Abstract
Background
Data on drug-induced reversible cerebral vasoconstriction syndrome (RCVS) are scarce. We aimed to describe RCVS characteristics with drugs previously identified as associated with RCVS and investigate potential signals related to other drugs.
Methods
VigiBase® was queried for all reports of RCVS until 31 May 2023. A descriptive study was performed on reports concerning drug classes known to precipitate RCVS. To identify new drugs, a disproportionality analysis was conducted.
Results
In total, 560 reports were included. RCVS occurred in patients aged between 45–64 years (40%) and 18–44 years (35%), mainly in females (72.5%). Drugs were antidepressants (38.4%), triptans (6.4%), nasal decongestants (3.7%) and immunosupressants (8.7%). In 50 cases, antidepressants were in association with drugs known to precipitate RCVS. The median time to onset was 195 days for antidepressants and much shorter (1–10 days) for triptans, nasal decongestants and immunosuppressants. The outcome was favorable in 87% of cases, and fatal in 4.4%. We found a disproportionality signal with 14 drugs: glucocorticoids, bupropion, varenicline, mycophenolic acid, aripiprazole, trazodone, monoclonal antibodies (erenumab, ustekinumab and tocilizumab), leuprorelin and anastrozole.
Conclusions
The present study confirms the role of vasoconstrictors in the onset of RCVS, particularly when used in combination and found potential signals, which may help clinicians envisage an iatrogenic etiology of RCVS.
Introduction
For nearly 30 years, reversible cerebral vasoconstriction syndrome (RCVS) has been referred to by various names (e.g. Call–Fleming syndrome, benign central nervous system angiopathy, benign acute cerebral angiitis, benign isolated cerebral vasculitis, migraine angiitis, migrainous vasospasm, headache in thunderbolt with vasospasm). The name RCVS was proposed in 2007 to group all similar cases reported in the observations of small prospective series (1,2) within a single nosological framework.
RCVS is a syndrome combining sudden onset thunderclap headaches, sometimes accompanied by focal neurological deficits and/or comitial seizures. Radiologically, the disease is characterized by multifocal vasoconstriction of the cerebral arteries that can lead to multiple infarctions and usually resolves spontaneously within three months. Clinically, headache is the most common and earliest symptom of RCVS (3). It is defined by the International Headache Society as a severe headache, of brutal installation with maximum intensity in less than one minute (4). The headache attacks are typically repeated in salvo over 1–4 weeks, with or without painful background intercurrent. Other types of headaches (moderate, progressive) can be observed but without sudden onset, and the RCVS hypothesis is called into question. Only retrospectively will the diagnosis will be definitively confirmed, by highlighting the reversibility of arterial lesions in one to three months. The most common complications are subarachnoid or parenchymal bleeding or infarction (5).
Although there are no validated criteria for RCVS diagnosis, a combination of patient history and non-invasive imaging will suffice to rule out other etiologies. Moreover, there exists no proven or established therapy for RCVS. Patient management is based first of all on discontinuation of vasoactive substances. Symptomatic treatments such as analgesics for headaches, antiepileptics for possible crises and monitoring of blood pressure with treatment in the event of hypertensive crises have been proposed. Calcium channel blockers such as nimodipine are most often used aiming to reduce vasospasms. Total duration of treatment ranges from 4–12 weeks (3). Corticosteroids are not recommended and appear to be associated with a worse prognosis (6).
In the absence of epidemiological studies, the exact incidence of RCVS in the general population is still unknown. Based on the few currently available population-wide data, the estimated annual age-standardized incidence of cases leading to hospitalization in US adults is 4.1 (95% confidence interval (CI) = 3.5–4.8) cases per million in women versus 1.2 (95% CI = 0.9–1.6) cases per million in men, with overall incidence of 2.7 (95% CI = 0.9–1.6) cases per million (7). In an Italian study, it was estimated at one in 900 in women in postpartum (8). Although RCVS affects patients with a mean age range of 42–45 years, clinicians have also seen it in children and adolescents (9). In adults, females are more commonly affected, with female-to-male ratios ranging from 2:1 to 10:1 depending on the case series (7,10–12). A review of pediatric RCVS found that most cases affected males (13).
