Abstract
Background
Parry-Romberg syndrome is a neuro-cutaneous disease characterized by progressive hemifacial atrophy. Although common, headache in this population is scarcely reported in the literature.
Objective
To evaluate the clinical features of headache in pediatric and adult patients with Parry-Romberg syndrome, and to discuss diagnostic and treatment approaches of headache in Parry-Romberg syndrome.
Methods
We conducted a systematic review in accordance with PRISMA guidelines. We searched the MEDLINE database to identify eligible studies and identified patients with Parry-Romberg syndrome and headache. We further reported a paradigmatic case with a complex headache disorder and described its management and outcome.
Results
We identified 74 articles, 41 of which were included in the analysis. A total of 52 patients (55.8% female) were included for data analysis. The main age at onset of headache was 20 years (SD 15.2; range 3–56). A diagnosis of migraine was made in 53.9%. Abnormal brain imaging was found in 82.2% of patients.
Conclusion
Long-term follow-up of patients is required, because headache may develop (and evolve) at any time over the course of the disease. Primary and secondary headaches often co-occur in patients with Parry-Romberg syndrome. Further research into the underlying etiopathogenesis and therapeutic targets would be recommended.
Introduction
Parry Romberg syndrome (PRS), also known as progressive facial hemiatrophy, is a rare neuro-cutaneous disease, characterised by progressive but self-limited hemifacial atrophy with unilateral tissue loss that may involve the skin and underlying tissues, most generally involving dermatomes of one or multiple branches of the fifth cranial nerve (1). The disease is usually diagnosed in childhood, and females are more affected than males (2). The etiology of PRS is still unclear. Possible factors in its pathogenesis include genetic predisposition, autoimmunity, trauma, vascular abnormalities, sympathetic nervous system dysregulation, anomaly of the trigeminal nerve, and infections (3–5). PRS is typically sporadic, and no definite genetic cause has been identified. To date, only two families with PRS have been described (6,7).
Extracutaneous manifestations, which mainly involve mucous membranes and the nervous system, are common and may appear at any phase of disease (8). Neurological manifestations occur in up to 15% of the cases, and include headache and facial pain, epilepsy, hemiparesis, movement disorders and neuropsychological symptoms (9). These symptoms are usually slowly progressive, and may remain stable for many months or years, or rapidly worsen following the underlying disease’s course. Imaging studies may reveal unilateral brain lesions on the side of the mucocutaneous atrophy and are of great value to support the diagnosis and assess the severity and extent of the disease (9).
Headache is frequent in patient with PRS (9). In a worldwide online survey on 205 patients with PRS, 52% declared that they suffered from concurrent migraine (10). Headache has a relevant impact on quality of life, adding to depression, anxiety, and antisocial personality traits caused by facial disfigurement (11). Despite its clinical and psychological burden, however, headache has been scarcely characterised in patients with PRS.
We report a patient with PRS as a paradigmatic case of multifaced difficult-to-treat headache disorder in this syndrome, and perform a systematic review of the literature on headache and PRS. Our aim is to increase awareness and clarify the clinical features of headache in patients with PRS.
Materials and methods
Design
We performed a systematic review to answer the following questions: What is the prevalence of headache in patients with PRS, and how do demographic factors affect its manifestation? What are the clinical characteristics and proposed treatments of headache in such a population? What are the main theories of its pathogenesis?
Database and search strategy
We searched PubMed (Medline) through 19 July 2020. The MEDLINE research query was: Parry-Romberg syndrome OR progressive hemifacial atrophy AND (headache OR migraine). We additionally searched for the references of the related published articles to identify potential additional papers. Two researchers (AR and TF) independently screened the reference lists of the eligible articles. Full texts of records deemed eligible were retrieved and independently assessed for inclusion by the same investigators. Any discrepancies were resolved by consensus.
Case selection criteria
Inclusion criteria for patients with PRS and headache were set a priori. We included original articles of retrospective cohort studies, case series, case reports, and short communications in the English language without limits on year of publication. Letters to the editor, reviews, scientific posters and congresses were excluded. The eligibility criteria for inclusion requested that PRS was the leading diagnosis of the patient(s). Finally, articles of PRS patients without headache, or in which the main diagnosis was linear scleroderma, or with insufficient data were excluded. The third edition of the International Classification of Headache Disorders (ICHD) (12) was applied to recognise and categorise headache disorders. All selected cases were reviewed from detailed information when available, including sex, age at PRS presentation, age of headache onset, headache types and features, additional neurologic symptoms or diseases, imaging data, treatment and outcome.
