Abstract
Background
Endometriosis and migraine frequently coexist, but only a limited number of studies have focused on their mutual association. The aim of our study was to investigate, in untreated women with comorbid endometriosis/adenomyosis and migraine, the correlation between headache features and endometriotic subtypes and their possible relationship with pain severity and disease disability.
Methods
Fifty women affected by endometriosis/adenomyosis and migraine matched (1:2) with 100 patients with endometriosis alone and 100 patients with only migraine were recruited and underwent pelvic ultrasound imaging and neurological examination.
Results
Severe adenomyosis, posterior and anterior deep infiltrating endometriosis (p = 0.027, p = 0.0031 and p = 0.029, respectively) occurred more frequently in women with migraine. Dysmenorrhea was the most commonly reported symptom in women with endometriosis and migraine and the mean VAS scores of all typical endometriotic symptoms were significantly higher in the presence of comorbidity. Women with both migraine and endometriosis reported significant higher pain intensity (p = 0.004), higher monthly migraine days (p = 0.042) and increased HIT 6-scores (p = 0.01), compared with those without endometriosis.
Conclusions
Our results demonstrated that the co-occurrence of migraine in untreated women with endometriosis is associated with more severe gynecological infiltrations and correlated with increased pain intensity and disease disability.
Introduction
Endometriosis and migraine represent two major chronic painful conditions of paramount relevance for the women’s physical and mental health.
Endometriosis is a gynecological inflammatory disorder affecting up to 10–15% of women of reproductive age and 35%–50% of infertile ones worldwide (1). The disease is defined by the proliferation of endometrial tissue outside the uterus and is characterized by recurrent “pain symptoms” including dysmenorrhea, dyspareunia, dyschezia, dysuria, and chronic pelvic pain as well as infertility (1,2).
With an estimated global prevalence of 14.7%, migraine, on the other hand, has been considered by the Global Burden of Diseases 2019 as the first leading cause of disability in the under 50s young women (3). Like endometriosis, migraine typically presents with episodic moderate-to-severe headache attacks, that can be associated with other manifestations such as light and/or sound hypersensitivity, nausea and vomiting (4).
In addition to clinical and epidemiological similarities, the two pathologies share several risk factors including early menarche, menorrhagia, and hormonal dysregulation (5,6). A recent study revealed an hormone-dependent misregulation of plasma calcitonin gene-related peptide (CGRP) as a possible pathophysiological similarity of this comorbidity (7). Notwithstanding, it is observed a missed or delayed care, leading to increased levels of disability and impacting negatively on all aspects of individuals’ life (8,9).
Previous observational epidemiological studies reported a mutual relationship between migraine and endometriosis (10,11). Some authors also noted a different prevalence of pain-related problems and/or disease phenotypes when migraine and endometriosis coexist (12–14). However, most of these evidences are based on post-surgically proven refractory endometriosis and/or self-administered questionnaires for headache evaluation, with mixed and heterogeneous results (10–14). Moreover, these previous studies do not differentiate between women with endometriosis and migraine on hormonal treatment and those without treatment, which could mask the pain symptom correlated with both diseases, and are commonly used for managing endometriosis (12–14).
In the last years, non-invasive techniques such as transvaginal ultrasound imaging (TVS) has been recognized for reliably detect the localization and severity of endometriotic lesions without the necessity of histological confirmation, especially in association with other painful symptoms (migraine), allowing an earlier disease diagnosis (15). Growing evidence suggests a high accuracy (80–100%) between ultrasound findings and surgical staging, that has been considered as the diagnostic gold standard in the past but is limited by increased operative risks with economic barriers (16,17). Moreover, deep infiltrating endometriosis of retroperitoneal spaces is identified with higher accuracy by imaging compared to diagnostic laparoscopy which does not investigate this localization (18–20).
