFP recordings (bandpass 0–100 Hz) obtained in homotopic sites of the ipsilateral (V1 L) and contralateral (V1 R) visual cortices after mechanical stimulation of the left S1 somatosensory cortex; (b) filtered AC recordings (bandpass 1–100 Hz) of the fragment shown in (a); (c) spectrograms for the fragment shown in (b). Calibration bars are the same as in Figure 1.
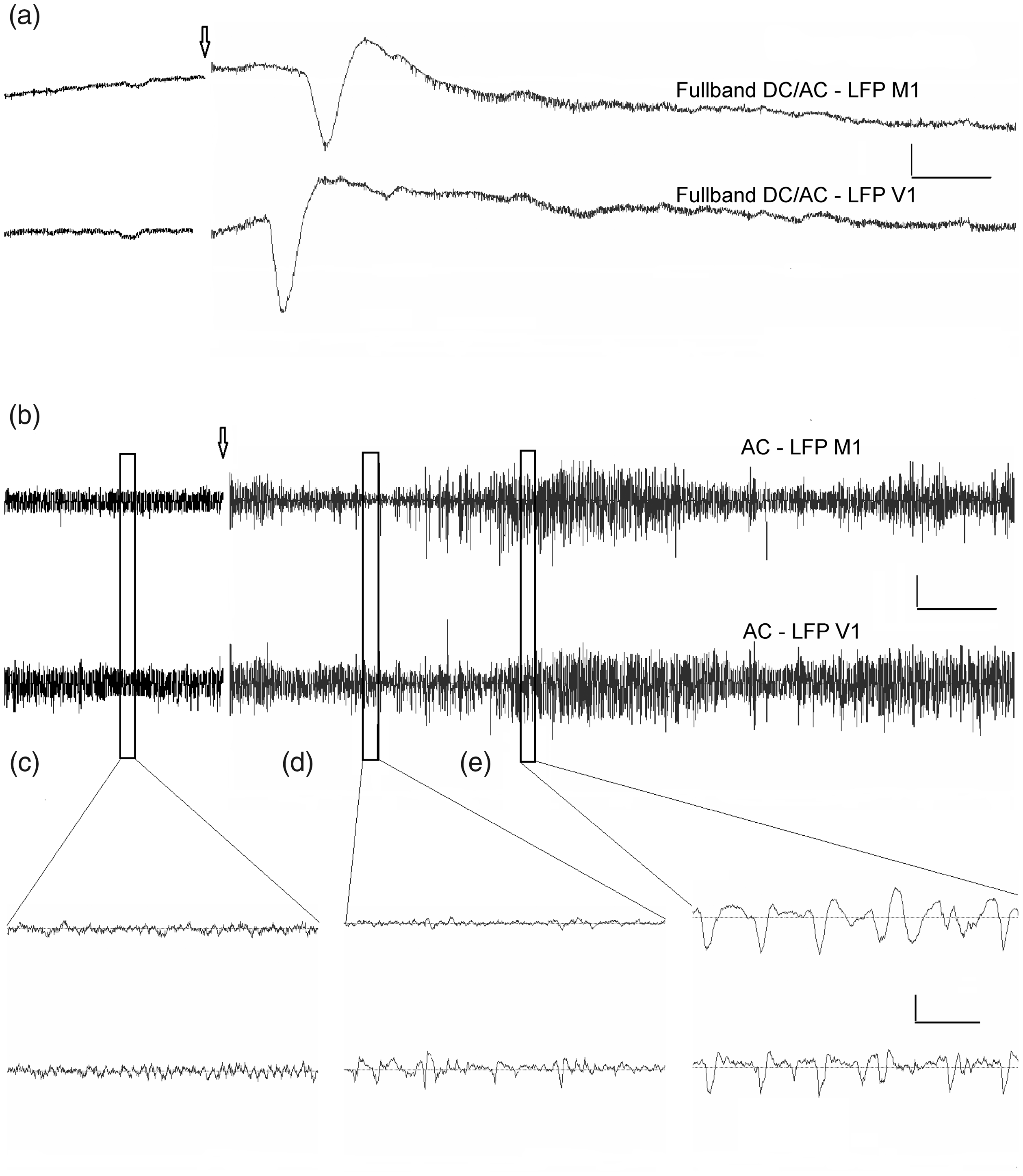
Spectral analysis of LFP signals from the homotopic cortical sites of the two hemispheres across rats (n = 7) showed that a single unilateral CSD elicited transient depression of high frequency oscillations in the affected cortex (Figure 3). A significant effect of CSD on LFP power was found only in the low gamma range (F = 5.68, p < 0.001, repeated measures ANOVA). The beta band showed a very slight tendency to depression after CSD (F = 1.422, p = 0.089).