Case report
We report one patient evaluated in the Pediatric Clinic of IRCCS San Matteo Hospital Foundation, Pavia (Italy) since 1992. Data collection was retrospectively obtained from medical records.
Case report
We report the case of a 28-year-old woman with PRS and typical neurological symptoms including difficult-to-treat headaches. She was born at term to a non-consanguineous couple with no family history of genetic or neurologic syndromes. She was referred to our observation at 12 months of age for a progressive sunken-in appearance of the left side of her face. Physical examination showed a left-sided facial asymmetry extending from the frontal bone to the mandibular region, homolateral iris dyschromia, microphthalmia and palpebral pseudoptosis, with a 1.0 × 1.5 cm hypopigmented patch of alopecia on the left frontoparietal region. Neurological examination revealed a right hemiparesis predominantly involving the upper limb. Brain magnetic resonance imaging (MRI) showed left cerebral hemi-atrophy, with compensatory external hydrocephalus, an enlarged left lateral ventricle and periventricular white matter T2-hyperintensity: A diagnosis of PRS was made.
At 1 year and 6 months of age, she experienced a focal to generalised tonic-clonic seizure and she was started on phenobarbital. Interictal electroencephalography (EEG) showed slow delta activity over the left frontal regions, and temporal spike-wave complexes. Ocular complications occurred, as she developed a left retinal detachment with extensive vision loss followed by phthisis bulbi at the age of three, leading to surgical enucleation of the left eye and cosmetic insertion of an ocular prosthesis.
At the age of 8, she begun to suffer from weekly severe headaches. Pain was localised in the left frontal-parietal region, lasted several hours, was not associated with typical migraine symptoms, and did not significantly improve after sleep or non-steroid anti-inflammatory drugs (NSAID) administration. An angio-MRI showed hypoplasia of the intracranial left carotid system. A diagnosis of tension-type headache was postulated, physiotherapy was implemented, and a symptomatic treatment with paracetamol was prescribed.
At the age of 13 she had severe chronic recurrent headaches, which did not respond to paracetamol or NSAID treatments. Clinical and radiological follow‐up showed progression of her left facial atrophy (Figure 1).

Fourteen-year-old girl with PRS. (a) Note the left-sided hemifacial atrophy with wasting of subcutaneous tissues and thinning of the skin. Hyperpigmentation is present in the left hemifacial region and left side of the neck (with permission from parents). (b) A coronal MRI image demonstrating atrophy of the left cerebral hemisphere, enlargement of the left pericerebral spaces, mild asymmetry of scalp thickness with relative paucity of subcutaneous fat on the left hemifacial side, absence of the left eyeball and left maxillary sinus hypoplasia. (c) A volume-rendered three-dimensional computer tomography image highlighting bony asymmetry of the face. The bony atrophy involves the left sided facial skeleton, particularly the left periorbital, maxillary, and mandibular areas.
