Abstract
Background
Comparative evaluations of preventive migraine treatments can help inform clinical decision making for managing migraine in clinical practice.
Methods
An anchored matching-adjusted indirect comparison analysis was conducted using pooled participant-level data from two phase 3 atogepant trials (ADVANCE and PROGRESS) and one phase 2/3 rimegepant trial (BHV3000-305) to evaluate the relative efficacy and safety/tolerability of atogepant and rimegepant as preventive migraine treatments. Participants receiving atogepant 60 mg once daily, rimegepant orally disintegrating tablet 75 mg once every other day, and placebo were included. Only participants meeting the BHV3000-305 inclusion/exclusion criteria were analyzed: ≥6 monthly migraine days and ≤18 monthly headache days at baseline. The primary efficacy assessment of interest was change in monthly migraine days across weeks 1–12.
Results
There were 252 participants in the atogepant group and 348 in the rimegepant group. Across weeks 1–12, atogepant 60 mg demonstrated a significantly greater reduction in mean monthly migraine days compared with rimegepant 75 mg (mean difference [95% CI]: −1.65 [−2.49, −0.81]; p < 0.001). Both atogepant and rimegepant demonstrated similar safety/tolerability profiles.
Conclusion
In this matching-adjusted indirect comparison analysis, oral atogepant 60 mg once daily demonstrated a significantly greater reduction in monthly migraine days compared with rimegepant 75 mg orally disintegrating tablet once every other day.
Introduction
Migraine is one of the most common neurologic diseases, the second leading cause of disability worldwide (1,2) and is generally categorized as episodic (<15 monthly headache days [MHDs]) or chronic (≥15 MHDs, including ≥8 migraine days) (3,4). The negative impact of migraine on functioning and quality of life (QoL) is related to attack frequency and severity (5,6). Migraine treatments are classified as either acute or preventive and can be used individually or combined based on the characteristics and impact of migraine (6). Preventive migraine treatments consist of traditional non-migraine specific drug classes (e.g., antiseizure medications, beta blockers) and novel migraine-specific drug classes targeting calcitonin gene–related peptide (CGRP) (5,7,8). Atogepant and rimegepant are oral small-molecule CGRP receptor antagonists used for the preventive treatment of migraine (9–13). Atogepant is indicated for the preventive treatment of episodic migraine (EM) and chronic migraine (CM), while rimegepant is indicated for the preventive treatment of EM and as an acute migraine treatment.
In the pivotal phase 3 ADVANCE clinical trial, adults with EM (4–14 migraine days per month) were randomized to receive once-daily oral atogepant 10 mg, 30 mg, or 60 mg, or placebo for 12 weeks (10). Regardless of dose, atogepant significantly reduced the number of migraine days compared with placebo, with similar incidences of adverse events observed across groups (10). The safety, tolerability, and efficacy of oral atogepant 10 mg once daily, 30 mg once daily and twice daily, and 60 mg once daily and twice daily was also demonstrated in a phase 2/3 trial (CGP-MD-01) for the preventive treatment of EM using the same criteria as ADVANCE (14). In the phase 3 PROGRESS clinical trial, adults with CM, defined as experiencing ≥15 MHDs including ≥8 migraine days, were randomized to receive atogepant 30 mg twice daily, 60 mg once daily, or placebo (11). Both doses of atogepant significantly reduced monthly migraine days (MMDs) compared with placebo and were well tolerated with no new safety findings identified.
Rimegepant is an orally administered, small-molecule CGRP receptor antagonist that is an efficacious, safe, and well-tolerated preventive treatment for EM (13). In a phase 2/3 randomized clinical trial (BHV3000-305), adults experiencing ≥4 and ≤18 moderate-to-severe migraine attacks per month over a 3-month screening period were subsequently randomized to receive oral rimegepant 75 mg or placebo every other day for 12 weeks following a 1-month observation period during which participants were required to experience ≥6 migraine days (13). Rimegepant was more effective than placebo at reducing the mean MMDs at weeks 9–12 and several secondary endpoints with the exception of monthly rescue medication use and migraine-related disability (13). The safety of rimegepant was similar to placebo and consistent with previous studies (13,15,16).