Based on this scenario, in the present study we aimed to investigate in non-hormonal-treated women with comorbid endometriosis/adenomyosis and migraine (EM-MG), compared to either women with only endometriosis/adenomyosis (EM-O) or women with migraine alone (MG-O): a) the different subtypes of endometriotic lesions and severity of adenomyosis; b) the different migraine features; c) their possible correlation with disease severity and disability.
Materials and methods
This prospective case-control study enrolled consecutive patients admitted to the Endometriosis Unit and the Headache Center at Tor Vergata University Hospital of Rome between January 2021 and July 2022. Within this cohort, women with endometriosis with concomitant migraine (EM-MG) were considered cases, meanwhile women with only endometriosis (EM-O) and women with migraine alone (MG-O) were regarded as two different control groups.
Women in premenopausal age (18–50 years) and not under hormonal therapy were included in the study. Exclusion criteria were: other concomitant neurological and/or psychiatric disorders; other primary form of headache; use of central nervous system-active medications; pregnancy at the time of examination; incomplete clinical reports; history of oncologic disease of genital tract; other chronic systemic, metabolic or autoimmune disorders that could have affected pain’s evaluation (i.e. fibromyalgia, inflammatory bowel diseases).
For each case of endometriosis with concomitant migraine (EM-MG), 2 controls with a defined diagnosis of migraine without endometriosis (MG-O) and a defined diagnosis of endometriosis without migraine (EM-O) were randomly enrolled querying the electronic database of both unit in the same study period. Controls were matched for age (+/− 5 years) and body mass index (BMI) (+/− 1).
All participants were evaluated at the time of enrollment by a gynecologist who confirmed the diagnosis of endometriosis based on the combination of clinical symptoms and transvaginal ultrasound imaging (TVS), as previously reported and validated (15). They were also examined by a headache expert neurologist, to confirm the diagnosis of migraine according to the International Classification of Headache Disorders 3rd edition (ICHD-3) (21).
The study was conducted in respect of Helsinki rules and all subjects signed a written consent before being included in the study. The Internal Committee of the University Hospital Tor Vergata approved the study with protocol number 119/21. The study database is available from the Corresponding Author on reasonable request.
Gynecological evaluation
For each patient, a detailed clinical history was assessed, collecting the following: date of birth and age at time of enrollment, body mass index (BMI Kg/m2), age of menarche, menstrual cycle characteristics, last menstrual period, parity, delivery modality, primary or secondary infertility, previous surgical interventions, other diseases, ongoing medications. Infertility was defined as no pregnancy after 12 months of intercourses. The following painful symptoms, compatible with endometriotic lesions and adenomyosis, were collected and scored according to a standard visual analogue scale (VAS): dysmenorrhea, dyspareunia, dyschezia, dysuria, chronic pelvic pain (CPP) intended as an unpleasant sensation experienced by the patient outside the menstrual cycle for at least 7 days and in 6 consecutive months. Bowel functional symptoms included symptoms such as constipation, cyclic diarrhea, non-cyclical diffuse abdominal pain, nonspecific gastrointestinal symptoms, nausea, and vomiting during the menstrual period. The amount and duration of menstrual bleeding and any episodes of intermenstrual bleeding were also investigated, asking patients to subjectively rate their menstrual flow (as being “poor”, “normal”, “abundant”). This subjective evaluation is reported as reliable and comparable to the pictorial blood loss analysis chart (PBCA) score (22).
