Abstract
Background
Medication overuse headache is a prevalent secondary headache due to the overuse of analgesics, mainly over-the-counter analgesics. Over-the-counter analgesics have been associated with disrupted male endocrinology, while the effects on female endocrinology remain nearly unknown. The aim was to understand the effect of long-term analgesic exposure in females with medication overuse headache on Anti-Müllerian hormone, a surrogate measure of female fertility.
Methods
Using a translational approach, an observational prospective clinical study was conducted to determine the effect of withdrawal therapy in females with medication overuse headache on Anti-Müllerian hormone levels, in combination with pre-clinical investigation of primary granulosa cells to understand the effects of analgesics on granulosa cell function.
Results
We included 21 females (mean-age 30.0 years; SD (7.3)) for Anti-Müllerian hormone -measurement. Anti-Müllerian Hormone increased by 21% from baseline (mean 20.1 pmol/L; SD (8.7)) after withdrawal of analgesics ((mean 24.3 pmol/L; SD (12.0)); p = 0.0023). Exposing primary granulosa cells to analgesics (acetaminophen (100 and 200 µM, n = 9–10) and ibuprofen (150 and 200 µM, n = 12–13)) did not reduce Anti-Müllerian hormone levels. In contrast, de novo DNA synthesis in GCs (n = 6) exposed to acetaminophen was reduced by 78% (p = 0.0036) compared to controls, suggesting that cellular proliferation was restricted.
Conclusion
We found that frequent use of over-the-counter analgesics was associated with repressed Anti-Müllerian Hormone levels, likely through disruption of granulosa cell proliferation. Further research is crucial to investigate a potential effect of analgesics on adult female reproductive endocrinology.
This is a visual representation of the abstract.
Keywords
Introduction
Over-the-counter analgesics (OTC) acetaminophen (N-acetyl-para-aminophenol (APAP) also known as paracetamol), acetylsalicylic acid and ibuprofen are considered first-line treatments for several headache and pain disorders (1,2). Many patients with medication overuse headache (MOH) are among those with a frequent use of OTC. MOH, affecting over 60 million people globally, is defined as a secondary chronic headache caused by regular overuse of analgesics for a period of at least three months (3,4). In MOH, the frequent intake of analgesics paradoxically contributes to worsening the burden of headache, instead of providing release, by lowering the pain perception threshold (5). The recommended treatment of MOH is withdrawal therapy of the analgesic-overuse that has proven effective in reversing the chronic headache to an episodic pattern (2,6,7). However, only a little attention has been paid to other side effects of the long-term frequent exposure to analgesic-use other than the chronification of headache.
There is an ongoing debate concerning the reasons for the continuously decreasing fertility (8). Increasing evidence shows that exposure to OTC can disrupt endocrinology during both fetal and adult life (9,10). Most patients with MOH are females of reproductive age (11–13). Nevertheless, studies investigating the effect of OTC on endocrinology among adults have solely focused on males, showing that, e.g., ibuprofen, is a powerful endocrine disruptor resulting in reduced levels of testosterone from testicular Leydig cells and compensated hypogonadism (9). The role of Anti-Müllerian hormone (AMH), a peptide growth factor of the transforming growth factor-β family, is clinically well characterized in females. AMH is mainly produced by the granulosa cells (GC) of small growing follicles in the ovaries and inhibits excessive follicular recruitment by follicular stimulating hormone (FSH) (14–17). Importantly, the level of AMH changes according to age as the ovarian reserve decreases; it rises in the early twenties, tops at the age of 25, and then declines to undetectable levels about the age of 50 (15,16). AMH is thus regarded as a reliable marker of ovarian reserve and is used in the clinic as a surrogate measure of female fertility preservation (18).
In this translational study, we therefore aimed to explore the effect of OTC analgesics on female adult endocrinology. We performed a combination of two interconnected approaches: (i) an observational prospective clinical study with females in the fertile age in which AMH levels were evaluated before and two months after complete withdrawal of analgesic use and (ii) an ex vivo cellular assay using human primary GCs to understand if OTC analgesics had any direct effect on cell function.
Methods
Study design
This is a translational study composed of a pre-clinical and an observational prospective clinical part. The study was conducted in close collaboration between the Danish Headache Center (DHC), Department of Growth and Reproduction and the Fertility Department at Copenhagen University Hospital – Rigshospitalet. The clinical part of the study was registered at ClinicalTrials.gov (Identifier NCT04090333).
