Abstract
Introduction
Serotonin (5-HT) plays a role in migraine pathophysiology, but whether brain 5-HT is involved in the conversion from episodic to chronic migraine is unknown. Here, we investigated brain 5-HT levels, as indexed by 5-HT4 receptor binding, in chronic migraine patients and evaluated whether these were associated with migraine frequency.
Methods
Sixteen chronic migraine patients underwent a dynamic PET scan after injection of [11C]SB207145, a specific 5-HT4 receptor radioligand. Data from 15 episodic migraine patients and 16 controls were included for comparison. Quantification of 5-HT4 receptor binding was used as a proxy for brain 5-HT levels, since 5-HT4 receptor binding is inversely related to brain 5-HT levels.
Results
Chronic migraine patients had 9.1% (95% CI: [−17%; −1.0%]) lower 5-HT4 receptor binding compared to controls (p = 0.039). There was no difference in 5-HT4 receptor binding between chronic and episodic migraine patients (p = 0.48) and no association between number of monthly migraine days and 5-HT4 receptor binding (slope estimate 0.003, 95% CI: [−0.004; 0.715], p = 0.39).
Conclusion
The finding of low 5-HT4 receptor binding suggests that cerebral levels of 5-HT are elevated in chronic migraine patients. This is in line with observations made in patients with episodic migraine. Elevated brain 5-HT levels may thus be an inherent trait of the migraine brain rather than a risk factor for conversion from episodic to chronic migraine.
Introduction
Chronic migraine (CM) affects 2% of the population worldwide (1). Chronic migraine patients have higher levels of disability and comorbidity compared to episodic migraine (EM) (2), leading to high levels of absenteeism and reduced productivity (3). Currently, CM is defined as a separate disease entity and is distinguished from EM by an arbitrary frequency limit of 15 headache days per month (4), while the neurobiological mechanisms underlying conversion from EM to CM are poorly understood (2). Putative mechanisms include a combination of increased excitability of neurons in central nociceptive pathways (central sensitization) and dysfunctional pain modulation (5). Serotonin (5-hydroxytryptamine, 5-HT) plays an important role in migraine pathophysiology (6) and, interestingly, 5-HT regulates both cortical excitability (7) and pain transmission (8).
The 5-HT4 receptor is inversely related to brain 5-HT levels, and 5-HT4 receptor binding assessed with PET neuroimaging can thus be used as a proxy for brain 5-HT levels (9). Using this novel neuroimaging method, we recently found that EM patients had lower 5-HT4 receptor binding compared to healthy volunteers (10), indicating higher brain 5-HT levels. These findings were partly in conflict with previous PET studies, which found a lower or no difference in 5-HT synthesis capacity between episodic migraine patients and controls (11,12). However, brain 5-HT levels have not previously been investigated specifically in CM patients and it is unknown whether alterations in brain 5-HT levels could reflect migraine severity and potentially serve as a biomarker of CM. In the present study, we hypothesized that CM patients have higher brain 5-HT levels; that is, lower 5-HT4 receptor binding, compared to both healthy volunteers and to EM patients. In addition, we hypothesized that migraine frequency would be directly related to 5-HT4 receptor binding.
Material and methods
Participants
All participants were recruited from a Danish website for recruitment of volunteers to health research (forsøgsperson.dk), through online adverts or from a local database. Specific inclusion criteria for CM patients were 1) A verified diagnosis of chronic migraine without aura according to the International Headache Society Criteria (13). All data from chronic migraine patients were collected from April 2016 to May 2017. Data from the EM and HC groups were collected from January 2015 to December 2015. Data on EM patients and controls included in this paper have been published previously (10). Briefly, specific inclusion criteria for EM patients were: a) A verified diagnosis of migraine without aura according to the International Headache Society Criteria (13); b) at least one migraine attack every other month but less than five migraine days per month; c) reported previous successful treatment of migraine attacks with sumatriptan. Specific inclusion criteria for healthy controls were having no history of any primary headache and no first-degree relatives with migraine. Furthermore, all participants were eligible for inclusion if they were between 18 and 65 years, did not suffer from psychiatric, cardio- and cerebrovascular diseases, were not pregnant or nursing, and had no daily intake of medication.
