Abstract
To investigate the role of plasma calcitonin gene-related peptide (CGRP) in paediatric migraine, we prospectively collected 134 blood samples during or between attacks from 66 migraine, 33 non-migraine headache (non-migraine) and 22 non-headache patients, aged 4–18 years. Plasma CGRP concentrations were measured by enzyme-linked immunosorbent assay and disability by Pediatric MIgraine Disability ASsessment (PedMIDAS) questionnaire. Migraineurs had higher plasma CGRP levels than non-migraine patients (P = 0.007). The attack level was higher than the non-attack level in migraine (P = 0.036), but not in non-migraine, patients. This was also revealed in paired comparison (n = 9, P = 0.015 vs. n = 4, P = 0.47). Using a threshold of 55.1 pg/ml, the sensitivity of the attack level in predicting migraine was 0.81, and specificity 0.75. The PedMIDAS score tended to be higher in the high CGRP (> 200 pg/ml, n = 7) group than in the low (< 200 pg/ml, n = 33) group (26.07 vs. 19.32, P = 0.16) using Mann–Whitney test. Plasma CGRP is useful for diagnosis in paediatric migraine.
Keywords
Introduction
Migraine is a complex, disabling disorder of the brain that is characterized by attacks of severe throbbing headaches with sensory sensitivity to light, sound and head movement (1). It is among the most common disorders and remains one of the leading causes of disability (2). The diagnosis of migraine is based on clinical features. The diagnostic biomarkers have rarely been established for clinical application. Especially for young children who cannot clearly describe their symptoms, the importance of developing a biomarker for the diagnosis and monitoring of migraine cannot be overemphasized.
Calcitonin gene-related peptide (CGRP), a 37-amino-acid neuropeptide, is one potential biomarker; extensive studies have been done to test its key role in the pathogenesis of migraine. It is the most potent peptidergic dilator of peripheral and cerebral blood vessels (3, 4). In the trigeminovascular system, trigeminal ganglionic cell bodies constitute the main source of CGRP and send axons peripherally to the adventitial–medial junction around the middle meningeal arteries (5). Thus, trigeminal activation may induce CGRP release from perivascular nerve endings, resulting in pia vessel dilation, which is believed to be a cause of migraine (6).
Clinically, in adult migraineurs, baseline CGRP levels have been found to be considerably higher than in healthy controls (7). CGRP, but not other neuropeptides, is elevated in external jugular venous blood during migraine headache (8), and its changes are correlated with headache intensity (7). Intravenous infusion of CGRP produced a migraine-like headache in migraine volunteers (9), implying that CGRP plays a causative role in migraine. Furthermore, the CGRP receptor antagonist, BIBN 4096 BS, is effective in treating acute migraine attacks (10). Triptans, a clinically effective antimigraine agent, has been found to reduce CGRP level in cats and humans (11).
There are rare studies exploring the diagnostic value of plasma CGRP in paediatric migraine. Gallai et al. (12) found that plasma CGRP levels during attacks were elevated compared with baseline levels in juvenile migraineurs. However, it is uncertain whether CGRP is useful in differentiating migraine from non-migraine headaches in children. Moreover, no data exist concerning the correlation between plasma CGRP level and migraine disability. Therefore, we investigated the value of plasma CGRP in the diagnosis and prediction of disability in paediatric migraine.
