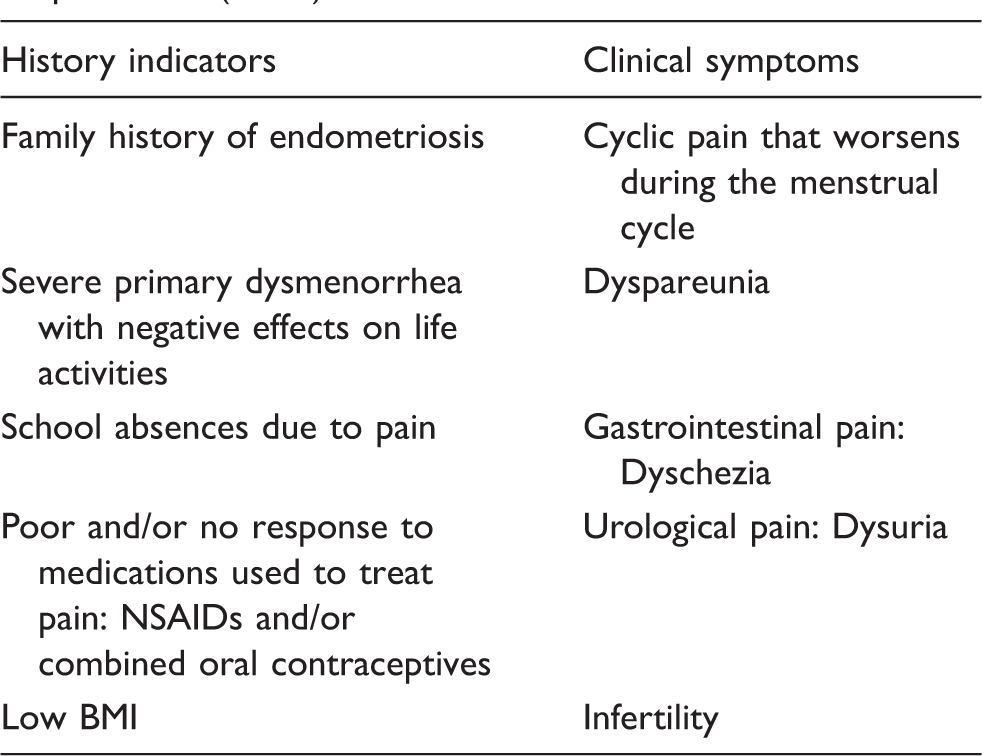
Abstract
Background
Studies have shown a significant association between migraine and endometriosis, but no study has explored the relationship between migraine and endometriosis phenotypes: Superficial peritoneal endometriosis, ovarian endometrioma, and deep infiltrating endometriosis.
Methods
We conducted a case-control study using data collected from 314 women aged 18 to 42 years who had undergone surgery for benign gynecological conditions between January 2013 and December 2015. All women completed a self-administered headache questionnaire according to the IHS classification. Cases (n = 182) are women with histologically proven endometriosis and controls are women (n = 132) without endometriosis. Occurrence of migraine was studied according to endometriosis phenotypes.
Results
Migraine prevalence in cases was significantly higher compared with controls (35.2% vs. 17.4%,
Conclusion
Our study shows a significant association between migraine and endometriosis. In clinical practice, women of reproductive age who suffer from migraine should be screened for endometriosis criteria in order to optimise the medical and therapeutic care of this condition.
Introduction
Endometriosis and migraine are two chronic benign disorders that share many similarities in terms of their clinical manifestations, epidemiology, and pathogenesis. Pain is the main symptom common to both conditions, and it is a source of frequent impairment of quality of life. These two conditions have a high prevalence in women of childbearing age. The overall prevalence of migraine is 15–18% and that of endometriosis is 5–10%, with a peak frequency between 30 and 40 years of age (1,2). Both of these conditions are characterised by a delay in their diagnosis and treatment, and they generally have major socio-economic repercussions (3,4).
The pathophysiological mechanisms underlying endometriosis and migraine have a high degree of similarity (5). They involve a hormonal influence and chronic inflammation phenomena. Indeed, mitogen-activated protein kinase (MAPK) pathways have been implicated directly in the regulation of the pathophysiology of endometriosis (6). Moreover, the synthesis of calcitonin gene-related peptide (CGRP), which plays an important role in the pathophysiology of migraine by a vasodilator effect on cerebral vessels, is thought to be regulated by the MAPK signaling pathway (7). It has also been hypothesised that there are common genetic influences and that cerebral hyperexcitability is involved (8).