Ultrasound imaging
The ultrasound examination was performed using a Voluson E6 or E8 device (GE Healthcare, Zipf, Austria) with a transvaginal probe. In pre-sexually active patients, the vaginal probe was used transrectally to evaluate the pelvic organs with the same accuracy of the transvaginal approach. Ultrasound examinations performed only transabdominally were not considered in this study. A two- and three-dimensional (2D and 3D) ultrasound with grey scale and power Doppler for assessment of the pelvis was performed. Scans evaluated the uterus, the adnexa, the pouch of Douglas, all other pelvic organs and sites. Endometriosis features were always carefully scanned using a previously published ultrasound mapping system (23). The ultrasound diagnosis of ovarian endometrioma was defined by the presence of a persistent unilocular or multilocular (less than five locules) cyst characterized by a homogeneous low-level echogenicity (ground glass echogenicity) of the cyst fluid and absent or moderate vascularization of the cystic walls (24). The diagnosis of deep infiltrating endometriosis (DIE) was made if at least one structure in the anterior, posterior or lateral compartments showed the presence of an abnormal retroperitoneal hypoechoic linear or nodular thickening with irregular contours and no or few Doppler signals, according to previously validated criteria (18–22). We reported the DIE localization in relation to the pelvic compartment: anterior (bladder, vescico-uterin septum), posterior (rectum, torus posterior vaginal fornix, RVS) and lateral (bilateral USL parametria, ureters). Sonographic findings of uterine adenomyosis were also recorded and its diagnosis was made when at least one direct imaging morphological sign was present according to MUSA criteria (24–26). Adenomyosis was classified as mild, moderate, or severe according to our recently published classification scoring system (26,27). According to available ESHRE Guidelines, these TVS direct features can be considered diagnostic and do not require a surgical confirmation (15).
Neurological examination
Patients were assessed by a headache expert neurologist with a face-to-face interview using a semi-structured questionnaire addressing clinical migraine characteristic, previous and current acute and preventive migraine treatments, comorbidities, and concomitant medications. All patients were asked to fill a prospective headache diary. The following parameters were collected: age at migraine onset and duration, monthly migraine days (MMDs); monthly days of painkillers intake (MPIs), intensity and duration of the headache attack, presence of associated symptoms (i.e. aura, cutaneous allodynia). Patients were also asked to rate pain severity using the Visual Analogue Scale (VAS) of the monthly most painful attack. According to the ICHD-3 criteria, patients were categorized as having: pure menstrual migraine (defined as migraine occurring exclusively on day 1 ± 2 of menstruation); menstrually-related migraine defined as migraine occurring on day 1 ± 2 of menstruation and additionally at other times of the cycle) or non-menstrual migraine (20). Migraine disability was measured using the next validated standard self-reported questionnaires: the Headache Impact Test (HIT-6) and the Migraine Disability Assessment Scale (MIDAS) (28,29). A detailed physical and neurological exam has been carried out to exclude presence of focal neurological deficit and/or secondary causes of headache.
Statistical analysis
The sample size was based on the following considerations, matching the comorbid women by 1:2 with each control group. Assuming a drop-out rate of 10% and a statistical power of 0.9 at a significance level of a = 0.05 (two-tailed) using the Kruskal–Wallis analysis of variance (ANOVA), we planned to enroll a final sample size of at least 50 cases (EM-MG), and respectively matched 1:2, 100 controls with only endometriosis (EM-O) and 100 patients with migraine alone (MG-O).
Statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS v.15.0; SPSS, Inc., Chicago, IL). For population characteristics all continuous variables were expressed in terms of mean ± SD, while categorical variables were indicated in terms of frequency and percentage. The statistical analysis initially assessed the general characteristics, prevalence of symptoms of endometriosis/adenomyosis and migraine, and transvaginal ultrasound findings of the study population EM-MG. General characteristics of EM-MG were compared with the two control groups: EM-O and MG-O. Prevalence rate of different types of endometriosis/adenomyosis and endometriosic symptoms have been compared between the study population and the control group EM-O. Prevalence rate of different migraine phenotypes and the impact on patient’s quality of life has been calculated between the study population and the control group MG-O. The characteristics between groups were compared using the independent samples t-test for continuous variables and chi-square tests for categorical variables. Fisher exact test was used to compare prevalence and a p < 0.05 was considered statistically significant.
