Abstract
Background
The present study aimed to determine whether machine-learning (ML)-based models can predict 3-, 6, and 12-month responses to the monoclonal antibodies (mAbs) against the calcitonin gene-related peptide (CGRP) or its receptor (anti-CGRPmAbs) in patients with migraine using early predictors (up to one month) and to create an evolving prediction tool.
Methods
In this prospective cohort study
Results
Three hundred thirty-six patients treated with anti-CGRP mAbs were included. The external cohort included 93 patients treated with anti-CGRP mAbs. We developed six models to predict 3- 6- and 12-month responses using early predictors. ML-based models yielded predictions with an accuracy score in the range 0.40–0.73 and an AUC-ROC score in the range 0.56–0.76 during internal testing and yielding predictions with an accuracy in the range 0.39–0.64 and an AUC-ROC score in the range 0.52–0.78 when tested against an external test cohort. Shapley Additive explanations summary plots were generated to interpret the contribution of each feature for each model. Based on these findings, a response prediction tool was developed. Each model was run through a backward feature selection to find the most relevant features for the models. The MHDs reduction of the previous data point tends to be the most relevant, while the migraine with aura indicator tends to be the least effective predictor.
Conclusions
The response prediction tool utilizing evolving ML-based models holds promise in the early prediction of treatment outcomes for patients with migraine undergoing anti-CGRP mAbs treatment.
Introduction
The monoclonal antibodies (mAbs) against the calcitonin gene-related peptide (CGRP) or its receptor (anti-CGRP mAbs) are increasingly used for the prophylactic treatment of migraine and have demonstrated effectiveness in patients with episodic migraine (EM) and chronic migraine (CM) (1).
Because of the widespread use of anti-CGRP mAbs for migraine prevention, their cost, the potential long-term treatment needed for numerous patients and the reimbursement rules still imposed by national health services in some countries, there is a growing emphasis on identifying response predictors to customize treatment approaches. Real-world studies have attempted to identify response predictors among different patient subgroups with inconsistent results (2).
A machine learning (ML) algorithm is a computer program specifically engineered to enhance its performance autonomously through data-driven learning. These algorithms employ statistical models to detect patterns and correlations within data and employ these patterns to formulate predictions or decisions. Furthermore, the iterative nature of ML models allows them to continuously learn and adapt, incorporating new information (3).
These ML algorithms are increasingly used to support the diagnosis, treatment and prognosis of several neurological disorders (4). Concerning migraine, some studies have employed ML techniques to classify primary headache disorders (5), and a limited number of investigations have focused on forecasting the response to onabotulinumtoxinA (BTX-A) and CGRPmAbs treatments (6,7). Furthermore, ML algorithms can also extract prognostic insights from demographic, clinical, and biochemical data (8).
The utilization of early predictive factors for assessing the treatment response to anti-CGRP mAbs could allow cost optimization and personalized treatment strategies, thereby favoring the use of these drugs in those patients who are likely to respond positively (9). To date, there are no ML algorithms that incorporate early predictive features to evaluate the effectiveness of anti-CGRP mAbs.
In the present study, we aimed to determine whether ML-based models can predict 3- 6- and 12-month responses to anti-CGRP mAbs in patients with migraine using early predictors (up to one month) and to create an evolving online prediction tool applicable in clinical settings.
Methods
Design, period and setting
In this observational analytic study with prospective cohort design, all consecutive patients treated with anti-CGRP mAbs (anti-ligand or receptor) over a period of 12 months at two outpatient headache centers (Careggi University Hospital in Florence and Policlinico Gemelli in Rome) were included. The inclusion period ranged from 1 December 2019 to 28 February 2023. The external test cohort included patients treated with anti-CGRP mAbs prospectively recruited from the same centers at a different time period; specifically, from 28 February 2023 to 1 March 2024.
Data were collected with standardized data collection instruments. All patients provided their written informed consent. The study was reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (i.e. STROBE) guidelines and is part of the Registro Italiano Cefalee (RICe) study, which was approved by the local Ethics committee (CEAVC Studio RICe, 14591_oss and subsequent amendments 2022–609).
Study participants, patients selection and study variables
The study participants were adult patients with migraine treated with anti-CGRP mAbs. Participants were included if they were older than 18 years, had a diagnosis of EM or CM according to the International Classification of Headache Disorders, 3rd edition (ICHD-3) criteria (10) with or without medication overuse (MO), and started a preventive therapy with an anti-CGRP mAb (erenumab 70–140 mg monthly; galcanezumab 240 mg first dose and 120 mg monthly; or fremanezumab 225 mg monthly) according to the local standard of care. Patients were excluded if they declined to provide informed consent. Medication overuse was codified according to ICHD-3 criteria, regardless of the diagnosis of MO headache. Before starting the first anti-CGRP mAb, all patients reported lack of effectiveness (no meaningful improvement in the frequency of headache after the administration of drugs for ≥3 months at appropriate dose) of at least three previous migraine preventive treatments, including β-blockers, tricyclic antidepressants, antiseizure medications and BTX-A.
Three months before the anti-CGRP mAb prescription and throughout the treatment period, patients completed a paper headache diary. This diary recorded the number of monthly headache days (MHDs) and the use of acute medication, including the absolute number of analgesics (AMNs) and the number of days with at least one analgesic [acute medication use (AMDs)] per month. A headache day was defined as any day a patient recorded experiencing any type of headache.
