Abstract
Objective
The objective of this study was to describe and discuss patterns of migraine medication use in the entire Norwegian population.
Methods
In this nationwide, observational study, all individuals with a migraine-related prescription between 2010 and 2020 were identified using the Norwegian Prescription Database. The outcomes of interest were the incidence and 1-year prevalence of migraine medication users, as well as individuals with triptan overuse. Patterns of medication use were statistically compared between women and men adjusted for age, year of treatment start, comorbidities and county of residence calculating adjusted odds ratios (aOR) with 95% confidence intervals (CI).
Results
We identified 327,904 migraine medication users. The incidence ranged from 0.39% to 0.46%, and the 1-year prevalence increased from 1.99% to 2.99%. Preventive use increased >50% during the study period. Preventives were significantly more often prescribed to women than to men (39.72% vs. 33.75%; aOR 1.41, 95% CI 1.38 to 1.44). Triptan overuse was significantly more common among women, but women with overuse were more often using preventives, as compared to men (56.64% vs 52.69%; aOR = 1.43, 95% CI 1.37 to 1.49).
Conclusion
The prevalence of medically treated migraine is low. Overuse of triptans is frequent, especially among women. Clinicians should be encouraged to try out different triptans, recognize triptan overuse, and prescribe preventives when indicated.
Background
While the global 1-year prevalence of migraine is estimated to be 15% (1), the prevalence of migraine in Norway may be somewhat lower, with population-based health survey estimates ranging from 11.2% to 13.2% (2). Worldwide, and in Norway, migraine is second among causes of disability overall, and the leading cause of disability among young women (1,3,4) —necessitating prompt and effective treatment.
Numerous pharmacological treatment options have been established as effective in managing migraine (5). Over-the-counter analgesics and triptans can be effective in aborting attacks (6), and drugs from a wide spectrum of pharmacologic classes can provide effective prophylaxis: reducing attack frequency, severity and duration, and improving quality of life (5,7,8).
National treatment guidelines in Norway are in line with the international literature (9). The guidelines recommend treating infrequent episodic migraine with over-the-counter analgesics such as non-steroidal anti-inflammatory drugs or paracetamol, and prescribing triptans if the former fails. Triptans are only available through physician prescription in Norway. Preventive treatment is recommended if acute treatment is insufficient or has unacceptable side-effects, if attacks are frequent (more than three monthly attacks), or attacks and auras are long-lasting or highly debilitating. The suggested first-line preventives are metoprolol, propranolol, candesartan, topiramate and amitriptyline. Calcitonin gene related peptide (CGRP) pathway monoclonal antibodies and botulinum toxin A are reimbursed only for individuals with chronic migraine (CM) that have failed other preventives from three or more different pharmacological classes.
Despite the many therapeutical options, we know that many individuals with migraine receive poor medical care (10), and that triptan and preventive treatment coverage is low (11,12). Moreover, we have limited knowledge of how the different available treatment options are utilized in a real-world setting in Norway. There is a concern that there are unjustified differences in the management of migraine among women and men (13,14). Also, headache experts in Norway have stated that more of those with frequent attacks should use preventive medication (15), and that the knowledge of treatment of medication overuse headache (MOH) should be enhanced (16).
The aim of this paper is to use prescription data to analyze the development over time in use of both acute and preventive medications across the entire Norwegian population and compare different sex and age groups. The results will be discussed in the light of established national and international treatment guidelines for migraine, followed by a discussion on how management strategies for migraine can be optimized.
Methods
Study design, setting and data sources
We conducted a nationwide, registry-based, observational study using data from the Norwegian Prescription Database from 01 January 2008 to 31 December 2020. We used the period between 2008 and 2010 as a wash-out period and studied incident and prevalent migraine medication users over an 11-year period, from 2010 to 2020. The Norwegian Prescription Database contains detailed information on all prescriptions filled in pharmacies nationwide since 2004. All pharmacies in Norway register prescriptions electronically. Each prescription includes the date of redemption, county of residence, year and month of birth and death, sex, reimbursement code, pack size, drug strength, drug name, defined daily dose (DDD) as defined by the World Health Organization Collaborating Centre for Drug Statistics Methodology, and the Anatomical Therapeutical Chemical (ATC) classification code (17). Over-the-counter drugs and medications used in hospitals and nursing homes are not registered in the prescription database.
The Norwegian Health Care System provides universal health and social security coverage, and public health insurance is automatic for all residents (18). About 10% of the population had private insurance in 2016, which mainly offers quicker access to outpatient services. Primary headache disorders are usually handled by primary care providers, while a smaller proportion of patients are referred to neurologists. Most migraine drugs are reimbursed with a cap on out-of-pocket contributions, and triptans are only available through physician prescription.