Average LFP power in the delta (1–4 Hz), theta (4–10 Hz), beta (10–25 Hz), low gamma (25–50 Hz) and high gamma (50–100 Hz) frequency bands at the homotopic sites of the visual and motor cortices of the ipsilateral (red) and contralateral (green) hemispheres after a single unilateral CSD (n = 7). The power was averaged across animals (mean ± SEM).
Changes in interhemispheric functional connectivity induced by unilateral CSD
Analysis of interhemispheric functional connectivity using a mutual information measure did not reveal significant alterations in hemispheric interactions during DC shifts, but showed a profound 2.5–3-fold drop in the coupling following CSD (Figure 4). A significant reduction in the connectivity was observed with a 1–1.5 min delay after the end of respective DC shifts (Table 1). ANOVA for repeated measures showed a main effect of CSD on the functional connectivity measure (p < 0.001, Table 2).

Post-CSD drop of interhemispheric functional connectivity estimated by mutual information measure. CSD-associated DC shifts and average mutual information values (mean ± SEM) for the visual and motor cortices after sham (green, n = 7) and CSD-induced (red, n = 7) stimulation. Vertical dashed lines indicate the DC shift onset and termination. The asterisks mark the significant differences between CSD and sham groups.
Results of repeated measures ANOVA for interhemispheric functional connectivity.

Bold values are significant at p < 0.05.
Phase synchronization measure confirmed the post-CSD reduction in interhemispheric functional connectivity. The temporal evolution of the CSD-induced changes in the cortical synchrony for specific frequency bands is shown in Figure 5. A single unilateral CSD was followed by a transient 2–3-fold loss of interhemispheric synchrony in a wide frequency band (1–50 Hz). The post-CSD decoupling developed gradually from 80–120 sec, peaked around 120–180 sec and terminated by 250–280 sec after CSD initiation (Figure 5, Table 1). The onset of the hemispheric functional disconnection in the delta and beta bands corresponded to the succession of CSD propagation over the cortex; that is, the disconnection started initially in the visual cortex and later in the motor cortex (Table 1). On the other hand, the termination of the hemispheric decoupling occurred near simultaneously in both cortical regions (Table 1, Figure 5).

Dynamics of interhemispheric functional connectivity estimated by phase synchronization measure (mean ± SEM) for delta (1–4 Hz), theta (4–10 Hz) beta (10–25 Hz), low gamma (25–50 Hz) and high gamma (50–100 Hz) frequency bands for the visual and motor regions of the cortex after sham (green, n = 7) and CSD-induced (red, n = 7) stimulation. The asterisks mark the significant differences between CSD and sham groups.
The most pronounced impairment of interhemispheric synchrony was detected for the beta frequency range in the visual cortex. The beta-band synchrony was significantly reduced not only during and after CSD but also before the arrival of CSD at the visual cortex, though the early reduction (before and during CSD) was subtle compared to the subsequent post-CSD hypo-connectivity (Figure 5).
ANOVA for repeated measures showed a significant effect of CSD on the interhemispheric connectivity for all frequency bands excluding high gamma (Table 2). In addition, the dynamics of cortical connectivity in the beta and gamma bands showed regional specificity. The effect of CSD on hemispheric connectivity in the beta-low gamma ranges was stronger in the visual cortex compared to the motor one (p = 0.041 and p = 0.006, respectively, Newman-Keuls test). CSD affected low gamma connectivity only in the visual cortex. On the other hand, the effect of CSD on high gamma synchrony was found only the motor cortex (p = 0.045, post-hoc Newman-Keuls test) where a slight increase in connectivity was observed at 80 sec.