Response rates were assessed based on reductions in MHDs, with categories defined as <25%, 25–50%, 50–75% and >75% reduction in MHDs (11). Additionally, because patients reported outcome measurements, they completed the Headache Impact Test (HIT-6) questionnaire on a monthly basis and the Migraine Disability Assessment (MIDAS) questionnaire every quarter. At baseline and every three months, patients were evaluated by a headache expert, who conducted an in-person clinical interview and collected a series of variables, including demographic, clinical, and treatment-related variables. Adverse events were systemically assessed and reported for all patients.
Patients who stopped the treatment due to ineffectiveness were categorized as unresponsive (non-responders) and included in the analysis. Patients who stopped the treatment for reasons that were not related to the treatment effectiveness (such as adverse events) were excluded from the model dataset.
Statistical analysis
There was no sample size estimation in advance, but based on prior studies (9,12), a sample size >300 was considered adequate. Demographic and baseline characteristics were summarized descriptively; namely as the mean ± SD or median interquartile range (IQR) for continuous variables and number (percentage) for categorical data. Normality assumption was assessed using the Shapiro–Wilk test. Missing data management is described in the paragraph below.
Machine learning analysis
Various classification models, such as gradient boosting trees, logistic regression, histogram gradient boosting classifier, decision trees and random forest, were trained using a training set and then tested and evaluated against a test set to find the most accurate model to fit the data. The random forest algorithm was chosen based on superior performance measured by average F1-score values across all models on the test data once each model was fitted on the training set. The F1-score is the harmonic mean of the precision and recall. It is used in statistics to measure the accuracy of two classifications and assume that recall and precision are equally important (13). Classification models were trained for the prediction of treatment response rates of <25%, 25–50%, 50–75% and >75% reduction in MHDs. The models were measured using performance metrics such as accuracy, precision, recall and F1-score. The models were one-hot encoded to generate a multi-class receiver operating characteristic (ROC) curve for each model to plot the true positive rates against the false positive rate to visualize how well the models distinguish between different classes and the area under the ROC curve (AUC-ROC) was measured. Additionally, Shapley Additive explanations (SHAP) summary plots were generated to interpret the contribution of each feature for each model's output. The SHAP summary plots indicate the importance of each input feature in predicting the outcome of the model. Each point in the graph signifies an individual data point. The horizontal axis corresponds to the SHAP value, and the color of each point indicates whether the observation possesses a higher or lower value relative to other data points. Features exhibiting more positive values denote a robust positive correlation with the output, whereas features with more negative values signify a pronounced inverse or negative correlation. Features with values closer to 0 suggest a weak correlation (14).
Each model was given 11 baseline and demographic data inputs that are readily assessable in clinical practice (type of anti-CGRP mAb, age, sex, migraine diagnosis, disease duration, aura, MHD, AMD, HIT-6, number of analgesics and chronicization onset) and month-based predictors for months 1, 3 and 6. The time-dependent features (HIT-6, AMD and MHD) were calculated as percentage change compared to baseline to normalize the range in values. The dataset was split into 80%-20% sets to train and test the models, respectively. The models were fitted using the training data and accuracy measures were evaluated on the results obtained by running the model against the test data. Each model was run through a sequential backward feature selector to train the model on the most significant variables (maximum of 7) associated with anti-CGRP mAbs treatment response for each response rate group and time point. Hyperparameters for each model were tuned using grid search cross-validation against the training data set to optimize for average weighted F1-score of the model.
In total, the following six models were trained:
Predicting response rate for month 3 using 1-month data; Predicting response rate for month 6 using 1-month data; Predicting response rate for month 6 using both 1-month and 3-month data; Predicting response rate for month 12 using 1 month data; Predicting response rate for month 12 using 1-month and 3-month data; Predicting response rate for month 12 using 1-month, 3-month, and 6-month data.
Numeric missing data were imputed using the k-nearest-neighbors algorithm to get the mean value of the five nearest neighbors. The data processing, analysis and ML models were developed in Python, version 3.6.1 (https://www.python.org) using the Scikit-Learn (version 1.2.2) library. Additional Python packages used for data handling and plotting of graphs include: NumPy (version 1.24.2), Pandas (version 1.5.3), Joblib (version 1.2.0), SHAP (version 0.42.0) and Matplotlib (version 3.7).
The models were then further tested against an external data set and the results have been posted and discussed as part of the second phase of testing the model.
Prediction tool development
Each model was built and saved in a cloud storage. These models are accessed by a custom Flask application that exposes an application programming interface (APIs) to query the models. There are two endpoints built for the application. The endpoint to GET the model’s input columns takes in the prediction model and the most recent month (either 1, 3 or 6 months) of patient data available and returns all the selected features for that model. The prediction endpoint takes in all the required input data based on the selected features for the model and returns the prediction probabilities for each output class. These endpoints are consumed by a custom user interface built in Angular. The web application requests the month to predict for and the most recent patient data (1 month, 3 months or 6 months) from the user. It then queries the API to display all the required fields for the model selected. Once all the inputs are filled in and meet the validation checks, the web application then displays the probabilities of headache reduction for each of the four classification labels. Both the Angular application and the Flask API are containerized using docker and are hosted using Google Cloud.