Study population
We identified eligible migraine medication users as individuals who, between 01 January 2010 and 31 December 2020, fulfilled at least one of two conditions: 1) had at least one prescription reimbursed for migraine (code G43 according to the 10th revision of International Statistical Classification of Diseases (ICD-10) (19); or code N89 according to The International Classification of Primary Care (ICPC-2) (20)); or 2) had at least two prescriptions of triptans (ATC code N02CC), regardless of reimbursement code. The latter condition was included because compliance with the registration of reimbursement codes in the early years of the prescription database was incomplete. Since triptans are used almost exclusively for the treatment of migraine in Norway, triptan prescriptions can be used to identify migraine patients with high specificity, but presumably with lower sensitivity as there are many migraine sufferers who neither use a triptan nor get a prescription for a migraine preventive (11). To further ensure that the study population was composed exclusively of migraine patients, we excluded patients who had at any time had a drug reimbursed for cluster headache (ICD-10 code G44.0-G44.2 or ICPC-2 code N90), and patients who received only one triptan prescription and never had a migraine reimbursement code during the study period. We also excluded patients <2 or >99 years old at the time of their first prescription. To increase the likelihood that we captured individuals’ first migraine related prescriptions, we omitted all patients who had received a migraine related medication in the two years prior to the start of the study period (the wash-out period) from the incidence calculations (Figure 1).
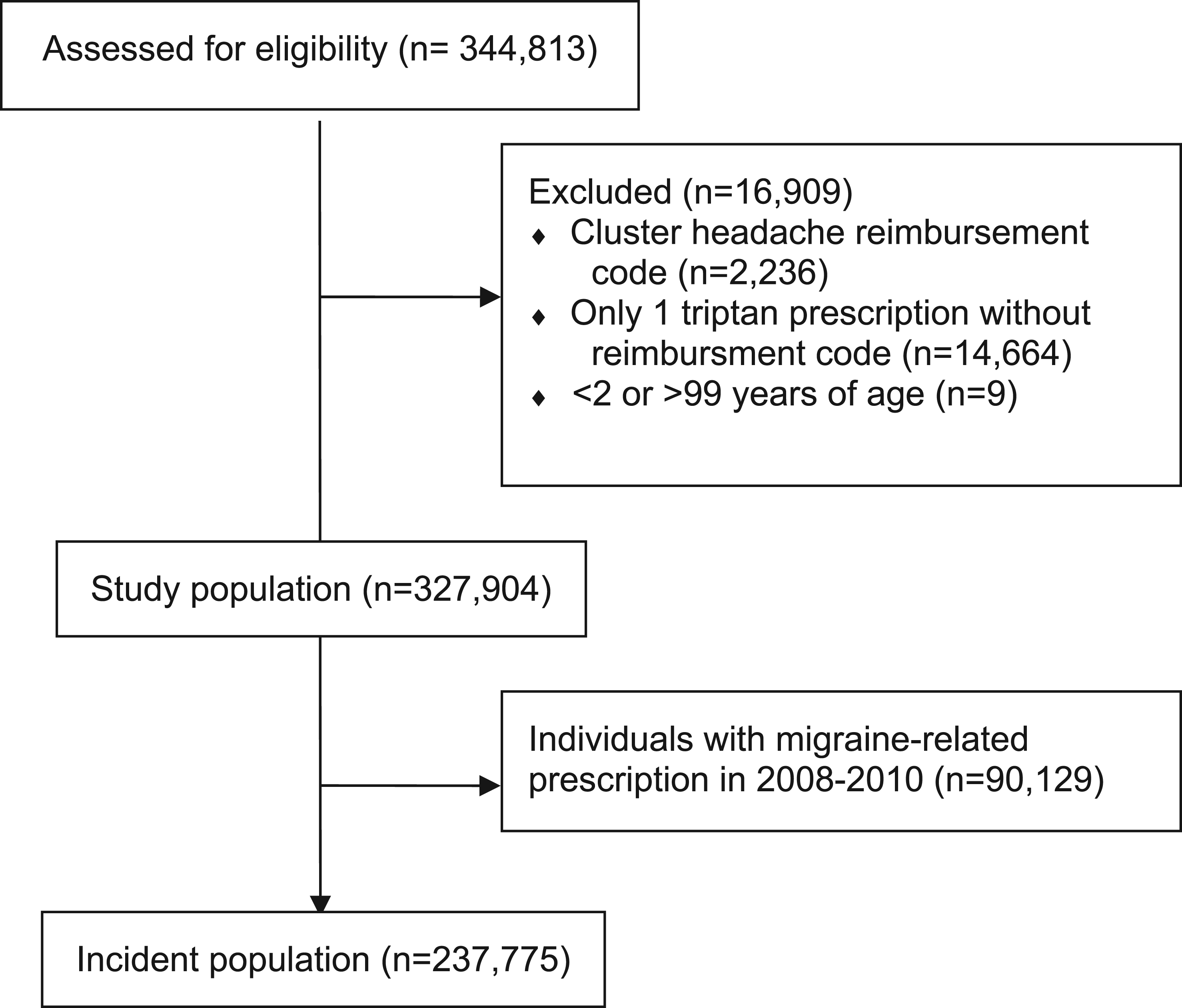
Flow chart of study population.