Several studies have found that there is a higher prevalence of migraine in women with endometriosis (9–11). On the other hand, it has also been reported that there is a higher prevalence of endometriosis in migrainous patients (12). Moreover, a recent study has shown that adolescents with endometriosis are more likely to experience migraines than adolescents without endometriosis (13).
To our knowledge, no previous study has specifically assessed the risk of the three different endometriosis phenotypes associated with migraine. A better knowledge of these links could lead to better care of these patients, who suffer from recurrent pain.
In the present study, we aimed to determine: a) The prevalence of migraine in women with endometriosis versus women without endometriosis; b) the risk of endometriosis in migrainous women compared to women without headache; c) the risk of endometriosis phenotypes (SUP, OMA, DIE) associated with migraine.
Methods
We conducted a case-control study using data prospectively collected in all non-pregnant patients who were less than 42 years of age and who had undergone surgical exploration by operative laparoscopy for a benign gynecological indication at our institution between January 2013 and December 2015. Women with cancer and/or who refused to consent to the study were excluded from this population.
The indications for surgery (possibly more than one per patient) included: Pelvic pain (defined as the presence of dysmenorrhea and/or intermenstrual pelvic pain and/or dyspareunia of moderate to severe intensity for at least 6 months) (17); infertility, defined as at least 12 months of unprotected intercourse not resulting in pregnancy (18); a pelvic mass (benign ovarian cysts, uterine myomas, etc); and other reasons (e.g. uterine bleeding, request for tubal ligation, infection, etc.). Patients were only deemed to have endometriosis when the lesions were histologically proven. Patients visually diagnosed with endometriosis but without histological confirmation were excluded from the study (19).
For the purpose of this study, the women were divided into two groups based on the histological findings: A control group of women without visual endometriosis lesions, as verified during the operative procedure by a thorough examination of the abdominopelvic cavity (referred to as the control group) and an endometriosis group of women with histologically proven and graded endometriotic lesions (referred to here as the case group).
Moreover, the endometriotic lesions were classified into the three types, SUP, OMA, and DIE. DIE was histologically defined when the muscularis (i.e. the bladder, intestine, ureter, etc) exhibited endometriotic lesions (16). As these three types of endometriotic lesions are frequently associated (20), endometriotic women were classified based on the worst finding. By definition, endometriotic lesions ranked from least to worst are SUP, OMA, and DIE (15).
For each patient, data were collected through face-to-face interviews conducted by the surgeon in the month preceding the surgery. For this, we used a previously published structured questionnaire (19). Briefly, for all of the patients, we collected general information such as their age, gravidity, height, weight, body mass index (BMI), age of menarche, the occurrence of infertility, pelvic pain, and lifestyle habits. Pelvic pain scores and associated symptoms (e.g. dysmenorrhea, deep dyspareunia, non-cyclic chronic pelvic pain, gastrointestinal symptoms, and lower urinary tract symptoms) were assessed preoperatively using a visual analog scale (21).
All of the included patients completed a self-administered questionnaire to assess the occurrence and the type of headache, with specific questions about their characteristics, according to the criteria of the International Headache Society (15). The Head-Hunt headache questionnaire (available in the Appendix) has been validated (22) and it has already been used in a published cross-sectional French study (23). Patients who answered “yes” to the question “Have you had headache in the past 12 months” were classified as headache sufferers. Based on the answers to specific clinical questions about the headache episodes, they were then classified into two groups, as either migraine or non-migrainous headache sufferers. The two diagnoses were mutually exclusive. Patients suffering from secondary headaches were excluded.
The pain intensities (for dysmenorrhea and headache) were evaluated with VAS scores. All of the answers were systematically checked by a physician and supplemented if necessary.
The baseline characteristics were analysed with the standard statistical tests used for cross-sectional data: Means ± the standard deviation (SD) for continuous variables and proportions for categorical variables. Variance analysis and the Chi-2 test were performed to compare quantitative or qualitative variables among groups. The t-test was used to compare quantitative variables between two groups. A two-tailed
The data were analysed for an association between endometriosis and migraine using multivariate analyses. Odd ratios (ORs) and 95% confidence intervals (CIs) were estimated with a logistic regression model using women with neither endometriosis nor migraine as the reference group. For phenotype analysis, a polytomous logistic regression model was used. The ORs were adjusted for age, body mass index, and family history of migraine. The statistical analysis used procedures available in SAS software (SAS Institute Inc, Cary, NC, USA).
Each woman included in the study provided written informed consent. The local ethics committee (CCPPRB: Comité Consultatif de Protection des Personnes dans la Recherche Biomédicale) of our institution approved the study protocol.