Results
Cohort characteristics
Overall, 336 patients with a 12-month follow-up were eligible and completed the study. An overview of the demographic and clinical features of the overall population at baseline is provided in the Supplementary material (Table S1). The mean ± SD age of the patients was 48.2 ± 13.0 years. The majority of the participants were female (80.1%). Chronic migraine was prevalent in 89.9% of the cases and 77.1% reported medication overuse. The mean ± SD duration of migraine was 38.1 ± 15.7 years, whereas the chronicization duration (if applicable) was 18.0 ± 15.4 years. On average, patients experienced 22.9 MHDs, with 19.6 days involving at least one analgesic use. The MIDAS score averaged 88.1 ± 61.7 and the HIT-6 score averaged 67.8 ± 6.4.
Feature selection and machine learning results
Figure 1 shows the flow diagram of the anti-CGRP mAbs response calculator tool at 3, 6 and 12 months. The baseline features, demographic inputs and month-based predictors for months 1, 3 and 6 were: the type of anti-CGRPmAb, age, sex, migraine diagnosis, disease duration, aura, MHDs, AMDs, HIT-6, number of analgesics and chronicization onset. Based on these features, the variables selected for each model by sequential backward feature selection are shown in Table 1. After training, the models were rerun against the training data to evaluate for overfitting, and the performance scores are documented in the Supplementary material (Table S2). When tested against the test data split, mean AUCs for the 3, 6 and 12 months were in the range 0.56–0.76 (Figure 2) and the accuracy was in the range 0.40–0.73. The accuracy, precision, recall, F1-score and AUC scores for each model against the test data are shown in Table 2. The confusion matrix of the model, showing the class-wise performance of the model and the average weighted values of each performance index, is provided in the Supplementary material (Table S3). SHAP summary plots for the selected features for each model were drawn (Figure 3; see also the Supplementary material, Figures S1 to S3); MHDs reduction of the most recent input month tends to have the strongest positive correlation with the output of the model. The number of years since the chronicization of headache also consistently has an inverse relation with the predicted outcome.
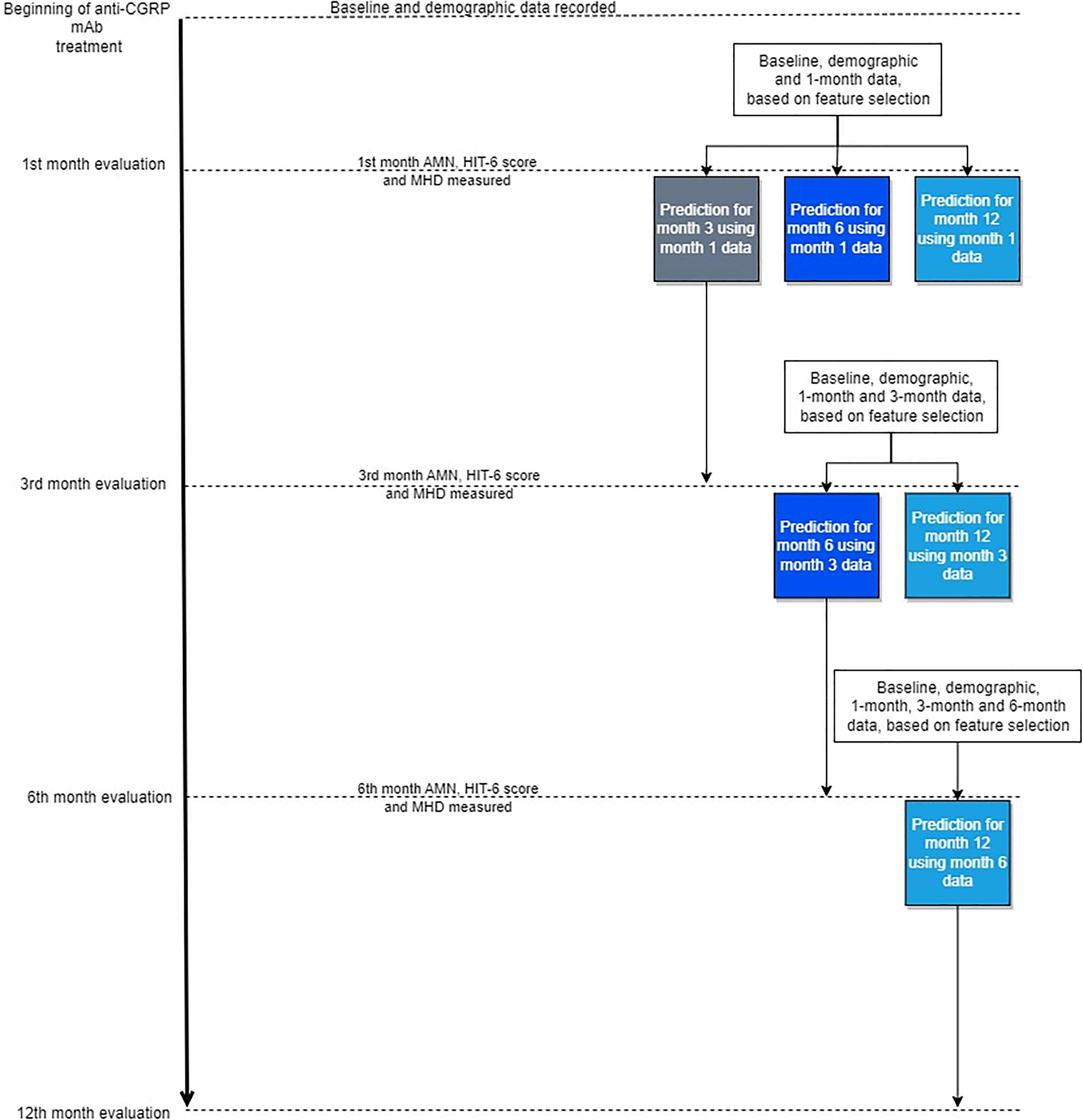
Flow chart of the anti-calcitonin gene-related peptide (CGRP) monoclonal antibodies calculator tool at 3, 6 and 12 months. AMN = absolute number of analgesics; CGRP = calcitonin gene-related peptide; HIT-6 = Headache Impact Test-6; mAb = monoclonal antibody; MHD = monthly headache days.