Behavioral changes induced by unilateral CSD
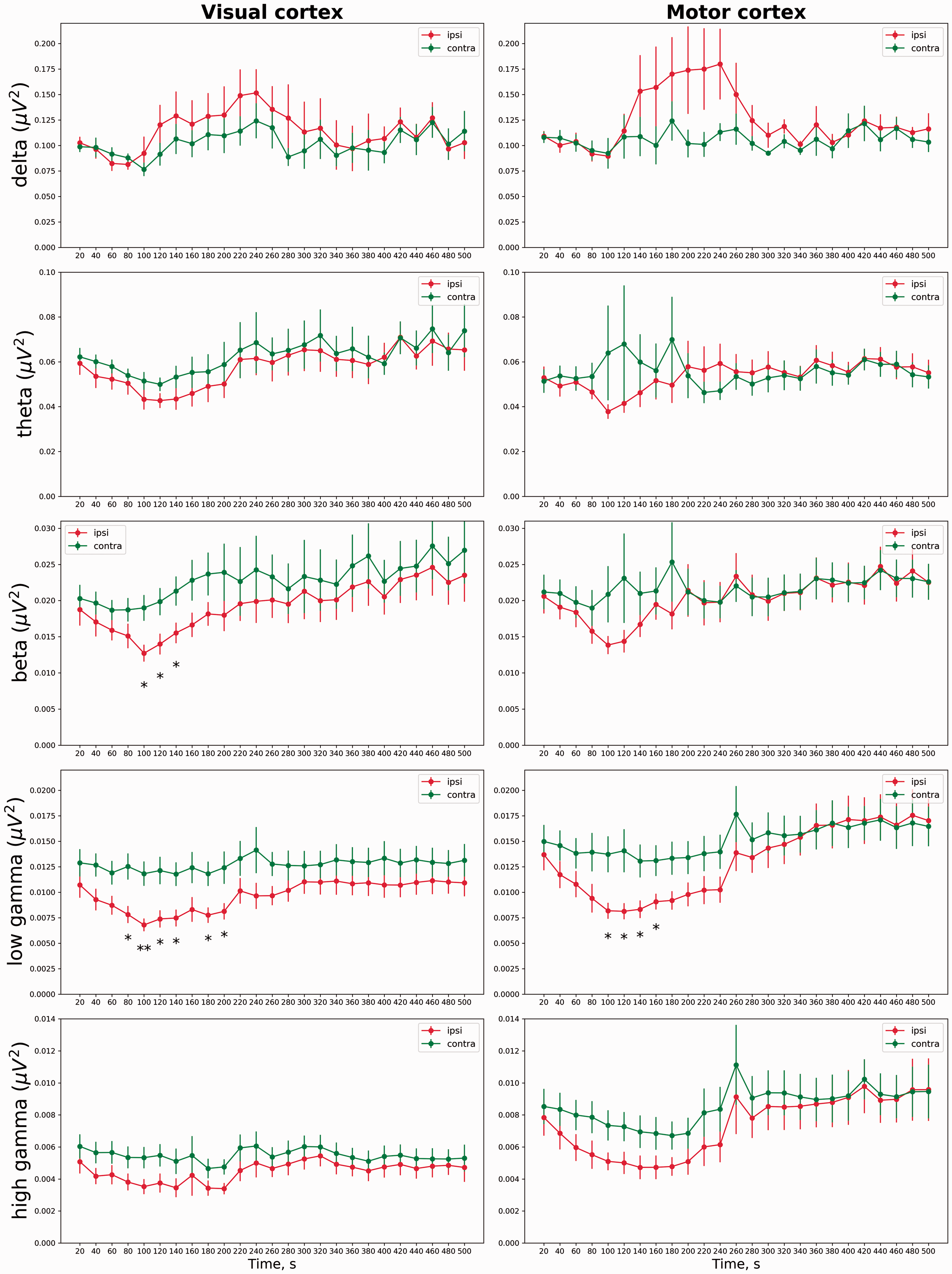
Comparison of spontaneous behavior after sham stimulation (n = 14) and a single unilateral CSD (n = 13) has shown significant enhancement of grooming during CSD (40–80 sec) and remote post-CSD (300–420 sec) behavioral activation (Figure 6). In addition to these changes in general behavior, atypical behavioral patterns were observed following CSD (Figure 6): Behavioral arrest (freezing-like episodes) with or without tremor (100–280 sec) and forced circling to the side contralateral to CSD (140–280 sec) heralded CSD penetration into the striatum, as we reported previously (10). The abnormal behavioral patterns were time-locked with the observed post-CSD hemispheric disconnection (Figure 6).