Results
Of the 314 consecutive women, 182 (58%) had histologically proven endometriosis and 132 (42%) had no visual endometriosis. In the endometriosis group, 82 patients had headaches, 64 of which were migrainous headaches (Figure 1). Migrainous headache was significantly more frequent in the women with endometriosis than in those without (35.2% vs. 17.4%, respectively, Flowchart inclusion of women.
Women characteristics according to endometriosis status.

The data are presented as means ± the standard deviation for quantitative variables or number (percentage) for qualitative variables.
When we compared the VAS scores for the women with endometriosis, the VAS score for chronic non-cyclic pelvic pain was higher in the women with migraine compared with the women without headache (3.6 ± 2.9 vs. 2.3 ± 2.8; respectively,
Endometriosis risk according to headache types.

The OR was adjusted for the body mass index, age and family history of migraine.
Risk of endometriosis phenotypes associated with migraine.
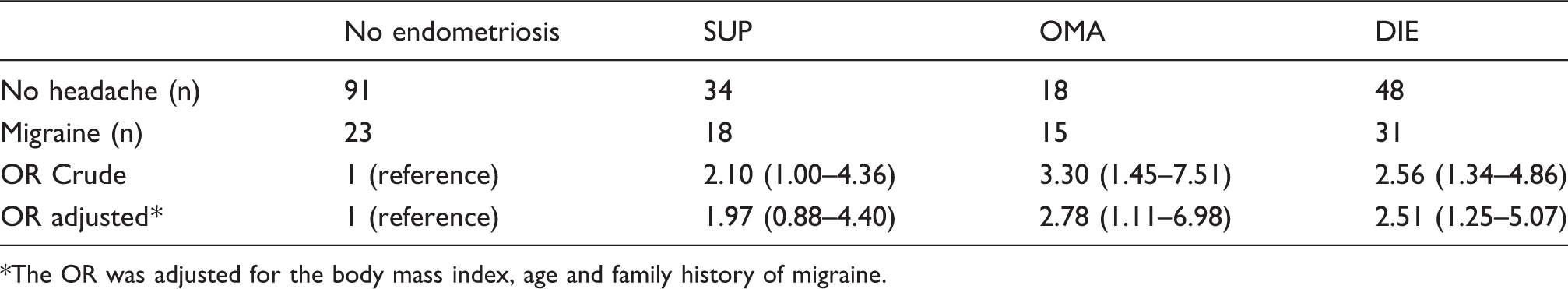
The OR was adjusted for the body mass index, age and family history of migraine.
Discussion
In our study, the prevalence of migraine was significantly higher in the endometriosis group compared with the control group. The risk of endometriosis was significantly higher in the women with migraine. To the best of our knowledge, our study is the first designated to explore the associations between migraine and endometriosis phenotypes. The risk of OMA and DIE was significantly higher in migrainous women, whereas the risk of SUP was not significant.
Our data confirm the results of two other studies: The Italian case-control study published in 2004 that assessed the prevalence of migraine in patients with histologically confirmed endometriosis (10) and a recent study in adolescent women (13). However, neither of these studies took into account endometriosis phenotypes. Unlike the Italian case-control study, very few women suffered from migraine with aura in our study. The very low number of women did not allow us to separately assess the risk of endometriosis associated with both types of migraines.
Our study has several methodological strengths. Firstly, like the Italian case-control study (10), all of the included women had histological evidence confirming the diagnosis of endometriosis. However, most of the studies to date that assessed this association relied solely on self-reported clinical criteria or on medico-administrative databases, which do not provide a validated diagnosis of endometriosis (11,24). Secondly, a self-administered “headache” questionnaire was used to determine the occurrence and the type of headache as either a migraine or a non-migrainous headache. The self-questionnaire had previously been validated for the diagnosis of migraine with a PPV of 84% and a VPN of 76% (21). The corresponding French questionnaire (available in the Appendix) was used previously in a cross-sectional French study (23). Thirdly, the prevalence of migraines (17.4%) observed in our control group is consistent with findings for the French general population, for which the prevalence of migraine in women has been estimated to be 17.6% (25).
Our study has some limitations. There was a possible selection bias due to the specificity of the study design, as it only included surgical patients in a referral center that specialises in endometriosis surgery. Therefore, women referred to our center may have suffered from particularly severe forms of endometriosis that are frequently associated with a high level of pain. Another limitation is the small number of patients in each phenotype of endometriosis.
The understanding of the pathogenesis underlying the association between endometriosis and migraine remains incomplete as the exact mechanisms of each of these two pathologies are not fully understood. The mechanisms by which migraine increases the risk of endometriosis remain speculative, but the following hypotheses are plausible.
