Abstract
Background
High-frequency episodic migraine (HFEM) has gained attention in the field of headache research and clinical practice. In this narrative review, we analyzed the available literature to assess the evidence that could help decide whether HFEM may represent a distinct clinical and/or biological entity within the migraine spectrum.
Methods
The output of the literature search included 61 papers that were allocated to one of the following topics: (i) socio-demographic features and burden; (ii) clinical and therapeutic aspects; (iii) pathophysiology; and (iv) classification.
Results
Multiple features differentiate subjects with HFEM from low-frequency episodic migraine and from chronic migraine: education, employment rates, quality of life, disability and psychiatric comorbidities load. Some evidence also suggests that HFEM bears a specific profile of activation of cortical and spinal pain-related pathways, possibly related to maladaptive plasticity.
Conclusions
Subjects with HFEM bear a distinctive clinical and socio-demographic profile within the episodic migraine group, with a higher disease burden and an increased risk of transitioning to chronic migraine
This is a visual representation of the abstract.
Introduction
Migraine was defined and classified in the first edition of the International Classification of Headache Disorders (ICHD) (1) as an idiopathic, recurring headache disorder manifesting in attacks lasting 4–72 hours with typical features such as unilateral location, pulsating quality, moderate or severe intensity, aggravation by routine physical activity, and association with nausea and/or vomiting, photo- and phono-phobia. In the first iteration of ICHD, chronic migraine (CM) was not considered. Indeed, a comment was made on the possibility of “coexisting migraine and tension-type headache”. For this reason, subjects with migraine presenting headache on more than 15 headache days received the dual diagnosis of migraine and tension-type headache (1). In the second edition of ICHD (2004), the concept of CM was included as a migraine complication (2) and defined as a headache present 15 or more days per month, with eight or more days having migraine features. In the last ICHD edition (ICHD-III; 2018), CM was classified as a separate clinical entity (3). Noteworthy, according to ICHD-III, the presence of CM excludes a concomitant diagnosis of tension-type headache (3). The evolution of the diagnostic criteria for CM led to several advantages: (i) improvement of the diagnostic sophistication; (ii) stimulation of research on the pathophysiological mechanisms underlying migraine chronification; and (iii) increased clinical and public health attention on a highly disabling migraine state. On the other hand, quite surprisingly the term of “episodic” migraine (EM), as opposed to CM, has never been mentioned specifically in the ICHD editions. Goadsby and Evers (4) provided an interim definition of EM pending the expected official definition to be delivered within the next ICHD edition, which has been widely adopted in the clinical and research area since the 1980s (5). The lack of alignment in nosology and clinical practice is partly a result of the use in ICHD of attack classification as a primary tool for disorder definition. The evolution of ICHD toward the identification of CM has favored research on the definition of migraine as a disorder, which has contributed to extraordinary advances in many fields of migraine medicine and science.
Migraine has indeed a dynamic nature that manifests in the varying recurrence of attacks, with some people with EM progressing to chronification or, vice versa, reverting to a lower attack load, either spontaneously or following effective treatments. The frequency of monthly headache days (MHDs) is a strong driver of migraine related disability (6) and it is a risk factor for migraine progression to the chronic type as underscored several years ago in the Frequent Headache Epidemiology study (7). According to the progression model proposed by Buse et al. (8), a subset of individuals with EM may face a gradual increase in attacks frequency over time, influenced by specific risk factors such as medication overuse, presence of depression, caffeine abuse and obesity. Subjects transitioning from a low frequency of MHDs to a higher frequency may ultimately evolve into CM. This model also emphasizes the bidirectionality of migraine frequency pattern that can also revert from a chronic to an episodic form over time. The data are consistent with migraine as a cyclic and dynamic disorder, a concept that was further reinforced by Serrano et al. (9), showing that most of subjects with CM fall below the diagnostic threshold of 15 headache days per month at least once over the course of a year. This dynamic pattern is however limited to a subgroup of individuals, as suggested by a more recent study showing that subjects with EM can be subdivided into two phenotypes, cyclic and non-cyclic, according to the fluctuations of migraine frequency across a one-year observation period (10). People with a cyclic migraine phenotype are more likely to face an increase in headache frequency within one year if not treated with migraine preventive medications. Thus, there is a subgroup of EM subjects with a more unstable phenotype of disease, bearing a higher risk of progressing to CM. The intrinsic dynamic behavior of migraine is also confirmed by its natural course across people's life. In children and adolescents, headache prevalence increases gradually up to 7–15 years of age, with a subsequent tendency to reduce spontaneously at a 20-year follow-up (11). In adults, migraine prevalence peaks in the third decade of life, with a tendency to decrease in the later stages of life (12).
The dual importance of the frequency of MHDs in defining the burden of migraine and in influencing negatively its temporal pattern has gained traction over the years. An increasing number of papers have adopted the clinical entity “high-frequency episodic migraine” (HFEM) to identify the subgroup of subjects with a number of MHDs varying from 7–10 to 14. These papers have provided information on the epidemiological, clinical and pathophysiological aspects of HFEM (Table 1). We are aware that (i) simply classifying people with migraine into episodic and chronic does not fully reflect the impact of the disease; (ii) a more accurate framing of migraine subtypes may contribute to further advance knowledge and optimize management of the disease; and (iii) headache frequency alone may be an inadequate, if not simplistic, means of defining migraine subtypes.
Summary of findings differentiating high-frequency migraine from low-frequency migraine or chronic migraine reported in papers focusing on sociodemographic feature, disease burden, quality of life or risk of progression

BMI = body mass index; CM = chronic migraine; EM = episodic migraine; HFEM = high-frequency episodic migraine; ICHD = International Classification of Headache Disorders (different editions = I, II or III); LFEM = low-frequency episodic migraine; MHDs = monthly headache days; MFEM = medium-frequency episodic migraine; MMDs = monthly migraine days; NSAIDs = non-steroidal anti-inflammatory drugs; TM = transformed migraine.
In this narrative review, we illustrate and analyze the available literature on HFEM, with a specific focus on the differences emerged with low-frequency EM or CM, aiming to verify whether there is sufficient evidence to propose a definition of HFEM for field testing and validation.
Methods
In this narrative review, we performed a structured literature search to include, organize and assess relevant papers discussing the topic of HFEM. We searched the PubMed database using combinations of the following keywords: “migraine”, “high frequency”, “low frequency” and “medium frequency”. The initial selection of articles was performed independently by three of the investigators (FC, MC and FB) using the “Rayyan – Intelligent Systematic Review” platform (https://www.rayyan.ai), an assisted tool that supports and facilitates literature review. In our narrative review, we used the tool for organizing the initial large volumes of articles and allow the simultaneous revision by multiple raters. Each rater adopted custom tags to describe the area of interest of each article.
We also analyzed the references of the selected papers for additional publications. The search yielded a total of 485 articles, with 20 duplicates being excluded.