Brain MRI imaging findings at the age of 14 highlighted multiple anatomical abnormalities and complications of PRS (Figure 2). Neither endovascular nor neurosurgical treatment options were available, therefore annual radiological follow-up by angio-MRI was established. Phenobarbital was stopped, and pregabalin (50 mg twice a day) was initiated in the attempt to control her pain, without response.
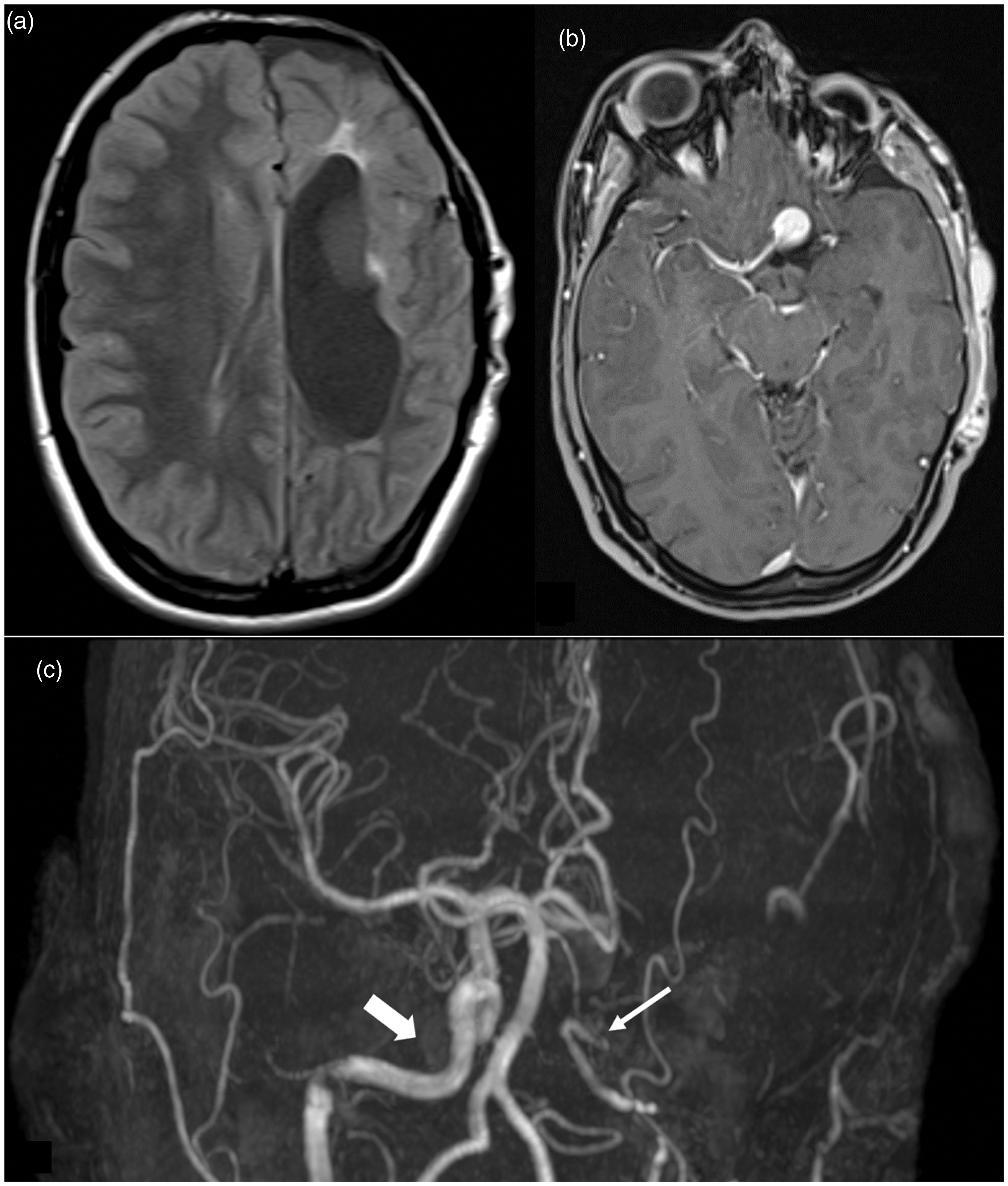
(a) A baseline axial fluid attenuated inversion recovery (FLAIR) MRI revealing several T2-hyperintense lesions, involving the left periventricular white matter and external capsule, with an associated pseudocyst (not shown), extending to the posterior limb of the internal capsule. All lesions are compatible with gliotic/malacic sequelae. An increased ex-vacuo dilatation of the left lateral ventricle was also noted. (b) An axial contrast enhanced MRI showed an intracranial aneurysm of the anterior communicating and left anterior cerebral arteries. (c) An angio-MRI showing an intracranial fusiform aneurysm of the anterior communicating and left anterior cerebral arteries (diameter: 10 mm) and hypoplasia of the intracranial left carotid system (thin arrow), with a compensatory overflow of the right carotid system (thick arrow) resulting in an asymmetric circle of Willis.
At the age of 19, left-sided headache attacks increased in frequency (two or three times per week) and duration (up to 12 h), now frequently associated with photophobia, vertigo and nausea, meeting diagnostic criteria for migraine without aura. Violaceus rashes of the face, blurred vision, and attention deficits at school occurred. Therefore, pregabalin was switched to topiramate (80 mg twice a day) 6 months later, with a satisfactory response.
One year later, hemifacial atrophy drastically and rapidly worsened and the patient was started on methotrexate. This treatment lasted 3 years and the girl responded with considerable aesthetical improvements (regression of alopecia with 70% of hair regrowth at the end of therapy). Meanwhile, after 2 years of well-controlled headaches, topiramate’s effect somehow waned over time. Migraine attacks often appeared after waking up in the morning, or they were provoked by acoustic stimuli such as a dog barking. The pain was moderate in intensity, constant and localised to the anterior left hemisphere, associated with photo- and phonophobia. She denied migraine aura-like symptoms, but she referred constant persistence of subjective vertigo with nausea and sporadic episodes of vomiting. Otolaryngology evaluation led to a diagnosis of a left vestibular deficit, and suggested lifestyle modifications including exercise and vestibular rehabilitation interventions. Therapeutic trials with carbamazepine and amitriptyline/chlordiazepoxide were performed. Follow-up visits showed stabilisation of the hemifacial atrophy, without further brain modifications on MRI.