Although atogepant and rimegepant have similar mechanisms of action and are both administered orally, there are no head-to-head studies comparing their efficacy, safety, and tolerability (9,12). Comparative evaluations of preventive migraine treatments can help to inform clinical decision making for managing migraine in practice. Due to differences in enrolled atogepant and rimegepant study populations, the potential for conducting standard indirect treatment comparisons using traditional methods is limited. Advanced population-adjustment methods that utilize participant-level data may provide a more precise estimate of the relative effect between treatments. The current analyses evaluated the relative efficacy, impact on QoL, safety, and tolerability of atogepant compared with rimegepant for the preventive treatment of EM using an anchored matching-adjusted indirect comparison (MAIC) analysis, an established means of comparing results from studies with different methodologies (17).
Methods
Study design
This MAIC analysis included pooled data from two phase 3 atogepant trials (ADVANCE [NCT03777059] and PROGRESS [NCT03855137]) and one phase 2/3 rimegepant trial (BHV3000-305 [NCT03732638]) (10,11,13). CGP-MD-01 was a dose-finding study that assessed unapproved doses, leading to more participants receiving atogepant than placebo compared with phase 3 trials, and used International Classification of Headache Disorders, 3rd edition (ICHD-3), beta diagnostic criteria, whereas the remaining studies used ICHD-3 criteria (14,18,19). CGP-MD-01 also did not include QoL outcomes. For these reasons, it was omitted from the base case analysis, although it was included in a scenario analysis (10,11,13). Additional details about the study designs are provided in Online Supplemental Table 1. Participants receiving atogepant 60 mg once daily and rimegepant 75 mg once every other day (administered as an orally disintegrating tablet [ODT]), were included. Participants receiving placebo were also included to allow for an anchored comparison.
Inclusion and exclusion criteria for baseline MMDs and MHDs varied across trials and are described in Online Supplemental Table 2 (10,11,13). These analyses included only those participants who met the BHV3000-305 inclusion and exclusion criteria of ≥6 migraine days and ≤18 migraine/non-migraine headache days during the four-week, pretreatment observation period (13) using pooled atogepant data from ADVANCE (EM) and PROGRESS (CM). Therefore, these analyses were conducted on a mixed population of individuals with EM or CM, as per the ICHD-3 criteria.
MAIC analysis
An anchored MAIC, a form of propensity score weighting, was conducted utilizing pooled participant-level data from the atogepant studies to make an adjusted comparison between the atogepant and rimegepant trial populations (Figure 1). Atogepant trial participants were assigned statistical weights using MAIC to adjust for their over- or under-representation relative to the average treatment-effect modifiers observed in the rimegepant trial. To identify treatment-effect modifiers, a subset of data for the baseline characteristic category of interest was taken, and separate univariate regression models (e.g., logistic regression for binary data, mixed-effects models for repeated measures for continuous data) were performed, including a treatment covariate within each subset. For example, to investigate the impact of sex on treatment effect, the subgroup of data for participants who were male was selected, and a regression model with treatment as the only covariate was performed. A separate regression (with treatment as the only covariate) for the subgroup of female participants was also performed. After the matching procedure was conducted and the weights were derived, outcomes were compared between balanced treatment groups using analyses that incorporated the derived weights between atogepant and rimegepant.

Study design of the matching-adjusted indirect comparison analysis.
Regression models that included treatment as a covariate and utilized the observed outcome data with the derived weights for the pooled atogepant population were used to estimate the adjusted treatment effect for atogepant compared with placebo. Odds ratios (ORs) were used to describe binary endpoints, and mean differences (MD) were used to describe continuous endpoints. To estimate the relative treatment effect between atogepant and rimegepant, a Bucher comparison was performed utilizing the aggregated data for the reweighted atogepant trial sample compared with the observed effects in BHV3000-305 using placebo as an anchor between studies. Confidence intervals (CIs) for the indirect estimate were derived. To account for uncertainty in the weight estimations, the bootstrapping approach was chosen over another commonly used approach, robust sandwich estimators, since it allows estimated weights to be subject to sampling uncertainty.
Endpoints
The efficacy assessments of interest were change in MMDs and differences in acute medication use days (MUDs), which were compared between atogepant and rimegepant across weeks 1–12, a frequently used timepoint to assess the efficacy of preventive migraine treatments. To match the assessment time for the rimegepant trial, efficacy endpoints for atogepant were also compared relative to rimegepant across weeks 9–12. Although treatment response rates (e.g., ≥50% reduction in MMDs) are also recommended as clinical trial outcomes for preventive migraine treatments (20), the definition used in the rimegepant trial (≥50% reduction in moderate or severe MMDs during weeks 9–12) (13) differed from the definition used in the atogepant trials (≥50% reduction in the 12-week average of MMDs) (10,11), so the decision was made not to include this outcome in this MAIC. Change from baseline in Migraine-Specific Quality of Life questionnaire version 2.1 (MSQ v2.1) Role Function−Restrictive (RFR) domain score was assessed at week 12. Safety and tolerability outcomes, such as treatment-emergent adverse events (TEAEs) and all-cause discontinuation, were evaluated across the 12-week treatment period.