Of the remaining papers, we rejected 371 after reading the abstract because they did not focus on HFEM, were written in languages other than English, or were abstract or editorials, whereas an additional 33 were excluded after full-text review because they did not provide specific information on HFEM. Ultimately, the narrative review included 61 papers that addressed different aspects of HFEM (Figure 1). Some of the papers also offered cues for the classification. After the assessment of the 61 articles, based on the topics addressed, we structured the review into four sections, namely “Socio-demographic features and disease burden”, “Clinical and therapeutic data”, “Pathophysiology” and “Classification aspects”, with the addition of a viewpoint section to provide a critical appraisal of available data.

PRISMA-like flow diagram.
Results
Socio-demographic features, disease burden, quality of life and risk of progression
The following sections provide a critical appraisal on the available data regarding socio-demographic aspects, disease burden, quality of life and risk of chronification of people with HFEM. More specifically, we summarize data from 14 studies (Table 1), involving 38,304 subjects with EM, 4512 with CM and 2507 with probable migraine or other diagnoses.
Socio-demographic features
Two main studies support the idea that HFEM is characterized by distinct demographic and socio-economic features. In 2011, Katsarava et al. (16) conducted a study based on the data from the German Headache Consortium survey and compared the epidemiological characteristics of people with CM, defined as having a minimum of 15 headache days per month, according to the Transformed Migraine criteria by Silberstein and Lipton (14), with those of individuals with low frequency (0–8 headache days per month) and those with a ‘critical’ frequency (9–14 headache days per month) (16). The results showed that the subjects in the critical frequency group were notably younger compared to people with CM (HFEM: 39.8 ± 13.4, CM: 46.2 ± 13.5). Interestingly, the authors showed a pattern of step-increasing gradient from low frequency to CM for low education (low frequency 58.8%, 1385 out of 2356; critical frequency 64.5%, 147 out of 228; CM 78.0%, 142 out of 185) and current smoking (low frequency 31.9%, 752 out of 2356; critical frequency 36.4%, 83 out of 228; CM 45.9%, 85 out of 185) (16). Of note, the differences observed were only significant when comparing the critical frequency group with the CM group, whereas they did not separate the low frequency and the critical frequency groups.
In 2020, Buse et al. (24) conducted an observational study on 11,603 subjects with migraine who participated in the 2005 American Migraine Prevalence and Prevention Survey. In this study, individuals were categorized into low-frequency episodic migraine (LFEM, less than four headache days per month), moderate frequency episodic migraine (MFEM, 4–7 headache days per month), HFEM (8–14 headache days per month) and CM. Persons with HFEM showed specific socio-demographic and clinical differences compared to subjects with MFEM. HFEM subjects had lower odds of having a degree (odds ratio (OR) = 0.76), of being employed (OR = 0.77) and, consequently, a lower chance to earn an annual income higher than $US 60,000 (OR = 0.81).
Disease burden and quality of life
In 2021, Ishii et al. (26) conducted a multicenter observational study on 836 migraine individuals with the primary aim of quantifying disability according to migraine frequency. They identified four groups of participants: group 1 (0–7 headache days per month, n = 286), group 2 (8–14 headache days per month, n = 180), group 3 (15–23 headache days per month, n = 153) and group 4 (≥24 headache days per month, n = 217). Using the Work Productivity and Activity Impairment (WPAI) and Pain Interference (PROMIS-PI) tools, there was a progression of disability moving from group 1 to 4. Of note, subjects belonging to groups 2 and 3 (i.e. subjects with HFEM or subjects with CM with less than 24 MHDs) showed a similar profile in terms of absenteeism, presenteeism, work activity impairment and work productivity reduction (26). These two groups also had similar pain interference on functioning and quality of life. By contrast, Group 1 and Group 2 Migraine Disability Assessment (MIDAS) scores, General Anxiety Disorder Scale 7-item (GAD-7), Patient Health Questionnaire-4 (PHQ-4), absenteeism, presenteeism and total work productivity were not different. When comparing subjects with HFEM with the persons with CM with ≥24 MHDs, a totally different profile was reported in terms of global disability, work impairment and pain interference on functioning and quality of life. Based on these findings, it was concluded that both HFEM and CM entities deserve specific consideration compared to other subgroups of EM subjects for their high burden of disease and treatment needs, although they provide scant evidence for a distinction between groups 1 and 2, consistent with a continuum of migraine rather than groupings. Recently, Shapiro et al. (27) conducted a large survey (n = 4429) to assess the association between migraine frequency and employment disability. They showed that headache frequency was higher in respondents who self-reported short- or long-term work disability. This latter was reported roughly twice as often among subjects with CM, relative to the matched control group. More specifically, self-reported short- or long-term employment disability was reported by 4.4% of subjects with LFEM (≤4 headache days per month), 6.4% of subjects with MFEM (5–9 headache days per month), 7.9% of subjects with HFEM (10–14 headache days per month) and 12.3% of subjects with CM. These figures clearly point to an effect of the frequency of headache days on employment, although it could be argued that MFEM and HFEM have much in common, than, for example, HFEM and CM.
In a relatively large population of 362 migraine subjects, Guglielmetti et al. (22) performed a secondary analysis of disability and quality of life by means of validated tools and described differences between HFEM and LFEM groups and between HFEM and CM groups. Indeed, subjects with HFEM scored worse than LFEM, but better than CM, in disability and quality of life. More specifically, the MIDAS score was 17.7 for LFEM, 47.3 for HFEM and 101.3 for CM, whereas the 12-item World Health Organization Disability Assessment (WHODAS-12) score was 21.2 for LFEM, 31.5 for HFEM and 41.2 for CM. Quality of life, detected with the Migraine Specific QoL (MSQ) scale, was better in HFEM compared to CM, but worse than LFEM. It is appropriate to add that the analysis was barely described, and thus the claimed outcomes need to be viewed with caution.
In the above-mentioned study by Buse et al. (24), HFEM subjects had a 12-fold risk of severe disability compared to the group of subjects with LFEM and MFEM taken together, whereas the difference was much less pronounced between MFEM and HFEM. In their cohort, HFEM were less likely to have severe disability when compared to CM .
Chalmer et al. (25) compared socio-economic impact on two groups of subjects with migraine from the Danish Headache Center. One group consisted of subjects diagnosed with CM according to the ICHD-III criteria, whereas the other included subjects with HFEM. There were no notable differences in the demographic profile between the two groups. The number of lifelong or annual attacks, comorbid diseases and self-reported effectiveness of triptans did not display any significant variances. However, subjects with HFEM purchased a higher quantity of triptans compared to subjects with CM. CM participants were more likely to receive early pension, but there were no significant differences between CM and HFEM groups in terms of sickness benefit, cash assistance, or rehabilitation benefit.
In the survey by Shapiro et al. (27), migraine-related disability, measured by MIDAS scores, consistently increased with increasing headache frequency. LFEM had the lowest MIDAS scores (11.4 ± 17.7), whereas CM had the highest (56.5 ± 58.5). Interestingly, a discrepancy was noted between self-reported short- or long-term work disability and MIDAS-assessed severe disability. This gap was already evident in LFEM, with 14.5% classified as severely disabled by MIDAS but only 4.4% self-reported work disability, but became more evident with increasing headache frequency, showing larger disparities between MIDAS-assessed severe disability and self-reported short- or long-term work disability (MFEM 36.9% vs. 6.4%, HFEM 49.5% vs. 7.9%, CM 64.0% vs. 12.3%).