Methods
Study subjects
We prospectively recruited 123 study subjects, aged 4–18 years, from January 2004 to December 2006 at National Taiwan University Hospital, a tertiary medical centre in Taiwan. Two were later excluded due to their regularly taking unknown herbal drugs. A total of 121 enrolled subjects were divided into migraine, non-migraine headache (non-migraine) and non-headache groups. The diagnosis of paediatric migraine was based on the criteria of the International Headache Society (13). Those patients who suffered from headaches without fulfilling the diagnostic criteria of migraine were classified into the non-migraine group, including primary tension-type headache, epileptic headache and headache secondary to sinusitis. The non-headache group included age-matched patients who visited our out-patient clinic without recurrent headache. Exclusion criteria of the study subjects consisted of the following conditions: herbal drug intake, intracranial mass, mental retardation, congenital or chromosomal anomaly, other major organ diseases and pregnancy. Headache condition, attack or non-attack, at blood sampling was recorded. The blood at attack was sampled at any time during the first 8 h of the headache episode. Non-attack was patients with headache free for ≥ 24 h before and after sampling. Data collected included age, sex, age of onset, history of allergy (allergic rhinitis, asthma or atopic dermatitis) or tics, and whether there was a family history of headache or migraine, memory impairment, food-related attacks (specific to ice, cheese, chocolate or monosodium glutamate), and headache release by sleep. Comprehensive cardinal symptoms of migraine were recorded, including presence of aura, unilaterality, throbbing headache, aggravation by activity, nausea, vomiting, photophobia, phonophobia and release by sleep. Other associated symptoms recorded included dizziness or vertigo, abdominal pain, chest pain, blurred vision, tinnitus, allodynia, pallor and sweating. Electroencephalogram (EEG), computed tomography or magnetic resonance imaging examinations, if necessary, were performed to exclude brain organic lesions. The study was approved by the Scientific Ethics Committee of National Taiwan University Hospital. Signed, informed consent was obtained from all patients and their parents.
Disability assessment
The Pediatric MIgraine Disability ASsessment (PedMIDAS) questionnaire, a six-question tool, has been proved to be a reliable assessment of the impact of paediatric headache in the 3 months prior to their presentation at hospital (14). A Chinese version of this questionnaire, prepared with the agreement of Dr Hershey (University of Cincinnati, Cincinnati, OH, USA), who developed PedMIDAS, was used to assess the level of disability of paediatric migraine. Those patients with migraine, with the help of their family, were asked to complete the questionnaire on the day of blood sampling.
Plasma CGRP measurement
Three millilitres of blood was sampled from the cubital vein of each patient and put into a 5-ml Lavender tube (BD vacutainer; Becton Dickinson, Plymouth, UK). Plasma samples were prepared by centrifuging (750 g, 15 min) the whole blood and then stored at −80°C. Plasma CGRP levels were measured by a commercial enzyme-linked immunosorbent assay (ELISA) kit (SPI-BIO, Montigny-le-Bretonneux, France). A plasma sample from the same volunteer was used in each ELISA plate as an internal control. The person conducting CGRP measurement was blind to the identity, diagnosis and attack status of the patients.
Statistical analysis
Data were expressed as means ±
Results
Patient characteristics
Of 121 study subjects, 66 were children (39 boys) with migraine, 33 (13 boys) were non-migraine headache (non-migraine) and 22 (13 boys) non-headache patients. All patients received one blood sampling either during or between attacks except for nine migraine and four non-migraine patients who received paired comparison in attack and non-attack states. In total, 25 samples were collected from migraineurs during attack and 50 between attacks; 13 were from non-migraine patients during attack and 24 between attacks. The time points of blood sampling from the onset of headache attack were similar in the migraine and non-migraine patients (2.3 ± 0.3 h vs. 2.4 ± 0.5 h). Either the migraine (11.6 ± 0.4 years) or non-migraine (9.9 ± 0.7 years) group was age-matched to the non-headache group (10.1 ± 0.8 years, P > 0.05), but migraineurs were significantly older than non-migraine patients (P = 0.023) by statistical post hoc analysis. These differences were attributed to the fact that the girls in the migraine group were older than boys in the non-migraine group (P = 0.001). The sex distribution in the three groups was, however, not significantly different.
Demographic features
The demographic features in the migraine and non-migraine groups are compared in Table 1. The percentage of migraineurs with maternal history of migraine or headache was higher than of the non-migraine patients (P = 0.043), whereas the difference was insignificant in paternal inheritance. Other features of family history, age of onset, and comorbidity with tics or allergy were not significantly different.