Outcomes
The incidence of migraine medication users was defined as the number of new individuals, as defined above, each year. Incidence was calculated for the population as a whole and for the 5-year age groups for women and men respectively. The incidence rate expressed as a proportion (%) of the population was calculated by dividing the number of new cases within a year by the total Norwegian population at the beginning of the year. Statistics Norway provided estimates of the size of the Norwegian population as a whole and by sex- and age-specific groups.
The 1-year prevalence of migraine medication users was defined as all individuals fulfilling criteria for migraine medication use as defined above, during a given year. Prevalence rates were also calculated as proportions of the Norwegian population.
We defined patients in remission as previous migraine medication users with no migraine related treatment (neither acute medication, preventive medications, nor any prescriptions with a migraine reimbursement code) for two or more consecutive years. The rationale for using two years as a cut-off was to account for patients who might have had a large quantum prescription at the end of one year that lasted the whole next year.
The following acute medications were identified based on their ATC code: sumatriptan, naratriptan, zolmitriptan, rizatriptan, almotriptan, eletriptan and frovatriptan (N02CC01-7). Preventives were pooled according to pharmacological class and the following were included: betablockers (propranolol C07AA05 and metoprolol C07AB02), lisinopril (C09AA03), candesartan (C09CA06), clonidine (N02CX02), CGRP-antibodies (fremanezumab, galcanezumab, erenumab, N02CD01-3), topiramate (N03AX11), amitriptyline (N06AA09), and botulinum toxin type A (Botox, M03AX01). Use of preventives were recorded regardless of their indication for prescription as long as the patient fulfilled the eligibility criteria for inclusion in the study (see above). The rationale for this decision was that practices and reimbursement regulations have changed over time. Thus, we captured prescriptions that require individual application for reimbursement (candesartan) or are not reimbursed (lisinopril). In addition, we captured the prescriptions with double indications (e.g., betablockers for both hypertension and migraine). Clonidine was used as a migraine preventive in Norway until it was withdrawn from the market in 2019. Lisinopril is listed as an option for prevention of migraine in Norwegian guidelines but is not reimbursed. The latter two are rarely or never used in other European countries. Calcium channel blockers (verapamil, flunarizine) and antidepressants other than amitriptyline (e.g., fluoxetine, venlafaxine) are very rarely used for migraine prevention in Norway and were therefore not included in this study.
To evaluate the use of different migraine-related medications we calculated the proportion of the Norwegian population using different triptans, and the proportion of the Norwegian population using the different pharmacologic classes of preventives. We also calculated the number of days from the date of a first triptan prescription to the date of a first prescription of a preventive, and the proportion of individuals receiving a preventive within the first year after a triptan prescription. We calculated proportions for the total population and for women and men separately.
To estimate the prevalence CM and medication overuse headache (MOH) we defined three different conditions based on monthly triptan use: 1) ≥ 8 DDD of any triptan in three consecutive months as a proxy for possible CM (assuming administration of only one triptan dose per day, indicating eight migraine days as per ICHD-3 criteria). 2) ≥ 16 DDD of any triptan in three consecutive months as a proxy for probable CM and/or probable MOH (assuming administration of one or two triptan doses on at least eight migraine days). 3) ≥ 30 DDD of any triptan in three consecutive months as a proxy for definite CM and MOH (assuming a minimum of 15 monthly migraine days requiring one or two triptan doses, thus fulfilling ICHD-3 criteria for both MOH and CM). It is not possible to differentiate between MOH and CM solely based on triptan use as an indicator of a migraine and/or headache days.
Covariates
Covariates included age, sex, county of residence, year of treatment start and selected comorbidities. Age, captured in the index year (first year of registration as a migraine medication user) was categorized in 5-year groups. Comorbidities were assessed through identification of reimbursement codes (ICD-10 or ICPC-2) from a list of predefined diseases (acute myocardial infarction, congestive heart failure, hypertension, heart rhythm disorders, renal disease, depression, anxiety disorder, use of nicotine replacement products, epilepsy, mood disorders, type 2 diabetes, and asthma). Covariates were captured in the index year, i.e., first year of registration as a migraine medication user according to the inclusion criteria. Age, prescribing year, geographical region and comorbidities were considered potential confounders when comparing migraine medication use in women and men as they are associated with both sex and migraine medication use. Age and comorbidities could influence choice of antimigraine therapy (21). Year of treatment start was included to account for disease duration and changes in prescribing patterns over different time periods (22). County of residence was included to adjust for the impact of sociodemographic differences and local prescription patterns across the country.
