Abstract
Background
Recent studies suggested that persons with migraine might be at higher risk of structural brain changes, including cerebral small vessel disease and atrophy. However, findings in the literature are inconsistent, with variations observed in the direction, magnitude, and population characteristics of reported effects, and large-scale population-based evidence remains scarce. Hence, we investigated the association of migraine with structural brain changes in a middle-aged and elderly population.
Methods
Within the population-based Rotterdam Study, lifetime history of migraine was assessed using a validated questionnaire between 2006 and 2011. Magnetic resonance imaging of the brain was performed in 4920 participants (median age 61.7 [IQR 45.5, 97.5] years, 55.4% female) to assess imaging markers of cerebral small vessel disease and brain atrophy. We used linear and logistic regression models to examine the cross-sectional association of migraine with brain volumes (total grey and white matter volumes in mL) and cerebral small vessel disease markers (white matter hyperintensity volume in mL, presence of lacunes and cerebral microbleeds). Adjustments were made for age, sex, intracranial volume and cardiovascular variables. Analyses were also stratified by sex and presence of aura.
Results
The lifetime prevalence of migraine was 15.3% (752/4920). In multivariable adjusted regression models, we found no statistically significant differences between participants with and without migraine in terms of total brain volume (mean difference [MD]: 2.21 mL, 95% confidence interval [CI]: −0.38 ; 4.81), grey matter volume (MD: 0.38 mL, 95% CI: −1.98 ; 2.74), white matter volume (MD: 2.19 mL, 95% CI: −0.56 ; 4.93), log white matter hyperintensity volume (MD: −0.04 mL, 95% CI: −0.10 ; 0.02), presence of lacunes (odds ratio [OR]: 0.82, 95% CI: 0.58–1.15), and presence of cerebral microbleeds (OR: 0.95, 95% CI: 0.76–1.18).
Conclusion
In this study, we found that middle-aged and elderly participants with migraine were not more likely to have structural brain changes on magnetic resonance imaging.
Introduction
Migraine is a common paroxysmal neurological disorder that poses a significant burden on individuals and society (1). The etiology of migraine is multifactorial and not fully understood, but two major pathophysiological mechanisms that are thought to play an important role include the sensitization of trigeminovascular pathways, and the dysregulation of vasodilator neuropeptides (2). Besides the acute attacks, it is thought that these mechanisms may have long-term detrimental effects on the brain (3,4). Several studies have used non-invasive neuroimaging in migraine patients to directly visualize potential changes in the brain (5), such as markers of cerebral small vessel disease (CSVD). Signs of CSVD on conventional magnetic resonance imaging (MRI) include white matter hyperintensities (WMHs), lacunes, cerebral microbleeds (CMBs), and atrophy (5–7). Although some studies reported a higher prevalence of CSVD markers in migraine patients compared to controls, others found no differences (7–10). Moreover, studies in the literature are mainly restricted to clinic-based settings, and relatively small samples of patients with comorbidities (5,7). This introduces potential issues, as the recruitment of participants from specialty care settings may inadvertently create selection bias due to the fact that only a minority of migraine patients consult a physician (11,12). Furthermore, those who do seek specialty care are more likely to have severe symptomatology, and multiple comorbidities. In contrast, population-based evidence remains scarce. To address these limitations, we investigated the association of migraine with structural brain changes in the population-based Rotterdam Study.
Materials and methods
Study setting and population
This study was conducted as part of the Rotterdam Study, which is a prospective population-based cohort study among middle-aged and elderly people living in the Ommoord district in the city of Rotterdam, the Netherlands (13). The study began in 1990 with the first cohort. Then an expansion (second) cohort was added in 2000, both cohorts included participants over the age of 55. In 2006, the study was expanded again with a (third) cohort including participants over the age of 45. The lifetime prevalence of migraine was assessed between 2006 and 2011 in a home interview or over the phone (for 634 participants due to logistical reasons) in a total of 7266 participants from all three cohorts.