Clinical part
Study population
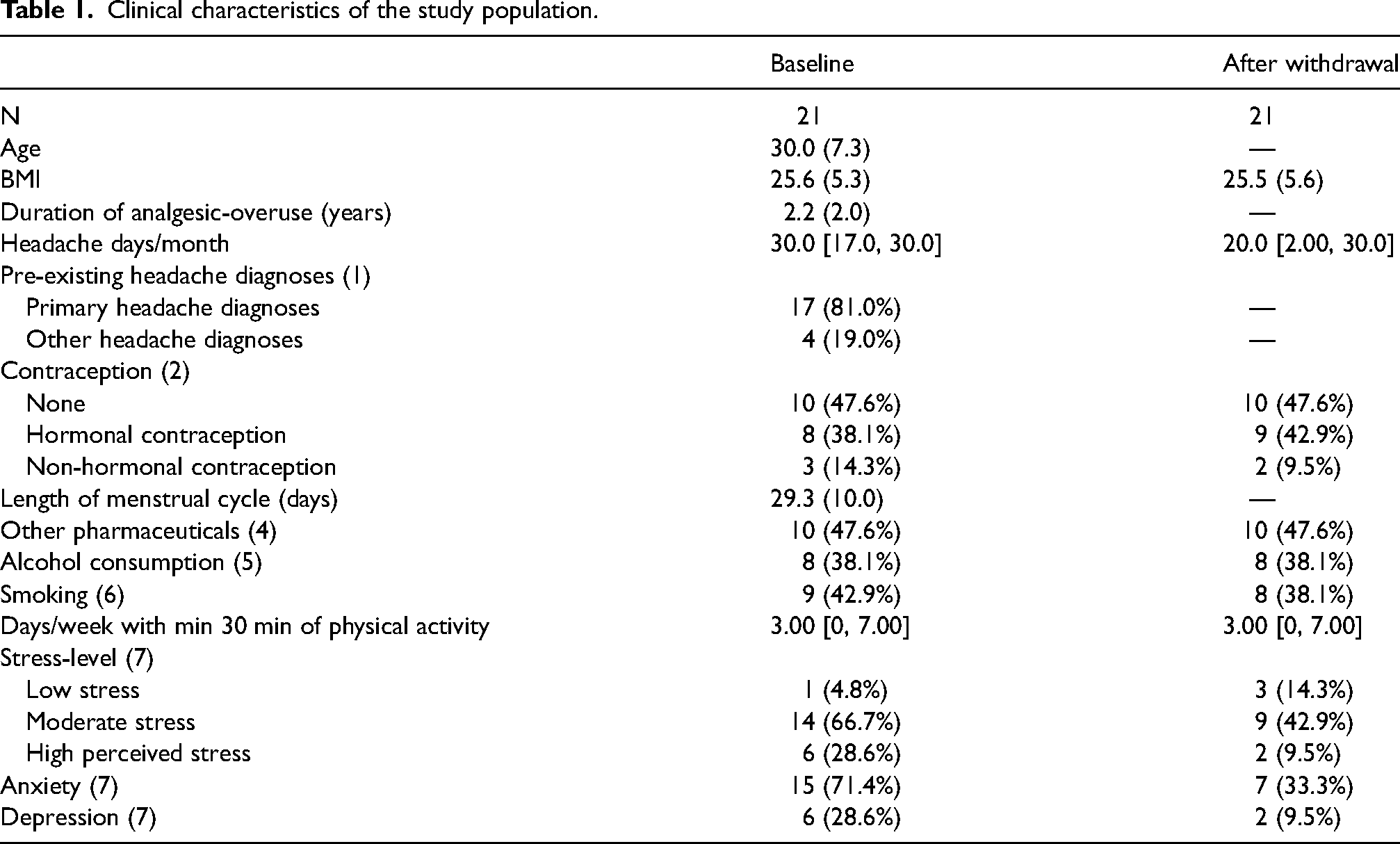
Females with MOH referred to a tertiary clinic, DHC, were invited to participate. Inclusion criteria were 1) confirmed MOH diagnosis according to International Classification of Headache Disorders 3rd version (headache occurring on ≥ 15 days/month for ≥3 months in a patient with a preexisting headache disorder; regular overuse of acute headache medication where overuse is defined as ≥ 15 days/month when taking simple analgesics and ≥ 10 days/month when taking triptans, ergotamines, opioids, combination analgesics or a combination of any of these analgesics without overuse of a single drug; not better accounted for by another International Criteria of Headache Disorders 3rd version diagnosis) (3) ; 2) age between 20–40 years; and 3) capable of providing and signing informed consent.
Exclusion criteria were 1) known history of polycystic ovary syndrome (PCOS); 2) planned pregnancy within the next two months; 3) current pregnancy or breastfeeding; and 4) inability to provide reliable information about medical history. Only patients who followed the withdrawal treatment protocol were included in the final analyses. The regional ethics committee in the Capital Region approved the study (VEK H-19023379), and all participants provided written informed consent.
Standard care withdrawal
Patients received standard care for MOH following the applicable guidelines that included withdrawal of analgesics for two months, with maximal use of analgesics being nine days per month (2). During the withdrawal process, all patients received prescriptions for ‘rescue medication’ in the form of tablet levomepromazine or tablet promethazine, both up to a maximum of 75 mg per day, and antiemetics in the form of tablet domperidone or tablet metoclopramide, both up to 30 mg per day.
Clinical characteristics and endpoints
Information about age, headache history, co-morbidities and previous history of fertility treatment were collected at baseline by a semi-structured interview of the clinician. Following information was obtained by the clinician both at baseline and after withdrawal: Body Mass Index (BMI; kg2/m), headache days and use of analgesics (including exact type and days of use per month) by fulfilled headache calendars, smoking habits (never, occasionally, daily, previously), intake of alcohol (defined as none, normal use (1–7 units of alcohol per week), overuse (8–14 units per week, and damaging use (over 14 units per week)), type of contraception, length of menstrual cycle, use of rescue medication during withdrawal and use of other pharmaceuticals. Furthermore, patients were asked to fill out two validated questionnaires at baseline and after withdrawal: The Hospitality Anxiety and Depression Scale (HADS) and the Perceived Stress Scale (PSS). HADS was used to identify patients at risk of having depression or anxiety (cut-off value of 8), while PSS estimated stress level (mild, moderate, or high perceived stress) (19–21). The primary endpoint was change in AMH levels before and after two months of withdrawal. A subgroup analysis of AMH level change was performed for patients age ≥ 25 years, corresponding to the point in life from which AMH will gradually decrease (22).