Demographic features in migraine and non-migraine headache patients
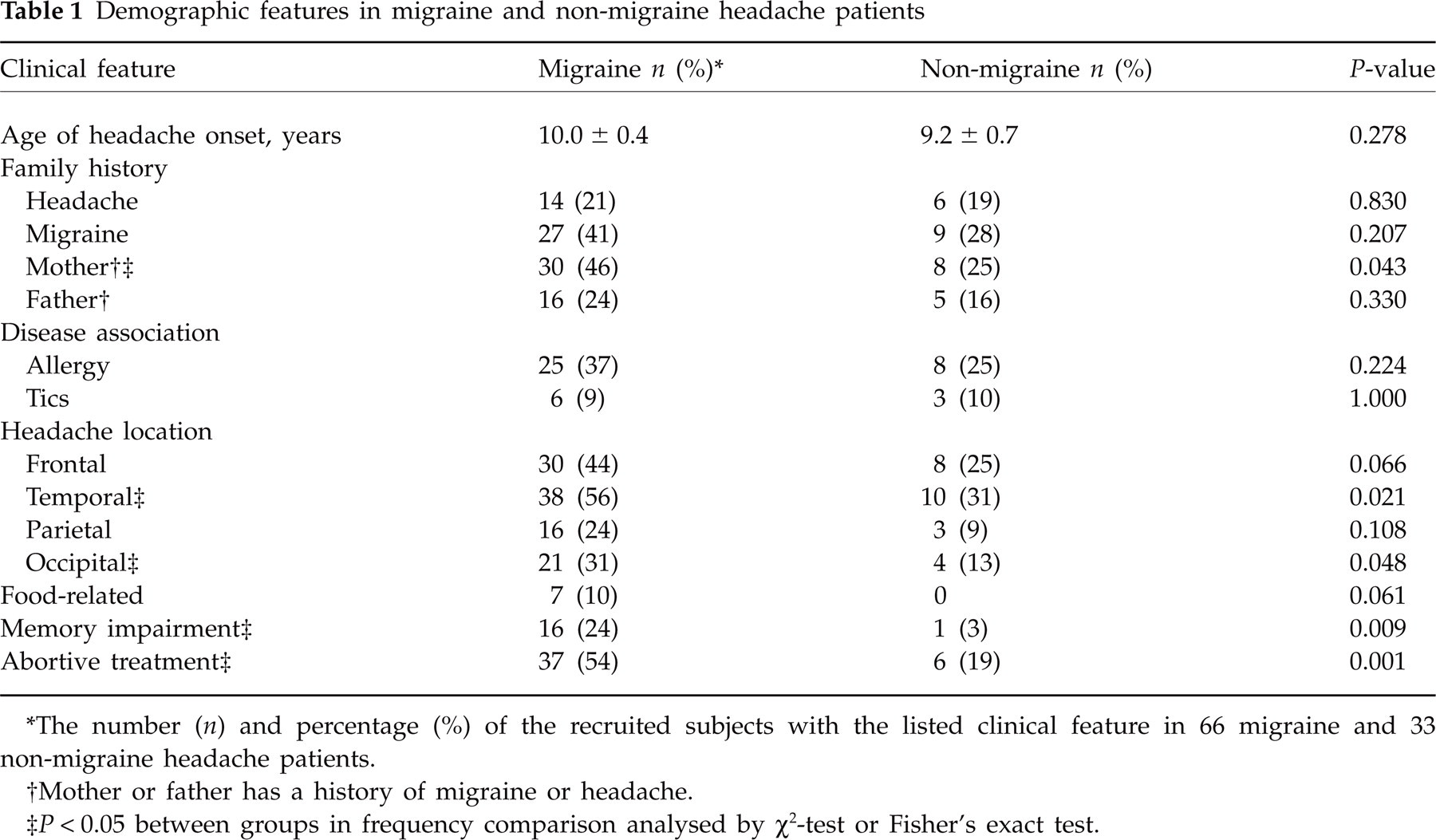
∗The number (n) and percentage (%) of the recruited subjects with the listed clinical feature in 66 migraine and 33 non-migraine headache patients.