Scenario analyses
Due to residual uncertainty, five additional scenario analyses (Online Supplemental Table 3) were performed to assess their potential impact on the results. Unless otherwise stated, all scenario analyses included variables that matched the base case (age, sex, race, primary migraine type, and baseline MMDs) with the exception of the unadjusted scenario analysis, which used a population without any adjustment for participant characteristics. The methodological scenario analysis was conducted using a simulated treatment comparison (STC) approach instead of an MAIC. The MAIC study scenario analysis included week 1–12 efficacy data from the phase 2/3 atogepant trial (CGP-MD-01), with the objective of examining a range of atogepant doses for safety, tolerability, and efficacy in the preventive treatment of migraine (14), in addition to data from ADVANCE, PROGRESS, and BHV3000-305. Atogepant 60 mg once daily was the only dose consistently reported across all atogepant trials and was therefore used in this analysis. QoL data were excluded from this scenario analysis, as CGP-MD-01 did not include QoL endpoints (14). The MAIC population scenario analysis did not exclude participants based on baseline MMDs or MHDs from the ADVANCE, PROGRESS, and BHV3000-305 studies. The MAIC characteristics scenario 1 analysis included two additional participant characteristic covariates: history of CM and moderate-to-severe headache days per month (atogepant trials) and moderate-to-severe attack days per month (rimegepant trial), which were not included in the initial analysis of ADVANCE, PROGRESS, and BHV3000-305. The MAIC characteristics scenario 2 analysis included two additional participant characteristic covariates: body mass index and ethnicity (Hispanic/Latinx or not Hispanic/Latinx), which were not included in the initial analysis of ADVANCE, PROGRESS, and BHV3000-305.
Results
Participants
A total of 230–259 participants were included in the pooled atogepant 60 mg once-daily group and 269–370 participants were included in the rimegepant 75 mg once-every-other-day group. Sample sizes varied for the efficacy and tolerability outcomes; the pooled atogepant 60 mg once-daily group included 252 participants for efficacy outcomes and 259 for safety/tolerability outcomes, whereas the rimegepant 75 mg once-every-other-day group included 348 participants for efficacy outcomes and 370 for safety/tolerability outcomes. Sample size for the MSQ v2.1 RFR outcome was 230 in the pooled atogepant 60 mg once-daily group and 269 in the rimegepant 75 mg once-every-other-day group. Baseline demographics among participants across trials prior to weighting are presented in Table 1. After weighting, average baseline characteristics of atogepant-treated participants were balanced with the rimegepant-treated participants (Online Supplemental Table 4).
Information reported in the intent-to-treat population (ITT) since the exploratory analyses included safety and tolerability endpoints (i.e., ITT population of analysis). The indirect comparison analyses were performed using the modified ITT population for consistency with the population of analysis from BHV3000-305.
Study does not report primary migraine type but does report with aura, without aura, and both separately.
BMI, body mass index; IQR, interquartile range; MMD, monthly migraine day; NR, not reported; QD, once daily; QOD, once every other day; SD, standard deviation.
Efficacy endpoints
Mean change in MMDs
Atogepant 60 mg once daily demonstrated a significantly greater reduction in mean MMDs across weeks 1–12 (MD [95% CI]: −1.65 [−2.49, −0.81]; p < 0.001) and weeks 9–12 (MD [95% CI]: −1.50 [−2.55, −0.43]; p < 0.01) compared with rimegepant 75 mg once every other day (Figure 2a and b).
