Abstract
Background
Human models of migraine have been used for the past 30 years to test putative ‘trigger’ molecules and ascertain whether they induce migraine attacks in humans. However, nocebo effects using this model have never been systematically explored.
Objective
To assess the nocebo response rate in randomised clinical trials conducted at the Danish Headache Center, and in which human models of migraine were used.
Methods
In this systematic review and meta-analysis, we searched PubMed for studies of human models of migraine with a randomised, double-blind, placebo-controlled, two-way crossover design that included data on the incidence of migraine attacks or headache after infusion of placebo. A total of 943 articles were screened by title and abstract. Of these, 27 studies met the inclusion criteria (published between 1994 and 2020) and were included in the qualitative and quantitative analysis. We performed a random effects meta-analysis for the incidence of migraine attacks or delayed headache after placebo infusion.
Results
Twenty-seven studies were eligible for inclusion: 12 studies reported data for adults with migraine (n = 182), whereas 16 studies reported data on healthy volunteers (n = 210). For adults with migraine, the incidence of migraine attacks after placebo was 8.1% (95% CI = 2.5–15.5%, I2 = 50.8%). The incidence of delayed headache was 25.9% (95% CI = 18.5–34.1%, I2 = 18.9%). For healthy volunteers, the incidence of migraine attacks after placebo was 0.5% (95% CI = 0.0–3.6%, I2 = 0.0%) while the incidence of delayed headache was 10.5% (95% CI = 4.8–17.6%, I2 = 45.2%).
Conclusion
The nocebo response in randomised, placebo-controlled two-way crossover trials with intravenous infusions of placebo in migraine is negligible. Future studies using human models of migraine can be conducted by assuming a nocebo response rate of 15.5%.
Introduction
Human models of migraine have been used for decades to study signaling pathways responsible for the genesis of migraine pain (1). The rationale is to investigate whether endogenous signaling molecules or other putative ‘trigger’ molecules can induce migraine attacks in humans (2,3). Interest in these ‘trigger’ molecules have directly facilitated the development of mechanism-based therapies, targeting calcitonin gene-related peptide (CGRP) or its receptor (3,4).
A randomised, double-blind, placebo-controlled, two-way crossover design has been the gold standard for human models of migraine (2). Participants are randomly allocated to receive a putative ‘trigger’ molecule or placebo (e.g. isotonic saline). A unique feature of these models is that only individuals with migraine develop “triggered” attacks, while no more than mild headache is reported by healthy volunteers (2). Numerous studies have been carried out with this design and it is, therefore, timely to ascertain the nocebo response rate in individuals with migraine without aura and healthy volunteers. Such efforts may improve the design of future human models of migraine and contextualise findings from previously published studies. Here, we provide a systematic review and meta-analysis of the nocebo response rate in randomised clinical trials (RCTs) conducted at the Danish Headache Center, and in which human models of migraine were used.
Methods
Data sources
No protocol was registered for this study. A systematic search was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guideline. The PubMed database was searched for article on human models of migraine on June 25, 2020, with the following search string and no restrictions in terms of date of publication: “Migraine AND Denmark”. We also searched ClinicalTrials.gov for published, unpublished, and ongoing RCTs. The search features were “Migraine”, “Danish Headache Center”, and “Denmark”. If available, we obtained missing or unpublished data by contacting the relevant study authors.
Eligibility criteria
The PICOS (population, intervention, comparison, outcome and study design) structured approach was used in this systematic review. The population of interest was adult individuals with migraine without aura and healthy volunteers. The intervention of interest was intravenous infusion of placebo (isotonic saline) and an infusion time of less than 120 minutes. The search was limited to studies conducted at the Danish Headache Center. Reference lists of identified primary articles were reviewed to identify studies that may have been missed in the search process. Studies were deemed eligible if they included data from randomised, double-blind, placebo-controlled, two-way crossover trials. Furthermore, we only included studies in which the study population of interest were adults with migraine without aura according to any edition of the International Classification of Headache Disorders (5–7) or healthy volunteers. The key outcomes of interest were the incidence of migraine attacks after intravenous infusion with placebo (isotonic saline) in individuals with migraine without aura and healthy volunteers.