Behavioral changes induced by a single unilateral CSD in freely moving rats. Locomotor activity and grooming behavior induced by a single unilateral CSD (red, n = 13) and sham treatment (green, n = 14) and abnormal behavioral patterns (episodes of behavioral arrest and forced circling) in the immediate post-CSD period. Vertical dashed lines indicate the periods of CSD propagation along the neocortex (0–100 sec) and post-CSD interhemispheric decoupling (100–300 sec). Behavioral activity (percent of rats exhibiting a specific behavioral pattern) was plotted in 20-sec time bins (as quantified by video analysis). The asterisks mark the significant differences between CSD and sham groups.
Discussion
The present study shows that in awake rats a single unilateral CSD is followed by transient interhemispheric functional decoupling, alongside dynamic changes in spontaneous cortical activity and animal behavior.
Electrographic effects of unilateral CSD in awake rats
We found that in awake rats unilateral CSD is followed by complex changes in cortical activity, including both depression and abnormal activation of the ipsilateral cortex. A single CSD transiently depressed high-frequency (gamma) cortical oscillations in the affected hemisphere of un-anesthetized, freely moving rats. A similar effect of CSD has been reported in the model of focal stroke (11). The mild suppressive influence of CSD on cortical activity in our study may be due to a lack of anesthesia and a short duration of CSD, which has been shown to correlate to depressive effects of CSD on electrocorticographic activity in patients (12).
The late excitatory period after CSD has been previously described both in in vitro and in vivo experiments (3,13,14). Even the first studies of the CSD phenomenon reported development of specific cortical activity resembling seizure patterns at the end of DC shift where, normally, depression of spontaneous activity is observed (3,15). The seizure-like patterns (synchronized polispike wave activity and isolated spikes) outlasted the DC shift by 7–10 min and propagated slowly with CSD over the cortex. The spreading convulsions have been recorded in cortical slices and anesthetized animals (3,13–15), mostly after repeated CSDs or cortical disinhibition, as well as in patients with subarachnoid haemorrhage (16). The post-CSD activation is supposed to be mediated by suppression of GABAergic inhibition (17) and changes in intrinsic neuronal properties following CSD (13,14). The post-CSD seizure-like patterns are unlikely to represent true epileptic activity but seem to reflect an enhanced propensity of the cortex to local network synchronization (18). Our study shows that in awake rats, CSD induces late cortical activation without preconditioning procedures such as pharmacological disinhibition or repeated induction of CSD.
The cortical hyperactivation following CSD may have detrimental effects and carry the risk of producing abnormal excitation (seizures) (10,18,19). Long-range excitatory trans-hemispheric connections may easily transfer the seizure-like activity to the unaffected contralateral cortex, but we found no signs of its abnormal activation. We suggest that the post-CSD interhemispheric decoupling described in the current study and observed during the same post-CSD period may represent an intrinsic protective mechanism precluding generalization of the abnormal activity to the contralateral cortex.
Effects of unilateral CSD on functional connectivity between hemispheres
Interhemispheric functional connectivity during unilateral CSD
The near-complete depolarization of neurons during CSD transiently inactivates mechanisms of action potential generation and synaptic transmission (4,16) and thus interrupts inputs/outputs of the depolarized cortex, including transcallosal pathways (20). It is reasonable to suggest that the most prominent disruption of bihemispheric interactions occurs during the depolarization phase of CSD (DC shift), but we observed slight alterations in hemispheric functional connectivity during the period. Only the visual cortex reduced beta synchrony during CSD, though the reduction was significantly less dramatic than subsequent post-CSD decoupling. Therefore, neuronal silence during the CSD-induced depolarization block of cortical activity appears to be a minor contributor to the changes in interhemispheric communication induced by CSD.