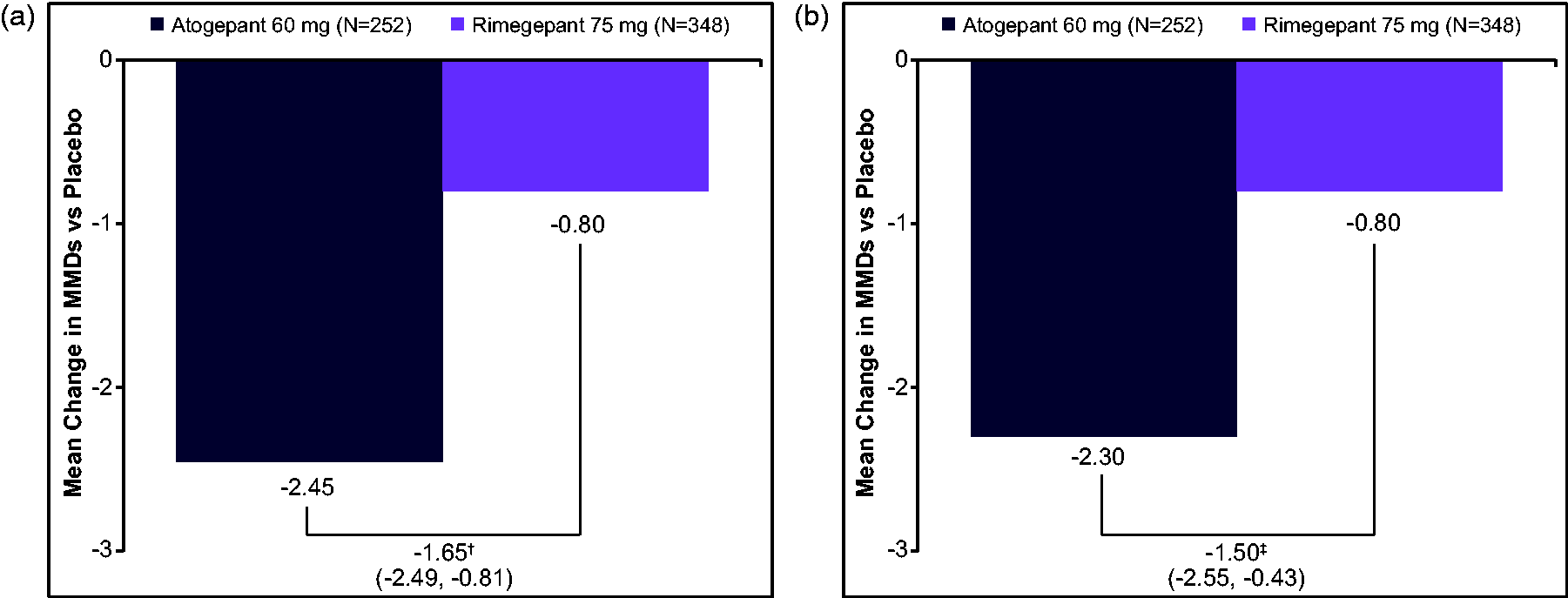
Mean differences vs placebo in MMDs: atogepant- vs rimegepant-treated participants across (a) weeks 1–12 and (b) weeks 9–12.
Reduction in acute MUDs
Atogepant 60 mg once daily also demonstrated significantly greater reductions in acute MUDs in relation to rimegepant 75 mg once every other day across weeks 1–12 (MD [95% CI]: −2.08 [−3.00, −1.16]; p < 0.0001) and 9–12 (MD [95% CI]: −1.79 [−2.78, −0.78]; p = 0.0005) (Figure 3a and b).

Mean differences vs placebo in acute medication use days: atogepant- vs rimegepant-treated participants across (a) weeks 1–12* and (b) weeks 9–12.
QoL endpoint
At week 12, atogepant 60 mg once daily demonstrated a significantly higher MSQ v2.1 RFR score compared with rimegepant 75 mg once every other day (MD [95% CI]: 7.36 [1.88, 12.82]; p < 0.01) (Figure 4).

Mean difference vs placebo in MSQ v2.1 RFR domain score: atogepant- vs rimegepant-treated participants at week 12.
Tolerability/Safety
Participants treated with atogepant 60 mg once daily had similar odds of experiencing a TEAE (OR [95% CI]: 0.91 [0.56, 1.45]; p = 0.6773) and numerically higher odds of discontinuing treatment for any reason (OR [95% CI]: 1.43 [0.69, 3.06]; p = 0.3284) compared with rimegepant 75 mg once every other day (Figure 5). However, neither difference was statistically significant.

Forest plots of odds ratio vs placebo for treatment-emergent adverse events and all-cause discontinuation: atogepant- vs rimegepant-treated participants.