Statistical analyses
All analyses were performed with STATA version 17. Categorical variables were presented as number and percentages, and continuous variables as means with standard deviations (SD) or medians with inter-quartile ranges (IQR) depending on distribution. The normality assumption was assessed based on visual inspection of histograms. Sex differences for dichotomous outcomes were compared using logistic regression adjusted for the covariates. Adjusted odds ratios (aOR) with 95% confidence intervals (CI) were calculated. Sex differences for the continuous outcome, i.e., time in days from first triptan prescription to first preventive prescription, was compared using linear regression adjusted for the covariates. The significance threshold was set to 0.05. No imputations were made as there were no missing data.
Results
Study population
The total population of Norway was 4,920,305 at the end of 2010, rising to 5,391,369 at the end of 2020 (Online Supplementary Figure 1). During the study period 327,904 individuals fulfilled the criteria for migraine medication users, and they filled 54 million prescriptions in total. Among these 90,129 patients had a migraine-related prescription between 2008 and 2010, resulting in an incident population of 237,775 patients (Figure 1). Table 1 describes the study population.
Patient characteristics.

Characteristics of men, women and total population summarizing all covariates except county of residence. Comorbidities are calculated based on medication reimbursement diagnostic codes. E.g. prescription of antihypertensives with an ICD-10 or ICPC-2 reimbursement code of hypertension are used to estimate the prevalence of hypertension. Patient characteristics were captured in the index year, i.e., first year of registration as a migraine medication user according to the inclusion criteria. See Online Supplementary Table 12 for complete background data.
SD = standard deviation.
Incidence, prevalence, and remission of migraine medication users
Over the 11-year study period the incidence of migraine medication use ranged from 0.39% to 0.46% (n = 20,458 to 23,172), the rate being relatively stable over the years. Age- and sex-specific incidence rates are shown in Figure 2. The 1-year prevalence of migraine medication use increased from 1.99% (n = 98,118) to 2.99% (n = 161,046) during the study period (Table 2), for women from 3.18% (78,218) to 4.84% (129,414), and for men from 0.81% (n = 19,900) to 1.16% (n = 31,632; Online Supplementary Table 1). During the study period, 36.69% (n = 120,323) of migraine medication users experienced disease remission (Table 2). Of those with a remission period, 24.24% (n = 29,166) were subsequently prescribed migraine medication at another time, indicating a relapse of their migraine (not shown in table).

Incidence rates (%) by age and sex.
Incidence, prevalence and remission of migraine medication users.
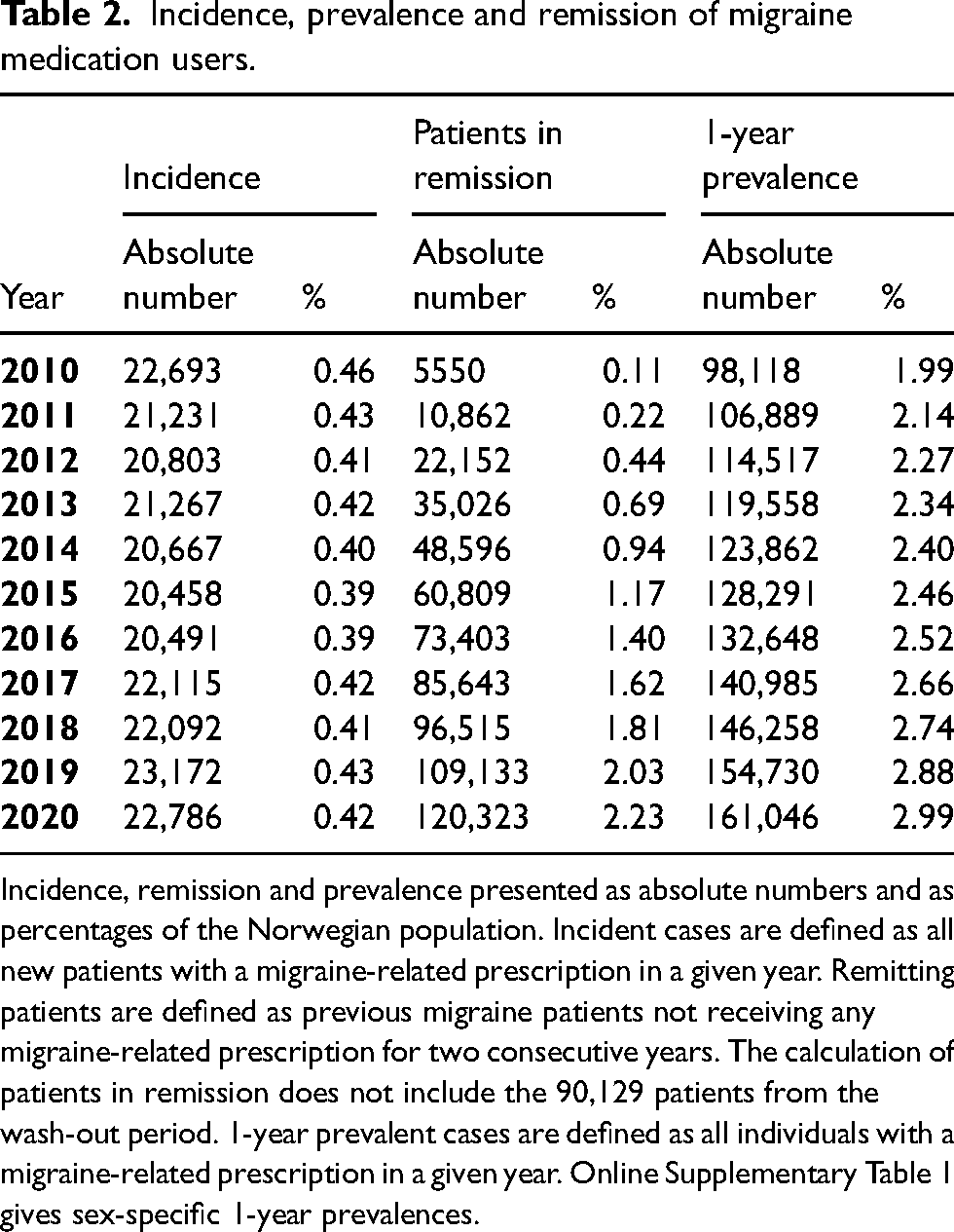
Incidence, remission and prevalence presented as absolute numbers and as percentages of the Norwegian population. Incident cases are defined as all new patients with a migraine-related prescription in a given year. Remitting patients are defined as previous migraine patients not receiving any migraine-related prescription for two consecutive years. The calculation of patients in remission does not include the 90,129 patients from the wash-out period. 1-year prevalent cases are defined as all individuals with a migraine-related prescription in a given year. Online Supplementary Table 1 gives sex-specific 1-year prevalences.