Remarkably, in the visual cortex, the functional decoupling in the beta band was detected before CSD arrival at the region, which matches clinical data about the beta frequency band having the highest sensitivity to CSD occurrence (21). In patients with acute traumatic brain injury, suppression of beta oscillations has been recorded not only during and after CSD but long before (2 h prior to) its appearance at the recording sites, which was interpreted as a result of trauma-induced cerebral ischemia (21). The small localized damage to the somatosensory cortex used to trigger CSD in our experiments was unlikely to elicit such global ischemia, although we cannot exclude some influence of the lesion on activity of the visual cortex, which is located slightly closer to the lesion site than the motor cortex. In the model of focal stroke, it has been shown that a local lesion can induce widespread impairments of intracortical interactions (11).
In the motor cortex, a brief increase in high gamma synchrony (50–100 Hz) was found at the onset of the depolarization phase of CSD. The slight short-lasting hyperconnectivity likely accompanies high-frequency (60–115 Hz) LFP oscillations that develop several seconds ahead of the negative DC shift and subside with its onset (22). The activation of local network synchronizing mechanisms in the front of spreading depression has been supposed to underlie the pre-SD oscillations. We find that CSD also activates region-specific mechanisms of long-range interhemispheric synchronization.
Post-CSD loss of interhemispheric functional connectivity
The most dramatic changes in interhemispheric functional connectivity followed DC shifts with a delay of tens of seconds. The functional communication between the two hemispheres reduced gradually, achieving its minimum at about 2–3 min and recovering by 4–5 min after CSD initiation. An irreversible reduction in interhemispheric connectivity after unilateral CSD has been previously reported for the somatosensory cortex in anesthetized mice (23). In the study, using optical intrinsic signal imaging, reduced connectivity was observed during the whole 20–70 min period of registration. Our study shows that in awake rats, a single unilateral CSD elicits reversible functional disconnection of the hemispheres that resolves within 5 min.
The post-CSD functional disruption of the hemispheric communication involved both the motor and visual regions of the cortex, although the visual cortex showed more pronounced impairments particularly for the beta frequency band that may reflect higher vulnerability of the visual cortex to CSD effects. Both local and remote mechanisms of the post-CSD interhemispheric imbalance may be involved. The relationship between the onsets of DC shift and delta/beta decoupling indicates participation of local mechanisms activated by CSD, while near-simultaneous termination of the decoupling in the visual and motor cortices suggests possible contribution of shared modulatory effects of other brain regions. Our findings suggest that CSD-induced hemispheric functional disconnection is an active process and not merely a result of cortical deactivation. Mild depression of high-frequency cortical activity elicited by unilateral CSD in awake rats is unlikely to produce such extensive wideband loss of hemispheric connectivity as was found in the current study.
CSD triggers multiple events in neuronal tissue, including disruption of ionic homeostasis, changes in local cerebral blood flow and metabolism, intracellular calcium loading, release of neuroactive substances, ultrastructure changes (dendritic beading, spine loss), and abnormal activity of glutamatergic pathways mediating long-distance synchronization (4,16,20). However, most of the changes peak during the depolarization phase of CSD and fade out rapidly after its termination. The dynamics profoundly differ from the time course of the post-CSD hemispheric disconnection, which starts only after the end of the depolarization phase of CSD (DC shift). A plausible mediator of the post-CSD functional decoupling is adenosine – a homeostatic regulator and ATP metabolite that accumulates in the extracellular space from 50 sec to 3–4 min after the DC peak (24). Cytotoxic edema, a reliable consequence of CSD (25), may also modulate interhemispheric interactions. The corpus callosum is highly vulnerable to cytotoxic edema. Callosal edema is supposed to underlie reversible lesions of the splenium of the corpus callosum in patients with various neurological conditions including sensory abnormalities, aphasia, hemiparesis, ataxia and seizures (26). Reversible callosal lesions and signs of transient cytotoxic edema have been described during aura in migraine patients (27,28). The lesions usually involve the posterior part of the corpus callosum, fibers of which connect parieto-occipital lobes of the two hemispheres, which matches our data about higher vulnerability of the occipital cortex to the post-CSD functional decoupling.