The pathophysiology hypothesis of RCVS involves acute and transient dysregulation in the control of brain vessel tone, which results in multifocal vasoconstriction and segmental dilatation. This would involve sympathetic hypertonia, endothelial dysfunction and oxidative stress. Endogenous (physical or emotional stress) and/or exogenous (toxic or drug-induced) sympathetic stimulation could trigger vasoconstriction of small arteries and bilaterally activate the protective trigemino-vascular reflexes against ischemia (3,14).
RCVS is probably multifactorial, involving genetic susceptibility and multiple environmental factors or medical conditions (pregnancy, peripartum period, trauma, head and neck surgery, transient hypertensive crises, catecholamine-secreting tumors, hemolysis elevated liver enzymes and low platelet count (i.e. HELLP) syndrome, antiphospholipid antibody syndrome, thrombotic thrombocytopenic purpura) (9,14). A sympathetic endogenous stimulus is reported in 80% of patients, such as sexual activity, physical exertion, defecation, urination, cough, sneezing, Valsalva maneuver, bath or shower and sudden head movement (3,15). A history of migraine is also confirmed to be a relevant factor, with a prevalence of 9.8–42% reported in the literature (16,17). Patients with proven RCVS frequently have a history of migraine. Acute headaches as a result of RCVS are sometimes mistaken for bad migraine attacks (18,19). However, migraine patients who have previously experienced RCVS recognized that thunderclap headaches are totally different from a migraine attack (5).
It may be secondary to the use of vasoactive substances such as sympathomimetics or serotoninergic drugs (antidepressants, triptans, nasal decongestants and noradrenaline) and/or recreational drugs (cannabis, cocaine, amphetamines). In the earliest series of 67 cases from patients seen between 2004 and 2007 at a French center and diagnosed with RCVS, 37% were spontaneous and 63% secondary. Of note, 37 patients had taken one or more vasoactive substances, including nine taking cannabis alone, nine taking a selective serotonin reuptake inhibitor (SSRI) antidepressant alone, five taking a nasal decongestant alone and one taking an interferon alpha. The SSRIs involved were paroxetine (n = 5), citalopram (n = 4), fluoxetine (n = 3) and sertraline (n = 1) (5). These results should be compared with those of Singhal et al. (15), who analyzed 139 cases of RCVS from two American hospitals and observed that vasoactive substances are involved in 42% of cases. They cited sumatriptan, SSRIs, serotonin and norepinephrine reuptake inhibitors (SNRIs), pseudoephedrine, amphetamines, and illicit drugs such as ecstasy, cocaine and marijuana. However, data on drug-induced RCVS are scarce and no study has defined the characteristics of reported RCVS in which these drugs are suspected.
In this context, we used the world's largest pharmacovigilance database worldwide, the World Health Organization (WHO) Global Individual Case Safety Report (ICSR) database, to first describe RCVS characteristics for drugs already identified as associated with RCVS in literature. Next, we generated disproportionality signals to identify new drugs potentially associated with RCVS.
Methods
The present study comprised a retrospective observational post-marketing study using VigiBase®, the WHO Global ICSR database (https://who-umc.org/vigibase). Among more than 34 million ICSRs, we selected in Medical Dictionary for Regular Activities those registered as Preferred Term “Reversible Cerebral Vasoconstriction Syndrome RCVS” until 31 May 2023. Because of the difficulty of diagnosis, only ICSRs reported by healthcare professionals were included. Among them, all ICSRs including nimodipine coded suspect and used to treat RCVS were excluded, considering a probable protopathic bias because it is used in the treatment of RCVS.
General data
We first described, for all ICSRs retained, the place of occurrence, the population characteristics (gender, age of patient and outcomes). RCVS notifications over time and the most suspected drugs were also described.
RCVS characteristics
To address the first objective, all ICSRs containing drugs already known to precipitate RCVS according to the lists cited by Ducros et al. (3,5,20) were analyzed. To investigate RCVS characteristics, the drugs were grouped by pharmacotherapeutic classes.
For each ICSR, we collected information on gender, age of onset, suspected drugs, associated drugs, time of onset and outcomes.