Use of preventive and acute medications
Figure 3 shows the 1-year prevalence of use of triptans, preventives and combination of triptans and preventives throughout the study period. Among all migraine medication users, use of preventives ranged from 23.52% to 31.78% (n = 23,082 to 51,181) throughout the study period. Of the whole Norwegian population, there was an 53% increase in use of preventives (either alone or in combination with triptans) from 0.68% to 1.04% during the period (Figure 3). Using a migraine prevalence in Norway was between 11.2% to 13.2% (2), and there were 51,181 preventive users in 2020, the overall preventive coverage was estimated at 7.1% to 8.3%. Likewise, 122,664 triptan users in 2020 translates to an overall triptan coverage in persons with migraine between 16.9% and 20.0%. Significantly higher proportions of women were prescribed preventives, as compared to men (Table 3, Online Supplementary Tables 2 and 3).

Use of triptans and migraine preventive drugs.
Results of regression analyses comparing men and women.

Results of regression analyses comparing men and women. Absolute numbers and percentages relative to the sex are provided. Odds ratios above 1 indicate higher probability among women. Analyses are adjusted for patient age, county of residence at treatment start, year of treatment start and number of relevant comorbidities.
OR = odds ratio; CI = confidence interval; DDD = defined daily doses; SD = standard deviation.
The 1-year prevalence of triptan users increased from 1.66% to 2.28% (Figure 3). The most prescribed triptan was sumatriptan, followed by rizatriptan and zolmitriptan (Online Supplementary Table 4). Only 3.11% of individuals with a migraine related prescription (n = 10,189) were prescribed three or more different triptans in the 360-day period following their first prescription of a migraine-related medication. Significantly more women than men were prescribed three or more triptans in the first year after first prescription (Table 3, Online Supplementary Tables 5 and 6).
Betablockers were the most used preventive followed by candesartan and amitriptyline (Online Supplementary Table 7). Candesartan had the largest increase in the number of prescriptions throughout the study period. Figure 4 shows the typical preventive drug journey for individuals with migraine illustrated as a Sankey diagram. Betablockers, candesartan and amitriptyline were the most common as the first and second drug, topiramate was the most common as the third, and CGRP-antibodies were prescribed as the fourth.

Sankey diagram illustrating patient preventive treatment journeys.
The mean number of days from first prescription of a triptan to the first prescription of a preventive was 1235 days (SD 1196.77, IQR 240 to 1978, Online Supplementary Table 8). This time was significantly longer for women compared to men (64.14 days, 95% CI 49.83 to 78.44). However, significantly more women than men were prescribed a preventive in the first year after their first triptan prescription (Table 3).