Results
The final sample consisted of 50 patients (mean age 33.46 ± 8.98) suffering from both endometriosis/adenomyosis and migraine (EM-MG) and two control groups of 100 women each one: EM-O (endometriosis/adenomyosis without migraine) and MG-O (patients with migraine without endometriosis/adenomyosis). The general characteristics of these three groups are reported in Table 1. In most patients (45/50, 90%), endometriosis was diagnosed after the migraine onset, with a mean time lag of at least 12 years. The remaining 10% of women received the diagnosis at the same age (2/50) or before (3/50) migraine presentation.
Demographic data.

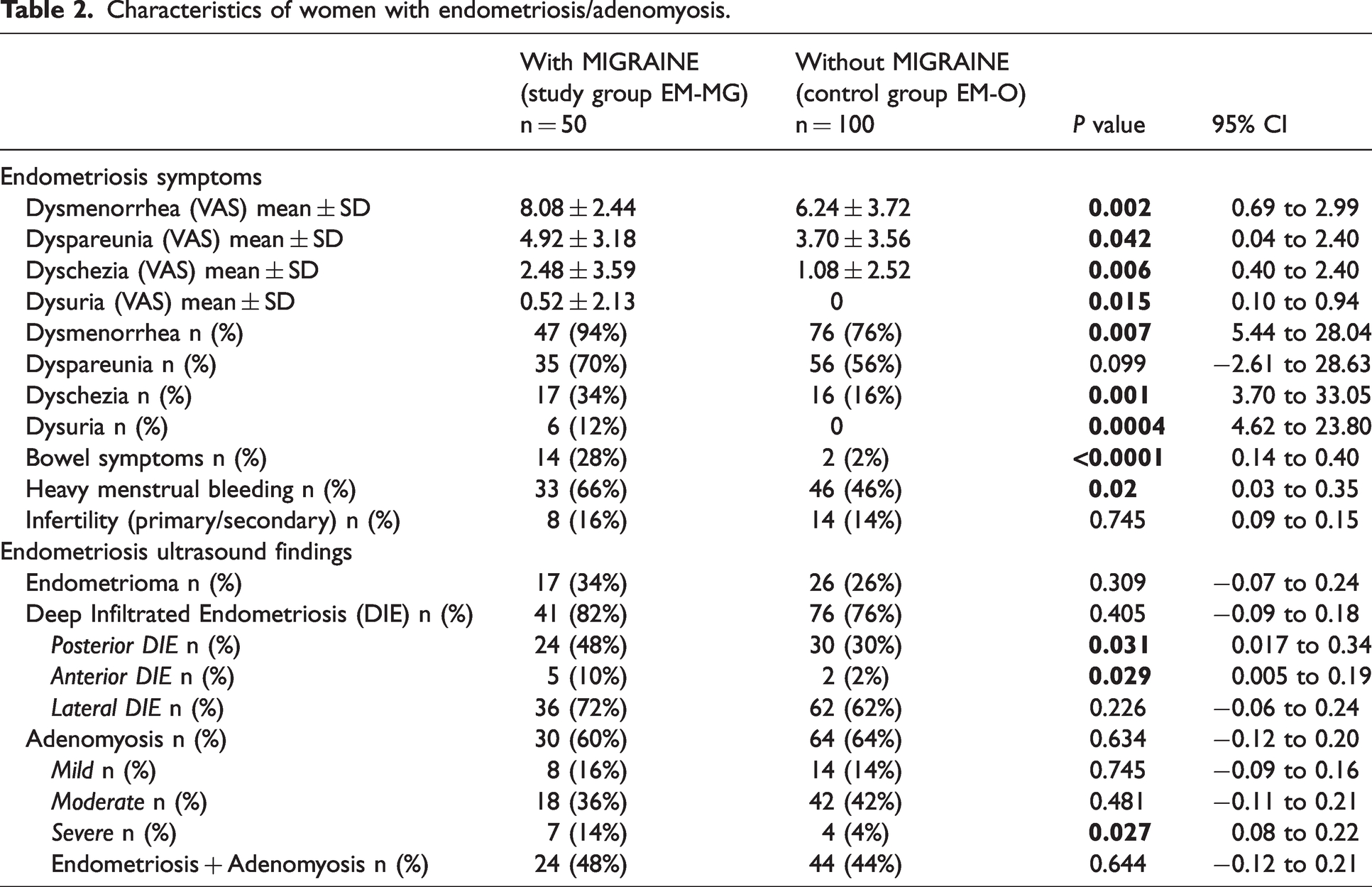
The comparison between EM-MG patients and EM-O control group is shown in Table 2. Mean VAS scores of all typical endometriotic symptoms (dysmenorrhea, dyspareunia, dyschezia, dysuria) were significantly higher in endometriotic women with migraine than in those without migraine. Dysmenorrhea was the most common symptom (94% vs 76%, p = 0.007, Table 2), reported during TVS in women with migraine than in those without migraine. Moreover, bowel symptoms (28% vs 2%, p < 0.0001) and heavy menstrual bleeding (66% vs 46%, p = 0.02) were more frequent in cases than controls. Only the incidence of infertility was similar in these two groups. Considering the specific localization of endometriotic lesions and the different types of adenomyosis, we observed a higher percentage of severe adenomyosis in the study group EM-MG compared with the control group EM-O (14% vs 4%, p = 0.027). Posterior and anterior DIE occurred more frequently in women with migraine EM-MG (48% and 10%, respectively) than in those without migraine EM-O (30% and 2%, p = 0.0031 and 0.029, respectively). The incidence of other phenotypes of endometriosis was similar between cases and controls (p > 0.05) (Table 2).
Characteristics of women with endometriosis/adenomyosis.
The migraine features of women with and without endometriosis (EM-MG versus MG-O) are summarized in Table 3. Both groups of patients had a long history of migraine (a long migraine duration), commonly starting before the age of 18 years old.
Characteristics of women with migraine.