Signal generation
To identify new drugs potentially associated with RCVS, we generated a disproportionality analysis, which is a set of methods largely used by researchers and regulatory/drug agencies to generate disproportionality signals, such as putative links between drugs and adverse drug reactions (ADRs). Disproportionality analysis compares the proportion of specific ADRs reported for a single drug with the proportion of the same ADR for all other drugs or for a selected panel of control drugs. Briefly, if the proportion of Y ADRs in patients exposed to drug X is greater than the proportion of Y ADRs in patients not exposed to drug X, this suggests an association between the specific drug and reaction and is a potential signal for safety (21). Disproportionality was calculated using the reporting odds ratio (ROR) and its 95% CI estimated with Woolf's method. To limit competition bias between drugs, we have selected only cases without co-suspected drugs already known to be associated with RCVS. For the comparator group, we have excluded COVID-19 vaccines, which, as a result of extensive media coverage, would be associated with a large number of reports, thereby generating a competition bias (22). In this present study, for each single drug, the ROR is the odds of exposure to the single drug among RCVS reports divided by the odds of exposure to all the other drugs, without COVID-19 vaccines, recorded in the database among all others ADRs reports during the same period. Thresholds used for signal detection was RORLB >1 and more than two cases recorded for each drug identified.
Results
We extracted 681 RCVS cases from the WHO pharmacovigilance database. After excluding cases not reported by healthcare professionals and cases suspecting nimodipine, 560 ICSRs were retained. They came principally from USA (n = 267, 47.7%), France (n = 114, 20.4%) and Japan (n = 55, 9.8%). They occurred mainly in females (n = 406, 72,5%). Males represented 105 persons (18,8%). Patients were most likely to be aged between 45 and 64 years (n = 226, 40.4%) and 18 and 44 years (n = 196, 35.0%). RCVS also occurred in children and adolescents between 2 and 17 years (n = 36, 6.4%). The proportion of women is higher than men, regardless of the age of onset, except for age group 12–17 years (Figure 1).

Sex distribution by age of occurrence of reversible cerebral vasoconstriction syndrome
All cases were serious: 359 (64.1%) hospitalizations, 22 (3.9%) incapacities or disabilities, 38 (6.7%) life-threatening events, 25 (4.4%) deaths and 116 (20.7%) other medically important conditions.
The first ICSR of RCVS in VigiBase® was reported in 2006, the second case in 2010, the third in 2012 and the next in 2013, and, up until 2022, the number continued to increase (118 reports).
The number of cases was the highest for SSRIs such as escitalopram (n = 45), fluoxetine (n = 41), sertraline (n = 32) citalopram (n = 28), SNRIs such as venlafaxine (n = 27) and duloxetine (n = 31), immunosuppressants such as tacrolimus (n = 33) and ciclosporin (n = 16), glucocorticoids such as methylprednisolone (n = 25) and prednisone (n = 18), bupropion (n = 33), and triptans such as sumatriptan (n = 21). Other drugs (mycophenolic acid and COVID-19 vaccines) were reported in 14 and 17 ICRSs, respectively. Recreational drugs were reported in 20 ICRSs with cannabis (n = 17), cocaine (n = 1), ethanol (n = 1), metamfetamine (n = 3), ecstasy (n = 1) and tobacco (n = 1). These recreational drugs were sometimes combined (e.g. cocaine and cannabis).
In total, 25 deaths were reported: 11 US cases (44%) involving escitalopram (n = 5) and citalopram (n = 6) occurred in the context of coronarovirus infection and six australian cases (24%), two of which involved sertraline, occurred in the context of a worsening of RCVS because of misdiagnosis and treatment with methylprednisolone. The eight other cases of RCVS with fatal outcome involved dihydroergotamine (n = 1), escitalopram (n = 2), venlafaxine and tuaminoheptane (n = 1), human normal immunoglobulin (n = 1), sumatriptan (n = 1), and the association of thalidomide, carfilzomib and cyclophosphamide in the setting of plasma cell myeloma (n = 2). For the analysis of ICSRs, we have retained all the drugs cited by Ducros et al. (3,5,20). They mentioned drug classes comprising antidepressants such as SSRIs, SNRIs and monoamine oxidase inhibitors, triptans, nasal decongestants such as pseudoephedrine, ephedrine and phenylpropanolamine, and ergot alkaloids such as ergotamine, methylergometrine, bromocriptine and lisuride. They also cited norepinephrine, isometheptene, tacrolimus, ciclosporin, fingolimod, cyclophosphamide, human normal immunoglobulin, erythropoietin, interferon alpha, interferon beta-1a, nicotine patches and illicit drugs as ecstasy, cocaine and marijuana.