Risk of progression
In the prospective German study conducted by Katsarava et al. (13), subjects with 10–14 MHDs (n = 123) had an OR of 20.1 for developing CM across one year, which was markedly higher than participants with 0–9 MHDs (n = 327, OR = 6.1). Similar findings were obtained by Bigal et al. (15) on a large population of 8219 subjects with EM with data extracted from the 2005 American Migraine Prevalence and Prevention Survey, where the frequency of headaches at baseline influenced incident CM. The primary aim of their study was to evaluate the likelihood of migraine evolution from EM to CM during a one-year follow-up period, with a specific attention to the class of acute medications used and the baseline migraine frequency (LFEM: 1–4 monthly migraine days (MMDs); MFEM: 5–9 MMDs; and HFEM: 10–14 MMDs). CM was defined according to the criteria of Silberstein and Lipton for transformed migraine (TM) (14). HFEM was found to be the only contributing factor to the transition from EM to TM (OR = 3.3). By contrast, the overall number of days of exposure to acute medications did not increase the risk of TM transition. Indeed, a high frequency of headache at baseline survived a multivariate analysis as a contributing factor to an increased risk of transitioning to TM (OR = 4.3), even when corrected for days of triptan intake per month and sex. Notably, the study unveiled a significant interaction between days of monthly intake of non-steroidal anti-inflammatory drugs (NSAIDs) and monthly headache frequency (OR = 1.93). This interaction indicates that the intake of NSAIDs offers protection against transitioning to TM at low-to-moderate monthly headache frequencies, but it becomes associated with an increased risk of transitioning to TM in people with HFEM. This observation suggests that subjects with HFEM are characterized by a specific susceptibility to acute drugs intake, which is not observed at lower migraine frequencies. The later is perhaps a function of headache day frequency rather than any biological effect of the medicines. In the above studies, the output may have been artificially influenced by the a priori stratification of subjects into groups based on the monthly frequency of headache days. A different approach, was adopted by Manzoni et al. (17) in a retrospective study on 348 subjects with EM to identify potential factors that contribute to the progression toward CM. Participants were stratified into two groups based on the subsequent natural course of their migraine: group A exhibited a favorable or stable evolution, whereas group B demonstrated an unfavorable evolution, characterized by the development of CM over time. Group B had a higher proportion of subjects with HFEM (40.3% vs. 21.0% in group A). This corroborates once again the idea that increased migraine frequency is a prognostic factor for migraine chronification.
Lipton et al. (19) applied factor mixture models to analyze a sample from the American Migraine Prevalence and Prevention (AMPP) study, including individuals with probable migraine (PM), EM (including HFEM) and CM. The factor mixture models approach assumed that the distribution of clinical features in the population is accounted for by two or more latent diagnostic categories, referred to as “taxa”. The study identified five distinct subgroups (taxa). Among these five taxa, taxon 2, which was the one with the highest percentage of subjects with HFEM (22.6%), was associated with the highest risk of developing CM over a longitudinal period of four years.
If these two studies further underscore the increased risk of progression to CM in subjects with a higher frequency of MHDs, taken alone, the data do not necessarily support a single cutoff for that progress.
The findings presented above focus on the unidirectional evolution of EM to CM, although we must also consider that migraine prevalence markedly decreases in the elderly. Migraine is therefore a dynamic condition featuring bidirectional fluctuations of frequency over time, with eventual pronounced reduction in frequency in the majority of subject. Gallardo et al. (10) conducted a prospective longitudinal study to track the frequency of headache using a web-based daily electronic diary. Participants were categorized into three groups: LFEM (4–7 headache days per month), HFEM (8–14 headache days per month) and CM (≥15 headache days per month). Migraine progression was observed in 9.5% (two out of 21) of people with LFEM and in 16.0% (eight out of 50) of people with HFEM. By contrast, 49.3% (35 out of 71) of subjects with CM improved at the end of the one-year observation period, showing less than 15 migraine headache days. Based on the observed pattern over the relatively short period of one year, 54.9% of subjects (78 out of 142) were categorized as having a non-cyclic pattern, whereas 45.1% (64 out of 142) displayed a cyclic pattern. Of interest for the scope of the present review, a higher rate of progression toward CM was detected in subjects with cyclic HFEM who did not receive preventive treatment.
In summary (Table 1), HFEM delineates a specific subgroup of subjects within the EM group who have particular socio-economic features and bear a predictable higher burden of disease, the later simply based on frequency of migraine. The increased risk of progression associated with HFEM underscores the importance of putting in place early and adequate care for HFEM subjects because their disease in many cases appears to be a pre-chronic stage across the natural history of migraine. On the other hand, one might argue that any sufficiently disabled patient, even if they do not qualify for being categorized as HFEM, should receive adequate treatment and care because headache day frequency is not the sole qualifier for migraine burden.
Clinical and therapeutic data
The following sections provide a critical appraisal on the available data regarding clinical and therapeutic features of people with HFEM. We summarize data from 16 studies (Table 2), involving 9258 subjects with EM, 2017 with CM and 253 healthy controls.
Summary of findings differentiating high-frequency migraine from low-frequency migraine or chronic migraine reported in papers focusing on clinical and therapeutic data of HFEM

CM = chronic migraine; HCs = Healthy Controls; HFEM = High Frequency Episodic Migraine; ICHD = International Classification of Headache Disorders (different editions = I, II or III); high-frequency episodic migraine; LFEM = low-frequency episodic migraine; MHDs = monthly headache days; MMDs = monthly migraine days; TM = transformed migraine.
Clinical features and comorbidities
Several studies successfully highlighted how HFEM showed specific clinical features when compared to either LFEM or CM.
Compared to people with CM, HFEM subjects report more frequently a unilateral pain localization with a pressing quality, a more intense pain during attacks and a better response to triptans. HFEM subjects report less frequently allodynia and unilateral cranial autonomic symptoms. Psychiatric comorbidities are also less frequent in HFEM than CM (39).
More, yet modest, differences emerged from the comparison of HFEM with LFEM. These include a higher occurrence of a pressing-type quality of pain, psychiatric comorbidities and dizziness in HFEM, and remarkably much more mild-to-moderate anxiety in the LFEM group (6).
Zwart et al. (28) investigated the association among migraine, non-migraine headache, headache frequency, and the presence of depression and anxiety disorders in their large-scale cross-sectional study involving a population of 51,383 individuals. Participants were categorized into three groups based on headache frequency: those experiencing headaches less than seven days per month, those with a frequency between seven and 14 days per month, and those with a frequency >14 days per month. The findings revealed that the association between migraine and depression/anxiety became more prominent as headache frequency increased. Specifically, individuals with 7–14 MHDs were more likely to have depression (OR = 3.4) or anxiety (OR = 4.9) compared to those with a lower headache frequency.
Torres-Ferrus et al. (6) found that, compared to the LFEM group, people with HFEM frequently reported more severe levels of anxiety, both state and trait, although much less mild-to-moderate anxiety, as well as moderate-to-severe depression.