Chronic migraine and medication overuse
During the whole study period, 14.69% (n = 48,179) of migraine medication users had at least one 3-month period with monthly triptan use ≥8 DDD; 10.30% (n = 33,790) had three consecutive months ≥16 DDD; and 5.57% (n = 14,989) had three consecutive months ≥30 DDD. The proportions of triptan overuse remained stable throughout the study period (Online Supplementary Tables 9 and 10). Triptan overuse was significantly more common among women than among men at all three cut-offs (Table 3).
Among those with a triptan use indicative of probable CM and/or MOH (≥16 DDD triptans monthly in three consecutive months) 55.92% received a preventive medication the same year (Online Supplementary Table 11). There were significantly more women with probable MOH or probable CM that received a preventive compared to men (Table 3).
Discussion
We estimated the incidence and prevalence of medically treated migraine across the entire Norwegian population over an 11-year period—one of the largest such cohorts to date. The incidence remained relatively stable throughout the study period ranging from 0.39% to 0.46%, while the 1-year prevalence increased from 1.99% in 2010 to 2.99% in 2020. The highest incidence was found among women aged 15–19, at 1.37%, more than three times that of the general population. The findings are comparable to what has been reported in other large cohorts (12,23). In a German study that identified 243,471 individuals with migraine through a Company Sickness Fund Database, the incidence and prevalence of migraine medication users was 0.267% to 0.589%, and 2.89% to 3.98%, respectively (23). In an Italian cohort of more than 12 million persons captured from a drug reimbursement prescription database the overall prevalence of triptan users in 2013 was 0.8% (12).
The prevalence of migraine medication use is much lower than the estimates of migraine prevalence from Norwegian population-based health surveys relying on self-reporting of migraine symptoms, at 11.2% to 13.2% (2). This discrepancy likely reflects that most of those with mild or infrequent migraine treat themselves with over-the-counter analgesics (24) and are often not seen by a healthcare professional (10). However, the discrepancy also indicates an underdiagnosis or undertreatment of the migraine population. We also identified that up to 2.23% of the population experienced migraine remission. However, these figures must be interpreted with caution as there may be other reasons than remissions for discontinuation of migraine medicines, such as development of contraindications, lack of treatment effect, or pregnancies.
In our cohort, nearly 40% of all migraine medication users received preventive treatment at some point, which is higher than has been observed in other comparable studies. In the Italian cohort, 19.1% of individuals using ≥4 triptan doses per month also used oral preventives (12). In the German study 29.1% of patients received at least one preventive (23). In the American Prevalence and Prevention Study 12.4% of those with migraine reported using migraine preventive medications, and an additional 17.2% were using medications with possible antimigraine effects (11). The higher proportion of preventive use observed in our study could be explained by an increased media awareness on correct treatment of migraine in Norway over the last one to two decades, including recommendations to use preventives. Indeed, this is reflected in the marked increase in preventive use throughout the study period. Essentially all preventives show an increase in use throughout the study period (Online Supplementary Table 7). Nevertheless, the overall preventive coverage remains too low. We estimated that just 8% of the entire migraine population were treated with preventives, yet guidelines suggest that more than 30% of those with migraine qualify for preventive treatment (25,26).
Triptan coverage among individuals with migraine has in other countries been estimated to be around 20% (11,23,27,28), similar to the findings in our study (between 16.9% and 20.0%). In Denmark, prevalence of triptan use was lower at 12.3%—the low coverage was deemed to likely be caused by unsatisfactory efficacy and/or unpleasant side effects (29). We found sumatriptan to be the most prescribed triptan, followed by rizatriptan and zolmitriptan. The same pattern was observed in Germany (23). Different triptans can have significant interindividual responsiveness and guidelines emphasize the importance of trialing different triptans (30). Yet only 11.98% of those starting a triptan were prescribed a second triptan, and only 2.59% were prescribed three or more triptans within the first year after being diagnosed with migraine. Network meta-analyses have suggested that eletriptan is significantly superior to several other triptans (31) yet it is rarely used in our population. These findings emphasize that clinicians should become familiar with the different types of triptans and evaluate other triptans as soon as the first one trialed is established as non-effective. Indeed, a Danish study form 2022 show that there is a low adherence to the therapeutic guidelines for the acute treatment of migraine (32). Moreover, the updated guidelines of the American Headache Society emphasize that triptans should be a first-line therapy for acute treatment (33). Considering the relatively low triptan coverage among individuals with migraine in Norway (16.9% to 20.0%), we argue that national guidelines should more clearly emphasize the need for early introduction of triptans. It is also interesting to note that there is a significant use of triptans among those <12 and those >65 years of age (Online Supplementary Table 2). Both these groups can be difficult to manage. Prescription of triptans to children <12 years of age, apart from sumatriptan nasal spray, is off-label, and there is limited evidence for efficacy of prophylactics in children. Also, it is recommended that triptans are used with caution in individuals with cardiovascular disease, which becomes more prevalent with increasing age (21,34). These drawbacks emphasize the need for new therapeutic options.