The characteristics of reported ICSRs associated with drugs already known to precipitate RCVS are detailed in Table 1. They represented 64.5% of reported cases. We observed several cases with fingolimod (n = 7), cyclophosphamide (n = 9), human normal immunoglobulin (n = 6), norepinephrine (n = 9), nicotine patches (n = 4), phenelzine n = 3), isometheptene (n = 1) interferon beta-1a (n = 1) and cocaine (n = 1). They are not detailed in Table 1. No cases have been found for fluvoxamine, monoamine oxidase inhibitors such as moclobemide and iproniazide, nasal decongestants such as phenylpropanolamine, ergot alkaloid derivatives such as ergotamine and lisuride, and interferon alpha and erythropoietin.
Characteristics of reported ICSRs associated with drugs already known to precipitate RCVS.
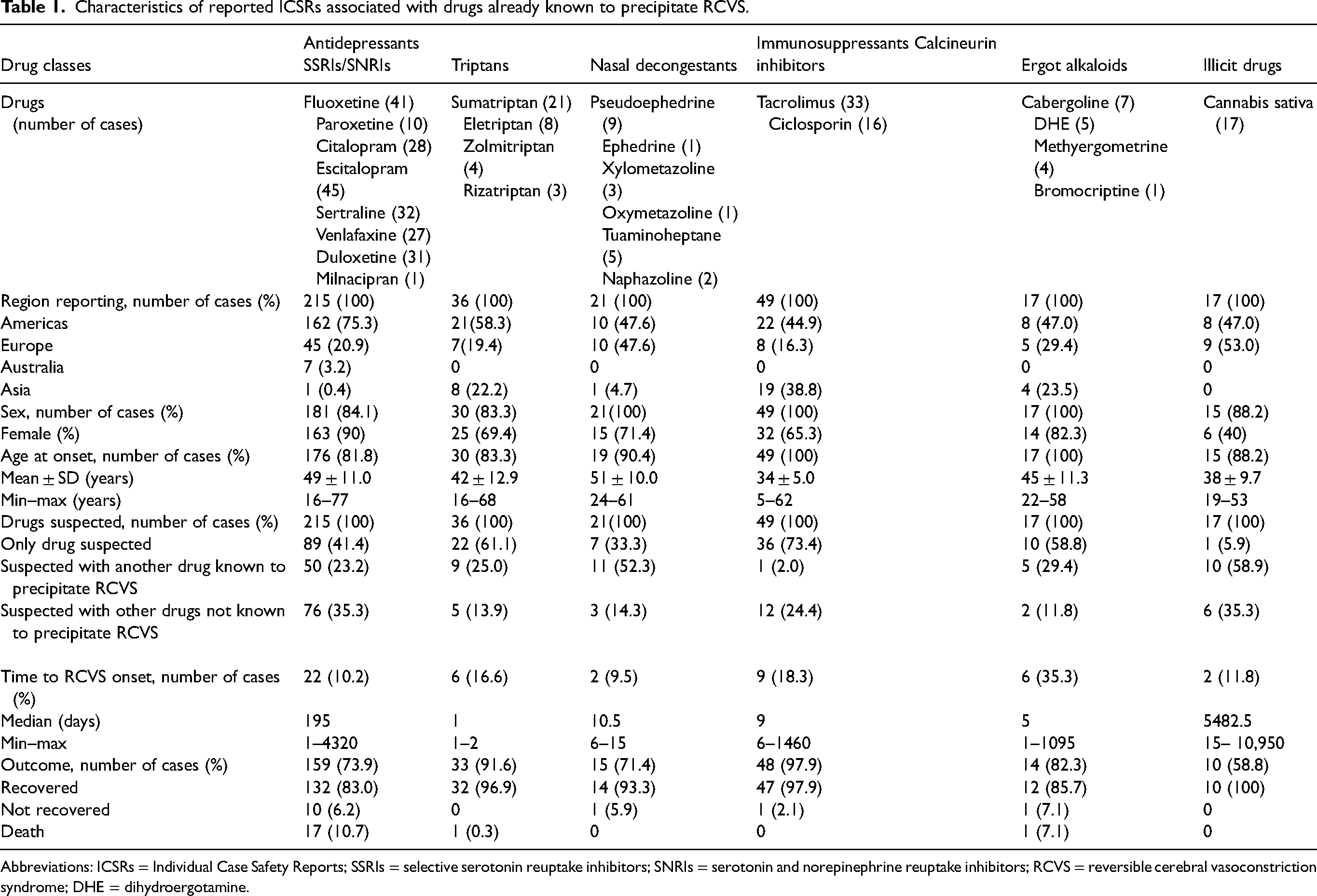
Abbreviations: ICSRs = Individual Case Safety Reports; SSRIs = selective serotonin reuptake inhibitors; SNRIs = serotonin and norepinephrine reuptake inhibitors; RCVS = reversible cerebral vasoconstriction syndrome; DHE = dihydroergotamine.