Assessment of migraine
Migraine was assessed by trained raters during a structured interview using a questionnaire based on the International Classification of Headache Disorders, second edition (ICHD-2) criteria (14). Items were modified from the questionnaire validated for use in the Genetic Epidemiology of Migraine (GEM) study in Leiden (15). The questionnaire had a sensitivity of 0.93 and a specificity of 0.36 (15). All participants who met the criteria for lifetime history of migraine were categorized as persons with migraine. As the questionnaire had a low specificity, in order to reduce the number of false positives, it was slightly modified from the ICHD-2 criteria in two ways. First, only participants who had reported a history of five attacks of severely (instead of moderately) painful headaches in the first question received the rest of the questions (i.e., the first question served as a screening tool). Second, the presence of aura symptoms accompanying migraine was defined as reporting at least five (instead of two due to the first question) attacks fulfilling the migraine without aura criteria, in combination with visual, sensory or speech-related aura symptoms (see online Supplementary Table S1 for the detailed modified criteria). Moreover, probable migraine was defined as meeting all criteria but one for migraine without aura. Participants who met the criteria for probable migraine were categorized as persons without migraine as the differential diagnosis of probable migraine is challenging (14). Migraine patients were further dichotomized as active (<1 year since last attack) or non-active (≥1 year since last attack) migraine.
Assessment of structural brain markers
All participants underwent brain MRI at various time points using a 1.5 Tesla MRI scanner (General Electric Healthcare, Milwaukee, WI) with an 8-channel head coil. In case a participant was scanned more than once, the MRI scan with the closest date to migraine interview was used in the analysis. No changes were made to the software or hardware during the course of the study. The scan protocol included T1-weighted, proton density-weighted, fluid-attenuated inversion recovery (FLAIR), and T2*-weighted gradient recalled echo (T2* GRE) sequences. A detailed protocol of Rotterdam Scan Study including quality control, was described previously (16). All scans were checked for quality and the presence of artifacts. An automated brain tissue segmentation was used to quantify brain volumetric measures. We employed a supervised atlas-based k-nearest neighbor algorithm (17) using the Approximate-Nearest-Neighbor (ANN) library (18), which was validated for cerebrospinal fluid (CSF), gray matter (GM), white matter (WM) and white matter hyperintensity (WMH) segmentation (19,20). These segmentations were also visually inspected and manually corrected as needed. Intracranial volume was calculated by summing supratentorial gray and white matter (normal-appearing white matter and white matter hyperintensity volume) and CSF volumes (20). Total brain volume was calculated by aggregating GM, normal-appearing WM, and WMH volume (21). Trained raters also visually evaluated all scans to assess for the presence of cortical infarcts, lacunes, and cerebral microbleeds. Each scan was assessed by a single rater, with cases meeting even a low threshold of uncertainty discussed in a consensus meeting led by an experienced neuroradiologist (M.W.V.). Lacunes were defined as focal lesions of noncortical tissue that were 3–15 mm in size with isointense signal intensity to cerebrospinal fluid on all sequences, and a hyperintense rim on FLAIR images when supratentorial (16). Cerebral microbleeds (CMBs) were identified on T2* GRE images as small, focal areas of very low signal intensity that did not exhibit any signal abnormalities on other sequences (16).
Other measurements
Information on the following covariables was collected via interview and physical examination: body mass index (weight in kg divided by height in m2), educational level (primary; lower/intermediate education; intermediate vocational or higher general education; and higher vocational education or university), physical activity (metabolic equivalent of a task in hours per week) (22) and smoking status (never, former and current smoker). Although not included in the adjustment set, data were also collected on the following variables. First, hypertension was defined as the average blood pressure of systolic ≥140 mm Hg or diastolic ≥90 mm Hg, across two measurements within two minutes with a random-zero sphygmomanometer in a sitting position at the right brachial artery, or the use of antihypertensive medication (23). Second, hypercholesterolemia was defined as total cholesterol of ≥6.2 mmol/L or the use of lipid lowering medication (23). Third, diabetes mellitus was defined as fasting glycaemia of ≥7.0 mmol/L or the use of antidiabetic medication. Fourth, the history of coronary heart disease (myocardial infarction or revascularization) and alcohol use (grams/day) were asked during a questionnaire.
Covariables were selected for being a potential cause of the exposure or the outcome (intracranial volume), or both (age, sex, smoking, body mass index and physical activity) (24,25), or for being a potential proxy (educational level (26,27)) of an unmeasured confounder (socio-economical status) (28).
Population for analysis
Participants with data on both migraine status, and a brain MRI scan were eligible for inclusion in the study (n = 5172). After the removal of 252 scans in quality control (due to motion artefacts or signal heterogeneity on MRI) (16), a sample of 4920 participants were available for the analyses with total brain volume, lacunes and CMBs. Moreover, participants with cortical brain infarcts on MRI (n = 180) were excluded from the analyses of GM, WM and WMH since the area of gliosis in these infarcts led to less reliable tissue segmentation (see Figure 1).