Blood samples
Blood samples were collected before and after two months of withdrawal. Blood samples were taken from the cubital vein in 9 ml EDTA tubes, centrifuged immediately after for 10 min at 15,000 rpm, after which EDTA plasma was pipetted in 1 ml Eppendorf tubes and frozen at −25 °C. All samples were thawed and re-frozen one time.
AMH plasma level measurements
In general, the investigation of hormonal disruption in females is difficult due to the complexity of varying hormone levels during the menstrual cycle. However, AMH is the only hormone that stays stable during the menstrual cycle, consequently plasma AMH levels are independent of the period of the menstrual cycle (15). AMH plasma levels (pmol/L) were analyzed at the Fertility Department at Rigshospitalet. The frozen EDTA plasma samples were thawed, and all AMH-analyses were conducted at the same time on Cobas 8000 e801 equipment. ELISA (Enzyme Linked Immuno Sorbent Assay) and RIA (RadioImmunoAssay) tests were performed for analyzing plasma levels of AMH.
Analysis of analgesics in plasma samples
The method was adapted from Norén et al. (23) Briefly, 100 µL of plasma, 0.01 mL of ammonium acetate (pH 6.5) and 0.01 mL glucuronidase (Escherichia coli, Roche Diagnostics, CAS No 9001-45-0) were added, and the solution was incubated at 37 °C for 90 min. After the addition of 0.025 mL of acetonitrile/MilliQ water (1:1), 0.025 mL of internal standard, and 0.2 mL of acetontirile, the plates were vigorously shaken for 30 min and centrifuged for 10 min at 3000 rpm. Extracts were analyzed for acetylsalicylic acid, 4-hydroxy-diclofenac, carboxy ibuprofen, APAP, sumatriptan, naproxen, carbamazepine, venlafaxine, sertraline, fluoxetine, and citalopram, using a Shimadzu UFLC system (Shimadzu Corporation, Kyoto, Japan) coupled to a QTRAP5500 equipped with a TurboIon Spray source (AB Sciex, Framingham, MA, USA). The limit of detection (LOD) ranged from 1 to 8 ng /ml and was defined as three times the standard deviation of the concentration corresponding to the peak at the same retention time as the analyte in the chemical blanks (n = 10).
Statistics
Statistical analyses were performed using statistical software R version (R Foundation for Statistical Computing, Vienna, Austria, version 1.1.463). We presented categorical variables as n (%). Continuous data were presented either as mean (SD) or median [min; max]. Comparison of the AMH level before and after withdrawal was performed by a paired t-test. Potential associations between change in AMH level from baseline to after withdrawal and following factors were analyzed using a simple linear regression model: 1) Change in frequency of analgesic-intake from baseline to after withdrawal; 2) change in headache frequency from baseline to after withdrawal; 3) change in plasma concentration of APAP from baseline to after withdrawal; 4) change in plasma concentration of ibuprofen from baseline to after withdrawal. Levels of analgesic concentrations that were “not detectable” were considered as “0” when conducting the data analysis. The level of significance was set to p < 0.05, and p-values were calculated as two-tailed.
The sample size calculations were based on an alpha-error on 5% and an 80% power. The outcome of interest was percentage change in AMH from baseline to after withdrawal. The clinically relevant difference is 9% with SD = 11%. However, we needed to add in some assumptions. First, we expected a drop-out rate on 15–20%. Additionally, we expected the study population to be more heterogeneous due to intake of distinct types of analgesics, and finally we decided to include patients with hormonal contraception. Initially, we accordingly estimated to include 40 females with MOH.
Pre-clinical part
Isolation and cultivation of GCs
The follicular aspirate was centrifuged for 10 min at 400g and the supernatant was discharged. The pellet was resuspended in 37°C phosphate-buffered saline (PBS, Thermo Fisher Scientific, Waltham, MA, USA) and purified by centrifugation for 30 min at 400g on a Lymphoprep gradient (StemCell Technologies, Vancouver, BC, Canada). The GCs were aspirated from the interface of the gradient with a Pasteur pipette and washed twice with 37°C PBS. Subsequently the GC were cultured in Minimum Essential Medium α (MEM α, Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 5% Human Serum Albumin (HAS, CSL Behring, King of Prussia, PA, USA), 1% Human Platelet Lysate (HPL, StemCell Technologies, Vancouver, BC, Canada), 1% Glutamax + (Thermo Fisher Scientific, Waltham, MA, USA), 1% penicillin–streptomycin (Thermo Fisher Scientific, Waltham, MA, USA), and 0.01% Insulin-transferrin-selenium (ITS-G, Thermo Fisher Scientific, Waltham, MA, USA) for 48 h. Upon exposure to mild analgesic, HPL and ITS-G were excluded from the culture medium. For the AMH measurements the GCs were exposed to control (DMSO), APAP or ibuprofen for 48 h prior to collection and storage (−80°C) of the culture media. The AMH content was measured with the Ultra-Sensitive Anti-Müllerian hormone/Müllerian inhibiting substance ELISA kit (US AMH/MIS). Click-iT Plus EdU Alexa Fluor 555 Cell Proliferation imaging kit (Thermo Fisher Scientific, Waltham, MA, USA) was used to quantify newly synthesized DNA and the GCs were exposed to control (DMSO) or APAP for 48 h with the addition of the thymidine analog EdU (10 µM) for the final 3 h of exposure. The use of surplus GCs was approved by the Research Ethics Committee of the Capital Region of Denmark (VEK H-20041816) and participating women signed informed consent prior to donation.