In the 2005 AMPP survey, Buse et al. (24) found that, compared to LFEM and MFEM, HFEM individuals were more likely to experience psychological and psychiatric comorbidities such as anxiety, depression and bipolar disorder. Notably, there was a significant trend in the severity of depression as measured by the PHQ-9 scale, with subjects with HFEM exhibiting more pronounced depressive symptoms compared to participants with LFEM and MFEM, and less severe levels of depression compared to CM. Remarkably, for psychiatric comorbidities there was a progression from LFEM to MFEM to HFEM to CM, suggesting a continuum of biology as opposed to a cut-point dichotomy (6).
The positive correlation between migraine frequency and psychiatric comorbidities was also observed in a cross-sectional study involving 588 migraine outpatients (44). Subjects with HFEM or CM exhibited greater disability and higher rates of anxiety and depression, as assessed by the Beck Depression Inventory and Hospital Anxiety and Depression Scale, compared to LFEM.
Higher migraine frequency is associated with increased rates of depression and anxiety, and this reflects in higher perceived stress levels, as shown in an observational study conducted by An et al. (35) on 577 clinical outpatients from a tertiary hospital in Taiwan. Interestingly, there was no difference in total Perceived Stress Score between HFEM and CM. A cross-sectional study conducted by Lin et al. (34) on 538 persons with migraine from a Taiwan headache clinic revealed that migraine frequency was an independent risk factor for suicidal ideation and attempts in subjects with migraine with aura. Noteworthy, in these subjects, a linear trend was identified where rates of suicidal ideation and attempts were highest in HFEM (ideation: 37.1%, attempts: 11.4%) and CM (ideation: 47.2%, attempts: 13.9%), compared to LFEM.
Regarding other comorbidities, the 2005 AMPP study showed that, compared to CM, subjects with HFEM had reduced likelihood of experiencing chronic pain, arthritis, high cholesterol and stomach ulcers (24). Again, these distinctions progressed through all the sub-groups used without showing obvious inflection points.
Lin et al. (30) delved into the bidirectional relationship between migraine frequency and disrupted sleep quality. They carried out a cross-sectional study involving 357 participants divided into four groups based on migraine frequency, with HFEM defined as having 9–14 migraine days per month. They found an association between reduced sleep quality and HFEM. Subjects with HFEM also reported a higher incidence of difficulties in falling asleep and staying asleep, leading to higher Pittsburgh Sleep Quality Index scores. These sleep alterations were comparable between people with HFEM or CM.
Another study found a link between restless leg syndrome and migraine frequency, with a higher prevalence in subjects with HFEM (9–14 MMDs). Notably, the only significant association was observed in subjects with migraine with aura, making the results somewhat challenging to interpret (31).
Regarding cognitive performance, in general, subjects with HFEM or CM showed poorer accuracy, increased switch cost (a measure of the time needed for the executive control processes to reconfigure the cognitive system to execute a new task), and reaction times compared to LFEM. In addition, HFEM had a lower switch cost and reaction time as compared to CM (40).
Therapeutic aspects
The vast majority of studies regarding therapeutic approaches in HFEM involved onabotulinumtoxinA (BoNT-A) or monoclonal antibodies targeting the calcitonin gene related peptide pathway (anti-CGRP mAbs). By contrast, scarce data are available regarding the role of oral preventive drugs or non-pharmacological approaches in HFEM.
Regarding the BoNT-A response to treatment in subjects with HFEM, it should be noted that only open-label trials are available. Alpuente et al. (33) conducted a prospective study aimed at evaluating the effectiveness of BoNT-A in a large cohort of 578 subjects with migraine (84% with CM and 16% with HFEM). The clinical response was assessed in terms of MHDs and MMDs reduction during a two-year treatment period of BoNT-A administered according to the PREEMPT protocol. According to an interim analysis conducted on subjects followed for 12 months, subjects with HFEM showed a reduction in MHDs and MMDs, as well as acute symptomatic intake, pain intensity, disability (MIDAS) and preventive therapy. Interestingly, the rate of improvement was different between HFEM and CM groups. Indeed, the percentage reduction in MMDs was lower in HFEM than in CM after six months of treatment (HFEM 34.2 ± 32.3% vs. CM 49.7 ± 34.9%), whereas it became comparable to subjects with CM after one-year follow-up (HFEM 41.2± 28.2% vs. CM 44.7 ± 33.4%). Martinelli et al. (45) conducted an open label, single-arm trial to evaluate the efficacy of a one-year treatment with BoNT-A in the prevention of HFEM (8–14 MMDs). With the limit of the open label approach, MMDs decreased by 3.7 days, with 39% of subjects with HFEM qualifying as 50% responders at 1 year. The same group applied machine learning algorithms to identify predictors of BoNT-A response in HFEM and CM. None of the selected clinical features was able to predict the outcome in the CM group. By contrast, an easy-to-use set of four baseline variables, namely age at migraine onset, opioid use, HADS-A and MIDAS scores, predicted the response in the HFEM subgroups at one year (42). It is worth noting that two randomized controlled trials aiming to assess the efficacy of BoNT-A in HFEM are currently in progress (NCT05028569 and NCT06047457). Until this later study is reported on, it remains speculative to consider what headache frequencies are affected by botulinum toxin, particularly because a phase two trial of prabotulinumtoxinA (ABP-450) in EM failed its primary end point (Aeon Biopharma Press Release, 13 November 2023).
Several double-blind phase 3 studies (namely EVOLVE-1, EVOLVE-2 and PROMISE-1) have demonstrated the efficacy of anti-CGRP mAbs, with a six-month follow-up in subjects with EM (37,38,46). The definition of HFEM adopted in the trials was slightly different as EVOLVE (which evaluated galcanezumab 120 or 24 mg subcutaneously) considered the HFEM group to have 8–14 MMDs, whereas, in the PROMISE trial (which evaluated eptinezumab 100 or 300 mg intravenously, the lower limit was set at 10 MMDs. It is worth noting that the parent studies recruited participants with frequencies down to four migraine days a month; these studies were all positive. No analyses have suggested that LFEM participants did not respond.
The EVOLVE post-hoc analysis was conducted on 1176 subjects with HFEM (36). Both 240 and 120 mg monthly galcanezumab administration showed early effects and maintained effectiveness in reducing MMDs in HFEM compared to placebo. Galcanezumab caused a switch from HFEM to LFEM (0–3 MMDs per month) soon after the first administration in a relevant portion of subjects (34.6% for the 120 mg dose and 38.7% for the 240 mg dose), whereas a higher percentage of participants achieved the status of LFEM after the third administration. Effectiveness was maintained during the whole six-month follow-up period. Disability monitored through MIDAS questionnaire improved accordingly, especially in participants switching to the LFEM status compared to subjects who persisted in the HFEM condition.