Study selection and data extraction
Two investigators (HG and AI) independently screened all articles by title and abstract. Articles deemed relevant were selected for full-text review to determine eligibility for inclusion. Data was independently extracted by two investigators (HG and AI) and cross-checked by a third investigator (TPD) to ensure the integrity of the contents. Discrepancies were resolved by discussion between the three investigators (HG, AI, and TPD). For each of the included studies, the investigators (HG, AI, and TPD) extracted the following data: Age, gender, experimental criteria for migraine attacks, personal and/or family history of headache disorders, medication history, headache prior to the experimental day, incidence of migraine attacks and delayed headache in after placebo in individuals with migraine without aura or healthy volunteers, and incidence of delayed headache after placebo in healthy volunteers.
Statistical analysis
We performed a random-effects meta-analysis for the primary outcome (incidence of migraine attacks after intravenous infusion with placebo (isotonic saline)) and secondary outcome (incidence of delayed headache after placebo) in both individuals with migraine without aura and healthy volunteers. Both outcomes were defined according to the authors’ case definition. Effect sizes were calculated using the inverse variance method on data transformed with Freeman-Tukey Double arcsine transformation while tau2 (the between-study variance) was calculated with the Restricted Maximum Likelihood method. The I2 statistic was used to assess between-study heterogeneity, with values ≥75% indicating considerable heterogeneity. Effect sizes are presented as the incidence (%) and the 95% confidence interval. Statistical analyses were performed using Microsoft Excel 2016 (Microsoft Corp) and R (version 3.6.3) with the “meta” (version 4.11) and “metaphor” (version 2.1) packages.
Risk of bias assessment
The risk of bias in the included studies was independently assessed by two investigators in accordance with the Cochrane Handbook for Systematic Reviews of Interventions (8). The potential risk of systemic error was categorised as low, high or unclear. The following seven factors, selection bias, performance bias, reporting bias, detection bias, attrition bias and ‘other’ sources of bias, were assessed.
Results
The database search identified 937 citations. An additional six articles were identified through a manual search of identified primary articles and the database search on ClinicalTrials.gov. A total of 943 articles were screened by title and abstract. Of these, 27 studies met the inclusion criteria (1994 and 2020) and were included in the qualitative and quantitative analysis (9–35) (Figure 1). Twelve studies (10 published and two studies under review) reported data on the incidence of migraine attacks after placebo in individuals with migraine without aura (9–17,31,33), whereas 10 studies did so in healthy volunteers (18,19,22,23,25,27,29,30,32,34). Furthermore, 16 studies reported data on the incidence of delayed headache after placebo in healthy volunteers (13,18–30,32,34). A summary of the included studies is presented in Table 1 and Table 2.
Summary of nocebo responses in trials with individuals with migraine.

*Baseline data; †Not reported; +Any headache in post-hospital phase considered as delayed headache.
Summary of nocebo responses in trials with subjects with healthy volunteers.