Case characteristics
Patient characteristics
Whatever the drug, RCVS cases largely concerned women (81.5%) (Table 1). Regarding ages at RCVS onset, patients treated with immunosuppressants were the youngest (median age = 34 years; ± 5 years). Immunosupressants cases came almost exclusively from Asian countries.
Drug exposure
In the majority of cases suspecting immunosuppressant calcineurin inhibitors, triptans and ergot alkaloids, the drug utilized was the only suspected one (73.4% cases involving immunosuppressants, 61.1% for triptans, 59.9% for ergot alkaloids). Conversely, RCVS was observed especially in the context of taking one or more others drugs for antidepressants SSRIs (58.6% of cases), nasal decongestants (66.6%) and cannabis (94.1%). These drugs were preferentially drugs known to precipitate RCVS. The most reported associations were those that combine an SSRI with another antidepressant (bupropion or SSRI) (n = 18), an SSRI with a nasal decongestant (mainly pseudoephedrine (n = 9)) or a triptan (n = 8) or an ergot alkaloid (n = 5) or cannabis (n = 5).
Time to onset
The time to RCVS was documented in only 13.5% of cases.
In the limited data observed, RCVS occurred within a median of 195 days after the first administration of antidepressants SSRIs and SNRIs (n = 22). It occurred earlier after administration of triptans: 1 day (n = 6), ergot alkaloids: 5 days (n = 6), nasal decongestants: 10 days (n = 2) and immunosuppressants: 9 days (n = 9). We observed that eight cases from the USA occurred in a context of fluoxetine overdose. In three French cases and one American case concerning SSRIs, the dose was increased 3 weeks (n = 1) and a few days (n = 3) before the RCVS occurrence.
Outcomes
When the information was available, patients recovered in 88.8% of cases. The outcome was fatal in 13% of ICSRs (n = 19), in which escitalopram (n = 7), citalopram (n = 6), sertraline (n = 3), venlafaxine associated with tuaminoheptane (n = 1), dihydroergotamine associated with escitalopram (n = 1) and sumatriptan (n = 1) were implicated.
Disproportionality analysis
The results of reporting odds ratio and main characteristics of cases are reported in Table 2.