All patients underwent a standardized interview including duration of disease (years), frequency (migraine days per month), maximum pain intensity of untreated headache as measured with the Numerical Rating Scale (NRS) (number 0–10), triptan use (days per month) and time since last migraine attack. We allowed CM patients to have headache on the day of the scan; in those cases, we registered pain intensity and type of headache (migraine or tension-type headache (TTH)). Medication intake was not allowed for any participant 24 hours before the scan. All participants had a normal physical and neurological examination and a normal structural brain MRI. All participants filled out the major depression inventory (MDI) (14) on the day of the PET scan.
The study was approved by the Ethics Committee of the Capital Region of Denmark and the Danish Data Protection Agency (H-6-2014-057). The study was registered at Clinicaltrials.gov (ID: NCT01896167). In accordance with the Declaration of Helsinki of 1964, with later revisions, all participants provided their written informed consent to participate in the study after detailed oral and written information and before any study-specific procedures.
Data acquisition
All participants were placed in a supine position on the scanner bed. A specialized head holder was used to reduce head movement. The radioligand, [11C]SB207145, was synthesized using an automated radiosynthesis (described elsewhere (15)). The radioligand was administered as an intravenous bolus injection over 20 s. Immediately after injection, a 120-minute dynamic scan was acquired using a high-resolution research tomography PET scanner (CTI/Siemens, Knoxville, TN, USA). Reconstruction was done as previously described (10). Briefly, a 3D-OSEM-PSF algorithm with TXTV based attenuation correction were used to reconstruct the scans into 38 frames (16–18). All subjects underwent a T1 and a T2 weighted structural MRI scan (Siemens Prisma 3T scanner, Erlangen, Germany), which were used for co-registration with PET and delineation of regions of interest (ROI).
Pre-processing and regions of interest
Each PET frame was aligned to the first five-minutes frame to correct for intra-scan movement using the AIR 5.2.5 software. PET images were then aligned and co-registered to the corresponding T1 weighted MR image using SPM8. Automatic delineation of ROIs was done in PVElab software (www.nru.dk) as previously described (19). Correct co-registration and ROI placement were ensured by visual inspection. Lastly, time-activity curves used for kinetic modeling and grey matter volumes for each ROI were extracted.
The non-displaceable binding potential (BPND) was estimated in MATLAB R2013a (8.1.0.604) 64 bit (Mathworks Inc, MA, USA) using the simplified reference tissue model (SRTM) with the cerebellum (excluding vermis) as reference tissue. This model has been validated for quantification of [11C]SB207145 in the human brain (15). The investigator performing the kinetic modeling was blinded to group status. A volume-weighted neocortical BPND based on 11 brain regions (occipital cortex, orbitofrontal cortex, superior, medial and inferior frontal gyri, insula, superior, medial and inferior temporal gyri, sensory motor cortex, parietal cortex) was calculated by volume weighting grey matter segmented brain region BPNDs:
Genotyping
Neocortical 5-HT4 receptor binding has been shown to be related to the tri-allelic 5-HT transporter-linked polymorphic region (5-HTTLPR) polymorphism (20). Therefore, participants were genotyped and categorized into two groups: 1. Carriers of the short allele (S-carriers) or the long LG allele (LG-carriers) (low-expressing alleles). 2. Homozygotes of the long LA allele (LA-homozygotes) (high-expressing alleles). Genotyping was performed on blood samples drawn at the time of PET scanning and immediately frozen at −20℃. Briefly, the genotyping was performed by PCR amplification from forward primer 5′-TAATGTCCCTACTGCAGCCC-3′ and reverse primer 5′-GGGACTGAGCTGGACAACC-3′ and subsequently digested by the restriction enzyme MspI and separated by gel electrophoresis.