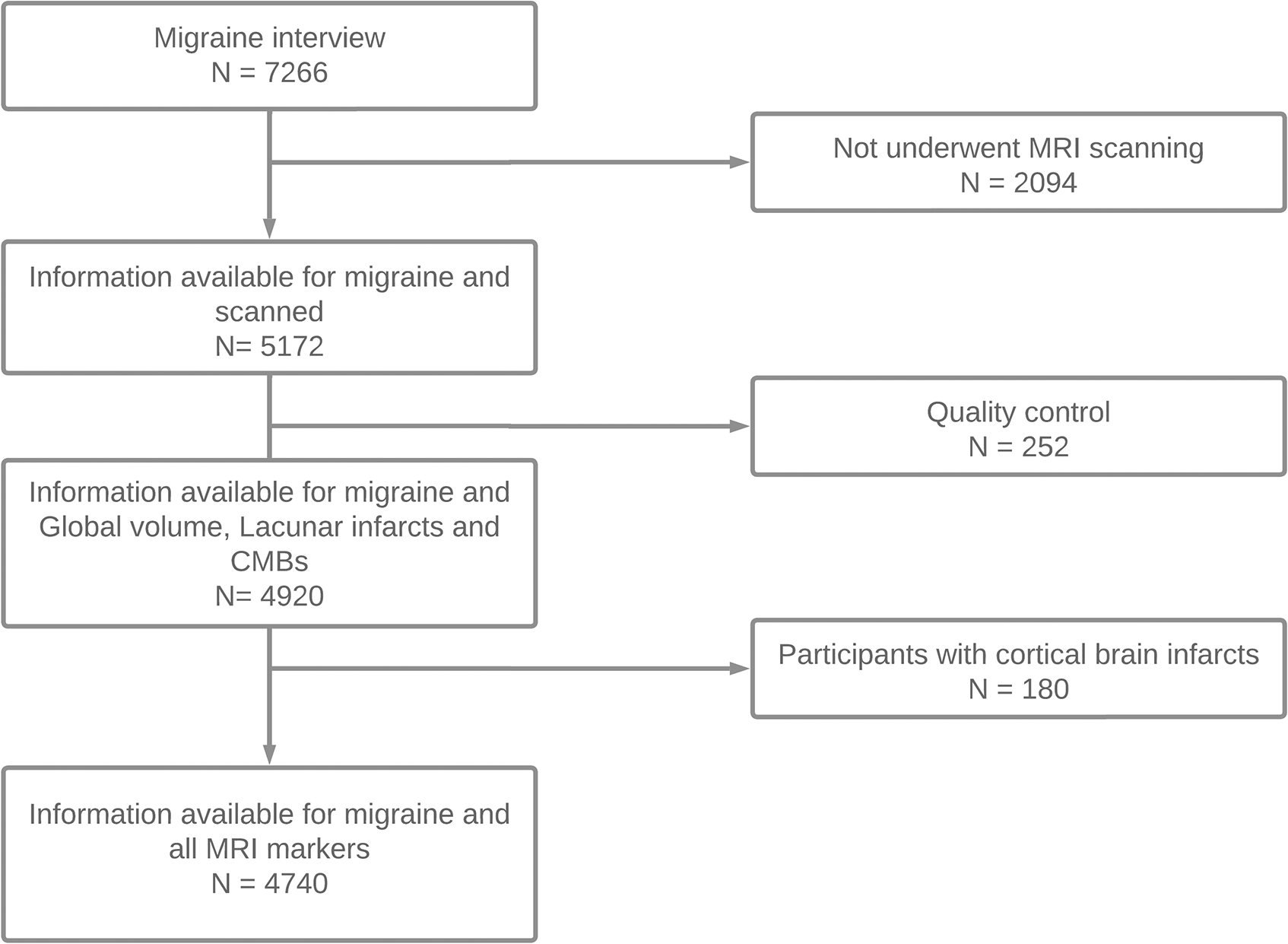
Flowchart of participant inclusion.
Statistical analyses
Missing covariable data were imputed using 5 multiple imputations by chained equation and 30 iterations based on all variables (missingness for each covariable presented under Table 1). WMH volume distribution was skewed and therefore natural log-transformed to better satisfy normality of residuals assumption of linear regression. IBM SPSS (Chicago, IL, USA) (29) version 25 and R (30) version 4.0.5 for Windows were used for the analyses and “mice” package (31) (version 3.13.0) in R was used for multiple imputation.
Descriptive characteristics categorized by migraine.