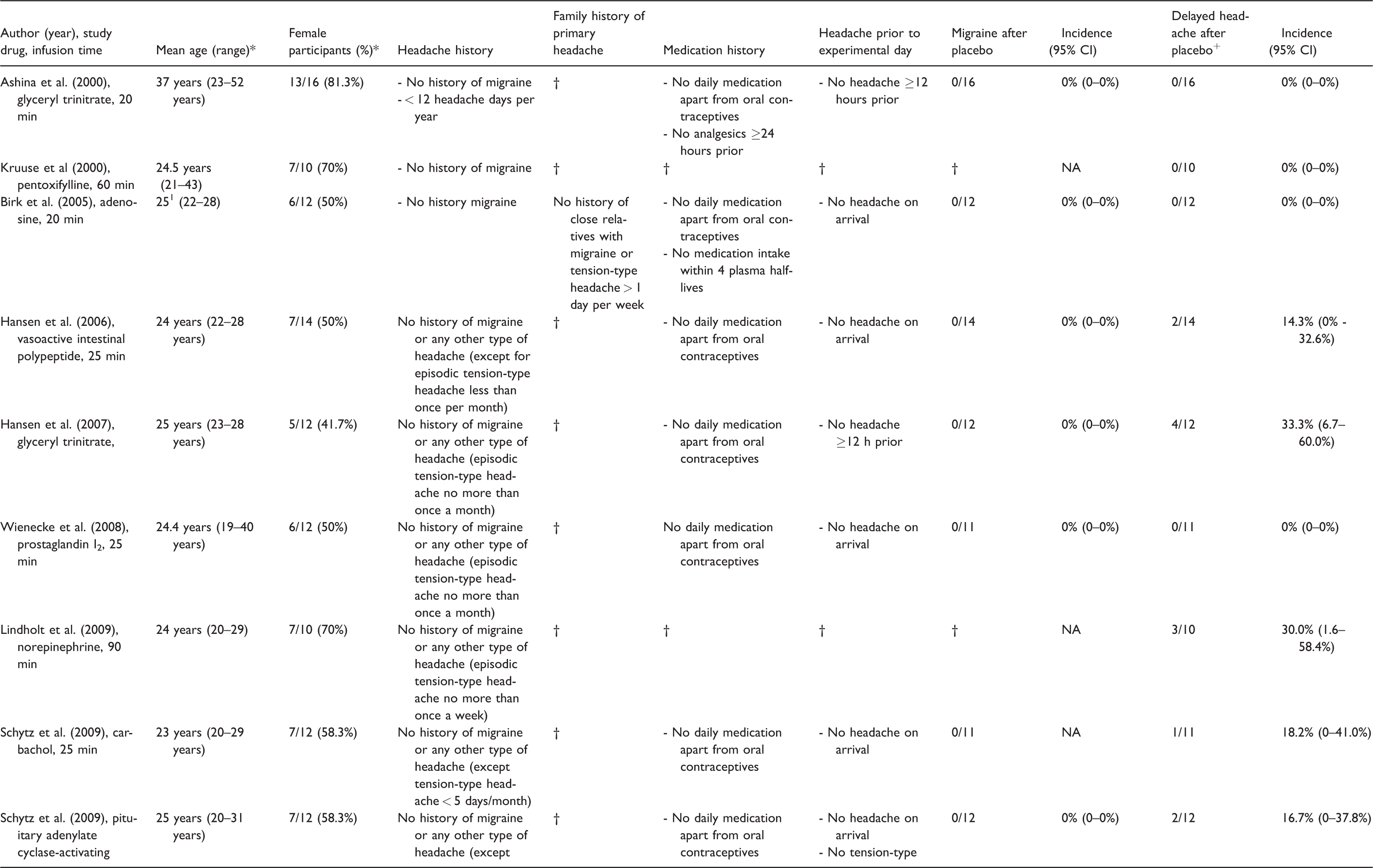
*Baseline data; 1Median; †Not reported; +Any headache in post-hospital phase considered as delayed headache.
Migraine attacks after placebo in individuals with migraine without aura
Twelve studies were included in the primary analysis, comprising a total of 182 adults with migraine without aura (9–17,31,33). Age ranged from 18–53 years while the proportion of female participants ranged from 67–100%. The random effects meta-analysis showed an incidence of 8.1% (95% CI = 2.5–15.5%, I2 = 50.8%). In terms of methodology and design, all studies required that participants were free of headache upon arrival on the experimental day and had no daily intake of medications other than oral contraceptives. Furthermore, all studies used randomisation with 1:1 allocation and a wash-out period of at least 7 days between the two experimental days. Carry-over and/or period effects were assessed in seven of 12 included studies, in which none were found (12,13,15–17,31,32) (Table 1) (Figures 2, 3).