Compared to women with only migraine (MG-O), those with EM-MG reported significant higher pain intensity (8.44 ± 1.18 vs 7.74 ± 1.47, p = 0.004) and higher monthly migraine days (MMDs 6.68 ± 4.01 vs 5.44 ± 3.21, p = 0.042). Monthly days with analgesic use tended to be higher in cases than in controls, without reaching the statistical significance (MPIs 4.88 ± 4.47 vs 4.12 ± 3.36, NS). Women mostly used non-steroidal anti-inflammatory drugs (NSAID) as acute medication, often self-administered without medical advice and at different dosage recommended for migraine. Only 4/50 EM-MG patients (8%) preferred triptans for acute migraine management, that had been prescribed by a previous headache specialist, as few of them were already examined previously by a neurologist and were recruited on their first visit in out headache center. In contrast, no women with migraine alone reported the use of prophylactic headache treatments, choosing non-pharmacological strategies such as nutraceuticals, osteopathy and acupuncture.
Of the EM-MG group, 20% (10/50) had pure menstrual migraine, 46% (23/50) had menstrually-related migraine and the remaining 17 patients (34%) had non-menstrual migraine. These percentages were similar to the MG-O control group.
The presence of migraine aura and/or cutaneous allodynia did not significantly differ between the two groups of patients (p > 0.05). HIT-6 score was significantly increased in the EM-MG group compared to the control group MG-O (62.33 ± 9.44 vs 57.38 ± 11.83, p = 0.01), meanwhile the MIDAS values were similar between them (see Table 3).
Discussion
This multidisciplinary study investigated the bidirectional relationship between a non-invasive diagnosed endometriosis and assessment of migraine in premenopausal female patients not on hormonal therapies. EM-MG group presented more severe symptoms and more frequent painful days than EM-O or MG-O. These alterations were more prominent in the presence of adenomyosis and/or deep endometriosis subtypes, and did correspond to personal perception of discomfort, with further negative effects on activities of daily life. Migraine onset together with dysmenorrhea often started in adolescence and preceded the diagnosis of endometriosis by several years, becoming underestimated and undertreated, with a possible delay in better pain management.