Scenario analyses
All scenario analyses conducted for each endpoint produced results that were consistent with the MAIC analysis (Online Supplemental Tables 5–9). For all scenario analyses, the ranges of MD change from baseline in MMDs were −1.30 to −1.86 (p < 0.01 for all analyses), acute MUDs were −1.79 to −2.36 (p < 0.01 for all analyses), and MSQ v2.1 RFR scores were 5.36 to 9.20 (p < 0.05 for all analyses except MAIC characteristics scenario 1). The ORs of experiencing a TEAE ranged from 0.88 to 1.06, and the ORs for discontinuing treatment for any reason ranged from 0.91 to 1.73 (differences were not statistically significant for all scenario analyses).
Discussion
In this MAIC analysis of phase 3 (atogepant) and phase 2/3 (rimegepant) clinical trial data, orally administered atogepant 60 mg once daily demonstrated significantly greater improvements in both efficacy and QoL compared with rimegepant 75 mg ODT once every other day. A previous analysis observed a decreased likelihood that individuals with migraine would try a second treatment option after initial treatment failure, highlighting the need to ensure the best possible outcome from initial treatment (22). Atogepant and rimegepant were associated with comparable levels of risk for experiencing TEAEs and treatment discontinuation for any reason. All scenario analyses produced results that were consistent with the base case analysis, thereby confirming the reliability of the MAIC approach that was utilized.
At the time these analyses were conducted, direct comparative data of newer preventive migraine treatments targeting CGRP receptors were not available. A phase 4 controlled trial evaluating galcanezumab and rimegepant as preventive treatments for migraine (CHALLENGE-MIG) was recently completed, and inclusion of data from this trial may be explored in any future analyses (23). As in other areas where head-to-head trial data are lacking, indirect comparisons of published preventive migraine treatment research, including MAICs and network meta-analyses (NMAs), have been conducted. An unanchored MAIC analysis was conducted with data from a rimegepant phase 2/3 open-label safety study, an erenumab phase 3 clinical trial, and two galcanezumab phase 3 clinical trials that assessed the effectiveness of these medications as preventive migraine treatments (24). To match the mAb studies, the rimegepant dataset included only the subset of participants experiencing 4–14 MMDs, and dosing schedules consistent with preventive use were analyzed (i.e., every other day or as needed). These analyses demonstrated that all active treatments significantly reduced MMDs and improved QoL, as measured by MSQ v2.1, compared with placebo. In general, no differences were observed among rimegepant, erenumab, and galcanezumab. However, rimegepant was shown to perform significantly better than erenumab on each MSQ v2.1 domain.
An NMA of 19 phase 3 clinical trials evaluated change from baseline in MMDs and the number of participants achieving a ≥50% reduction in MMDs for atogepant, rimegepant, and CGRP mAbs as preventive treatments for EM and CM (25). This NMA demonstrated that these therapies were efficacious as preventive migraine treatments compared with placebo and generally showed similar effects when compared with one another. Additionally, another NMA using the same group of 19 phase 3 studies demonstrated that the CGRP mAbs, atogepant, and rimegepant were generally safe and well tolerated for the preventive treatment of EM, despite some slight differences in tolerability that were observed (e.g., a higher risk of discontinuation due to adverse events with eptinezumab) (26).
The clinical significance of these results is difficult to determine due to the challenge of comparing indirect analyses. In the rimegepant MAIC, the authors reported MDs in MMDs for rimegepant of 0.59 (95% CI: −0.13, 1.32) compared with galcanezumab and −0.06 (95% CI: −0.61, 0.50) compared with erenumab (24). The NMA also reported a trend toward higher MDs in MMDs for atogepant 60 mg once daily (−1.35 [95% CI: −1.85, −0.85]) compared with rimegepant 75 mg once every other day (−0.80 [95% CI: −1.56, −0.04]) (25). In contrast, both base case analyses of the current MAIC reported differences in MMDs of −1.65 (95% CI: −2.49, −0.81) and −1.50 (95% CI: −2.55, −0.43), favoring atogepant 60 mg once daily over rimegepant 75 mg once every other day. The differences in treatment response between rimegepant and atogepant in our MAIC and the previous NMA could be explained by the different methodologies of these indirect comparisons. For example, only two atogepant studies (i.e., MD-01, ADVANCE) were used in the NMA. MAIC analyses have grown in popularity recently and are now accepted by the National Institute for Health and Care Excellence (27). They have the potential to compare treatments using different populations, but the details of the methodology should be considered when interpreting their results. The potential of inadvertently introducing bias into MAIC analyses is their major limitation, so the steps taken by the researchers to avoid this bias are important to know. A critical review of nine MAIC analyses in spinal muscular atrophy identified characteristics to consider when assessing the quality of MAICs (28), including providing justification for using MAIC; fully reporting key details; using studies with comparable populations, designs, and outcomes; identifying and accounting for effect modifiers; and reporting baseline characteristics pre- and post-adjustment.