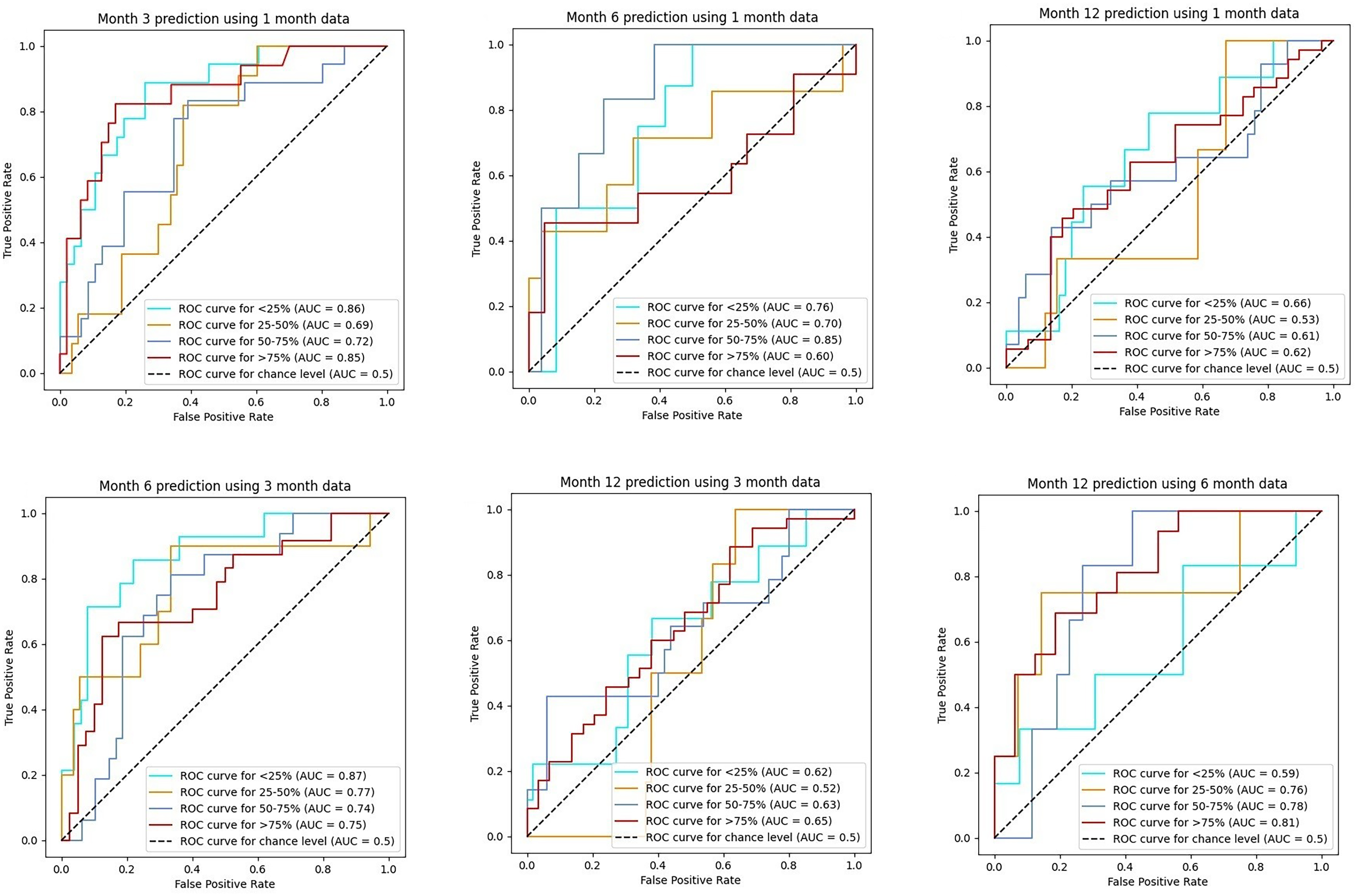
Receiver-operating characteristic (ROC) curves at 3, 6 and 12 months. AUC = area under the curve.