The patient is currently seizure free, although EEG shows persistent anomalies in the left fronto-parietal regions. She is still in therapy with amitriptyline/chlordiazepoxide and carbamazepine, with recurrence of mild migraine attacks once every 3–4 months, well controlled by symptomatic NSAID. She has mild cognitive impairment and suffers from anxiety and mild depression.
Results
Literature search
The PubMed search yielded 53 returns, of which 50 were in English. All 50 references were reviewed in full by the search authors. The references of these 50 were reviewed by hand to identify further references, leading to the identification of 21 additional ones, all of which were reviewed. We retrieved 41 articles meeting the eligibility criteria (PRISMA flow diagram is reported in Figure 3).
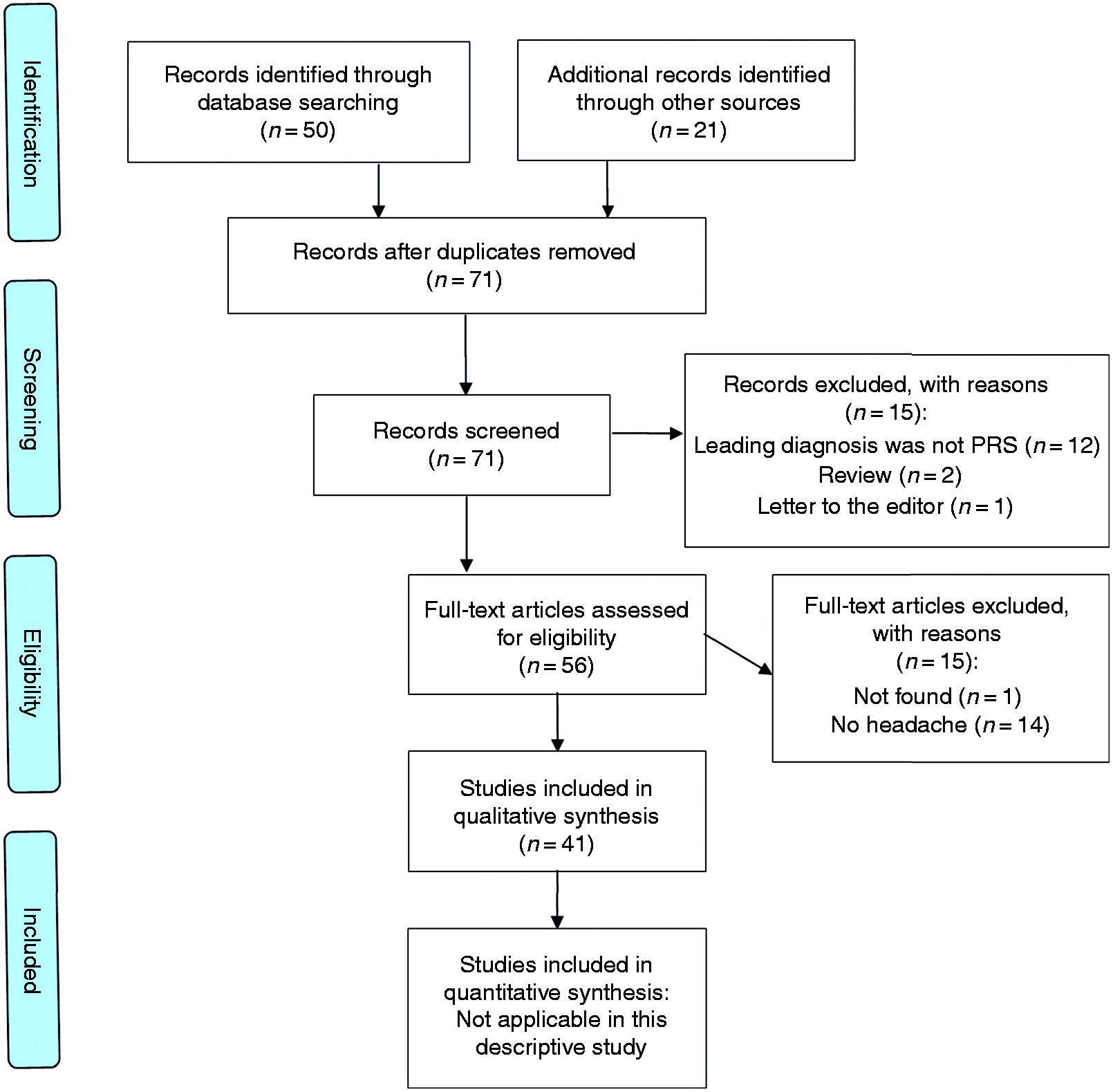
Flow diagram of study selection.