A post-hoc analysis of the PROMISE clinical trials focused on migraine improvement in subjects classified in four groups based on migraine frequency (47). Noteworthy, in the PROMISE-1 trial, up to 46.2% of participants enrolled qualified as HFEM (10–14 MHDs). After one month of treatment, the percentage of subjects with HFEM was reduced to 15.8% and it further reduced to a 10% at month 6. The effectiveness/efficacy of anti-CGRP mAbs in HFEM has been reported in several other studies (48,–50). In their real-life multicenter study, Barbanti et al. (51) confirmed the efficacy of fremanezumab in 148 participants treated for a minimum of 24 weeks. From weeks 21 to 24, fremanezumab significantly reduced MMDs, MHDs, analgesic medication use, headache intensity through numerical rating scale (NRS), Headache Impact Test-6 (HIT-6) and MIDAS scores in both HFEM and CM compared to baseline. Responder rates of ≥50%, ≥75% and 100% were reported and were similar between groups: 75.0%, 30.8% and 9.6% for HFEM, and 72.9%, 44.8% and 1% for CM during weeks 21–24. Significant improvements were seen as early as week 4 with responder rates at week 4 of 67.6%, 32.4% and 11.8% for HFEM, and 67.3%, 40% and 1.8% for CM.
Vernieri et al. (52) evaluated the effectiveness of 1-year treatment with galcanezumab in a real-life study with 191 participants with HFEM and CM. After one year, there was a reduction of 6.0 days in MMDs for HFEM and 11.9 days for people with CM. NRS scores and monthly acute medication intake decreased accordingly in both groups.
Regarding non-pharmacological treatments, an open-label study assessed the efficacy of a double stimulation delivered at the cervical level using a non-invasive vagal nerve stimulation device in the acute migraine treatment in a cohort of 50 migraine subjects: 36 with CM and 14 with HFEM (8–14 MHDs) (29). More than one third of subjects (39.6%) reported pain freedom at 2 hours. Noteworthy, in the subgroup analysis, subjects with HFEM had a better response than CM, with a two-hour pain freedom rate of 50%, whereas it was 35.3% for CM. Another open label retrospective study conducted on a small population of subjects with HFEM (9–14 MHDs) suggested the effectiveness of osteopathic manipulative therapy on mood disorders and migraine-related impact (32). The analysis did not involve comparative groups with different frequency of headache days and therefore these findings have a limited clinical relevance.
The findings of the papers addressing clinical and therapeutic data of HFEM are summarized in Table 2.
Predictors of treatment effectiveness
Some studies have focused on the identification of potential predictors of treatment effectiveness.
A multicenter study conducted by Barbanti et al. (39) evaluated whether clinical features could act as predictors of anti-CGRP mAbs response after a 24-week treatment in subjects with HFEM with at least eight MMDs. Unilateral pain and unilateral cranial autonomic symptoms, both suggesting trigeminal sensitization, reliably predicted 50% and 75% response and survived a logistic regression analysis. These features could thus characterize a specific HFEM phenotype with an excellent response to therapy, although it is not established to be specific for HFEM, and not seen in LFEM.
Argyriou et al. (43) aimed to determine predictive factors, including socio-demographic and phenotypic variables, associated with the response to fremanezumab in a real-world population of 204 subjects with HFEM or CM. HFEM diagnosis, along with strict unilateral pain and pain location in the ophthalmic trigeminal branch were associated with a positive response to fremanezumab.
Pozo-Rosich et al. (41) evaluated the role of baseline CGRP salivary levels as a predictor of the three-month response to erenumab treatment in a population of people with HFEM or CM. Higher baseline CGRP levels were associated with a better outcome in people with HFEM, but not in CM.
The large amount of available data witnesses the efforts and the interest of the scientific community in searching for treatments and predictors for HFEM. At state of the art, treatment with target-specific drugs, such as anti-CGRP mAbs and hopefully soon with BoNT-A, were found to be effective in HFEM.
Pathophysiological data
The following sections provide a critical appraisal on the available data regarding the pathophysiological characterization of subjects with HFEM. We summarize data from eight studies (Table 3), involving 463 subjects with EM, 112 with CM and 91 healthy controls.
Summary of findings differentiating high-frequency migraine from low-frequency migraine or chronic migraine reported in papers focusing on pathophysiological aspects of HFEM
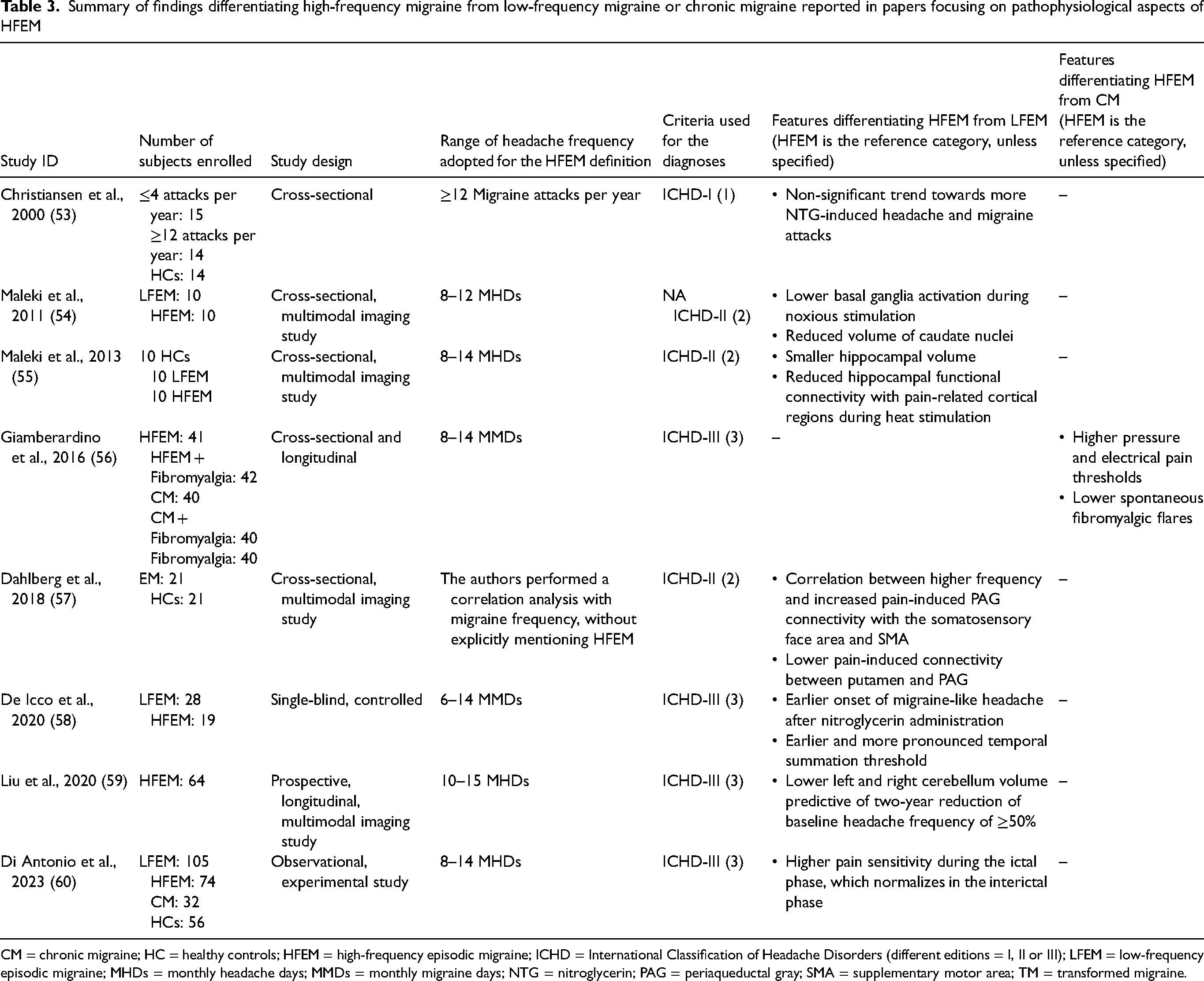
CM = chronic migraine; HC = healthy controls; HFEM = high-frequency episodic migraine; ICHD = International Classification of Headache Disorders (different editions = I, II or III); LFEM = low-frequency episodic migraine; MHDs = monthly headache days; MMDs = monthly migraine days; NTG = nitroglycerin; PAG = periaqueductal gray; SMA = supplementary motor area; TM = transformed migraine.