Among migraine medication users there was a substantial overuse of triptans. Approximately 10% had triptan use indicating probable CM and/or MOH (≥16 DDD triptans for three consecutive months), and nearly 5% had triptan use indicating definite CM and MOH (≥30 DDD triptans for three consecutive months). Similarly, in the Italian study, 10% had CM as indicated by triptan use (12). The global prevalence of chronic migraine has been estimated at 1.4 to 2.2% (35), while the prevalence of medication overuse headache has been estimated at around 2% in several population-based surveys (36,37). Our overall population estimate of probable CM and/or MOH (≥16 DDD triptans for three consecutive months) was 0.65% (Online Supplementary Table 9) which is somewhat lower than published estimates of MOH and CM prevalence. However, 0.65% is a conservative estimate as many individuals with MOH are not (over)using triptans, but rather non-steroidal anti-inflammatory drugs and/or paracetamol, which we could not assess from the prescription registry. Concerningly, only around half of those with probable or definitive CM in our population received a preventive in the first year after triptan overuse. Proper management of frequent acute medication use and instituting preventive treatment of high-frequency migraine are essential to avoid MOH and transformation to CM (38,39), especially viewed in light of the limited knowledge of management of MOH among Norwegian general practitioners (16).
We also identified important differences in prescription patterns between women and men. Overall, women had higher coverage of preventives, similar to what has been observed in other cohorts (11,23,27). Women were also more often prescribed multiple triptans in the first year after diagnosis. This discrepancy could reflection differences in health-care seeking behavior between men and women (14). Men should therefore be encouraged to see healthcare professionals for headache to optimize acute and preventive treatment. Pharmacokinetics could also explain some of the observed sex differences. A systematic review found that women, despite using higher and more frequent doses of triptans, did not have higher response rates (40). This could, at least in part, explain women's higher prevalence of medication overuse and CM, in spite of higher preventive coverage. In general, women have a much higher level of migraine related disability (4,14,41). Therefore, early preventive treatment must be particularly encouraged among women to prevent and avoid medication overuse. Next, there are differences in comorbidities between the sexes that could influence the patterns of migraine medication use. Notably, women more frequently suffer from depression, and comorbid treatment with antidepressants could in part contribute to the higher prevalence of migraine preventive users among women. On the other hand, men have higher prevalence of cardiovascular diseases. This could in part contribute to the lower use of triptans among men. Future studies should explore in-depth how the relationship between sex and comorbidities influence treatment patterns. Finally, the use of contraceptives could have influenced the patterns of medication use seen in our material. Contraceptives can sometimes have beneficial effects on menstrual migraine, thus impacting use of other preventives and triptans among women (14). Future studies should investigate factors such as practitioner habits, socio-economic status, comorbidities and concomitant medication use (especially contraceptives and hormonal replacement therapy) that may further explain the differences in prescription patterns between women and men.
The preventive prescription patterns identified in this study are in line with national and international guidelines. Propranolol, candesartan, and amitriptyline are the recommended first-line therapies, which is reflected in our findings (Figure 4). The first clinical trials establishing candesartan as a migraine preventive were conducted in Norway, likely explaining the relatively frequent use. In the European Consensus Statement endorsed by the Danish Headache Society, European Headache Federation and European Academy of Neurology, the recommended first line prophylactics are beta-blockers, topiramate, and candesartan (5). The American Academy of Neurology, supported by the American Headache Society, recommends divalproex sodium, sodium valproate, topiramate, metoprolol, propranolol and timolol as preventive treatments (42). Recently candesartan has also been included on their list of treatments with established efficacy (33). In Norway CGRP-antibodies were introduced in 2018, and are only reimbursed for those with chronic migraine that have tried three prophylactics from three different medication classes, thus explaining the limited use in this material. 3.94% of migraine medication users used CGRP-antibodies in 2020. Concurrently there was a slight decrease in overuse of triptans from 2019–2020 (Online Supplementary Table 9) which could be an impact of treating CM and MOH patients with CGRP-antibodies. Future studies with a longer observation period after the introduction of CGRP-antibodies could possibly better demonstrate such an impact on triptan use. Finally, a recent study of a similar data material identified simvastatin to have an effectiveness in line with CGRP-antibodies, possibly broadening the spectrum of options to combat migraine (43).
Strengths and limitations
This study has important strengths, the main being the nationwide coverage, capturing of the real-world prescription patterns for all Norwegian citizens without attrition or selection bias. The study also has a long observation period enabling identification of important temporal usage patterns for acute and preventive medications.