†Mother or father has a history of migraine or headache.
‡ P < 0.05 between groups in frequency comparison analysed by χ2-test or Fisher's exact test.
More frontal and temporal headaches were observed in both groups. Headaches located in the temporal or occipital, but not frontal or parietal, regions were more frequently observed in the migraine compared with the non-migraine group. Ten per cent of the migraineurs suffered from food-related attacks, whereas none was affected by food in the non-migraine group. The probability of memory impairment was significantly higher in the migraine group. More migraineurs than non-migraine patients needed abortive therapy.
Plasma CGRP and migraine diagnosis
When all blood samples were pooled, the plasma CGRP level in the migraine group (154.7 ± 26 pg/ml, n = 75) was significantly higher than in the non-migraine (49.9 ± 4.3 pg/ml, n = 37, P = 0.007) and non-headache (52.4 ± 5.6 pg/ml, n = 22, P = 0.033) groups. The CGRP levels during attack (236.8 ± 43.3 pg/ml) were significantly higher than the non-attack levels (113.6 ± 31.1 pg/ml) in migraineurs, and the attack (55.0 ± 9.2 pg/ml) or non-attack (47.1 ± 4.0 pg/ml) levels in the non-migraine group (Fig. 1). The non-attack CGRP levels in migraineurs did not significantly differ from those in the non-migraine group (open bars, Fig. 1). In the non-migraine group, attack and non-attack CGRP levels were insignificantly different (Fig. 1). In patients who underwent paired comparison, the attack CGRP level was higher than the non-attack level in the migraine group (197.8 ± 79.2 vs. 46.2 ± 6.4 pg/ml, P = 0.015), but not in the non-migraine group (43.6 ± 2.3 vs. 40.8 ± 8.9 pg/ml, P = 0.47) (Fig. 2B). In seven of nine migraineurs, the CGRP level during attack was > 30% higher than that between attacks (Fig. 2A). CGRP levels in patients with different migraine subtypes, migraine without aura (MoA) (124.2 ± 38.0 pg/ml, n = 40), basilar type (BT) (255.8 ± 51.7 pg/ml, n = 20) and migraine with aura (excluding BT) (MA) (101.2 ± 30.7 pg/ml, n = 15), tended to be different (P = 0.059;

Plasma calcitonin gene-related peptide (CGRP) levels in the migraine and non-migraine headache groups. Plasma CGRP levels during attack (filled bars) or between attacks (open bars) in the migraine and non-migraine groups are compared. The n number in each group is depicted on the bar. P-values analysed by post hoc Bonferroni test are denoted if < 0.05.

Paired comparisons of attack and non-attack plasma calcitonin gene-related peptide (CGRP) levels. Individual (A) and average (B) plasma levels in nine migraineurs (M1–M9) and four non-migraine children (H1–H4) during (filled bars) and between (open bars) attacks are compared. Note that in seven of nine migraineurs, the CGRP level during attack is > 30% higher than that between attacks. Wilcoxon signed rank test was used.
The diagnostic values of plasma CGRP and demographic variables in predicting migraine were analysed by binary logistic regression. This revealed, first, that plasma CGRP level was the only significant predictive factor for the diagnosis of migraine (P = 0.021), but not the other variables, including age, sex, family history, allergy or tic association, food-related attacks, memory impairment, EEG abnormality, headache locations, and associated symptoms. Furthermore, migraine diagnosis in the attack and non-attack patients with migraine or non-migraine headache was associated with the logarithm of the CGRP levels of attack (P = 0.013) and non-attack (P = 0.031) samples, respectively. The degree of association between CGRP levels and the diagnosis of migraine, expressed by odds ratio, was 39.5 and 13.6 for attack and non-attack blood samples, respectively. These results suggest that the probability of migraine diagnosis increases as plasma CGRP levels elevate, and the diagnosis of migraine is more strongly associated with the attack CGRP level than the non-attack level. ROC curve analysis indicated that the best positive predictive threshold value of the attack CGRP level in diagnosing migraine was 55.1 pg/ml, with sensitivity of 0.81, specificity 0.75, and positive likelihood ratio 3.24, whereas that of the non-attack level revealed indistinctive findings.