The output of the literature search yielded data mostly from human experimental models, neurophysiological studies, and neuroimaging/brain connectivity studies.
Human experimental models and neurophysiological investigations
Migraine models are important tools to explore migraine pathophysiology under well-controlled conditions. The nitroglycerin (NTG) migraine model has been widely used as an animal and human experimental tool to investigate migraine mechanisms and mediators (61,62).
Christiansen et al. (53) assessed the differences in the migraine induction rate of NTG according to migraine frequency, namely in subjects with rare (≤4 per year) and frequent (≥12 per year) migraine attacks (15 and 14 subjects, respectively). This study was based on the first ICHD classification (1), and considered “frequent migraineurs” as subjects who correspond more to the definition of LFEM adopted in later studies. Remarkably, no significant differences were found between the headache frequency of participants and any features of the triggered attacks.
De Icco et al. (58) explored the modifications induced by the NTG challenge on the temporal summation threshold, an electrophysiological measure of spinal nociceptive sensitization, in subjects with LFEM (1–5 MMDs), HFEM (6–14 MMDs) and healthy controls. Although confirming the specific temporal facilitation of nociceptive pathways common to all migraine subjects exposed to NTG (63), it was found that subjects with HFEM had an earlier and more pronounced reduction of the temporal summation threshold (suggestive of spinal sensitization), compared to LFEM. In LFEM, spinal sensitization developed only in those subjects who developed a migraine-like attack after NTG administration; by contrast, an altered spinal sensitization was present in all HFEM after NTG exposure. This observation was later confirmed in a separate population of subjects with HFEM (64).
Altogether, these results suggest how the exposure to a high number of migraine attacks in HFEM appears to make these subjects more sensitized in the explored nociceptive pathway.
This is also suggested by a study conducted on 203 participants with HFEM (8–14 MMDs) or CM with or without fibromyalgia aimed at investigating pain threshold through electrical and pressure stimulations (56). A higher migraine frequency was associated with lower pain thresholds, with a significant difference between HFEM and CM, as well as between HFEM comorbid with fibromyalgia and CM comorbid with fibromyalgia. Notably, fibromyalgic flares increased with headache frequency and were significantly more frequent in CM comorbid with fibromyalgia than in HFEM comorbid with fibromyalgia.
Di Antonio et al. (60) studied pain sensitivity in the trigeminocervical area by means of quantitative sensory testing and pressure pain threshold in LFEM, HFEM and CM. Compared to healthy controls, subjects with HFEM or CM were characterized by higher pain sensitivity during the ictal phase, which normalized in the interictal phase. LFEM showed an opposite behaviour, namely higher pain sensitivity in the interictal migraine phase and normalization in the ictal phase. Thus, HFEM featured a pain sensitivity profile more comparable to CM.
Functional connectivity studies
Several studies have investigated the presence of possible alterations in functional connectivity between cortical and subcortical structures in people with migraine, and how these alterations can vary depending on the frequency of migraine attacks.
Maleki et al. (55) investigated the possible association between migraine frequency and hippocampal changes, assessed with structural magnetic resonance imaging (MRI) and functional MRI during noxious sensory stimulation. They selected participants with migraine (according to the ICHD-II criteria) and divided them into HFEM (n = 10; 8–14 MHDs) and LFEM (n = 10; 1–2 MHDs). They found that HFEM had a reduced hippocampal functional connectivity with other pain-related cortical regions during heat stimulation, possibly representing a maladaptive plasticity as consequence of the more frequent attacks. With a similar experimental paradigm applied to 20 subjects with migraine, 10 of whom with HFEM, the same study also observed that functional and structural properties of the basal ganglia change in relation to headache frequency. Indeed, HFEM showed lower activation of the basal ganglia during noxious stimulation (54).
The impact of headache frequency on functional connectivity assessed during noxious stimuli was also investigated by Dahlberg et al. (57). Twenty-one subjects with EM (diagnosed according to the ICHD-II criteria) with a wide range of headache frequency were evaluated. A positive correlation was found with migraine frequency and the connectivity between the periaqueductal gray matter (PAG) and supplementary motor area (SMA), whereas the correlation was negative between the PAG and left putamen.
Finally, several structural studies have evaluated whether changes in migraine frequency can be reflected in volumetric alterations affecting some nervous structures potentially involved in its pathophysiology. Structural MRI findings during noxious sensory stimulation in the study by Maleki et al. (54,55) showed that the hippocampus and caudate nuclei volume was smaller in HFEM compared to LFEM. Once again, this volumetric shrinking was interpreted as a result of maladaptive plasticity as a result of more frequent attacks.
Another prospective study explored the MRI morphological features in 122 migraine subjects (diagnosed according to the ICHD-II criteria) categorized into eight groups of at least 10 subjects according to headache frequency: groups 1 (1–2 MHDs), 2 (3–4 MHDs), 3 (5–7 MHDs), 4 (8–10 MHDs), 5 (11–14 MHDs), 6 (15–19 MHDs), 7 (20–24 MHDs) and 8 (25–30 MHDs). It was found that the hippocampal volume varied across the frequency range, and in their cohort the greatest volumes were in participants with 5–7 days per month, immediately below the cutoff generally adopted for the definition of HFEM (65). The volume/headache days correlation for the amygdala and the hippocampus were generally positive for subjects with a lower frequency of headache days (groups 2–3) and negative for the subjects with a higher number of headache days.
In a similar study, Liu et al. (59) studied 56 subjects with HFEM (diagnosed according to the ICHD-III criteria, selecting patients with more than 10 MHDs), and focused on the possible baseline predictive features of a good two-year outcome in a real-life setting (defined as a reduction of baseline headache frequency of ≥50%). They found that a baseline lower volume over the right and left cerebellum was predictive of a good outcome in this cohort of subjects.
Other investigations
In 2016, a large cross-sectional observational study focused on the correlation between migraine frequency and hormonal changes in fertile, premenopausal, perimenopausal and menopausal women (66). Perimenopausal women had increased odds (1.42) of having HFEM. The same possibility was not confirmed in menopausal women. When considering that the perimenopausal period is characterized by wide fluctuations of estrogen levels, this observation may suggest a possible role of hormonal changes in the transition from low frequency to HFEM.
The findings of the papers addressing the pathophysiological aspects of HFEM are summarized in Table 3.