CSD-induced behavioral/neurological deficits
Given that long-range synchronization across hemispheres is essential for formation of large-scale network activity during sensory perception, sensorimotor integration and cognitive function, CSD-associated interhemispheric hypoconnectivity found in the current study might be relevant to behavioral and sensorimotor deficits induced by CSD. Our study showed the highly dynamic character of CSD-induced behavioral changes – an early brief period of enhanced grooming during CSD (40–80 sec), late post-CSD behavioral activation (300–420 sec), episodes of atypical behavior such as tremor, freezing and forced circling (100–300 sec). Similar behavioral changes, including grooming during CSD, episodes of freezing, circling and locomotor activation after CSD, have been described in other studies in awake rodents (10,29,30). The only change in spontaneous behavior, observed during CSD traveling over the neocortex in our study, was increased grooming, which is known to be associated with facial pain in rodents (31) and is produced by CGRP administration (32).
The post-CSD drop of hemispheric communication coincides with the occurrence of freezing episodes (10,29,30) and development of mild neurological deficits (contralateral sensory neglect and motor impairment) described after CSD in experimental animals, including models of familial hemiplegic migraine (4,30,33). Impaired perception of sensory stimuli received by the contralateral side of the body is thought to underlie the neurological deficits induced by CSD (4). Close relationship between the timing of the changes and post-CSD decoupling allow speculation that the observed network alterations may contribute to the behavioral impairments and sensory-motor deficits following unilateral CSD.
Relevance to migraine aura
Altered patterns of functional and effective connectivity have been reported in migraine patients during the interictal period (34,35), but ictal changes in cortical connectivity are less understood. Our study shows that a single unilateral CSD, the electrophysiological correlate of migraine aura, transiently disrupts normal interhemispheric interactions (particularly in the beta band), rendering them less effective for several minutes. The reversible post-CSD trans-hemispheric functional disconnection might underlie the unilateral sensorimotor deficit during migraine aura. The higher vulnerability of the visual cortex to the CSD-related network impairments compared to the motor cortex may contribute to the prevalence of visual aura symptoms in migraine patients.
We suggest that the findings might provide some clues for interpretation of CSD-related mechanisms of aura generation. Visual aura symptoms are traditionally attributed to the depolarization phase of CSD and/or CSD-induced hemodynamic changes. However, some features of CSD do not match clinical symptoms of aura; for example, the pre-CSD excitation of cortical neurons, which is thought to underlie positive aura symptoms, seems to be very brief (2–4 sec) to explain the symptoms (6). Our study in awake rats shows that CSD is followed by aberrant cortical activation and hemispheric functional disconnection that last several minutes. Overlapping of the CSD consequences during propagation of CSD over the large cortical areas might underlie the concomitant occurrence of different aura symptoms described in patients. The gyrencephalic cortex of humans promotes reverberating and spiral patterns of CSD propagation (36) that may further prolong CSD-induced changes in cortical activity.
Conclusion
The study provides the first evidence that unilateral CSD induces dramatic reversible loss of functional hemispheric connectivity in the cortex of awake animals. The experimental findings may enhance our understanding of neuropathological mechanisms of migraine with aura. The post-CSD short-term changes in cortical interactions should be taken into consideration when CSD features are translated to clinical symptoms of migraine aura.
Article highlights
A single unilateral cortical spreading depression (CSD), the electrographic correlate of migraine aura, induces a dramatic reversible loss of interhemispheric functional connectivity in the visual and motor cortices in awake, freely behaving rats. The hemispheric functional disconnection starts after the end of the CSD-associated DC shift, progresses gradually and terminates by 5 min after CSD initiation. The network impairment had region- and frequency-specific characteristics and was more pronounced in the visual cortex than in the motor cortex. The impaired synchrony of cortical networks paralleled aberrant electrographic activation of the ipsilateral cortex and occurrence of abnormal behavioral patterns. The post-CSD network changes may contribute to neuropathological mechanisms of aura generation.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Russian Foundation for Basic Research, grant 18-015-00418.