Immunofluorescence microscopy and imaging
GCs were grown on glass coverslips, washed twice in PBS, fixed for 15 min with 4% paraformaldehyde (PFA) solution, washed twice (3% BSA in PBS), permeabilized for 20 min (PBS with 0.5% Triton-X100) and the following EdU detection were performed as described in the protocol of the manufacture (Thermo Fisher Scientific, Waltham, MA, USA). An Olympus BX63 upright microscope with an Olympus DP72 color, 12.8-megapixel, 4140 Å∼3096 resolution camera (Olympus, Tokyo, Japan) was used to obtain the immunofluorescence images later analyzed for EdU intensity. ImageJ (Bethesda, MD, USA) were used to outline the nucleus (DAPI, Thermo Fisher Scientific, Waltham, MA, USA) as well as quantify the EdU intensity along with background readings. The mean fluorescence was calculated by subtracting the corresponding mean background value. An average of approximately 60 cells per condition were analyzed based on 3–5 representative images (40x) of each sample.
Data analysis and statistics
Data analyses were performed with GraphPad Prism 9-10 (San Diego, CA, USA). For the GC experiments the ‘n’ signifies the number of donors. The specific statistical test used to analyze the given data is stated in the figure legends. Data is represented as the mean ± SD or SEM and p < 0.05 was considered statistically significant.
Results
Study population
Females with MOH were recruited from DHC, consecutively from July 2019 to August 2020. The final follow-up visit occurred in October 2020. Of 24 females fulfilling the criteria, 21 (mean-age 29.6 years; SD (7.2)) were included in the final analyses, while three were excluded (two did not follow the withdrawal protocol and one was excluded due to outlier AMH-value at baseline). Inclusion of patients was complicated by the COVID-19 epidemic. Since we had a lower drop-out rate (12.5%) than expected, and since all the below-mentioned factors, which could have influenced the AMH level, remained stable, we decided to stop inclusion before reaching the estimated sample size.
Clinical characteristics