Analysis of all cases
A total of 52 patients (51 from literature and the one reported here) were included for data analysis. Most were reported as small series, or single cases. The clinical features of the patients (27 children (51.9%), 25 adults (48.1%)) are summarised in Tables 1 and 2.
Summary of the characteristics of children included in the review.

AZA: azathioprine; BTX-A: botulinum toxin type-A; CBZ: carbamazepine; CS: corticosteroid; ECDS: en coupe de sabre; F: female; HCQ: hydroxychloroquine; HM: hemiplegic migraine; IVIG: intravenous immunoglobulins; L: left; LEV: levetiracetam; LTG: lamotrigine; M: male; M: migraine; MA: migraine with aura; MMF: mycophenolate mofetil; MO: migraine without aura; MTX: methotrexate; N: patient number; NR: not reported in original article; NSAIDs: nonsteroidal anti-inflammatory drugs; OXC: oxcarbazepine; PRS: Parry-Romberg syndrome; R: right; TPM: topiramate; TTH: tension-type headache; VPA: valproic acid.
Summary of the characteristics of adults included in the review.

ASA: aspirin; CBZ: carbamazepine; CS: corticosteroid; ECDS: en coupe de sabre; F: female; HM: hemiplegic migraine; L: left; LEV: levetiracetam; M: male; M: migraine; MA: migraine with aura; MO: migraine without aura; MTX: methotrexate; N: patient number; NR: not reported in original article; NSAIDs: nonsteroidal anti-inflammatory drugs; PRS: Parry-Romberg syndrome; R: right; STA-MCA: superficial temporal artery to middle cerebral artery; TPM: topiramate; TTH: tension-type headache; VPA: valproic acid.
Twenty-nine patients (55.8%) were female. Age at onset of PRS was reported in 42/52 (76.9%) cases, with a mean age of 14.2 years (SD 12.8; range 1–45 years). Of the remaining 10 cases, seven were described among pediatric cohorts, three of whom had symptomatic onset in infancy or early childhood, while data are missing for the three remaining patients (20,21,35).
Age at onset of headache was reported in 32/52 (61.5%) cases and was 20 years (DS 15.2; range 3–56). Of these, 12 patients had their first headache attack in the first decade of life, eight in the second, four in the third, while the age of onset of headache was described as between the fourth and the sixth decades of life in the remaining eight patients.
A diagnosis of migraine was made in 28/52 (53.9%) patients. Type of migraine is specified in 10 cases: Three had migraine without aura, five had migraine with aura (four of whom developed hemiplegic migraine), one suffered from chronic migraine, while another case developed a status migrainosus.
Lateralisation: The atrophic hemiface was the left side in 30/52 (57.7%) patients. In 16 subjects, a unilateral headache is reported: In 14/16 (87.5%) cases, pain involved the same side as hemifacial atrophy (the left side in 10 and the right side in 4). Bilateral or alternating sides were described in two patients (19,35).
Pain features: Pain localisation was reported in 8/52 (15.4%) patients, including our case. Pain intensity was only explicitly described in 6/52 (11.5%) patients and was generally severe (5/6); it is referred as mild only in one patient. Pain quality was reported in only 4/52 (7.7%) cases as throbbing or stabbing.
Sensory and motor associated symptoms: In 6/52 (11.5%) cases, dysesthesia and motor deficits in the opposite side to that affected by hemifacial atrophy were also reported. Pichiecchio et al. (37) reported a woman with hemiplegic migraine, characterised by attacks of paresthesia in most of her right hemisoma, weakness and visual scotomas lasting un to 60 min, followed by contralateral headache with photophobia, nausea and vomiting. Instead, Cory et al. (14) reported dysesthesia in the right hand and leg, followed by transient and completely reversible hand paresis and clumsiness. In one case of hemiplegic migraine, hemisensory loss was reported along with intermittent right hemiplegia followed by generalised throbbing headache (36). Kacinski et al. (21) described a 12-year-old boy with intense headaches and a constellation of associated symptoms including numbness of the right leg and motor weakness of both legs, sometimes followed by tonic-clonic seizures. Finally, Zakkiriah et al. (30) described a 14-year-old boy with left frontotemporal headache lasting 5 min during transient stroke-like attacks (right hemisoma numbness and heaviness).
Other accompanying symptoms: Photophobia was noted in 8/52 (15.4%) patients including our case, associated with phonophobia, nausea and vomiting in six cases. Gastrointestinal symptoms (nausea and/or emesis) are described in 8/52 (15.4%) cases, including our patient. Blurred vision (21,50), diplopia (21,35), scotoma (37), visual auras (45) or intermittent visual loss (50) were noted in some cases. In one case, the individual had a progressive loss of vision with impaired ocular movements due to a thrombosed giant aneurysm of the cavernous carotid artery of the same side (35). One patient also noted disturbed balance, reddening, sweating of the face, and hot flushes (21). Three patients experienced aphasia (14,34,36), while one reported dysphasia (13).