Reporting odds ratio of the association between drugs and RCVS in vigibase®
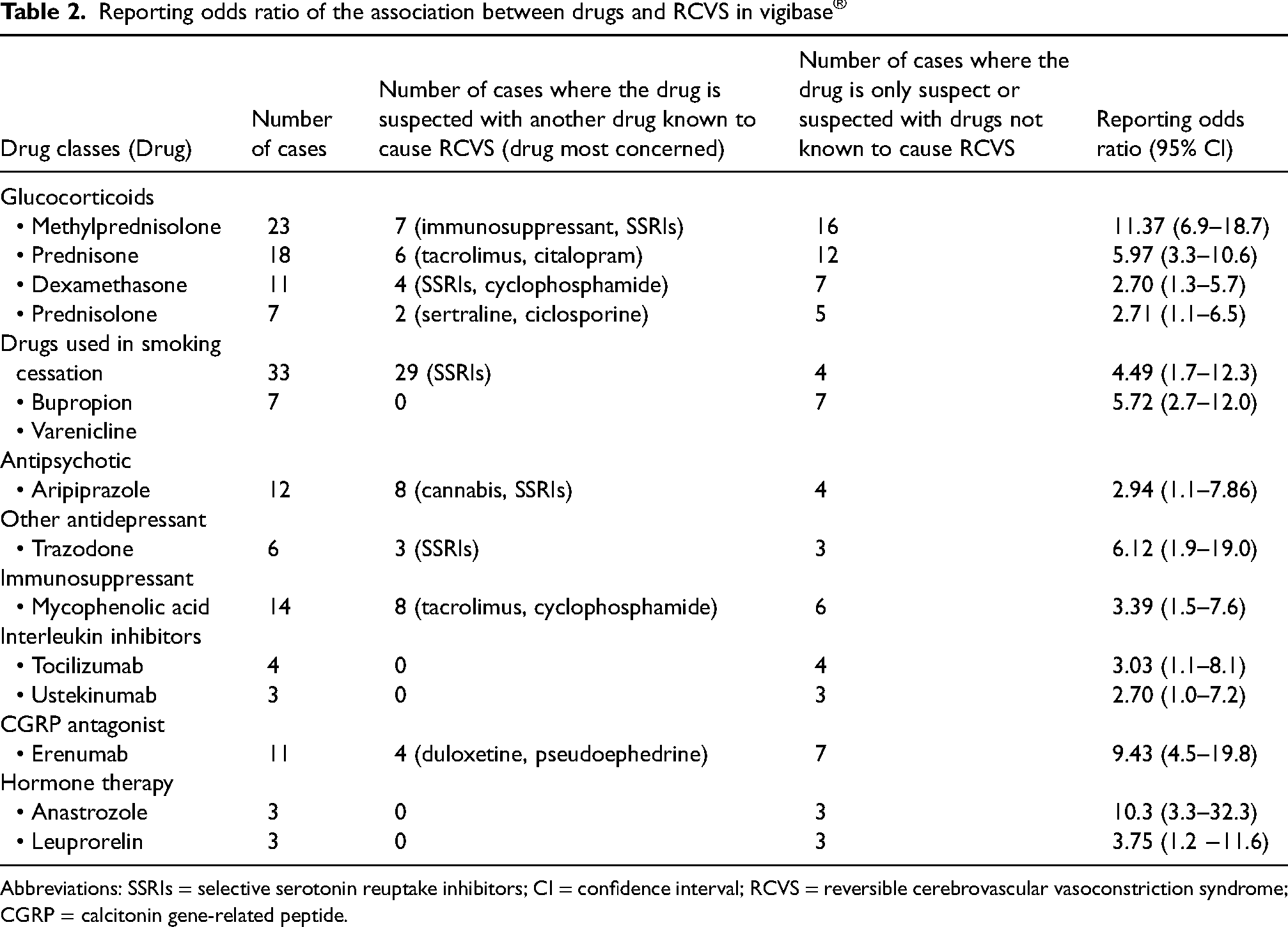
Abbreviations: SSRIs = selective serotonin reuptake inhibitors; CI = confidence interval; RCVS = reversible cerebrovascular vasoconstriction syndrome; CGRP = calcitonin gene-related peptide.
We have identified 14 signals with drugs that may be associated with the occurrence of RCVS. A disproportionality signal was observed for glucocorticoids, drugs used in smoking cessation such as bupropion and varenicline, an antipsychotic drug (aripiprazole), another antidepressant (trazodone), a selective immunosuppressant (mycophenolic acid), interleukin inhibitors (tocilizumab, ustekinumab), drugs used in migraine such as erenumab, a calcitonin gene-related peptide antagonist, as well as hormone therapies such as anastrozole and leuprorelin.
RCVS characteristics were fairly documented concerning median time of onset (62 cases out of 83 were not documented) and median dose used (65 cases out of 87 were unknown). For more than half, glucocorticoids (25 cases out of 41) and mycophenolic acid (four cases out of six) were associated with another glucocorticoid and/or an anticancer drug. Methylprednisolone was suspected in two cases of misdiagnosis that led to worsening of RCVS leading to death.
Discussion
To our knowledge, this is the first study to systematically retrieve and assess all RCVS cases reported in a large pharmacovigilance database. We selected ICSRs reported by healthcare professionals to ensure RCVS validity. The most widely reported drugs suspected in RCVS onset were SSRIs, SNRIs, immunosuppressant calcineurin inhibitors, triptans and nasal decongestants, which represent more than half of notifications.