Although endometriosis and migraine commonly co-occur in women of reproductive age (5,6), data exploring the specific correlation between the two disorders are not consistent across the studies, most probably due to the different methods of disease evaluation and high variability of patient cohorts (10–14). Maitrot-Mantelet and colleagues found a higher prevalence of ovarian endometrioma and deeply infiltrating endometriosis (DIE) in women with associated migraine compared to those without migraine (12). In contrast, Pasquini et al. showed no difference of endometriosis phenotypes between female patients with and without migraine (13). The main limitation of their studies was that these alterations were observed mostly in women with post-surgically proven drug-resistant endometriosis, by using self-administered headache reports and/or without considering the influence of hormonal contraceptive treatments on pain perception. Surgical laparoscopy has long been considered the gold standard for diagnosing endometriosis, but it is limited by increased operative risk, high financial costs, failure to evaluate the retroperitoneal spaces and underestimation of DIE (16,17). Recently, the European Society of Human Reproduction and Embryology guidelines recognized non-invasive techniques including TVS, as a valid tool for an earlier diagnosis of endometriosis in young patients, particularly in the presence of other suspected pain diseases such as migraine, without the necessity of laparoscopic/histological confirmation and with high accuracy (80–100%) compared to surgical staging (15,18–20).
In our case-control study, we confirmed a substantial correlation between migraine and endometriosis, demonstrating a difference in both the intensity of all typical painful symptoms and the disease severity between EM-MG and the other 2 groups. In fact, the VAS scores were significantly higher in women with associated endometriosis and migraine compared to women with only one of the two disorders. Furthermore, migraine and pain manifestations were more prevalent in advanced endometriosis, especially when coexisting with severe adenomyosis and deep infiltrating types. These results are in line with those of Wu et al, who suggested that moderate-severe stages of endometriosis, in particular of adenomyosis, were significantly associated with the presence of migraine in women undergoing surgery (13). In this light, pain and its features may be an early indicator of comorbid disorders and more complicated disease course, supporting the importance of a concurrent gynecological ultrasound screening in women with migraine and, on the contrary, of the clinical migraine search in patients with endometriosis.
Focusing on the different pelvic involvement of ectopic tissues by ultrasound imaging, we also reported that endometriosic patients with migraine had considerably higher prevalence of both posterior (p = 0.031) and anterior (p = 0.029) deeply infiltrating phenotypes than those without migraine. Previous preclinical evidence highlighted that CGRP-positive sensory nerve fibers are more abundant around lesions of deep endometriosis and create a pro-inflammatory state (28). This micro-environment is able to promote the survival and progression of endometrial cells and to trigger painful symptoms due to heightened levels of neuropeptides (29,30). At the same time, the release of CGRP and the so-called neurogenic inflammation in the trigeminovascular system are also independently crucial for migraine generation (30,31). Thus, we speculate that in comorbid women, severe adenomyosis and DIE might potentiate both abdominal symptoms and headache due to most pronounced CGRP release compared to patients EM-O or MG-O. This hypothesis is also supported by the fact that only 8% of our EM-MG used triptans, a migraine-specific acute therapy acting as potent agonists of 5-HT receptors that appear to be co-expressed with CGRP in trigeminocervical ganglion cells (31). Human CGRP levels were reduced by triptans, coincident with pain relief, at both central and peripheral afferent terminals (31,32). Consequently, we suggest that the scarce triptan use in our comorbid women who are hormonal-drug free, might further enhance the inflammatory setting CGRP-mediated and its persistent excitotoxic activities both in the uterus and in the brain, sustaining more painful and frequent attacks through a self-perpetuating vicious circle (Figure 1). Interestingly, a recent study demonstrated that in women with the comorbidity of migraine and endometriosis CGRP levels rise, with the drop of estrogens, during menstruation. While, in healthy females decrease of CGRP during the menstrual period was observed (7). However, other shared pathogenetic mechanisms should be considered in the strict complex relationship between migraine and endometriosis, including the genetic ones (6).