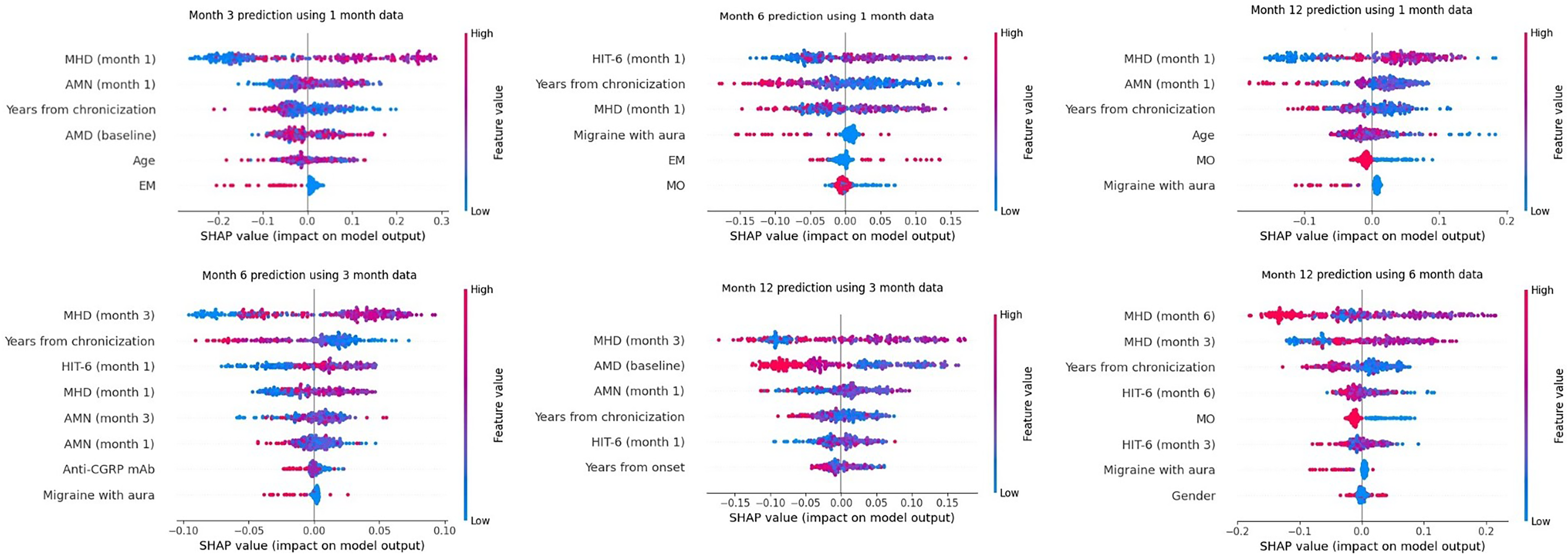
Shapley Additive explanations (SHAP) summary plots to predict anti-calcitonin gene-related peptide (CGRP) monoclonal antibodies response at 3, 6 and 12 months for the outcome measure 50–75% reduction in monthly headache days (MHDs). AMN = absolute number of analgesics; CGRP = calcitonin gene-related peptide; EM = episodic migraine; HIT-6 = Headache Impact Test-6; mAb = monoclonal antibody; MHD = monthly headache days; MO = medication overuse.
Features selected for each model
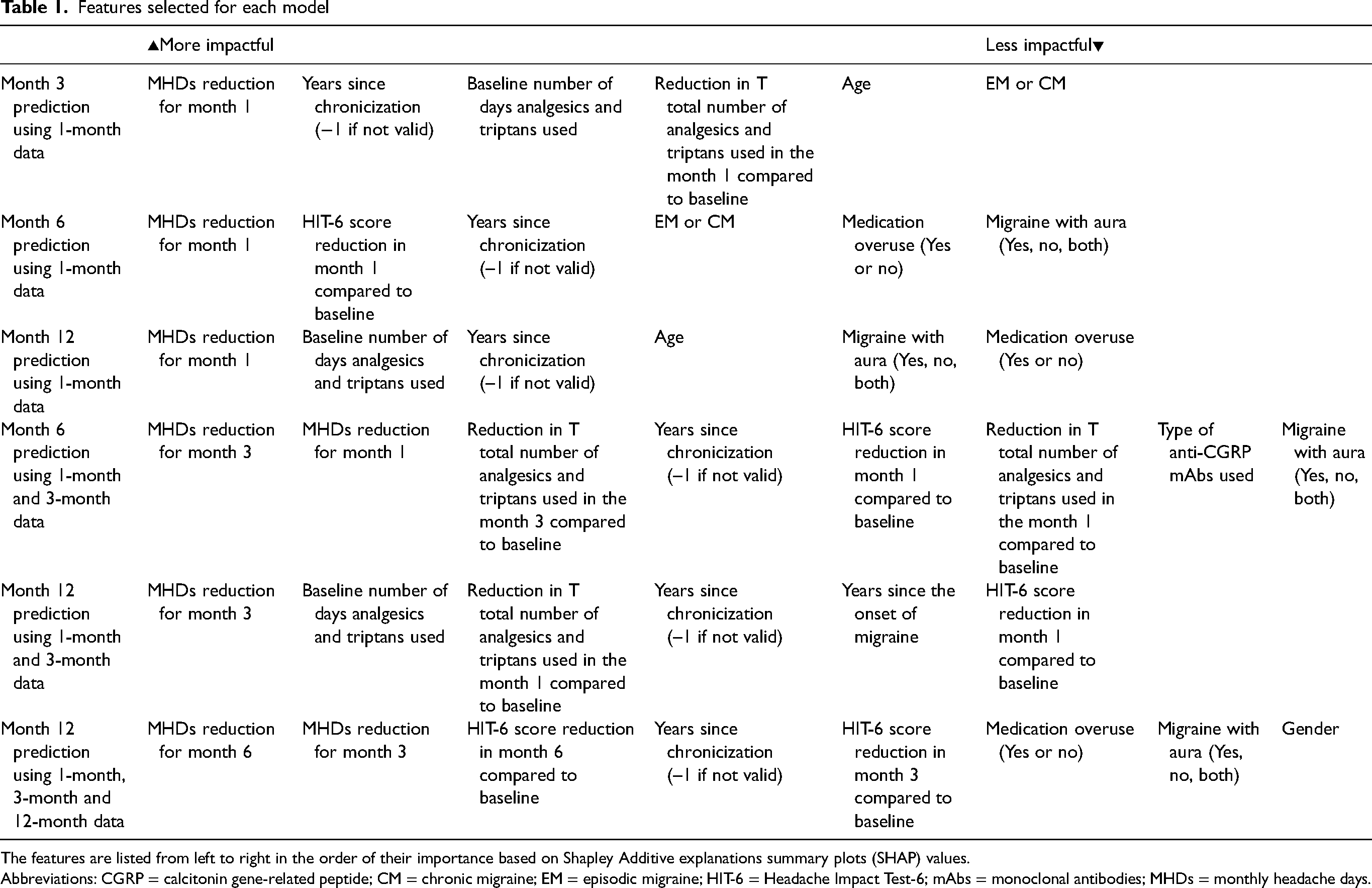
The features are listed from left to right in the order of their importance based on Shapley Additive explanations summary plots (SHAP) values.