Table 1 presents the characteristics of the study population. None of the participants had a history of PCOS, fertility problems or had undergone fertility therapy. BMI, physical activity, alcohol consumption and smoking habits, remained stable (Table 1). Only two shifted contraception during the study period: One from copper IUD (intrauterine device) to mini-pills; and one from copper IUD to none. Also, the level of other medication intake did not change remarkably, as only one stopped metoprolol, one started amitriptyline and one started candesartan during the study period, and these changes were in relation to the headache treatment. No other headache preventive medications were used. At baseline, six (28.6%) patients had a high perceived stress level, 15 (71.4%) had symptoms of anxiety and six (28.6%) had symptoms of depression. However, only 14 patients out of 21 answered the follow-up PSS and HADS questionnaires after withdrawal making it difficult to interpret the results completely.
Clinical characteristics of the study population.
Analgesic intake and withdrawal therapy
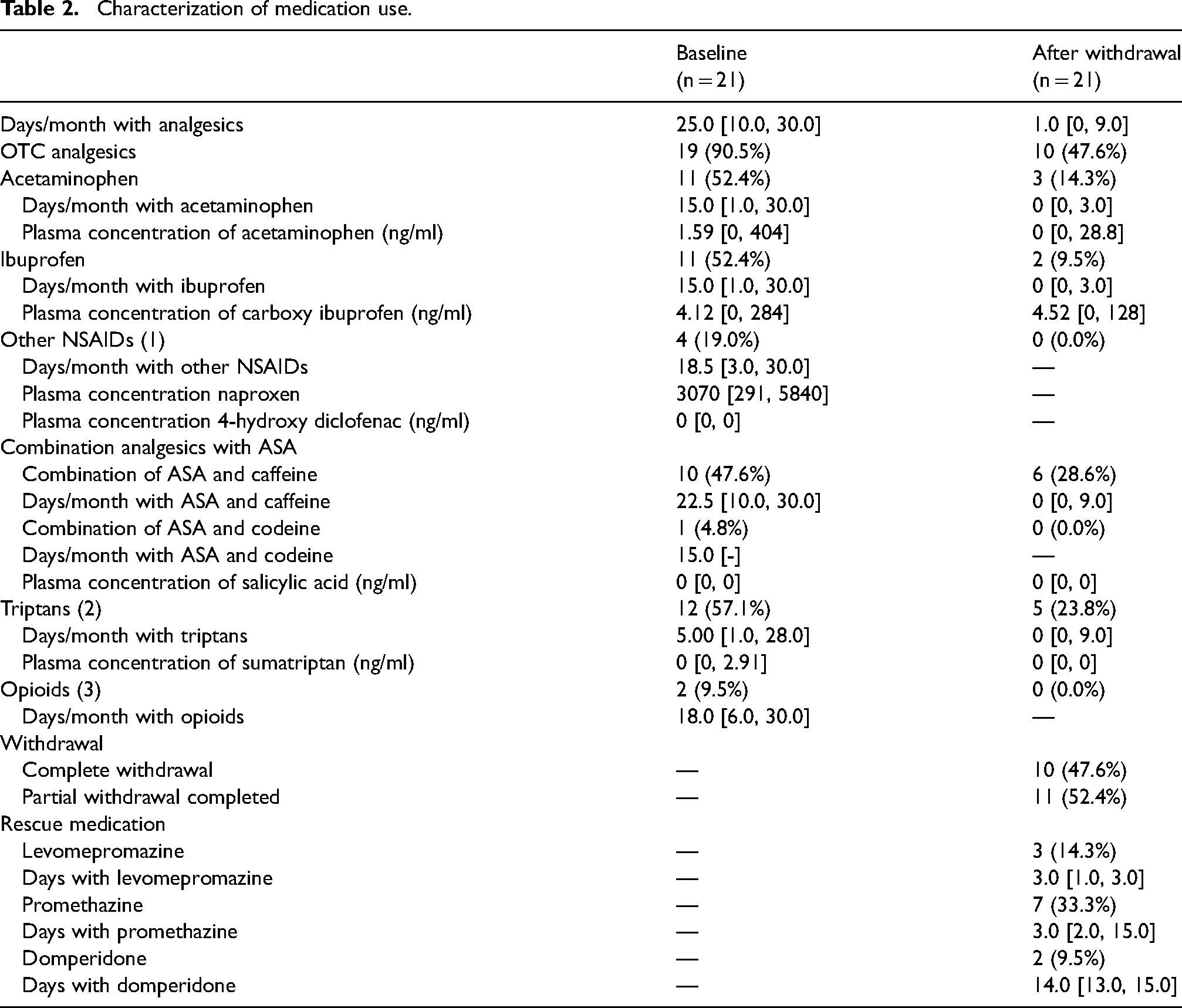
Almost all patients (n = 19; 90.5%) used OTC at baseline (Table 2), whereof the most frequently used drugs were APAP (n = 11; 52.4%), ibuprofen (n = 11; 52.4%) and combination-analgesics containing ASA and caffeine (n = 10; 47.6%). The rest used opioids (n = 2; 9.5%) or triptans (n = 12; 57.1%). As expected, total use of analgesics decreased from 25.0 days/month [10.0–30.0] at baseline to 1.0 days/month [0.0–9.0] after withdrawal (Table 2). Almost half of the patients (47.6%) completely stopped taking any analgesics for two months (complete withdrawal), and the rest used restricted analgesics for a maximum of nine days per month (partial withdrawal). The number of headache days decreased from 30 [17.0, 30.0] days per month to 20 [2.00, 30.0] days per month in the same time interval. Plasma concentrations of analgesics (Table 2) represented analgesic-intake within the last hours up to the blood samples. Therefore, plasma concentrations may not reflect the long-term intake of analgesics.
Characterization of medication use.
Change in AMH plasma level