All of these drugs have been widely reported in the literature as being predisposing factors to RCVS according to several studies (3,5,20,23).
We observed significant differences between countries regarding the drugs involved in reported cases. This is probably a result of the different pharmacovigilance systems and the availability of drugs in different countries.
Most cases of RCVS had a favorable outcome. Deaths observed were mostly because if coronarovirus infection and errors in diagnosis leading to a worsening of the RCVS. According to Breville et al. (24), fatal outcomes are rarely reported.
Observed data are limited to trends of time to onset for each drug class (around six months for antidepressants, and a few days on average for nasal decongestants, triptans, ergot alkaloids derivatives and immunosuppressants).
More precise data concerned four cases with SSRIs. The results are in agreement with Lange et al. (25) who recently hypothesized a temporal association if the precipitating condition occurred within four weeks prior to RCVS onset. We may add that increased dosage a few days or weeks before RCVS onset in a patient treated with an SSRI or SNRI for several years may be a predisposing factor. In addition, we observed that association with an SSRI or SNRI was the most common among all drugs known to precipitate RCVS. Consumption of a nasal decongestant or triptan in patients on long-term SSRI or SNRI therapy may be an additional risk factor. Some drugs such as oral contraceptive pills, antiemetics (ondansetron, granisetron and metoclopramide), alpha interferons, erythropoietin and clonazepam, as well as two serotoninergic drugs, tramadol and linezolid, described as precipitating drugs of RCVS in several studies (3,23), are not signaled in Vigibase®. We can hypothesize that the published lists of drugs known to precipitate RCVS appear to be established from the pharmacodynamic properties of the class drug rather than based on concrete cases. We identified 14 signals with drugs that may be associated with the occurrence of RCVS. We observed a signal of disproportionality with glucocorticoids, which are largely noted in VigiBase®. Glucocorticoids are considered not only as iatrogenic factors for RCVS, but also they may contribute to disease worsening according to Singhal et al. (6). In their study on 162 patients with RCVS, out of the 23 patients with clinical worsening, 17 received glucocorticoids (15 within the preceding days). In the present study, among the cases with an unfavorable outcome described in VigiBase®, two fatal RCVS were reported in patients for whom vasculitis was initially diagnosed and who were treated with methylprednisolone. They were also mentioned by Kunckok et al. (26), who reported two other fatal cases of misdiagnosis leading to aggravated RCVS and death. These results confirm the difficulty of diagnosing RCVS and underscore the importance of early diagnosis and avoidance of potentially harmful corticosteroid treatment. Concerning the drugs used in smoking cessation, De la Torre-Colmenero et al. (27) reported a case of RCVS after sudden cessation of smoking and treatment by bupropion. Although smoking cessation may be a triggering mechanism, bupropion has the ability to alter adrenergic and dopaminergic metabolism and, consequently, can also trigger RCVS. Although no cases have been published with regard to varenicline, seven have been observed in VigiBase® where it is the only suspected drug. Varenicline is a nicotinic acetylcholine receptor agonist for which stimulation, among other effects, increases the release of dopamine in certain brain structures, at least partially explaining addiction. It has the same mechanism of action as nicotine, which is a known RCVS trigger.
Aripiprazole has been implicated in VigiBase® in four non-informative cases of RCVS in young adults. The main evoked mechanism is its action on the serotonin system. The six cases concerning mycophenolic acid involved patients who were being treated for scleroderma or lupus, which are pathologies described as causing RCVS. Concerning trazodone, implicated in three non-informative cases, it is an antidepressant that can potentiate serotonergic activity, which can precipitate RCVS.
We found a signal of disproportionality with erenumab involved in seven cases of RCVS where it was the only suspected drug. Erenumab is a new type of migraine-preventive medication that blocks calcitonin gene-related peptide. It is a potent vasodilatator of the cerebrovascular system. Rozen et al. (28) reported a case of RCVS occurring after erenumab treatment They hypothesized that blocking calcitonin gene-related peptide was a trigger for cerebral artery vasospasm in patients at risk of developing RCVS (i.e. migraine patients). Tocilizumab as used in rheumatoid arthritis was implicated in four cases after eight months of treatment for two patients in Vigibase® and ustekinumab was implicated in three cases in patients with a history of Crohn's disease. The first case of RCVS possibly associated with tocilizumab was reported by Gonzales-Martinez et al. (29) and another case was reported after ustekinumab intake (30). The mechanism has not been elucidated.