Statistical analyses
Based on a previous study (21), a sample size of n = 15 was deemed sufficient to detect a 15% difference between groups with a power of 0.80 in very large brain regions (>50 cm3) such as, for example, the neocortex. Differences between groups in demographics, genotypes and migraine characteristics were evaluated using Mann-Whitney or Fisher’s exact test as appropriate.
For our group comparisons, our primary outcome measure was log-transformed neocortical BPND. Based on the model fit (examination of quantile probability plots, distribution of the residuals, and predicted values plotted against residuals), we found that this was the appropriate way to model our data so as to not violate the assumption of normality. Our first primary endpoint was to assess group effects (CM vs. HC) on neocortical 5-HT4 receptor binding. To account for the heteroscedasticity within the data used for this analysis, we fitted a model allowing for unequal variance for the two groups using generalized least square. Group status (CM vs. HC) was included as the predictive variable. Since 5-HT4 receptor binding is affected by 5-HTTLPR status, sex, and age, these were all added as covariates to the model (20,22). Our second primary endpoint was to assess the difference between CM and EM patients. This was done using a general linear model including log-transformed neocortical BPND as our primary dependent variable and group status (CM vs. EM) as the predictive variable, and lastly adding 5-HTTLPR status, sex, and age as covariates. No model assumptions were violated using this approach. The differences between CM and HC and CM and EM are reported as percent difference, since this measure is easier to interpret than the corresponding β estimates (relative percent difference = 1−exp(β)). Thirdly, to evaluate the association between the number of migraine days and 5-HT4 receptor binding, data for both EM and CM patients were pooled and analyzed using a general linear model including neocortical BPND as our primary dependent variable and migraine days per month as the predictive variable. 5-HTTLPR status, sex, and age were added as covariates. Again, no model assumptions were violated using this approach. Post hoc, the association between BPND and years with migraine and time since last migraine attack were evaluated using the same general linear model as above (changing the predictive variable accordingly). All statistical analyses were done in R Studio 3.2.3. The significance threshold was set at p < 0.05, and no corrections for multiple comparisons were performed.
Results
Participant characteristics.

Fisher’s exact test. +Mann Whitney. NA: not applicable.
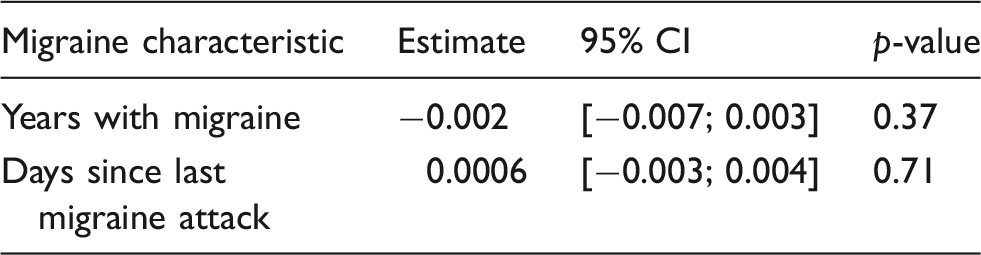
In comparison to healthy controls, chronic migraine patients had significantly lower neocortical BPND (9.1%, 95% CI: [−17%; −1.0%], p = 0.039). This corresponded to a mean BPND ± SD of 0.62 ± 0.09 in the CM group and of 0.67 ± 0.04 in the HC group (Figure 1). We found no difference in 5-HT4 receptor binding between CM and EM patients (4.8 % higher BPND in CM vs. EM, 95% CI [−8.5%; 20%], p = 0.48). Correlation analysis combining CM and EM data revealed no association between the number of migraine days and neocortical BPND (slope estimate 0.003, 95% CI: [−0.004; 0.715], p = 0.39) (Figure 2). Post hoc analyses showed no associations between BPND and years with migraine or time since last attack (Table 2).