Delayed headache after placebo in individuals with migraine without aura
Eleven studies were included in the primary analysis, comprising a total of 163 adults with migraine without aura (9–17,31,33). The random effects meta-analysis showed that the incidence of delayed headache after placebo was 25.9% (95% CI = 18.5–34.1%, I2 = 18.9%). The applied methodology and design were similar in the studies, in which the incidence of migraine attacks had been investigated in adults with migraine without aura (Table 1, Figures 4, 5).
Migraine attacks after placebo in healthy volunteers
Eleven studies were included in the primary analysis, comprising a total of 138 healthy volunteers (13,18,19,22,23,25,27,29,30,32,34). The random effects meta-analysis showed that the incidence of migraine attacks after placebo was 0.5% (95% CI = 0.0–3.8%, I2 = 0.0%). In terms of methodology and design, all studies required that participants were free of headache upon arrival on the experimental day and had no personal history of primary headache disorder, except episodic tension-type headache less than 5 days per month. In addition, all studies used a 1:1 randomisation and 7-day wash-out period (13,18,19,22,23,25,27,29,30,32,34) (Table 2) (Figures 4, 5).
Delayed headache after placebo in healthy volunteers
Seventeen studies were included in the primary analysis, comprising a total of 210 healthy volunteers (13,18–30,32,34). The random effects meta-analysis showed that the incidence of delayed headache after placebo was 10.5% (95% CI = 4.8–17.6%, I2 = 45.2%). The applied methodology and design were similar to the aforementioned studies, in which the incidence of migraine attacks had been investigated in healthy volunteers. Carry-over and/or period effects were assessed in nine of 17 included studies, in which none were found (13,19–21,23,24,27) (Table 2) (Figures 4, 5).
Discussion
This is the first systematic review and meta-analysis to ascertain the incidence of migraine attacks and delayed headache after intravenous infusion of placebo. In individuals with migraine without aura, the incidence of migraine attacks was 8.1% after placebo, whereas the corresponding incidence for delayed headache was 25.9%. In healthy volunteers, the incidence of migraine attacks was 1.5%, whereas the corresponding incidence for delayed headache was 10.5%.
Our data demonstrate that nocebo effect is a confounding factor in delayed incidence of headache in human migraine model. We found that nocebo effect is more noticeable in individuals with migraine compared to healthy volunteers. Interestingly, the incidence of migraine attacks remains low after infusion of placebo. Our study shows that nocebo effect clearly lacks migraine-inducing properties. Furthermore, the magnitude of nocebo response differs across the studies reviewed in this study, thus showing variation in the ways that participants respond to nocebo effect, and with proper methodological strategies these effects can be mitigated. Recently, the effect of nocebo on generation of headache pain was reviewed (35). This study shows that psychosocial factors might play an important role in nocebo response (35). Here, we provided the first empirical evidence that nocebo affects individuals with migraine. Greater attention is needed to reduce the magnitude of nocebo in the experimental setups and clinical practice in individuals with migraine.