Abbreviations: CGRP = calcitonin gene-related peptide; CM = chronic migraine; EM = episodic migraine; HIT-6 = Headache Impact Test-6; mAbs = monoclonal antibodies; MHDs = monthly headache days.
Summary table of performance scores across all models for test dataset and external test cohort
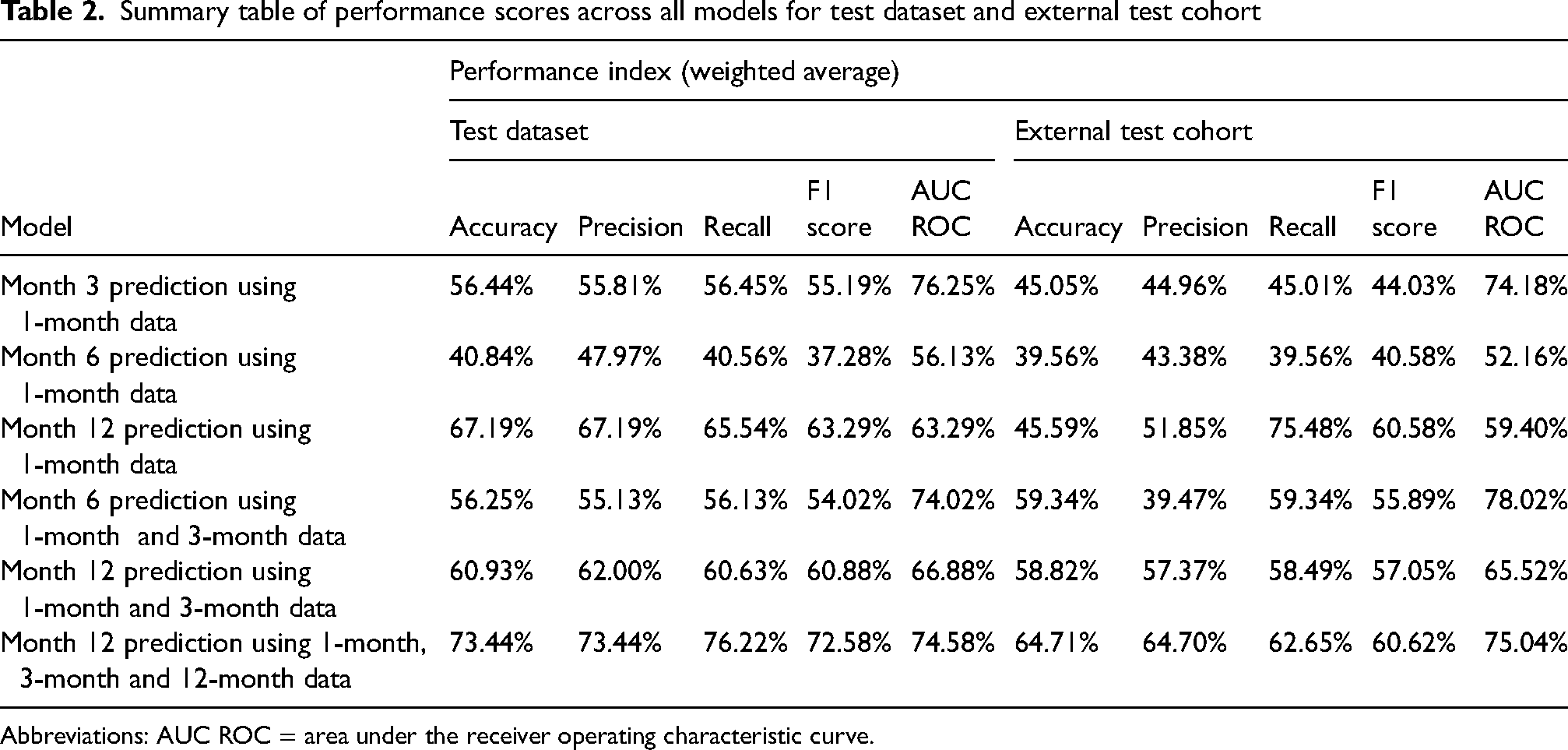
Abbreviations: AUC ROC = area under the receiver operating characteristic curve.