A first possibility is the role of estradiol in the modulation of pain. This is supported by the fact that endometriosis and migraine are two chronic pain conditions with peak prevalence between puberty and menopause, thus suggesting a key role of estradiol in their pathophysiology. Indeed, several studies have shown that women have a lower pain threshold than men. In addition, estradiol may act on peripheral and central pain signaling pathways, thus modulating the perception of pain through neurotransmitters such as endorphins, dopamine, serotonin, and gamma-aminobutyric acid (GABA) (26).
A second possibility is that inflammation and the MAPK signaling pathway play a key role. The synthesis of CGRP, which plays an important role in the pathophysiology of migraine by a vasodilator effect on cerebral vessels, is thought to be regulated by the MAPK signaling pathway (7). It has been suggested that activation of the MAPK signaling pathway can increase the expression of the CGRP gene during a migraine attack (5). Moreover, endometriosis has been shown to be associated with elevated levels of pro-inflammatory cytokines (6). Among these cytokines, polymorphisms of the interferon gamma (IFNγ) gene are thought to be involved in the severity of endometriosis. IFNγ activates the MAPK signaling pathway, thereby inducing the proliferation and survival of ectopic endometrial cells. However, it should be noted that no study to date has demonstrated a direct link between the secretion of an inflammatory mediator by ectopic endometrial tissue and the onset of migraine.
A third possibility is a central and peripheral hypersensitivity, as suggested by an increasing number of studies (27). On the one hand, in migraine there is a decrease in the threshold for triggering pain during seizures as well as during the crisis-free interval (28). On the other hand, immunohistochemical studies suggest a determining role of neurotrophins and central hyperexcitability in the genesis of pain in endometriosis (29). Central and peripheral hypersensitivity may be promoted by prostaglandin and NO, which are common biochemical mediators both in endometriosis and in migraine. Indeed, prostaglandins play an important role in triggering migraine (30) and their production is increased in endometriosis (31). Furthermore, NO is a key molecule in the regulation of cerebral blood flow, angiogenesis, and nociception that has been centrally implicated in the pathophysiology of headaches (32). There is also enhanced production of NO in endometriosis (33). Moreover, repetitive and persistent stimuli contribute to central sensitisation. We can make the hypothesis that the effect of central sensitisation due to pain could explain the association between migraine and endometriosis.
A fourth hypothesis is a shared genetic susceptibility. An Australian study investigated whether the combination of the two pathologies is the result of chance, selection bias, or common environmental and genetic factors. The authors investigated the co-occurrence of both pathologies among a cohort of 931 families with at least two sisters with surgically confirmed endometriosis, 815 pairs of monozygotic binoculars, and 415 pairs of dizygotic binoculars. The results of this study suggest that endometriosis and migraine have common genetic predispositions (8). To date, the genes responsible remain unknown. A number of authors have suggested a possible role for the estrogen receptor gene (34).
Finally, recent results from metabolomic analyses have suggested that the ketone pathway may be disrupted in both endometriosis and migraine (35,36). Ketones are involved in dopaminergic and GABAergic activation pathways in mice and rats. These neurotransmitters are involved in pain signaling pathways.
In our study, the most significant risk occurred in the subgroup of women with endometrioma. The hypotheses of alteration of ovarian endocrine functions in the presence of endometrioma or modifications of vascular growth factor expression such as vascular endothelial growth factor (VEGF) in the surrounding ovarian tissues could contribute to explaining our results (37,38).
Another explanation is that associated ovarian endometrioma is a marker for the severity of the DIE (39). The intensity of the pain is proportional to the penetration depth of the DIE lesions (40). Women suffering from migraine also experience severe pain. When we compared the VAS scores among the women with endometriosis, the VAS scores for chronic non-cyclic pelvic pain were higher in the women with migraine compared with the women without headache. These results support the hypothesis of central heightened pain sensitivity in women suffering from these two conditions. Indeed, chronic pain conditions can cluster, and having one pain condition can increase the likelihood of developing another.
In conclusion, our study shows a significant association between migraine and endometriosis. In clinical practice, women of reproductive age who suffer from migraine should be screened for endometriosis criteria to improve the delay in the diagnosis of endometriosis and to optimise the medical and therapeutic care of this condition. Further epidemiological studies including a larger number of patients and fundamental research are needed to confirm these results and to better understand the pathophysiological mechanisms underlying endometriosis and migraine.
Footnotes
Clinical implications
Prevalence of migraine is significantly higher in women with endometriosis as compared with women without endometriosis.
The higher risks appear in migrainous women with deep infiltrating endometriosis or endometrioma.
Women who suffer from migraine should be screened for endometriosis criteria.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.