Decreased 5-HT4 receptor binding in chronic migraine patients compared to controls. CM patients have lower neocortical 5-HT4 receptor binding compared to controls after adjusting for age, sex and 5-HTTLPR genotype (mean ± SD: 0.62 ± 0.09 vs. 0.67 ± 0.04). Black bars represent mean ± SD. Association between monthly migraine days and 5-HT4 receptor binding. In the sample of 31 migraine patients, frequency of migraine (migraine days pr. month) was not associated with adjusted neocortical 5-HT4 receptor binding (p = 0.39). Green dots represent EM patients. Red dots represent CM patients. Dashed line represents estimated regression line. Grey shade represents 95% confidence interval. Associations between migraine characteristics and BPND after adjusting for age, sex and genotype.

Discussion
An important novel finding of the present study was that CM patients had lower 5-HT4 receptor binding compared to controls but did not differ from EM patients. In addition, there was no association between 5-HT4 receptor binding and monthly migraine days. The inverse relationship between 5-HT4 receptor binding and stable brain 5-HT levels has been corroborated in several both preclinical and clinical studies, showing that pharmacological increases in 5-HT downregulate the 5-HT4 receptor whereas the receptor is upregulated when 5-HT levels are reduced (9,23–26). Additionally, a post-mortem study in suicidal depressed patients showed increased 5-HT4 receptor density in the caudate nucleus and frontal cortex, which were related to decreased 5-HT levels (27). Thus, we interpret our findings of low 5-HT4 receptor binding as reflecting high brain 5-HT levels in the migraine patients.
5-HT has for a long time been implicated in migraine pathophysiology (6), with the main hypothesis being that migraine patients have low brain 5-HT levels between attacks. However, previous PET studies, using uptake of α-[11C]-methyl-L-tryptophan as a surrogate marker of 5-HT synthesis, found similar synthesis rates in EM patients compared to controls (11,12), indicating no differences in brain 5-HT levels. In addition, we recently demonstrated increased brain 5-HT levels, as indexed by low 5-HT4 receptor binding, interictally in EM patients (10). Interestingly, in the current study we found that CM patients also had lower 5-HT4 receptor binding compared to controls, indicating higher brain 5-HT levels in this patient group as well. The precise mechanism of 5-HT in migraine pathophysiology is not fully understood. 5-HT is involved in pain modulation and is generally considered analgesic (28), but in 5-HT depleted animals pain responses are attenuated, indicating that 5-HT is also involved in central pain facilitation (29,30). 5-HT is also present in the descending pain modulation system, and dysfunction of this system, leading to increased facilitation and decreased inhibition of pain, may contribute to persistent pain (31). Thus, the high level of brain 5-HT found in migraine patients could contribute to the underlying pathophysiology of migraine through increases in pain facilitation. In the present study, we therefore also speculated that brain 5-HT levels would be higher in CM patients compared to patients with EM. However, we found no difference in 5-HT4 receptor binding between EM and CM patients and no evidence for frequency of migraine attacks being associated with 5-HT4 receptor binding. Thus, a high level of brain 5-HT could be an inherent trait of the migraine-susceptible brain in general rather than a risk factor for conversion from episodic into chronic migraine.
Although CM patients did not differ from EM patients regarding brain 5-HT4 receptor levels, the composition of other 5-HT receptors might be different in CM patients compared to EM patients. In a recent PET study, we demonstrated that EM patients had lower 5-HT1B receptor density in pain-modulating regions compared to controls and that 5-HT1B receptor binding in the raphe nuclei decreased with proximity to the last migraine attack (32). Pontine dysfunction has been reported interictally in CM patients (33,34), while brain stem activation has been reported in EM patients during attacks (35,36). It would be relevant to explore whether CM and EM patients differ in terms of 5-HT1B receptor binding, especially in the pontine raphe nuclei. Rather than differences in brain 5-HT levels, alterations in 5-HT receptor composition could be the underlying mechanism for differences in cortical excitability between CM and EM patients (5,37). Given that some serotonergic receptors are excitatory and some inhibitory, the action of 5-HT may have differential effects on the cortical neurons and their excitability, depending on which receptor subtype is activated (7).