The authors of the unanchored rimegepant MAIC noted several limitations, including steps taken to account for differences in study design with the phase 3 mAb trials (24). The rimegepant data were collected from an open-label, single-arm safety study, which may have biased the results due to the lack of a comparator. Only a select population was used from the rimegepant study, whose dosing schedule and migraine frequency most closely reflected those in the mAb trials, which also could have affected the results. For the erenumab trial, the authors were unable to match populations for disease duration and the presence of aura because these data were not reported (29). To manage scenarios where matching was not possible, separate MAIC analyses were conducted for these variables.
Results from our MAIC analysis differ from previously conducted analyses by demonstrating that atogepant was associated with significantly better efficacy compared with rimegepant. A strength of our study was the use of an anchored MAIC analysis to generate comparative treatment-effect estimates that overcame between-study heterogeneity for key treatment-effect modifiers. In networks consisting of only one or two trials per treatment, indirect comparisons are highly vulnerable to systematic variation (bias) resulting from imbalances in effect modifier distributions. Under these circumstances, population-adjusted methods have a distinct advantage. Thus, by using an MAIC, we were able to reduce the bias that may have resulted from a standard indirect treatment comparison. Additionally, the scenario analyses supported the base case analyses, suggesting validity for the methodology used. However, as is inherent in indirect comparisons, there are several limitations to our MAIC analysis that are worth noting. Given the differences in MMD and MHD inclusion and exclusion criteria across trials, this analysis excluded participants who did not meet BHV3000-305 criteria (≥6 migraine days and ≤18 migraine/non-migraine headache days during the four-week observation period). By excluding some participants (e.g., those with four to five MMDs in ADVANCE and ≥18 MHDs in PROGRESS), randomization was broken. However, applying population-adjusted methods of MAIC/STC to both treatment arms is expected to adjust for imbalances in these effect modifiers. This is reflected within the population scenario analysis, which produced results consistent with the base case analyses for all endpoints. In the rimegepant trial, efficacy by prior preventive treatment failure was not reported and could not be matched. Therefore, the MAIC was conducted only among the overall rimegepant trial population; thus, appropriate conclusions can be drawn for only this population. Notably, the difference in MMDs observed in the original rimegepant publication was consistent with the difference observed in our MAIC (−0.8 vs placebo), while the difference for atogepant increased by only <1 MMD, which may be a result of the nature of the MAIC analysis and other factors such as combining the CM and EM populations for atogepant (10,11,13).
Although multiple atogepant doses were used in the atogepant trials, atogepant 60 mg once daily was the only dose analyzed and reported within the MAIC, since it was the common dose between trials. Efficacy data timepoints varied between the atogepant and rimegepant trials, in which efficacy was analyzed at 1–12 weeks for atogepant and 9–12 weeks for rimegepant. However, this variation was assessed by analyzing data during both periods, and the results were consistent with the base case for all endpoints. The lack of availability of certain efficacy data among the trials prevented matching data within the MAIC analysis. For example, baseline MHDs, acute MUDs, and change from baseline in MHDs were not reported in the rimegepant trial, and some baseline migraine characteristics (onset, duration, and severity of migraine attacks per month, and primary migraine type) were not included in the atogepant trials. The current MAIC would have also benefited from including a ≥50% responder analysis, but the definition in the rimegepant trial differed from the definition used in the atogepant trials to a degree that precluded conducting these comparisons. Despite these limitations, the results from our MAIC analysis add to the current literature by demonstrating greater efficacy with atogepant compared with rimegepant, which may help to guide clinical decision making. In addition to conducting head-to-head trials, future research comparing preventive migraine treatments would benefit from including analyses that identify participant characteristics that are associated with symptomatic improvement to one treatment option over another.
Conclusion
Overall, atogepant 60 mg once daily resulted in greater improvements in efficacy and QoL in comparison with rimegepant 75 mg ODT once every other day with comparable safety profiles.