Classification aspects
According to our literature research, three studies focused on the identification of the cutoff for a clinical meaningful subdivision of migraine according to the number of MHDs.
More specifically, in the comparative analysis of their LFEM, HFEM and CM populations, Torres-Ferrús et al. (6) attempted to enhance patient categorization based on emotional and functional disability. This novel model identified the threshold of 10 or more MHDs for classifying subjects. Compared to the existing classification model, the new model demonstrated a slightly higher, though not significant, predictive ability (72.8% vs. 71.8%), but it provided a better definition for variables related to disability and impact, such as MIDAS (sensitivity 62.3%, specificity 70.6%) and HIT-6 scales (sensitivity 50.8%, specificity 74.7%).
Chalmer et al. (25) compared subjects with CM with a group of HFEM subjects. There were no between-groups differences in the demographic features, lifelong or annual attacks, comorbid diseases, and self-reported effectiveness of triptans, sickness benefit, cash assistance or rehabilitation benefit between CM and HFEM groups. Although caution is needed when interpreting these findings becuase they are partly based on recalled data and partly on measures of disability not validated for migraine, they once more suggest a striking similarity of clinical manifestations between HFEM and CM, which led to the proposal to lower the threshold to eight days per month for the classification of CM.
Ishii et al. (26) tackled the question about the adequacy of the cutoff of 15 MHDs in differentiating CM from EM. As previously discussed, they demonstrated that subjects with HFEM (8–14 MHDs) and subjects with CM with up to 24 MHDs were comparable regarding absenteeism, presenteeism, work activity impairment, work productivity reduction, interference on functioning and quality of life (26). By contrast, these values were completely different when the comparison was carried out with CM subjects with ≥24 MHDs. Finally, the probability of moderate or severe disability was lower only in subjects reporting less than eight MHDs. Based on these observations, a cuttoff of eight MHDs was suggested for capturing disability better across the migraine spectrum.
Viewpoint
ICHD-III enucleates CM from the general migraine group with diagnostic criteria requiring 15 or more headache days per month, of which at least 8 bear migraine features, for at least three months (3). Intuitively, the subjects with migraine not responding to the diagnostic criteria for CM qualify as EM. Although the definition “episodic” does not formally appear in ICHD-III, the terminology EM has been widely used in multiple studies and randomized controlled trials conducted in past 5–8 years.
Monthly migraine frequency has an impact of disease-associated disability (6), although other dimensions contribute to burden (pain intensity, associated symptoms, response to acute treatment, etc.). Several years ago, Lipton et al. (67) proposed a model based on four stages of migraine progression, which include no migraine, LFEM (with up to 9 MHDs), HFEM (with 10–14 MHDs) and CM (15 or more MHDs). Transitions between these states can be influenced by specific risk factors and may occur in either directions, resulting in increased or decreased headache frequency. Other studies have suggested the opportunity to further categorize EM into LFEM (0–3 MMDs), intermediate-frequency EM (4–7 MMDs) and HFEM (8–14 MMDs) to assist in guiding management (68).
In the previous sections, we have presented the output of a range of studies conducted to provide evidence for distinctive features of HEFM within the EM spectrum and against CM. The output of these studies presents a variable pattern comparing HFEM and lower-frequency migraine, some of which are directly frequency based. The areas covered include socio-demographic factors, comorbidities load, disability level and pathophysiological aspects (Figure 2, upper). Although migraine-associated disability requires attention and management also at frequencies below those adopted for defining HFEM, the disability level and the comorbidities load that characterize HFEM call for specific and more aggressive treatment approaches, which should also include integrated mental health support when considering the high load of psychiatric comorbidity. Regarding the pathophysiological aspects, the analyzed evidence, although still limited, underscores peculiar traits of HFEM that may have important implications for further advances in our understanding of migraine mechanisms underlying the dynamic nature of the disease. Cortical and spinal pain related pathways, assessed with different experimental set-ups, show alterations that are specific to HFEM. These are also reflected by the studies on brain connectivity, which suggests altered connectivity between areas involved in pain circuitry as a function of headache frequency. It is generally speculated that these findings may represent maladaptive plasticity, although they may also have a predictive value for the natural history of the disease (59,65). What remains unresolved is what frequency cutoff offers either pathophysiological insights or patient management benefits.

Main findings from the assessed papers, comparing subjects with high-frequency episodic migraine (HFEM) to subjects with low-frequency episodic migraine (LFEM) (upper panel) or chronic migraine (CM) (lower panel).
More blurred is the edge between HFEM and CM (Figure 2, lower). The number of studies dedicated to this topic is small, but some of them suggest that, although HFEM subjects have intermediate features between LFEM and CM, for many aspects they are closer to CM than to LFEM. It is worth noting that many of the studies discussed in the previous sections rely on the ICHD-II definition of CM, which is “pain and associated symptoms of migraine without aura for 15 days or more per month over 3 months or longer, without medication overuse”. This definition requires more migraine days than the actual ICHD-III criteria and may have led to the identification of a subgroup of CM subjects that are phenotypically more similar to HFEM than those with a phenotype closer to the ‘transformed migraine’ of Manzoni et al. (69), where migraine features are lost on many MHDs (70).
This consideration brings about the fact that, in most studies focusing on HFEM, the diagnostic definition relied on the number of monthly headache days, not of monthly migraine days. This approach may assume that subjects with HFEM only have migraine days, which is not necessarily true. Unfortunately, the literature lacks data confirming or refuting this hypothesis. Further elaboration on this aspect needs evidence based on prospective diary data recording in subjects with HFEM.
A recent systematic review and meta-analysis of observational cohort studies has identified a “high” level of evidence for monthly headache day frequency ≥ 10 in predicting progression from EM to CM (risk ratio = 5.95) (71). Although part of the risk may be related to acute medication overuse, which was associated with an even higher risk (8.82) although with a very low level of evidence, this observation seems extremely important and, by itself, justifies the need to draw attention to HFEM by singling it out of the larger group of EM for several reasons. First, the different iterations of ICHD have favored tremendous advances in headache medicine and science by introducing shared criteria for different entities. This has happened for CM, for which the final diagnostic criteria required evidence and expert consensus to achieve their final version (72,73). Although the 15 MHDs threshold for CM definition is mostly arbitrary, it has served well the purpose of providing precise boundaries for focusing attention on a more severe form of migraine. This approach not only boosted research on CM, but also, and more importantly for clinicians and people with migraine, prompted the acknowledgement and the recognition of the entity by regulators and by the healthcare systems. Along this line of reasoning, defining HEFM as a separate entity would attract attention on its high burden and negative prognostic value, facilitating access to a possibly wider range of treatment options and resources for people with migraine, clinicians and researchers. Second, it would stimulate research in an area that holds the promise of providing very important insights on different aspects of migraine, from the pathophysiology to the identification of novel specific treatments. Third, it would provide ground for preventing CM by treating more aggressively subjects with HFEM to avoid their progression. This would represent a tremendous breakthrough in the clinical approach to CM, which is presently based on the attempt to reverse its temporal pattern from chronic to episodic, rather than intercepting and preventing the episodic-to-chronic progression.