We acknowledge some limitations of the present study. Firstly, even though no previous findings indicate that that the 5-HT4 receptor is involved in migraine pathophysiology, we cannot exclude that low 5-HT4 receptor density is specific for migraine patients. It could be either genetically determined or congenital and thus independent of brain 5-HT levels. The physiological consequences of low 5-HT4 receptors are unknown. Stimulation of the 5-HT4 receptor has a positive influence on cognition and memory (38), and a low density of the receptor could therefore potentially have negative effects on both. To the best of our knowledge, the 5-HT4 receptor does not play a prominent role in pain modulation, and it is therefore unlikely that a low density of the receptor affects pain modulation in migraine patients. However, since the 5-HT4 receptor is regulating firing rates of 5-HT neurons of the dorsal raphe nucleus in animals (38), it can be speculated that changes in 5-HT4 receptor density affect the overall 5-HT signaling in the human brain. Secondly, our study is cross-sectional and does not allow us to investigate the temporal course of the changes in brain 5-HT levels. Longitudinal studies are warranted to clarify these remaining questions. Thirdly, whether a high level of brain 5-HT is specific for migraine or is present in other pain conditions is currently unknown. Increased plasma 5-HT levels have been found in complex regional pain syndrome, whereas low plasma 5-HT levels have been found in fibromyalgia (8). However, plasma levels do not reflect brain 5-HT levels (39) and 5-HT does not cross the blood-brain barrier (40).
Conclusion
In conclusion, the present results suggest that low 5-HT4 receptor binding, indicating high brain 5-HT levels, may be a trait marker of the migraine brain rather than a risk factor for conversion from EM to CM. While the pathophysiological consequences are not clear, increased brain 5-HT levels could enhance the susceptibility to migraine attacks, while other mechanisms determine frequency and severity. Consequently, reduction of brain 5-HT levels could be effective in the treatment of migraine. Future studies involving other 5-HT receptor subtypes and modulation of brain 5-HT levels in migraine patients are needed to further clarify these mechanisms.
Key findings
Chronic migraine patients have lower 5-HT4 receptor binding, interpreted as higher brain 5-HT levels, compared to controls. Episodic and chronic migraine patients have similar brain 5-HT levels, and migraine frequency is not related to brain 5-HT levels. High brain 5-HT levels may be a trait marker of the migraine brain.
Footnotes
Acknowledgements
We thank all participants for volunteering for this study. Brice Ozenne is gratefully acknowledged for statistical counseling and Bente Dall, Lone Ibsgaard Freyr, Martin Korsbak Madsen, Erik Perfalk, Linda Boje Dalsgaard and Gerda Thomsen are gratefully acknowledged for their excellent technical assistance. The John and Birthe Meyer Foundation is gratefully acknowledged for sponsoring the HRRT scanner.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Gitte Moos Knudsen has received honoraria as a council member of The Brain Prize and the Elsass Foundation and she is on the advisory board for the Kristian G. Jebsen Foundation and field editor for Int J Neuropsychopharm. Messoud Ashina is a consultant and/or scientific adviser/speaker for Allergan, Amgen, Alder, Eli Lilly, Novartis and Teva. All other authors report no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Migraine Research Foundation, the Lundbeck Foundation [R180-2014-3398], Innovation Fund Denmark, the A.P. Møller Foundation for the Advancement of Medical Science, and the Cool Sorption Foundation. The funding sources had no role in the study.