Figure 1 shows the individual change in AMH plasma level from baseline to after two months of withdrawal. AMH increased from baseline (mean 20.1 pmol/L; SD (8.7)) to after withdrawal (mean 24.3 pmol/L; SD (12.0)); p = 0.0023. This corresponds to an increase of 21%. When only including patients aged ≥ 25 years (n = 14), we still find a significant increase in AMH from 18.3 pmol/L (SD 6.9) at baseline to 21.6 pmol/L (SD 8.2) after withdrawal; p = 0.037, corresponding to an increase of 22%.We did not find any association between either change in AMH, headache days/month (p = 0.33), intake of analgesics (p = 0.52), APAP plasma concentration (p = 0.97) or carboxy ibuprofen (a ibuprofen biomarker) plasma concentration (p = 0.24).
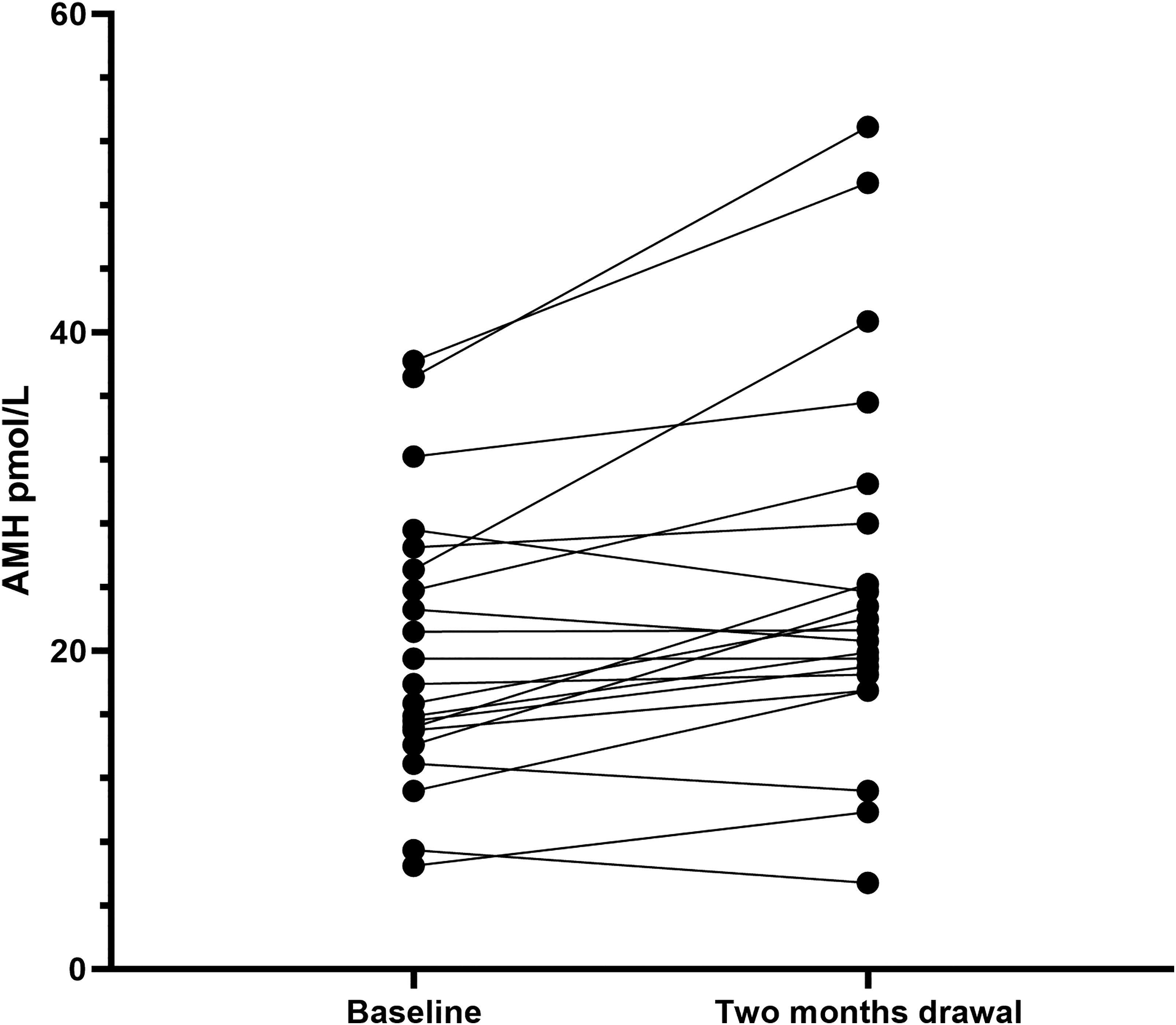
The plot represents changes in AMH level (pmol/L) from baseline to two months follow-up after withdrawal therapy for each participant.
Mild analgesics and granulosa cell function
The change in circulatory AMH levels suggests that analgesic use might affect the function of GCs. To investigate the direct effect of analgesics on cellular function, we isolated surplus GCs from females undergoing fertility treatment. Exposing cells to the two most used compounds among the patients, APAP (100 and 200 µM, n = 9–10) and ibuprofen (150 and 200 µM, n = 12–13), did not result in any decrease in AMH secretion from the cells after normalization to the DNA content (Figure 2(a) and (b)). These data suggest that APAP and ibuprofen do not have a direct effect on transcription, translation, or secretion of AMH.
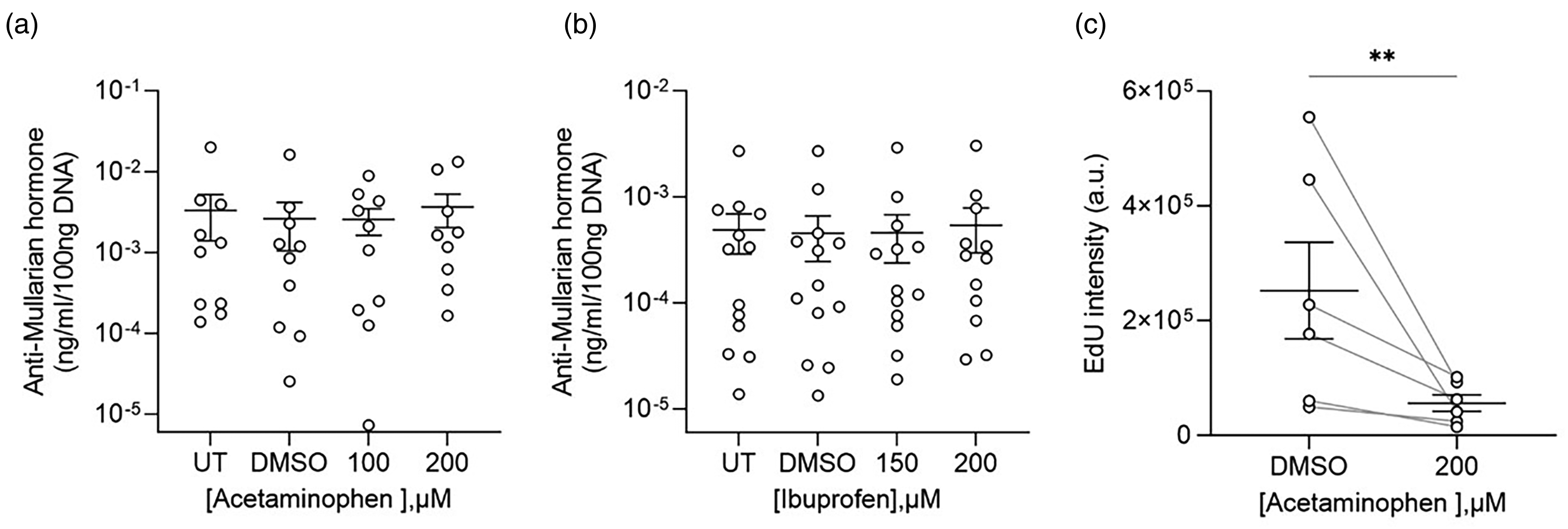
Granulosa cell function after exposure to mild analgesics. (a) AMH secretion from untreated (UT), DMSO (control) and APAP (100 and 200 µM, n = 9–10) exposed GCs. (b) AMH secretion from untreated (UT), DMSO (control) and ibuprofen (150 and 200 µM, n = 12–13) exposed GCs. C) Quantification of the EdU fluorescence intensity from DMSO (control) and APAP (200 µM, n = 6) exposed GCs. Statistical significance, for the AMH secretion, was tested with Kruskal–Wallis one-way analysis followed by Dunn's multiple comparison post hoc test. Statistical significance, for the EdU intensity, was tested with a ratio paired two-tailed t-test. Data points are represented as mean ± SEM. **P < 0.01.