A total of 25/52 (48.1%) patients exhibited one or more neurological signs on examination: Hemiparesis or monoparesis were described in 8/52 (15.4%) cases, lateralised sensory deficits (primarily hypoesthesia or paresthesia) in 6/52 (11.5%), while ophthalmic manifestations occurred in 15/52 (28.9%) patients. In particular, 6/52 (11.5%) patients were noted to have enophthalmos, 3/52 (5.8%) noted ptosis, while progressive monolateral vision loss was described in 3/52, hemianopia in another 3/52 and intermittent monolateral mydriasis in 1/52 (1.9%).
Seizures were reported in 12/52 (23.1%) patients.
Brain imaging, including brain CT and/or MRI, was available in 45/52 (86.5%) cases, and abnormalities were found in 37/45 (82.2%) patients: CT of the head was performed in 18/52 (34.6%) and 16/18 (88.9%) patients had abnormal brain findings, while brain MRI was performed in 39/52 (75.0%) and was abnormal in 32 (82.1%). Among the patients with abnormal brain MRI, 11 (34.4%) subjects underwent CT, resulting abnormal in all cases, with subcortical calcifications as the most common abnormalities (5/11; 45.5%). The most common MRI abnormalities were T2 hyperintensities of the subcortical white matter, which could be localised or diffuse, and when localised were more frequent in the frontal lobe. In addition, cerebral atrophy (mainly ipsilateral to hemifacial alterations, and hemispheric) and calcifications were described as common associated features, reported in 7/45 (15.6%) and 8/45 (17.8%) cases, respectively. Cerebral aneurysms were reported in 6/45 (13.3%) cases.
Information about anti-headache treatment was only specified in 15/52 (28.9%) patients: Symptomatic NSAIDs and paracetamol were proposed as first line therapy in 7/15 (46.7%) cases, and were reported as mostly ineffective. Data on headache outcome were reported in 18/52 (34.6%) patients, in 3/18 (16.7%) of whom attacks were poorly controlled, with a tendency to deteriorate and increase in frequency over time. All subjects had tried various drugs with limited success. An improvement in frequency and/or intensity or a relief of headache is reported in 8/18 (44.4%) and 7/18 (38.9%) cases, respectively.
Discussion
PRS is a rare progressive disease causing hemifacial atrophy and a variety of neurological symptoms over time. Incidence ranges from 0.3 to 2.5 cases per 100,000 population per year (9,52), with a 3:1 female:male ratio (1,9). Even though late-onset cases have been reported (10), PRS typically begins in the first decade of life (1), and slowly progresses over 2–20 years before stabilising (9).
Although certainly rare, PRS is possibly underdiagnosed due to a lack of standardised criteria for diagnosis, and the overlapping features with linear scleroderma (“en coup de sabre”, ECDS) (3,53,54). At present, there is a tendency to consider them variants of localised scleroderma, although these conditions may also coexist in the same patient (3). ECDS is characterised by unilateral band-like sclerosis on the frontoparietal area, usually preceded by induration of the skin and not extending below the eyebrow. Instead, in PRS, there is paramedian atrophy of the face without overlying skin induration. In PRS, histopathological features include atrophy of subcutaneous and underlying fat, muscle, and osseocartilaginous tissues (3,53). Although headache is a possible manifestation of both entities, Paprocka et al. (53) also described a more varied spectrum of neurological symptoms in PRS than in ECDS, which include vertigo, trigeminal neuralgia, unilateral facial palsy, and cognitive abnormalities. Since no consensus exists, for the purpose of this review we only considered PRS patients (possibly with concomitant ECDS), aiming to describe a more homogeneous sample.
Neurologic involvement is not infrequent among the PRS population and headache is one of most frequent reported in the literature (9,10). In a review of neurologic manifestations in adults and children with PRS, pain was described in 57/129 cases (44.2%): Of these, headache was reported in 46/57 cases (80.7%), while facial pain occurred in 11/57 cases (19.3%) (9).