Plasma CGRP and migraine disability
Of 66 migraineurs, 40 children (61%; mean age 11.9 ± 0.5 years) completed the PedMIDAS questionnaire. The patients were divided into high (n = 7) and low (n = 33) CGRP groups with a cut-off value of 200 pg/ml, and their PedMIDAS scores were compared. The mean score in the high CGRP group was higher than that in the low CGRP group (78.4 ± 39.9 vs. 26.2 ± 7.6, P = 0.24) using Student's t-test. Since the data distribution was not even, Mann–Whitney test was used to avoid Student's t-test being sensitive to the extreme values. The mean rank was higher in the high than in the low CGRP group (26.07 vs. 19.32, P = 0.16). However, the differences were not statistically significant.
Plasma CGRP and demographic variables
The plasma CGRP levels in patients with and without various demographic variables in both migraine and non-migraine groups are compared in Table 2. On univariate regression analysis, plasma CGRP levels significantly correlated to familial headache or migraine of paternal inheritance, but not maternal inheritance. CGRP levels were also correlated to allergy association, frontal and occipital headaches. Among the evaluated cardinal and associated symptoms of migraine, only aggravation by activity was correlated to CGRP level. Furthermore, patients with headache aggravated by activity had a higher CGRP level than those without this symptom (P = 0.005; Student's t-test). The correlation of plasma CGRP levels to the requirement of abortive therapy was significant. Plasma CGRP levels in patients requiring abortive treatment were significantly higher than those not (P = 0.018; Student's t-test). Simultaneously, we recruited the demographic variables into a multivariate linear regression model. It revealed that the plasma CGRP level correlated to the diagnosis of migraine (P = 0.014) only by controlling age, sex, age of onset, family history, comorbidity of tic or allergy, food-related attacks, memory impairment, and EEG abnormality.
Univariate analysis of the correlation between each demographic variable and plasma calcitonin gene-related peptide (CGRP) levels taken from both migraine and non-migraine headache patients

∗The plasma CGRP level (mean ± S.E.) in migraine and non-migraine headache patients with (positive) or without (negative) the listed clinical feature.
† P-value and
¶ P < 0.05 between positive and negative groups in CGRP level comparison analysed by univariate regression.
‡Mother or father has a history of migraine or headache.
§The number of blood samples.
∗∗ P < 0.05 between groups in CGRP level comparison analysed by Student's t-test.
Discussion
We found that plasma CGRP levels in children with migraine were higher than in those with non-migraine headache. In the migraine group, the attack CGRP levels were significantly elevated compared with non-attack levels. The PedMIDAS score tended to be higher in patients with higher CGRP levels. These data suggest that the plasma CGRP level can be of help in differentiating migraine from non-migraine headaches in children during headache attacks. To the best of our knowledge, this is the first study to assess the value of the plasma CGRP level in the diagnosis of migraine in children.
In children with migraine, we found that plasma CGRP levels during attack were elevated compared with non-attack levels, similar to what has been reported for juveniles (12) and adults (7). However, the finding that non-attack CGRP levels were not significantly different between children with migraine and non-migraine headaches differs from results reported for adults, whose baseline CGRP levels were considerably higher in migraineurs (7). This difference may be attributed to the fact that migraine is less severe in childhood in terms of having shorter duration, less frequent attacks and better response to treatment (16). This suggests that a higher plasma CGRP level during headache attack, but not between attacks, can be used to differentiate migraine from non-migraine headache children. Analysis of the odds ratio also showed that the diagnosis of migraine is more strongly associated with attack CGRP levels compared with non-attack levels. ROC curve analysis further suggests that the plasma CGRP level is a good predictor in the diagnosis of paediatric migraine in terms of sensitivity and specificity. However, CGRP levels from two (M1 and M3) of the nine migraineurs with paired comparison were not elevated as were the other seven patients (Fig. 2). It suggests that there may be a CGRP-independent population in the migraine group. This inference was also supported by the partial clinical effectiveness of CGRP-regulating agents. The CGRP receptor antagonist BIBN 4096 BS is effective in only 60% of the migraine population (10), as well as sumatriptan, treating migraine through reducing CGRP release (17). Therefore, some migraine attacks are not CGRP mediated.