Some limitations must be considered when interpreting the results of this study. First, failure to adhere to the reimbursement codes by healthcare professionals could lead to underestimation of incidence and prevalence. Second, our estimates of CM and MOH are based solely on triptan use, hence the findings must be interpreted with due caution. Third, individuals diagnosed with both cluster headache and migraine were excluded. Yet, migraine and cluster headache may be comorbid, and individuals with both conditions that were not prescribed any drugs with cluster headache indication would not be detected in this study. This small subgroup may have contributed to some of the MOH cases as triptans are often overused during bouts or in chronic cases. Fourth, individuals with migraine prescriptions between 2008–2010 were not considered in the incident population, yet some individuals were likely diagnosed prior to 2008 without having prescriptions in this period and thus erroneously recorded as an incident case if they were prescribed a migraine-related drug during the observation period—inflating the incidence in the first period of the study. Finally, there may be unmeasured confounders that influence the observed differences in prescription patterns between women and men, still, the aim of this paper is to describe associations rather than causality.
Conclusion
In this nationwide, registry-based, observational study we found a relatively low incidence and 1-year prevalence of both triptan and preventive migraine medication users, suggesting that a large proportion of those with migraine do not seek medical care. The choice of medications was in accordance with national and international guidelines. Encouragingly, the use of preventives increased markedly during the study period but is still much lower than recommended. Overuse of triptans was frequent, especially among women, and nearly half of those with overuse were not on a preventive treatment. Clinicians should be encouraged to trial different triptans, recognize triptan and medication overuse, and prescribe preventives to those who need them. Men should be encouraged to seek health care professionals for headaches. Finally, proper treatment of young women with migraine must be prioritized, especially since women significantly more often overuse triptans and in general have a higher migraine disability burden. The findings in this study support several action points for clinicians, policymakers and stakeholders.
The incidence and prevalence of medically treated migraine in Norway is low. The use of preventives increased ≥ 50% from 2010 to 2020 but is still lower than recommended. Triptan overuse is common, especially among women. Moreover, most of triptan overuses do not use preventives. Clinicians should be encouraged to trial different triptans, recognize medication overuse, and prescribe preventives when indicated.
Supplemental Material
sj-docx-1-cep-10.1177_03331024241268212 - Supplemental material for Patterns of migraine medication use in Norway: A nationwide registry-based observational study
Supplemental material, sj-docx-1-cep-10.1177_03331024241268212 for Patterns of migraine medication use in Norway: A nationwide registry-based observational study by Anker Stubberud, Solveig Borkenhagen, Francisco Oteiza, Aud Nome Dueland, Christoffer Bugge, Erik Magnus Sæther, Erling Tronvik, Lars Jacob Stovner and Marte-Helene Bjørk in Cephalalgia
Footnotes
Authors contributions
AS: data analysis, data interpretation, writing and revising the manuscript; SB, FO: data acquisition, data analysis, data interpretation, and revision of the manuscript; AND, CB, EMS: research design, data acquisition, data interpretation, and revision of the manuscript; ET, LJS: study conception, research design, data interpretation, and revision of the manuscript; MHB: study conception, research design, data acquisition, data interpretation, and revision of the manuscript. All authors reviewed the final version.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AS has received speaker honoraria from Teva.
SB is affiliated with Oslo Economics and has completed consultancy assignments for several pharmaceutical companies in recent years. FO is affiliated with Oslo Economics and has completed consultancy assignments for several pharmaceutical companies in recent years. AND has received speaker honoraria and/or consultancy honoraria from AbbVie, Lilly, Lundbeck, Novartis, Pfizer, Roche, Teva. CB is affiliated with Oslo Economics and has completed consultancy assignments for several pharmaceutical companies in recent years. EMS is affiliated with Oslo Economics and has completed consultancy assignments for several pharmaceutical companies in recent years. ET reports personal fees from lectures/advisory boards: Novartis, Eli Lilly, Abbvie, TEVA, Roche, Lundbeck, Pfizer, Biogen. Consultant for and owner of stocks and IP in Man & Science. Stocks and IP in Nordic Brain Tech. Stocks in Keimon Medical. Non-personal research funding from several sources, including EU, Norwegian Research Council, Dam foundation, KlinBeForsk. Commissioned research (non-personal): Lundbeck, Pfizer. LJS reports no disclosures relevant to the manuscript. MHB has received speaker honoraria and/or consultancy honoraria from Teva, Eisai, AbbVie, Pfizer, Novartis, Lundbeck, Angelini Pharma, Jazz pharmaceuticals, and Lilly during the last five years some of which are marked authorization holders of acute and/or preventive migraine drugs. She is affiliated with university of Bergen that has received fees from the marked authorization holders of valproate for a post authorization study of valproate.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was funded by The Research Council of Norway through NorHead Norwegian Centre for Headache Research. The data analysis was partially funded by Norges Forskningsråd, Novartis.
Ethical approval
Ethical approval and a waiver of informed consent was granted by the Regional Committees for Medical and Health Research Ethics (242126).
Availability of data and materials
The datafiles used in this study are not openly available for data protection reasons. However, similar data material is available upon application to the relevant regulatory authorities and data holders.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