Migraine is a common disabling primary headache disorder (55). Any link between migraine among young subjects (especially women) and PRS is rather questionable, though it seems more frequent in PRS than in the general population (affecting 52% of 205 patients with PRS surveyed using the internet) (10). Our retrospective analysis identified a diagnosis of migraine in 53.9% of patients with PRS and headache. Prospective studies focused on migraine in the PRS population are needed to confirm this data. Severe migrainosus states, with frontoparietal white matter lesions on MRI, have also been described (10,19). In our patient, after a first period of tension-type headaches, she clearly developed migraine without aura, and probably she still experienced both headache types during adulthood. However, it is important to consider that hemifacial skin atrophy with underlying tissue loss can be an independent etiologic factor for secondary headache (12). In most patients with PRS, headache episodes arise after the onset of progressive hemifacial atrophy. In this review, age at onset of PRS was 14.2 years, while age at onset of headache was 20 years, and headache attacks anticipated facial manifestations in only two cases. This may suggest a causal relationship between headaches and facial manifestations. Interestingly, in the majority (59.4%) of PRS patients with headache, onset of cephalalgia occurred during childhood or adolescence. Moreover, the secondary nature of headaches in patients with PRS might be supported by the fact that headache is often localised and ipsilateral to hemifacial atrophy. In this review, headache was ipsilateral with the hemifacial atrophy in 87.5% of patients. New onset chronic headache and abnormal neurological examination are well-defined alerts for a secondary origin of a headache, especially in patients with PRS. When this occurs, a brain imaging (MRI or CT scan) should be promptly performed.
In our review, brain imaging (CT scan and/or MRI) had been performed in 90% of the patients (Table 3 and 4) and was abnormal in 82.2% of the cases. In a recent review of patients with PRS and neurological manifestations, only 25.4% of the subjects had a normal brain MRI. Indeed, brain MRI frequently revealed abnormalities, mostly involving the hemisphere ipsilateral to the skin atrophy.
Neuroimaging findings in children included in the review.

ASL: arterial spin labeling; CT: computed tomography; DSA: digital subtraction angiography; FLAIR: fluid attenuated inversion recovery; HI: hyperintensity/hyperintensities; ICA: internal carotid artery; MCA: middle cerebral artery; MRA: magnetic resonance angiography; MRI: magnetic resonance imaging.
*Soft tissue and calvarial abnormalities were not uniformly described by all authors and are not shown. Only central nervous system alterations are reported in this table.
§Ipsilateral and controlateral always refer to the side of the facial hemiatrophy.
Neuroimaging findings in adults included in the review.

CT: computed tomography; FLAIR: fluid attenuated inversion recovery; HI: hyperintensity/hyperintensities; ICA: internal carotid artery; MCA: middle cerebral artery; MRA: magnetic resonance angiography; MRI: magnetic resonance imaging; PCA: posterior cerebral artery.
*Soft tissue and calvarial abnormalities were not uniformly described by all authors and are not shown in this table.
§Ipsilateral and contralateral always refer to the side of the facial hemiatrophy.
Cerebral aneurysms are often described as infrequent in patients with PRS (20,26,39,44). In our review, 11.5% of patients with PRS and headache had at least one aneurysm. However, the actual incidence remains unknown, since not all the patients underwent head CT scan, brain MRI, or cerebral angiogram. Cerebral aneurysms are rarely symptomatic and in this case they present with sudden headache due to rupture and subarachnoid haemorrhage (44). However, some pathogenic hypothesis have tried to link headache with unruptured aneurysms, including: a) Local thrombosis, stretching or dissection in the aneurysm wall; b) intramural bleeding; c) localised meningeal inflammation; d) occult or sentinel haemorrhage; e) perivascular sensory nerve terminals around the aneurysm rising central nervous system sensitisation; f) expansion or inflammation of the aneurysm (56). Whether cerebral aneurysms act as a mere bystander or as a direct cause for some headaches remains unclear, and we do not know if any treatment of such unruptured aneurysms might lead to any symptomatic improvement (56,57).
Other headache attributed to cranial and/or cervical vascular disorder can be due to spontaneous carotid-jugular fistula or carotid dissection (35,44). In our patient, vascular alterations were detected as soon as she was 3 years old. In fact, a supra-aortic trunk echo-Doppler revealed hypoplastic left internal and common carotid arteries with increased flow resistance in both arteries, suggesting a concomitant hypoplasia of the respective intracranial districts. An angio-MRI confirmed the presence of a hypoplastic left internal carotid artery, with absence of flow in its petrous-cavernous-supraclinoid tracts, and a dramatic left middle cerebral artery hypoplasia. Even if PRS may involve the cerebral or cervical vessels, symptomatic ischemic strokes are infrequent, and were only found in 3.9% of the patients. A similar percentage (5.7%) has been found by Vix and colleagues (9) among patients with PRS and neurological manifestations. In any case, a stepwise approach is essential to avoid missing secondary headaches in patients with PRS.