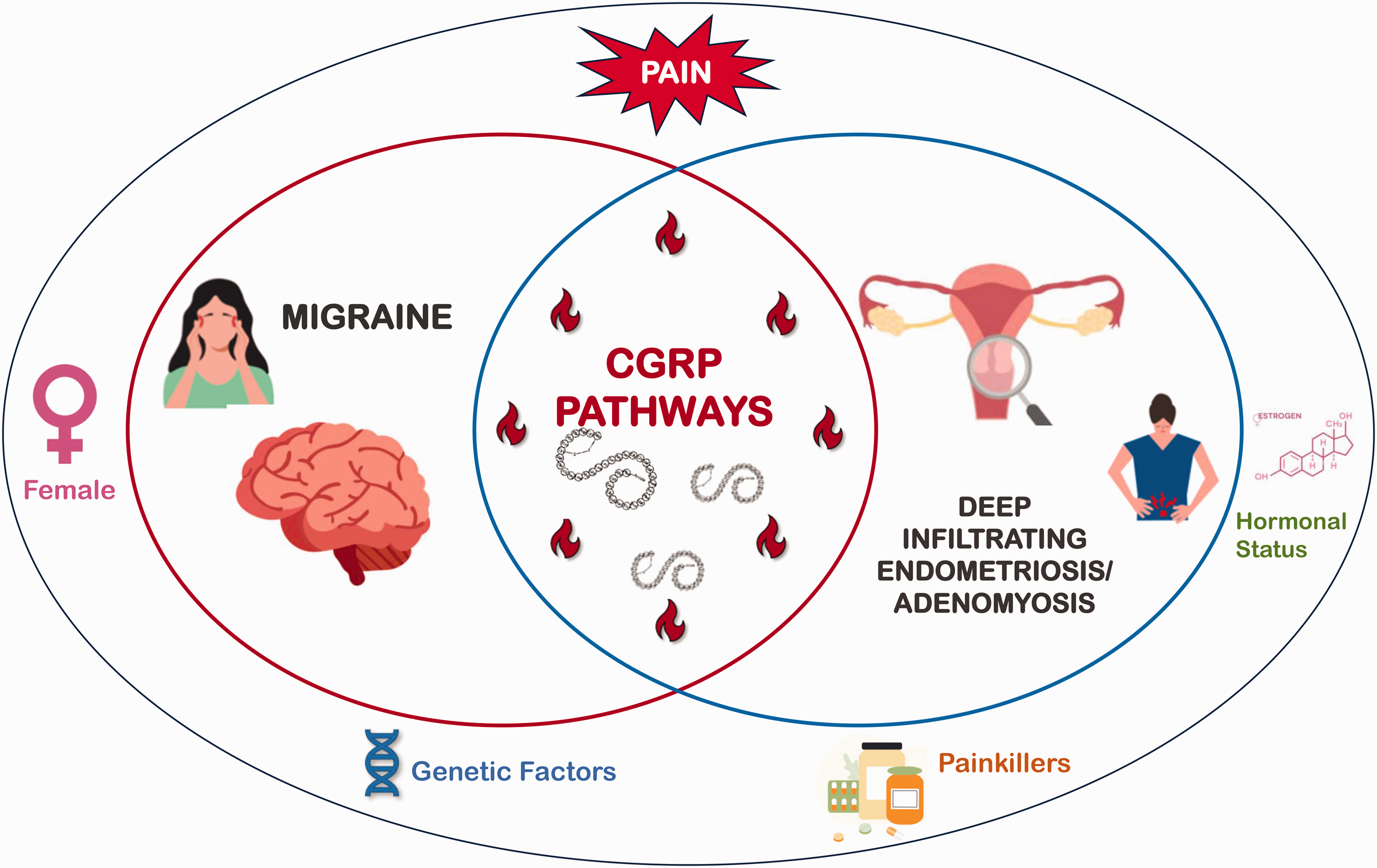
Mutual relationship between migraine and deep infiltrating endometriosis/adenomyosis.