As the proliferation of GCs during follicle maturation is critical, we next investigated whether APAP, the most used compound among the females, affected de novo DNA synthesis. To determine the effect of APAP on DNA synthesis, GCs were exposed to control (DMSO) or 200 µM APAP for 48 h with the thymidine analogue EdU (5-ethynyl-2´-deoxyuridine) added for the final 3 h to quantify DNA synthesis (Figure 2(c), n = 6). The de novo DNA synthesis in the APAP exposed GCs (mean 56,273 arbitrary units (a.u.); SD (35,804)) was lower compared to control (mean 252,411 a.u.; SD (206,548)); p = 0.0036, corresponding to a decrease of 78%. This significant drop in DNA synthesis suggests that APAP affects GCs ability to proliferate.
Discussion
The results of our study suggest that analgesics, e.g., APAP, can have a direct negative effect on female ovarian endocrinology and potentially oocyte quality, essential for female fertility (24,25). We measured the AMH plasma-level in females of fertile age with long-term frequent exposure to analgesics before and after withdrawal of the frequent analgesic intake, and found that a biomarker for female fertility, the granulosa cell hormone AMH, increased after withdrawal. Moreover, we found that frequently used analgesics APAP and ibuprofen did not have a direct effect on AMH secretion, but APAP instead inhibited GC proliferation. These data therefore suggest that frequent analgesic-use might decrease granulosa cell proliferation, resulting in changed follicular output of AMH.
Potential clinical factors for AMH-increase
AMH is considered to remain stable during the menstrual cycle (15). However, the AMH level can be affected by several distinct factors, e.g., hormonal prevention, which may lead to a reversible decrease on 30–50% in AMH level (26,27). Other factors that may affect the AMH level are other pharmaceuticals, environmental factors, PCOS, BMI, among others, in line with the current ongoing fertility debate (8). In our study, all the factors mentioned that could have influenced the AMH level, were stable during the study period. The only major change that we observed was the marked reduction in analgesic intake according to the intended withdrawal treatment of MOH. This, therefore, strongly suggests that the increase in AMH is related to the MOH-treatment and associated analgesic withdrawal. We have previously found that healthy male volunteers developed compensated hypogonadism after daily intake of 600 mg ibuprofen for two weeks in a randomized placebo-controlled trial (9). In compensated hypogonadism, the testosterone production is compromised but remains normal due to a compensatory increased level of stimulating Luteinizing Hormone (LH). Importantly, in this study we found a reduction in AMH levels by 9% after 14 days and by 7% after 44 days of ibuprofen intake. Together with the present data, this suggests that analgesic-use has a direct effect on AMH levels that cannot be compensated by increased gonadotropin secretion from the pituitary. Our present data from ex vivo GCs together with our previous data from men strongly suggest that the increase in AMH levels after withdrawal is likely not only linked to a better physiological state but directly a reduced level of disruption of granulosa or Sertoli cell function.
Is the AMH change clinically relevant?
From the age of 25 years AMH level in females is expected to decrease by approximately 2 pmol/L per year (16). Therefore, an increase of 4 pmol/L on average, corresponding to the observed 21% increase, within a two-month period seems to be of major interest and of clinical relevance. Moreover, when only looking at women aged ≥ 25 years, we find an increase of approximately 2 pmol/L, still corresponding to 22%, reflecting the reduced AMH level due to aging.
The ongoing debate concerning reasons for the continuously decreasing fertility includes changed lifestyle, direct and indirect contamination of xenobiotic chemicals, e.g., pollution, chemicals in food, plastics, and pharmaceuticals (8). Our results are, to our knowledge, the first to indicate a direct effect of long-term frequent exposure to analgesics and the repression of AMH levels in women in the fertile age bracket. Importantly, the present data also show that withdrawal of these compounds lead to an increase in AMH levels, indicating it is a reversible process. These data follow other studies that have indicated that analgesics-exposure during fetal life can have damaging effects on endocrine and reproductive functions, e.g., resulting in congenital cryptorchidism (10,28,29). Future studies should investigate the use of analgesics by females planning to or actively involved in fertility treatment for further understanding and guidance of a potential direct role of analgesics on fertility and subsequent pregnancy.