Furthermore, an important question raised by our findings is whether migraine attacks reported after placebo can be attributed to methodological limitations or simply the probability of developing a spontaneous attack within a 12-h observational period. Human models of migraine use a randomised, double-blind, placebo-controlled, two-way crossover design (2). This approach eliminates the between-subject variability and is the best suited design to assess short-lived effects of an intervention (36). A well-described limitation of crossover designs is, however, the potential influence of carry-over and period effects. In this context, it should be highlighted that such effects have never been reported in human models of migraine despite consistent testing in most of the included studies (13,19–21,23,24,27). This could be attributed to the standardised use of a 7-day wash-out period, but a remaining unknown is then whether a shorter wash-out period might suffice. Furthermore, we systematically assessed the risk of bias in the included studies. Unclear reporting of blinding of participants/personnel and allocation concealment were the main reasons for unclear risk of bias. However, it seems that proper and effective blinding has been carried out, but the authors did not properly document it in the method section. No high risk of bias was detected in the studies. The overall risk of bias in the studies remained low. In this review, only studies with intravenous administration of substances in migraine without aura and healthy volunteers were included. All participants were informed and gave their informed consent to receive an active and a placebo infusion. However, a hidden (infusion pump) design was used for infusion of substances in the cross-over design. Interestingly, previous studies showed that a hidden design might minimise the placebo/nocebo responses in clinical trials (37–39). Taken together, the lack of carry-over and period effects suggest that methodological issues are less important reasons, if at all, for the development of migraine attacks after placebo. It is, therefore, relevant to consider what the probability would be for developing a spontaneous attack within a 12-h observational period. Of note, all included studies had exclusively enrolled individuals with episodic migraine, who reported between six and 76 attacks per year (9–35). Thus, it may be hypothesised that the incidence of migraine attacks after placebo is comparable to the probability of developing spontaneous attacks within the same observational period. This possibility should be further explored and would require theoretical modelling, in which the conditional probability of a migraine attack within a given observational period is calculated. Equivalence of the calculated probability with the observed incidence would yield a discussion of whether a placebo arm is even needed. This is truly an intriguing matter, but such theorical models are currently not available and a placebo arm is likely to be considered a necessity until otherwise proven. To date, most studies have assumed a 20% nocebo response rate; that is, a 20% induction rate of migraine attacks after placebo in individuals with migraine without aura (9–35). This should be revised considering our findings, and an assumption of a 15.5% nocebo response rate would be appropriate for future investigations. The implications would then maximise feasibility and minimise costs.
Limitations and strengths
Our study has some limitations. We have included studies with intravenous infusion only, which were performed in our centre. All studies were performed with slightly different definition of migraine frequency using ICHD I-III. Our findings were also limited to the small trial sample size. Furthermore, individual studies were performed using different migraine trigger substances with different physiological, biochemical and pharmacological properties, which may possess differential placebo/nocebo effects. Despite these limitations, our study is the most comprehensive analysis of nocebo effect in the human models of migraine.
Conclusions
Migraine attacks are seldom reported by individuals with migraine without aura after intravenous infusion with placebo and never by healthy volunteers. Future sample size considerations should assume a nocebo response of 15.5% in those with migraine.
Article highlights
In individuals with migraine without aura, the incidence of migraine attacks after placebo was 8.1%. In healthy volunteers, the incidence of migraine attacks after placebo was 0.5%. An assumption of a 15.5% nocebo response rate (upper limit of the 95% confidence interval) should be used for future investigations using human models of migraine.

Flow chart of the study selection process.

Visual abstract showing migraine attack induction rate after placebo infusion.