Values are presented either as mean (SD), median [IQR] or as number (percentage). Number of missing values before imputation: 2 (0.06%) for body mass index, 3 (0.06%) for smoking status, 11 (0.22%) for alcohol consumption, 22 (0.45%) for hypertension, 38 (0.77%) for hypercholesterolemia, 40 (0.81%) for educational level, 46 (0.93%) for diabetes mellitus, 581 (11.81%) for physical activity, and 646 (13.13%) for coronary heart disease history. CHD, coronary heart disease (myocardial infarction or revascularization); MET, metabolic equivalent of tasks. For numeric variables, a two-sample t-test was used, while for categorical variables, a chi-square test of independence was employed.
Initially, to compare the main demographic and clinical characteristics between participants with and without migraine, we used two-sample t-test for numeric variables and chi-square test of independence for categorical variables. To determine the association between migraine and structural brain changes we used the following strategy. First, we constructed multivariable linear regression models to examine the association between migraine and total brain, GM, WM and log WMH volumes. Second, we constructed multivariable logistic regression models to examine the association between migraine and the presence of lacunes and CMBs. Both the linear and the logistic regression analyses included the same set of variables and each included two models with increasing levels of adjustment. Model 1 was adjusted for age, sex and intracranial volume. Model 2 was further adjusted for educational level, smoking status, BMI and physical activity.
Third, we conducted sensitivity analyses to further explore the effect of debated confounders, sex and presence of aura; on the association between migraine and brain structure. In the main analyses, any variable (hypertension (32), diabetes mellitus (33), coronary heart disease (4), hypercholesterolemia (34) and alcohol consumption (35)) that was thought to be on the causal pathway between migraine and brain structure, was excluded from the main adjustment set to avoid overadjustment bias. However, it can be argued that some of these variables may instead be confounders; therefore, we fitted separate models with these variables included. Next, to further explore the effect of sex and the presence of aura on the association between migraine and brain structure, we fitted separate models stratified by sex and the presence of aura. Additionally, we repeated some analyses considering the number of lacunes and microbleeds (as a measure of disease severity) as the outcome using Poisson regression. These models were also adjusted for the same covariables as in Model 2.
Results
Table 1 shows the descriptive characteristics of the study population. In total, 752 (15.3%) participants had a lifetime history of migraine. Among those, 309 (41.1%) had their last attack less than 12 months ago (i.e., active migraine) and in 150 (19.9%) of them aura symptoms were also present. The majority of migraine patients were female (78.9%). Participants with migraine (compared to those without) were less likely to be highly educated, to be current smokers (13.3 vs 17.3%) and had lower prevalence of chronic heart disease (CHD) history (2.8 vs 4.3%), hypertension (62.2 vs 64.7%), and diabetes mellitus (7.0 vs 11.2%). Moreover, they reported lower levels of alcohol consumption, alongside higher levels of physical activity on average, with the exception of a slightly higher prevalence of hypercholesterolemia (51.3 vs 49.3%).
Analyzing the associations between migraine status and brain volumes (Table 2) using multivariable-adjusted linear regression models, we found no statistically significant differences in terms of total brain volume (mean difference [MD]: 2.21, 95% confidence interval [CI]: −0.38; 4.81), GM volume (MD: 0.38, 95% CI: −1.98; 2.74), and WM volume (MD: 2.19, 95% CI: −0.56; 4.93). Moreover, using multivariable linear and logistic regression models, we found no associations between migraine status and imaging markers of CSVD (Table 2) such as: log WMH volume (MD: −0.04, 95% CI: −0.10; 0.02), presence of CMBs (odds ratio [OR]: 0.95, 95% CI: 0.76–1.18), and presence of lacunes (OR: 0.82, 95% CI: 0.58–1.15).
Cross-sectional associations between migraine and, brain volumes and cerebral small vessel disease (CSVD) markers.

Model 1: adjusted for age, age2, sex, intracranial volume; Model 2: Model 1 + adjusted for smoking, body mass index, educational level, physical activity. WMH, White Matter Hyperintensity.
Estimates obtained with N = 4920.
Estimates obtained with N = 4740.