PedMIDAS was developed by Hershey et al. (14) to assess the overall impact of headaches in relation to headache frequency and pain intensity over the 3 months before compilation. It provides a developmentally sensitive, reliable and valid assessment of the disability of childhood and adolescent headaches. We found that the mean score and mean rank of PedMIDAS score were higher in patients with higher CGRP levels, although not significantly so. This statistical insignificance in difference may result from large variance and small case number, especially with only seven patients in the high CGRP group. A chronic biomarker, in addition to CGRP, which might be an acute biomarker, is warranted for better prediction of the PedMIDAS score.
We have made the interesting finding that patients with a positive paternal migraine or headache history have higher CGRP levels, suggesting that paternal transmission is related to a more severe phenotype. On the other hand, our data show that maternal inheritance is more obvious in the migraine group (Table 1). This might be attributed to the two- to threefold higher prevalence in women than in men (18, 19).
The CGRP levels also correlated to allergy comorbidity by univariate analysis. This is in agreement with the involvement of neuroinflammation in the pathogenesis of migraine. CGRP could activate the dural mast cells, critical players in allergic reactions, located perivascularly near the trigeminal nerve terminals, leading to inflammatory mediator secretion, thereby contributing to migraine pathogenesis (20). On the other hand, the tic association was comparable in both groups (9–10%) but moderately higher than in the normal population of Taiwanese children (5.5%) (21). Nevertheless, our results show that both allergy and tics were insignificant confounding factors to the prediction of migraine by plasma CGRP levels in the multivariate regression analysis.
Our findings of headache predilection over frontal and temporal regions in migraine children are compatible with what has been reported (16). It is of interest that subjects with occipital headaches tended to be migraineurs and had higher plasma CGRP levels. However, CGRP levels in the basilar subgroup of migraineurs were marginally higher than those in the other subgroups. This discrepancy may be attributed to the higher CGRP levels of patients suffering from occipital headaches in the non-migraine group. It is very likely that these patients with occipital headache might evolve into migraineurs in the future.
Some limitations of this study need to be addressed. First, diagnosis of migraine in young children is difficult because these patients may not be able to describe the subjective characteristics of their headaches precisely, despite parental help. This may explain why migraineurs in this study were older than in the non-migraine group. Second, patients with mild symptoms of headaches would not seek medical attention. Therefore, it is possible that only patients with greater headache intensity were included in this study. Third, to specify the onset and duration of headache on blood sampling was hard for paediatric patients, especially for those with multiple episodic attacks during 1 day. The duration effects, which might cause variation in attack levels, had not been controlled in this study. The evolution of plasma CGRP over time during headache in children remains to be elucidated.
In conclusion, the plasma CGRP level during headache attack is valuable in differentiating migraine from non-migraine headache in children. These results may provide new insight into the diagnosis of migraine in children.
Footnotes
Competing interests
None to declare.
Acknowledgements
The authors thank Mr Wei-Nong Chang, Mr Chi-Ping Chuang and Ms Sui-Han Chen for their technical support. This study was supported by the grants from National Taiwan University Hospital (NTUH94M28), National Science Council, Taiwan (NSC93-2314-B002-179, NSC 95-2320-B002-095-MY3), National Health Research Institutes, Taiwan (NHRI-EX96-9506NI) and Department of Health, Taiwan (DOH96-NNB-1027).