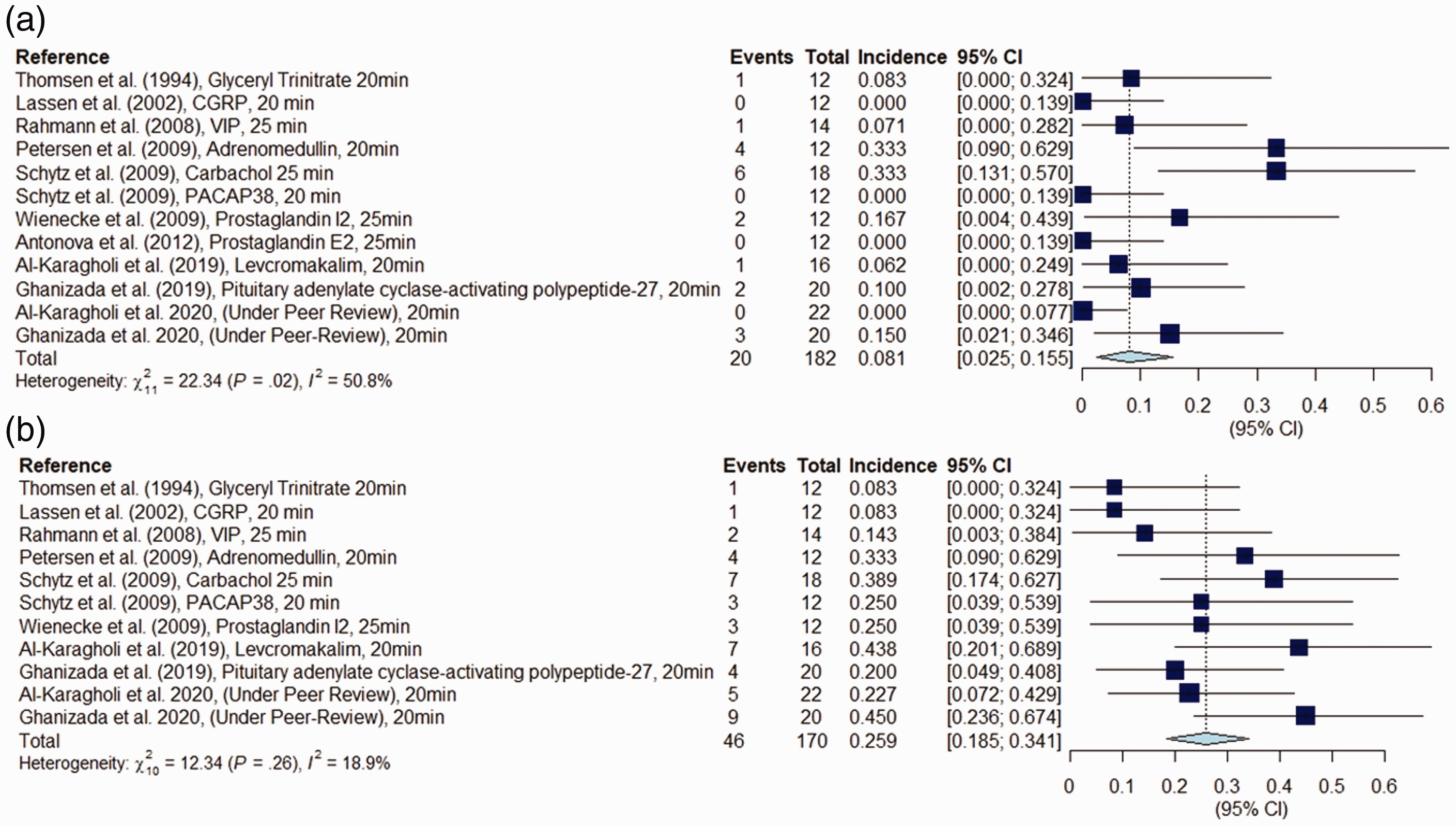
Forest plot: (a) Migraine attacks after placebo in individuals with migraine without aura. (b) Delayed headache after placebo in individuals with migraine without aura.

Visual abstract showing delayed headache induction rate after placebo infusion.

Forest plot: (a) Migraine attacks after placebo in healthy volunteers. (b) Delayed headache after placebo in healthy volunteers.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MA is a consultant, speaker or scientific advisor for Allergan, Amgen, Alder, Eli Lilly, Lundbeck, Novartis, and Teva, primary investigator for Alder, Amgen, ElectroCore, Novartis and Teva trials. MA has no ownership interest and does not own stocks of any pharmaceutical company. MA serves as associate editor of Cephalalgia, Headache, and the Journal of Headache and Pain. MA is the President of the International Headache Society. MAK has acted as an invited speaker for Novartis and received travel grant from ElectroCore and FMA is a lecturer or scientific adviser for Novartis and Teva. HG, AI, HA, and TPD declare they have no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study received financial support from Lundbeck Foundation (R155-2014-171).