No difference between cases and controls was noted concerning the prevalence of cutaneous allodynia. We have no reliable explanation for this finding, probably biased by the small number of women with chronic migraine included in the study (MMDs ≥15 in the last three months), of which allodynia is a common marker of disease progression (33). Other factors that have been reported to increase the likelihood of having allodynia during migraine episodes are: high body mass index, headache-specific features such as high frequency of aura attacks, and comorbidity with depression and anxiety, that were not present in our cohort of patients (32).
Additionally, our EM-MG patients exhibited higher HIT-6 scores than MG-O, pointing that the disease global burden could increase when both conditions occur together. On the other hand, the MIDAS values were similar among groups of women. Although HIT-6 and MIDAS are complimentary in measuring migraine-related disability, they also show some substantial differences (28–34).
The HIT-6 depends more on headache severity and explores patients’ impressions on how migraine is affecting them (28,34). The total score is obtained by summing the weighted responses to all six questions and a value of ≥60 reflects a greater disability, as reported only in our EM-MG patients (28,34). Unlike the HIT-6, MIDAS is basically a function of migraine frequency and relies on concrete variables (i.e., days of work or school missed) (30,35). A score ≥11 is a marker of negative impact of migraine on various aspects of women’s lives, with pain that can be disabling and compromising family, work, school and social relationships (29,34). Although been pathological in our women with migraine, the mean MIDAS score was similar between patients with and without endometriosis. Consequently, we cannot exclude that it is also somehow related to the societal normalization of women’s pain and stigma around menstrual issues (35). Women often feel ashamed of their condition and the taboo around topics like menstruation or painful sex can prevent them from taking time off from work, seeking help or discussing symptoms with and receiving support from friends, family, employer and colleagues (9,35). Many may be suffering in silence at work, resulting in loss of significant productivity and in missed or delayed diagnosis and treatment. This phenomenon might account for the very high percentage of EM-MG patients in our sample (46/50, 92%) who self-medicate pain without specialist-care for several years before being diagnosed.
Our data were limited by the non-quantitative measurement of inflammatory, genetic and hormonal mediators for exploring their direct impact on the phenotype and severity of the two clustering conditions. Further studies are needed to certify this association, to fully assess their changes in female patients and to develop more individualized therapeutic plans.
Conclusions
Our findings further confirm the comorbidity of endometriosis and migraine and indicate that pain intensity is correlated with disease severity and disability. A detail evaluation of pain problems, clinically and by using non-invasive techniques such as TVS, may represent a powerful tool for earlier diagnosis and an opportunity to monitor disease course, to prevent chronicization and to reduce socio-economic burden, the main unmet needs of affected women. This is of particular relevance in relation to innovative available pharmacological classes of drugs targeting the CGRP pathways (i.e monoclonal antibodies anti-CGRP, antagonists of CGRP receptors and/or agonists of the 5-HT1F receptor) (36), that have recently revolutionized the migraine management and may also be beneficial for comorbid endometriosis.
Clinical implications
Women with endometriosis and migraine presented more severe symptoms and more frequent painful days than patients with only endometriosis and/or migraine alone. These alterations were more prevalent in the presence of adenomyosis and/or deep endometriosis subtypes, leading to increased levels of disability. Early diagnosis of migraine and endometriosis (and of their association), clinically and by using non-invasive ultrasound techniques, could significantly improve the patient management and overall health status.
Footnotes
Author contributions
AS: ultrasonographic scan, design and conceptualized study and interpreted data; CE: ultrasonographic scan and revised the manuscript for intellectual content; CR: ultrasonographic scan; CO: role in technological execution; IM: role in technological execution; GM: ultrasonographic scan; FGM: revised the manuscript for intellectual content; NBM: revised the manuscript for intellectual content; MA: clinical neurological examination and revised the manuscript for intellectual content.
All the authors discussed the results and commented on the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