In the sensitivity analyses, separate models where we additionally adjusted for previously excluded variables (hypertension, diabetes mellitus, coronary heart disease, hypercholesterolemia and alcohol consumption) yielded unchanged results (online Supplementary Tables S2 and S3). Further analyses did not show evidence for an effect modification by sex (Table 3) or aura presence (Table 4) except for the presence of CMBs. We found that migraine and the presence of CMBs were positively associated in females (OR: 2.05, 95% CI: 1.20–3.62) but negatively associated in males (OR: 0.55, 95% CI: 0.33–0.88; Table 3). However, there were no sex differences in terms of number of CMBs (females incidence rate ratio [IRR]: 1.07, 95% CI: 0.85–1.37; males IRR: 0.92, 95% CI: 0.75–1.11; Table 5). Moreover, sensitivity analyses showed that history of migraine was associated with a lower number of lacunes (IRR: 0.75, 95% CI: 0.57–0.97) and migraine with aura was associated with a smaller log WMH volume (MD: −0.14, 95% CI: −0.27; −0.01).
Cross-sectional associations between migraine and, brain volumes and CSVD markers stratified by sex.

Models were adjusted for age, age2, intracranial volume, smoking, body mass index, educational level, physical activity. WMH, White Matter Hyperintensity. Bold values denote statistical significance at the p < 0.05 level.
Estimates obtained with N = 4920.
Estimates obtained with N = 4740.
Cross-sectional associations between migraine and, brain volumes and CSVD markers stratified by aura presence.

Models were adjusted for age, age2, sex, intracranial volume, smoking, body mass index, educational level, physical activity. WMH, White Matter Hyperintensity. Bold values denote statistical significance at the p < 0.05 level.
Estimates obtained with N = 4920.
Estimates obtained with N = 4740.
Poisson Regression analyses on the association between migraine and the number of CMBs and lacunes.

Models were adjusted for age, age2, sex, intracranial volume, smoking, body mass index, educational level, physical activity. Bold values denote statistical significance at the p < 0.05 level.
Estimates obtained with N = 4920.
Estimates obtained with N = 4740.
Discussion
In our population-based cohort study, we found no relationship between migraine and structural brain changes, including cerebral small vessel disease (CSVD) and cerebral atrophy.
Regarding the link between migraine and CSVD, findings in the literature are inconsistent. First, in some of the previous research, volumetric changes in GM and WM have been reported in individuals with migraine compared to controls (5). In contrast, other studies found no difference in GM (36) and WM (36,37) volumes, and one study even reported a positive association between migraine and GM density (38). Of note, we utilized whole-brain automated segmentation for volumetric analysis (16), contrasting with the regional voxel-based approach often used in literature (5). While not directly comparable, our approach showed good accuracy and reproducibility after extensive evaluation (17,19). Moreover, regional volumetric analysis, focusing on specific brain regions, may overlook global structural changes, limiting insights into overall brain morphology. Second, the results of two meta-analyses investigating the relationship between migraine and WMH are conflicting (7,39). Third, despite some studies showing an association between migraine and lacunes (40), in a meta-analysis, no association was found between migraine and lacunes (7). Fourth, the association between migraine and the presence of CMBs has only been explored in a single clinical study, which reported a higher frequency of CMBs in migraine patients exclusively in infratentorial regions but there were no differences in other regions (6). Overall, our findings suggest the absence of a link between the lifetime history of migraine and structural brain changes within the general population. In the sensitivity analyses, we found that female migraine patients have a higher chance of having CMBs than controls, whereas male migraine patients have a significantly lower chance. Moreover, sensitivity analyses showed that participants with migraine with aura symptoms tend to have a smaller volume of WMH. Since these observations were made in a sensitivity analysis, care should be taken when interpreting them from a causal perspective. We believe further studies are needed to draw firm conclusions regarding possible sex differences and effect of the aura on WMH progression.
Besides the findings of the present study, previous research within the Rotterdam Study also suggested that migraine may not have long-term consequences detrimental to the brain. Specifically, participants with migraine reported to have higher cerebral blood flow (41), show better performance on tests of executive function and fine motor skills (42), and have less arteriosclerosis in intracranial carotid arteries (43), compared to controls. Furthermore, between these groups, no statistically significant differences have been observed in terms of retinopathy (44) and stroke risk (45). These findings and the results of the present study, necessitate the consideration of the possibility of migraine not being as detrimental to the brain as thought previously. In fact, it has been hypothesized that migraine attacks can be a neuroprotective mechanism against oxidative stress (46), which plays an important role in brain aging (47) and the development of CSVD (48). However, it is important to note that the evidence supporting this hypothesis mainly comes from basic neuroscientific research on the separate components of migraine pathophysiology, and this hypothesis needs further testing in migraine-focused research.