Based on these results, a tool to predict the treatment response of the anti-CGRP mAbs at 3, 6 and 12 months of treatment was generated. The tool calculated the response rates in the ranges of <25%, 25–50%, 50–75%, and >75% reduction in MHDs. The prediction tool can be found at: http://anticgrptool.com (see Supplementary material, Figure S4).
External test cohort
Each model was then validated against an external test dataset of 93 patients undergoing treatment with anti-CGRP mAbs for the month 3 and month 6 prediction models and 68 patients for month 12, accounting for patient drop-off.
The demographical and clinical characteristics of the external test cohort are reported in the Supplementary material (Table S4). Table 2 reports performance scores across all models for the external test cohort. A confusion matrix for each model and the accuracy, precision, recall and F1-score of each model for each output class, are provided in the Supplementary material (Table S5). The weighted accuracy was found to be between 0.39 to 0.64. The mean AUC scores ranged from 0.52 to 0.78.
Discussion
The present study applied ML algorithms using early predictors to assess treatment response to anti-CGRP mAbs in patients with migraine at 3, 6 and 12 months using demographical and clinical variables. Furthermore, based on the data, an online tool to predict anti-CGRP mAb response was developed that implements an iterative approach that can be further improved upon as more data are collected or ML algorithms are improved. Our algorithm employs six different models to predict treatment response to anti-CGRPmAbs at 3, 6 and 12 months. Each model considers 11 baseline data inputs, including demographic data, migraine characteristics, type of anti-CGRP mAb, diagnosis, disease duration, onset of chronicization, HIT-6 score and information on acute medication intake. ML algorithms are increasingly utilized to support the diagnosis, treatment and prognosis of several neurological disorders (4). Concerning migraine, a few studies have employed ML algorithms to classify primary headache disorders. Liu et al. (5) applied ML algorithms using demographic data and headache characteristics in the differential diagnosis between migraine and tension-type headache with high diagnostic accuracy.
In a recent study focused on the response to BTX-A, the routine anamnestic features acquired in real-world settings predict the treatment response to BTX-A in high-frequency EM but not in CM patients (6). A limited number of studies employed ML approaches to predict treatment response in migraine patients. Gonzalez-Martinez et al. (7) developed the first ML model for predicting response to anti-CGRP mAbs and proposed a predictive tool. This ML algorithm included baseline headache variables (frequency of headache days and migraine days per month), change in the number of headache or migraine days per month from baseline to months 3 and 6, and the HIT-6 score at months 3 and 6, to predict response rate at 6, 9 and 12 months in patients with migraine receiving anti-CGRP mAbs. The model yielded an accuracy of over 70% in each model developed (7). The algorithm employed in this study utilizes a reduced set of variables commonly assessed in clinical practice to facilitate its use. However, demographic variables, data on migraine characteristics, history and diagnosis, and information on acute medication were not included. Furthermore, their tool is designed to provide predictions no earlier than the sixth month of treatment and uses three months of treatment as the earliest outcome (7). This can pose practical challenges in settings where the assessment of treatment response is required at month 3 of treatment to determine its continuation (15).
Although numerous, all variables included in the ML model are simple and can be effortlessly collected during routine ambulatory visits, allowing widespread use of the algorithm. An additional and innovative feature of our model is its capability to predict treatment response not only at months 6 and 12 but also as early as month 3. This prediction is achieved using month-based predictors for months 1, 3 and 6 of treatment. For example, the algorithm can provide a treatment response prediction at month 12 using data from the first month. Considering the costs of anti-CGRPmAbs, favoring the use of these drugs in responder patients represents a priority for a tailored therapeutic approach and health resource optimization. With the widespread use of anti-CGRP mAbs in the prophylactic treatment of migraine, there has been a growing focus on identifying predictors of treatment response in order to tailor and personalize treatment approaches. Indeed, the use of response predictors may identify patients who may need more intensive therapy (e.g. polytherapy) and may prevent the use of these expensive drugs in patients who will not benefit from them, providing potentially most effective alternatives. Several real-world studies attempted to identify predictors of response to anti-CGRP mAbs at different treatment time points and in different subgroups of patients (16–20). Other studies evaluated the predictive factors of sustained response to anti-CGRP mAbs after discontinuation (21). However, the results are inconsistent among studies (2). The advantage of ML techniques is that they are unbiased and very sensitive in detecting patterns arising from small changes across a large number of variables. Their potential to be improved over time with new and more numerous data is one of the main differences compared with standard statistical analysis, as the iterative nature of the models allows them to continuously learn and adapt, incorporating new information as it becomes available. Possible pitfalls are that building reliable classifiers requires large amounts of well-selected data and extensive validation (3).
The study has several strengths and limitations. This is a multicentric study with data collected prospectively monthly, with careful collection of acute medication use and included disability questionnaires. There are also some limitations to acknowledge. First, the model is built with the selection of MHDs over MMDs, which was necessitated by data availability within clinical practice settings. Second, the data set has class imbalances that skew toward patients being highly responsive to the treatment. This imbalance causes the model to have better performances at predicting patients who will be responsive to anti-CGRP mAbs. Lastly, the results from the external test cohort indicate that some models demonstrate a level of accuracy that still requires implementation. Furthermore, the models are also susceptible to overfitting, and the generalizability of the results has yet to be evaluated. Thus, we can consider it as an evolving model that will require further optimization and validation to be a practical aid to support clinical decision-making.