The etiopathogenetic mechanism for progressive facial atrophy and neurological manifestation remains controversial. Verma et al. (45) hypothesised that a compromised trigeminal-vascular system could be involved in both headache and facial atrophy. On the other hand, Cory et al. (14) have postulated that a hyperactivity of the sympathetic nervous system may act as the common cause of progressive hemiatrophy and headache. In the literature, not only neural dysfunction but also local trauma and infection (particularly neuro-Lyme disease) have been suggested as possible causative factors (42,58).
Mood disorder is a frequent comorbidity in headache (59), and depression due to facial disfigurement should always be considered as a triggering or worsening factor of headache in episodic and chronic headache sufferers with PRS. In this context, administration of selective serotonin reuptake inhibitors (SSRIs) and tricyclics as prophylactic treatments for both disorders could be a therapeutic strategy. Indeed, treatment with amytriptiline/chlordiazepoxide provided satisfactory long-term headache control in our patient.
Finally, specific treatment guidelines for headache in patients with PRS are lacking. NSAIDs and triptans are the mainstays of acute treatment of migraine (60). In particular, ibuprofen (10 mg/kg) is recommended as an initial treatment in both children and adolescents. For adolescents, triptans are also recommended, specifically sumatriptan/naproxen tablets, sumatriptan or zolmitriptan nasal spray, rizatriptan oral dissolving tablets, and almotriptan tablets (61,62). However, these drugs should be cautiously administered in patients with PRS because of their potential vessel instability and risk of vasospasm. Moreover, in the setting of vascular abnormalities, such as aneurysms, safety issues of triptans still need to be fully addressed (56). According to our review, NSAIDs are the most-used symptomatic drugs in patients with PRS and acute headache attacks. Headache prophylaxis includes beta blockers such as propranolol (33,41,45), tricyclic antidepressant amitriptyline used in our patient, calcium-channel blockers such as flunarizine (45), and anti-seizure drugs such as valproate (36) and topiramate (23,29). Injections of onabotulinumtoxin A (Botox) about every 12 weeks and topiramate are the only currently available treatments with high-quality evidence for the prophylaxis of chronic migraine from more than one randomised controlled trial (63). This treatment has been also described for various symptoms of PRS including headache, jaw and facial pain, and dystonia in pediatric and adult populations (31,64,65), but further studies on its efficacy and safety for these indications are needed. Immunosuppressive drugs used in the therapy of skin and underlying tissue lesions may also have some impact on headache control, as well as surgical reconstruction of progressing hemifacial atrophy to treat deformities after at least 2 years of disease stability. One of the limits of most studies is that patients received two or more medications at the same time, limiting the assessment of efficacy of any single management independently.
We are cautious about the conclusions of this review not only because of the retrospective nature of all reports, but also for a tendency to preferentially publish the most challenging cases, especially case reports, and this may not be representative of the people with PRS. In most cases, headache was not the main focus of the description. Therefore, information regarding headache features was often limited, and there was a tendency to report other specific aspects of this complex disease, such as radiological features, facial disfigurement, and surgical reconstructive techniques. As a result, data concerning headache could potentially suffer from publication bias. Taken together, these limitations strongly underline the need for a better description and classification of headache and associated neurological conditions in both adults and children with PRS.
Conclusions
The shared features of the 52 cases summarised in this systematic review allow for a description of the clinical phenotype of patients with PRS and headache. Headache appears to be not infrequent, but probably under-reported and therefore possibly underestimated in people with PRS. This happens despite headache control being among the most challenging clinical problems faced by these patients and their physicians. Long-term follow-up of patients with PRS is required, given the evidence that headache may develop (and evolve) at any time over the course of the disease. The underlying cause of headache often remains unclear, therefore further research into the underlying etiopathogenesis and therapeutic targets would be strongly recommended.
Clinical implications
What is the prevalence of headache in patients with PRS, and how do demographic factors affect its manifestation? What are the clinical characteristics and proposed treatments of headache in such population? What are the main theories of its pathogenesis?
Footnotes
Ethical statement
We confirm that we have read the Journal’s position on issues involved in ethical publication and affirm that this report is consistent with those guidelines. Our patient gave their informed consent for inclusion before she participated in the study. Informed consent was obtained from the participant, and consent to use images was also included in the study. Informed consents will be available upon request to the corresponding author.
Acknowledgments
AR and TF equally contributed to study conception, data extraction, risk of bias rating and manuscript writing. SS contributed to study conception and design, analysis, and interpretation of data. AR and TF conducted the article retrieval. SS revised the manuscript critically for important intellectual content. All authors approved the final version of the manuscript, including the authorship list.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