The current study has a number of strengths, including validated migraine ascertainment, a population-based design with a sufficient sample size, a clearly defined control group and a broad age range. However, there are also some limitations to consider. First, we assessed migraine using a slightly altered version of the ICHD-2 criteria. As a result, migraine patients with less than severe headaches may have been misclassified as participants without migraine. However, because it is unrelated to brain structure, this would be a non-differential misclassification. Second, the low specificity of the questionnaire might have led the migraine group to include participants with non-migraine headache disorders. This would have reduced the statistical power to detect potential differences in brain structure between participants with and without migraine. Likewise, participants without migraine were, on average, older, had a higher prevalence of diabetes mellitus, and consumed more alcohol compared to those with migraine. There might be several reasons why participants without migraine are older, including the decreased prevalence of migraine with age (24), potential higher mortality in migraine with aura patients (4), and differences in our study cohorts, where our third cohort recruited into the Rotterdam Study is younger and thus had a higher prevalence of migraine. Since diabetes prevalence increases with age (49), a higher prevalence of diabetes mellitus can be explained by age difference, and participants with migraine may avoid alcohol due to it being a potential migraine trigger (35). We also fitted separate models that were statistically adjusted for diabetes mellitus and alcohol consumption (see online Supplementary Materials), and the results remained unchanged. However, it is important to acknowledge the possibility of residual confounding. Third, it would be interesting to see the effect of duration of disease and frequency of migraine attacks on the brain structure, however, we did not collect detailed data on exact frequency of attacks and migraine onset. Similarly, it would have been interesting to investigate differences in terms of WMH number and location, however, due to the lack of a main effect we decided not to proceed with these analyses. Fourth, although we did not find any association between migraine and some of the global CSVD measures, we cannot exclude the possibility of structural differences in specific brain regions. Fifth, despite migraine being a chronic disorder with an early onset in life therefore probably preceding the CSVD changes in the brain, the cross-sectional nature of our analyses does not inform in the direction of the causality. Further longitudinal studies are needed to elucidate the relationship between migraine and structural brain markers as the current literature reports varied courses of brain changes in migraine patients (10,50,51). Additionally, more population-based research is essential to capture a more representative sample of migraine patients with varying severities and comorbidities.
In conclusion, our population-based study found no significant association between migraine and cerebral small vessel disease (CSVD), as indicated by various structural brain markers. These results challenge prior assumptions about the detrimental impact of migraine on brain health.
Public health relevance
The study's findings suggest that there may be no significant association between migraine and cerebral small vessel disease (CSVD) or atrophy in the brain, based on structural imaging markers. This challenges prior assumptions about the potential long-term neurological consequences of migraine.
Clinicians can cautiously incorporate these findings into their discussions with migraine patients, acknowledging the limitations of cross-sectional data and the need for further research to elucidate the relationship between migraine and brain health over time.
From a public health perspective, while these results imply a potential reassurance for individuals with migraine, the findings underscores the importance of continued research to comprehensively understand the impact of migraine on brain structure and function.
Supplemental Material
sj-docx-1-cep-10.1177_03331024241266951 - Supplemental material for Migraine and brain structure in the elderly: The Rotterdam Study
Supplemental material, sj-docx-1-cep-10.1177_03331024241266951 for Migraine and brain structure in the elderly: The Rotterdam Study by Cevdet Acarsoy, Mohammad Kamran Ikram, Mohammad Arfan Ikram, Meike W. Vernooij, and Daniel Bos in Cephalalgia
Footnotes
Author contributions
All authors have made a substantial intellectual contribution to conception and design of the study, acquisition of data (C.A., M.W.V.), analysis and interpretation of data (C.A., M.K.I., D.B.), drafting the article (C.A.), or drafting a significant portion of the article or figures (C.A., M.W.V., M.A.I.). All authors approved the final version of the article for publication. All authors had full access to the data in the study and took responsibility for data integrity and accuracy of data analysis.
Acknowledgements
The authors are grateful to the study participants, the staff from the Rotterdam Study, and the participating general practitioners and pharmacists. Special thanks to Elizabeth A. Loehrer and Joyce van den Ende for their contribution to data cleaning.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Rotterdam Study is funded by Erasmus Medical Center and Erasmus University, Rotterdam, Netherlands Organization for the Health Research and Development (ZonMw), the Research Institute for Diseases in the Elderly (RIDE), the Ministry of Education, Culture and Science, the Ministry for Health, Welfare and Sports, the European Commission (DG XII), and the Municipality of Rotterdam.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