Conclusions
The ML-based tool holds promise in the prediction of treatment outcomes for patients with migraine undergoing anti-CGRPmAbs treatment using early predictors. The ongoing development and implementation of ML algorithms and tools in clinical practice have the potential to improve migraine care, yielding personalized precision medicine.
The ML-based tool holds promise in the prediction of treatment outcomes for patients with migraine undergoing anti-CGRP mAbs treatment using early predictors; ML-based algorithms may aid in clinical decision support and cost-optimization with the potential to improve migraine care.
Supplemental Material
sj-png-1-cep-10.1177_03331024241262751 - Supplemental material for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine
Supplemental material, sj-png-1-cep-10.1177_03331024241262751 for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine by Marina Romozzi, Ammar Lokhandwala, Catello Vollono, Giulia Vigani, Andrea Burgalassi, David García-Azorín, Paolo Calabresi, Alberto Chiarugi, Pierangelo Geppetti and Luigi Francesco Iannone in Cephalalgia
Supplemental Material
sj-jpg-2-cep-10.1177_03331024241262751 - Supplemental material for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine
Supplemental material, sj-jpg-2-cep-10.1177_03331024241262751 for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine by Marina Romozzi, Ammar Lokhandwala, Catello Vollono, Giulia Vigani, Andrea Burgalassi, David García-Azorín, Paolo Calabresi, Alberto Chiarugi, Pierangelo Geppetti and Luigi Francesco Iannone in Cephalalgia
Supplemental Material
sj-jpg-3-cep-10.1177_03331024241262751 - Supplemental material for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine
Supplemental material, sj-jpg-3-cep-10.1177_03331024241262751 for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine by Marina Romozzi, Ammar Lokhandwala, Catello Vollono, Giulia Vigani, Andrea Burgalassi, David García-Azorín, Paolo Calabresi, Alberto Chiarugi, Pierangelo Geppetti and Luigi Francesco Iannone in Cephalalgia
Supplemental Material
sj-png-4-cep-10.1177_03331024241262751 - Supplemental material for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine
Supplemental material, sj-png-4-cep-10.1177_03331024241262751 for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine by Marina Romozzi, Ammar Lokhandwala, Catello Vollono, Giulia Vigani, Andrea Burgalassi, David García-Azorín, Paolo Calabresi, Alberto Chiarugi, Pierangelo Geppetti and Luigi Francesco Iannone in Cephalalgia
Supplemental Material
sj-docx-5-cep-10.1177_03331024241262751 - Supplemental material for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine
Supplemental material, sj-docx-5-cep-10.1177_03331024241262751 for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine by Marina Romozzi, Ammar Lokhandwala, Catello Vollono, Giulia Vigani, Andrea Burgalassi, David García-Azorín, Paolo Calabresi, Alberto Chiarugi, Pierangelo Geppetti and Luigi Francesco Iannone in Cephalalgia
Supplemental Material
sj-docx-6-cep-10.1177_03331024241262751 - Supplemental material for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine
Supplemental material, sj-docx-6-cep-10.1177_03331024241262751 for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine by Marina Romozzi, Ammar Lokhandwala, Catello Vollono, Giulia Vigani, Andrea Burgalassi, David García-Azorín, Paolo Calabresi, Alberto Chiarugi, Pierangelo Geppetti and Luigi Francesco Iannone in Cephalalgia
Supplemental Material
sj-docx-7-cep-10.1177_03331024241262751 - Supplemental material for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine
Supplemental material, sj-docx-7-cep-10.1177_03331024241262751 for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine by Marina Romozzi, Ammar Lokhandwala, Catello Vollono, Giulia Vigani, Andrea Burgalassi, David García-Azorín, Paolo Calabresi, Alberto Chiarugi, Pierangelo Geppetti and Luigi Francesco Iannone in Cephalalgia
Supplemental Material
sj-docx-8-cep-10.1177_03331024241262751 - Supplemental material for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine
Supplemental material, sj-docx-8-cep-10.1177_03331024241262751 for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine by Marina Romozzi, Ammar Lokhandwala, Catello Vollono, Giulia Vigani, Andrea Burgalassi, David García-Azorín, Paolo Calabresi, Alberto Chiarugi, Pierangelo Geppetti and Luigi Francesco Iannone in Cephalalgia
Supplemental Material
sj-docx-9-cep-10.1177_03331024241262751 - Supplemental material for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine
Supplemental material, sj-docx-9-cep-10.1177_03331024241262751 for An evolving machine-learning-based algorithm to early predict response to anti-CGRP monoclonal antibodies in patients with migraine by Marina Romozzi, Ammar Lokhandwala, Catello Vollono, Giulia Vigani, Andrea Burgalassi, David García-Azorín, Paolo Calabresi, Alberto Chiarugi, Pierangelo Geppetti and Luigi Francesco Iannone in Cephalalgia
Footnotes
Acknowledgements
The “Società Italiana per lo Studio delle Cefalee” (SISC) is acknowledged for the “Registro Italiano delle Cefalee (RICe)”.
Author contributions
All authors critically reviewed the manuscript, agreed to be fully accountable for ensuring the integrity and accuracy of the work, and have read and approved the final version of the manuscript submitted for publication.
Data availability
Data supporting the findings in the present study are reported in the article and in the supplementary materials. The raw data collected and analyzed are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical statement
The study is part of the Registro Italiano Cefalee (RICe) study approved by the local Ethics committee (CEAVC Studio RICe, 14591_oss and subsequent amendments 2022-609).
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