Finally, we found a signal of disproportionality with leuprorelin and anastrozole, each of which, according to VigiBase®, was implicated in three cases. A previous study (20) indicated that acute estrogen reduction because of factors other than the postpartum state, including oral contraceptive pills, can cause RCVS. Hiroki et al. (31) reported a case of RCVS following pseudo-menopause therapy with leuprorelin acetate injection for uterine adenomyosis. It was suggested that the reduction of estrogen as a result of treatment with leuprorelin may lead to cerebral vasoconstriction (31). A case has been reported with anastrozole in the treatment of breast cancer (32).
The present study has several biases and limitations.
First, there is a bias of under-reporting, which is an inherent and systematic limitation to this type of pharmacovigilance study, particularly regarding RCVS notifications. Furthermore, the detection of a “statistically significant” disproportionality signal should not be intended as an ADR or a confirmed drug event association, but rather as an alert requiring additional characterization (case-by-case assessment), including biological plausibility and analytical pharmaco-epidemiological investigations, when needed. Indeed, positive prognostic values of disproportionality analyses to detect true ADR are estimated between 0.2 and 0.6 according to method and threshold; thus, a large number of false-positive signals is expected (33). Using Vigibase® also exposes to notification bias depending on the country: in low- and middle-income countries, there are very low spontaneous reporting rates with poor quality reports hindering robust signal detection analyses (34).
Second, this syndrome is considered as an increasingly recognized but is a still under-diagnosed condition. Headache is the main symptom and often remains the only manifestation of RCVS. Acute headaches as a result of RCVS are sometimes mistaken for bad migraine attacks for migraine patients. RCVS and primary angeitiis of central nervous system have not been recognized as two distinct disorders and several clinicians are wary of missing a diagnosis of angiitis and delayed treatment (35). In severe forms of RCVS, patients may present convulsions and neurological deficit, making it difficult to differentiate from posterior reversible encephalopathy (PRES). Moreover, PRES and RCVS can coexist (RCVS includes PRES in 10% of cases) and there is an overlap between comorbidities and associated toxicities. This suggests a common underlying pathophysiology (36). Possible confusion may concern immunosuppressants and neoplastic drugs that are known to cause PRES. Third, the triggering factors of RCVS are numerous, leading to confusion bias. In addition to drugs, environmental factors such as psychological stress, acute emotion (announcement of a death, strong fear, etc.), the pathology for which the drug is administrated, such as migraine, depression and anxiety, and autoimmune diseases such as lupus erythematous, multiple sclerosis and systemic scleroderma are conditions associated with RCVS. The completeness of the information collected in VigiBase® is not always guaranteed because the patient's medical history or parameters related to the drug of interest or co-suspected (i.e., dose, time to onset) are sometimes missing or incompletely documented. Any conclusions about temporal relationships should be treated with caution. The use of recreational drugs was not a criterion systematically sought, which also limits the interpretation of confounding bias in this retrospective study. The term RCVS has been known only since the 2010s and was introduced into the MeDRa dictionary of databases recently. Consequently, identified cases of RCVS are not queried. Finally, once the diagnosis of RCVS is established, the drug is not always suspected by the clinician as a triggering factor because it has been taken for a long time and it is not clear why a sudden reaction to a drug would finally, unexpectedly have occurred.
Conclusions
In the present study, we have confirmed the potential role of vasoconstrictor drugs in the onset of RCVS, particularly when used in combination. In addition to these drugs, which are well known to be associated with RCVS, we have found a disproportionality signal with 14 other drugs. However, the results of the present study must be interpreted as an exploratory analysis. Continuous monitoring through spontaneous reporting systems together with future pharmacoepidemiologic studies will contribute to precise our data.
This is the first pharmacovigilance study that has explored drugs potentially associated with RCVS from the global pharmacovigilance database. We have confirmed the role of vasoconstrictor drugs in the onset of RCVS, particularly when used in combination. We have found a disproportionality signal with drugs that were not previously known to be associated with increased reporting of RCVS
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