Potential mechanisms behind AMH-increase
Our clinical data strongly suggest that OTC analgesics repressed AMH production. This could potentially be due to at least three different reasons that are not necessarily independent of each other: (i) Gene/protein level: Repression of AMH transcription and/or translation; (ii) Cellular level: Repression of granulosa cell growth/maturation during follicle development, resulting in fewer GCs to secrete AMH; (iii) Organ level: Repression of follicular recruitment, resulting in fewer antral follicles to produce AMH. Our data from the clinical study cannot distinguish between the chronic long-term effects and the potential acute effects on AMH production of the OTC analgesic-use. Moreover, we observed no associations between the change in AMH to either change in frequency of analgesic-use or change in plasma concentrations of analgesics (APAP and ibuprofen), suggesting that there might not be a direct ‘dose-response curve’ in vivo. However, our preclinical data indicate that there is no direct effect on protein secretion from GCs of ibuprofen and APAP, indicating that these compounds do not repress gene function. In contrast, we found a marked direct effect on DNA synthesis of GCs after exposure to 200 µM APAP, strongly suggesting that APAP repressed cell division. We have previously found an average of ≈40 µM in follicular fluid after standard administration of 1g APAP, indicating that the level of 200 µM is physiologically relevant (not yet published: https://www.researchsquare.com/article/rs-3685511/v1). Moreover, the effect on DNA synthesis is in line with other data that show that APAP blocks pre-implantation embryonic development through inhibition of proliferation mediated by blocking DNA synthesis. Together these indicate that a response at the cellular level (scenario two) might be a reason for the reduced levels of AMH among the females. Whether such repression of cell growth through DNA synthesis inhibition might also reduce follicle recruitment remains to be answered.
Strengths and limitations
A major strength of our study is the translational design, including both in vivo and in vitro results. Data were collected prospectively, using very standardized methods and patients acted as their own controls. The study was also designed specifically to measure levels of sex hormones, i.e., AMH. Noteworthy, due to the standard withdrawal program we decided to measure the AMH level again after two months before the patients started taking analgesics again. Normally, recruiting new follicles takes at least three months which means that we probably could have seen an even higher increase of the AMH level if the withdrawal period and follow-up period had been at least three months.
It is well known that hormonal prevention can lower the AMH level significantly and could have been a major limitation of the study. Fortunately, only one patient decided to make changes in hormonal prevention during the study, which may not have affected the study results.
Due to practical issues, it was not possible to collect fasting blood samples for AMH measurement, which potentially could be a limitation. Moreover, there might be inter-cyclic variation of the AMH levels, even though AMH is considered to be relatively stable.
An important limitation of the data in the present study is that general pain relief when treating the headache by withdrawing the analgesic overuse might also have contributed to increasing AMH levels. The influence of fasting is yet unknown and in real life in fertility clinics the AMH level measurements are not based on blood samples collected from fasting patients. However, the analysis of AMH level was conducted simultaneously avoiding batch effect and was conducted by specialists at the Fertility Department. Included patients were carefully diagnosed with MOH by headache specialists at the DHC, also ensuring a precise history of the long-term use of analgesics. Unfortunately, due to the Covid pandemic and other obstacles, the sample size of the study population was lower than the initially estimated, but factors that could have affected AMH level, remained stable throughout the study period. Nevertheless, it may also be considered as a limitation that seven patients did not answer the PSS and HADS questionnaires after withdrawal, since it is well known that withdrawal contributes to decreasing symptoms of depression, anxiety and stress. On the other hand, the relation of AMH level and these psychological symptoms remain undescribed and may not interfere with our results. Finally, it could be seen as a limitation that the GCs only were exposed to APAP when investigating the cell proliferation. However, this was a conscious choice due to limited resources and the fact that APAP was the most common drug used among our MOH patient population.
Conclusion
In conclusion, our study suggests that long-term and frequent use of OTCs, e.g., APAP and ibuprofen decrease the AMH level in females in the fertile age bracket and affects ovarian endocrinology. The use of analgesics may compromise follicular functional integrity by lowering the number of GCs and thereby reducing the circulating levels of AMH. It is crucial to further investigate the potential negative effects of analgesics on adult female reproductive endocrinology, especially in females undergoing fertility therapy, and to provide advice about the positive effect of withdrawal from analgesics.
Clinical implications
Females with medication-overuse headache had repressed levels of Anti-Müllerian hormone (AMH), a well-established biomarker for fertility. The mechanism behind reduced AMH is likely through disruption of granulosa cell proliferation.
Footnotes
Acknowledgements
A special thanks goes to project nurses Helle Schlie Andresen and Mette Bisgaard and headache nurses Annette Fjeldborg Jonasson, Annette Vangaa Rasmussen, Malene Danø and Maren Østergaard Eriksen for helping with managing the treatment programs. Thanks to Sabrina Lauritzen for contributing to the work with GCs.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L.N.C. has received funding from the TrygFoundation. L.N.C. has also given lectures for Novartis, Allergan and TEVA, and has provided consulting advice to Lundbeck. C.R. has received a 12-month scholarship from Novo Nordisk Foundation. R.H.J. received grants from ATI, Lundbeck Foundation, The Medical Society in Copenhagen, Novo Nordisk Foundation, TrygFoundation and University of Copenhagen. R.H.J. has also conducted clinical trials for ATI, Electrocore, and Eli-Lilly; given lectures for Allergan, ATI, Merck, Novartis, Pfizer, and TEVA; has been trustee in International Headache Society, Director of Lifting Thetherden, and current Director of the Danish Headache Center. D.M.K. has received a grant from the Lundbeck Foundation. B.S.N., M.R.P., C.H.L., A.M.K., C.S.J.W., J.V., L.B.J., S.G.K., S.Z. have nothing to declare. The authors declare that all research conducted, its contents and results are free and independent from the funders.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the Lundbeck Foundation (R324-2019-1881). Data and program codes will not be available. The analysis plan was preregistered at ClinicalTrials.gov.