Several migraine-specific preventive drugs have become available in the past few years. They are more expensive than the traditional oral preventive treatments, but evidence is accumulating to show that effective treatment of migraine brings about enormous direct and indirect cost savings, in addition to improving the life of sufferers. Siersbæk et al. (74) investigated the economic implications of administering anti-CGRP mAbs to 362 participants with LFEM, HFEM and CM. Health economic savings after the initiation of treatment amounted to €1179, €264 and €175 per year in people with CM, HFEM and LFEM, respectively. Furthermore, socio-economic gains associated with anti-CGRP mAb treatment totaled an average gross domestic product gain of €13,329, €10,449 and €9947 for subjects with CM, HFEM and LFEM, respectively.
Bearing in mind the possible positive implications illustrated above and the abundance of data discussed in this review, we feel that the identification of HFEM as a migraine subtype may have several beneficial consequences. According to data from literature, the lower threshold of 8 MHDs appears to capture the intrinsic features of HFEM. Of note, the same value has been also proposed as the lower limit for the definition of resistant and/or refractory migraine (75). An important point emerging from our critical appraisal of the literature is the lack of consensus on the use monthly headache days or monthly migraine days for the definition of HFEM. As for CM, we are aware that in HFEM some headache days may not display clear migraine features. Therefore, when considering that ICHD-III requires approximately that 50% of the minimum number of MHDs (8 out 15) qualify as MMDs for the diagnosis of CM, in our proposal of diagnostic criteria for HFEM, we require 4 MMDs out of the minimum number of 8 MHDs needed for the diagnosis. We hereby propose a working definition of HFEM to prompt a consensus definition to be tested in clinical practice and in research studies in the next few years (Table 4).
Proposed definition of HFEM and associated diagnostic criteria

HFEM = high-frequency episodic migraine; ICHD = International Classification of Headache Disorders.
According to this proposal, the migraine chapter would be re-arranged as suggested in Table 5.
Proposed reorganization of “1. Migraine” chapter at the first and second digit level.

The proposed definition of HFEM is of course amenable for discussion and further improvement and it will hopefully stimulate validation studies on large datasets, supported by data-driven, rather than hypothesis-driven, modeling approaches to identify headache day frequency and/or disability cutoffs reliably associated with pathophysiological aspects or patient management benefits.
Limitations
The present study presents several limitations. Although a structured research strategy was adopted, as a narrative review only a single database was used for the selection of articles. Our review included retrospective studies or data collection based on self-administered questionnaires; these methodological approaches may suffer recall biases as well as poor reliability. Additionally, several results derived from post-hoc or sub-group analyses, with only a part of the revised studies originally conceived and powered for the study of HFEM. Furthermore, the lack of a consensus definition of HFEM, led to subjective and variable identification of the cutoff of days for defining HFEM in the different papers. This prevented de facto any comparison between homogeneous groups and may have yielded some overlap across cohorts.
Conclusions
An increasing amount of evidence supports the idea that HFEM is a clinical entity deserving a close monitoring and a tailored therapeutic approach. HFEM indeed identifies a subgroup of people with EM characterized by higher disability, clinical peculiarities and an increased risk of negative outcome. Here, we endeavored to offer a revision of the pertinent literature and a tentative definition, with associated criteria, to be tested for a possible future integration in the classification. Taking this step will hopefully foster multiple advances in different areas of headache medicine. In the research area, it will allow the mechanisms of migraine progression to chronicity or, equally interesting, the opposite process of reversion from HFEM or CM to low-frequency EM to be tackled. In the clinical field, it will prompt the possibility of preventing migraine chronification. In the public health arena, it will attract focus and resources for the better management of a severe and highly disabling condition.
Singling HFEM out of the EM frequency spectrum does not imply that LFEM deserves less attention because disability is not simply the sum of MMDs or MHDs, in that it encompasses multiple factors (attack intensity, associated symptoms, response to acute or preventive medications). Indeed, having four or more days of migraine per month is the threshold for starting preventive treatment, and this is already correlated with disability, as well as healthcare and resources utilization (20). HFEM identification comprises an important step toward a more personalized approach for patients who bear the same diagnosis but different expression of the disease.
Clinical implications
High-frequency episodic migraine (HFEM) has gained the attention of clinicians and researchers despite the lack of a consensus definition. Several clinical and pathophysiological features differentiate HFEM from low-frequency episodic migraine and, to a lesser extent, from chronic migraine. Within the episodic migraine spectrum, people with HFEM are characterized by a higher disability, a more severe socio-economic burden and an increased risk of progression toward chronic migraine. A consensus definition of HFEM can facilitate a better understanding of the mechanisms underlying migraine frequency spectrum and foster more adequate public health approaches.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article (past 4 years). FC: None. RDI received personal fees for participating in speaking at scientific events from Eli-Lilly, TEVA and Lundbeck. He has participated in an advisory board for Pfizer and AbbVie. MC: None. FB: None. GV received personal fees for participating in speaking at scientific events from Lundbeck. DM reports support from Abbvie for participating in advisory boards; consulting fees from LifeSciences Consultants, fees from Medscape for organizing educational programs; and personal fees from Lundbeck for lecturing at symposia. PP-R reports support for the present study from AbbVie and personal fees for consulting from AbbVie, Eli Lilly, Lundbeck, Medscape, Novartis, Pfizer and Teva. She has received personal fees for speaking from AbbVie, Dr Reddy's, Eli Lilly, Lundbeck, Medscape, Novartis and Teva, and has had grants paid to her research group from AbbVie, AGAUR, EraNet Neuron, FEDER RIS3CAT, Instituto Investigacion Carlos III, International Headache Society, Novartis and Teva. She is a member of the Scientific Advisory Board for Lilly Foundation Spain.
PJG reports, over the last 36 months, grants from Celgene and Kallyope; personal fees from Aeon Biopharma, Abbvie, Amgen, eNeura, CoolTech LLC, Dr Reddys’, Eli-Lilly and Company, Epalex, Linpharma, Lundbeck, Man&Science, Novartis, Pfizer, Sanofi, Satsuma, Shiratronics and Teva Pharmaceuticals; personal fees for advice through Gerson Lehrman Group, Guidepoint, SAI Med Partners and Vector Metric; fees for educational materials from CME Outfitters; as well as publishing royalties or fees from Massachusetts Medical Society, Oxford University Press, UptoDate and Wolters Kluwer. CT reports support from Abbvie and Novartis for investigator-initiated trials; consulting fees from Abbvie, Eli Lilly, Dompé, Ipsen, Lundbeck, Pfizer, Teva and Medscape for participating in advisory boards; support from Abbvie, Eli Lilly, Dompé, Ipsen, Lundbeck, Pfizer and Teva for attending meetings; and personal fees from Abbvie, Eli Lilly, Lundbeck, Pfizer and Teva for lecturing at symposia. She is principal investigator of clinical trials sponsored by Abbvie, Biohaven, Eli Lilly, Ipsen, Lundbeck, Pfizer and Teva. She has received research grants from the European Commission, the Italian Ministry of Health and the Migraine Research Foundation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: support was provided by the Ministero della Salute (grant number Ricerca Finalizzata GR-2021-12372429).