Clinical implications
Atogepant 60 mg once daily resulted in greater improvements in efficacy and quality of life in comparison with rimegepant 75 mg once every other day, with comparable safety profiles. Our MAIC analysis adds to the current literature by demonstrating improved efficacy with atogepant compared with rimegepant, which may help to guide clinical decision making.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024241235156 - Supplemental material for Comparative efficacy, quality of life, safety, and tolerability of atogepant and rimegepant in migraine prevention: A matching-adjusted indirect comparison analysis
Supplemental material, sj-pdf-1-cep-10.1177_03331024241235156 for Comparative efficacy, quality of life, safety, and tolerability of atogepant and rimegepant in migraine prevention: A matching-adjusted indirect comparison analysis by Cristina Tassorelli, Kateryna Onishchenko, Rashmi B. Halker Singh, Molly Duan, Laure Dupont-Benjamin, Matthew Hemstock, Corey Voller, Peter McAllister, Stephanie J. Nahas, Pranav Gandhi and Jessica Ailani in Cephalalgia
Footnotes
Acknowledgments
The authors would like to thank Karen Carr for her contributions.
Author contributions
Study concept and design K.O., M.H., L.D.B., P.G., C.V., M.D.
Acquisition of data C.T., L.D.B.
Analysis and interpretation of data K.O., C.T., M.H., L.D.B., P.G., C.V., P.M., M.D., J.A., S.J.N., R.B.H.S.
Drafting the manuscript K.O., L.D.B.
Revising the manuscript for intellectual content K.O., S.J.N., C.T., M.H., L.D.B., P.G., C.V., P.M., M.D., J.A., R.B.H.S.
Final approval of the completed manuscript K.O., S.J.N., C.T., M.H., L.D.B., P.G., C.V., P.M., M.D., J.A., R.B.H.S.
Data sharing statement
AbbVie is committed to responsible data sharing regarding the clinical trials we sponsor. This includes access to anonymized, individual, and trial-level data (analysis data sets), as well as other information (e.g., protocols, clinical study reports, or analysis plans), as long as the trials are not part of an ongoing or planned regulatory submission. This includes requests for clinical trial data for unlicensed products and indications.
These clinical trial data can be requested by any qualified researchers who engage in rigorous, independent, scientific research, and will be provided following review and approval of a research proposal, Statistical Analysis Plan (SAP), and execution of a Data Sharing Agreement (DSA). Data requests can be submitted at any time after approval in the US and Europe and after acceptance of this manuscript for publication. The data will be accessible for 12 months, with possible extensions considered. For more information on the process or to submit a request, visit the following link: ![]() then select “Home.”
then select “Home.”
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.T. has participated in advisory boards for AbbVie, Dompé, Eli Lilly, Lundbeck, Pfizer, and Teva. She has lectured at symposia sponsored by AbbVie, Eli Lilly, Lundbeck, Novartis, and Teva. She is principal investigator or collaborator in clinical trials sponsored by AbbVie, Biohaven, Eli Lilly, Lundbeck, and Teva. She has received research grants from the European Commission, the Italian Ministry of Health, the Migraine Research Foundation, and the Italian Multiple Sclerosis Foundation. She serves as an associate editor for Cephalalgia and The Journal of Headache and Pain. K.O., M.D., and P.G. are employees of AbbVie and may hold AbbVie stock. R.B.H.S. serves on the editorial board of Current Neurology and Neuroscience Reports, serves as deputy editor for Headache, and previously received grants for research support from Amgen and Eli Lilly. L.D.B. was an employee of AbbVie at the time of study conduct and may hold AbbVie stock. M.H. and C.V. were employees of Lumanity at the time of study conduct. Lumanity was reimbursed by AbbVie as a consultancy for time spent planning, reviewing, and reporting the statistical analyses. P.M. has received research funding from AbbVie, Amgen, Biogen, Biohaven, EMD Serono, Lundbeck, Novartis, and Teva, and has received personal compensation for advisory boards and/or speakers bureaus for AbbVie, Aeon, Amgen, Biohaven, Lilly, Lundbeck, Revance, and Teva. S.J.N. has received honoraria for consulting from Allergan/AbbVie, Axsome (ended January 2022), BioDelivery Sciences (ended February 2022), Eli Lilly, Lundbeck, Pfizer, Teva, Theranica, and Tonix; and has received honoraria for work in education or publishing from American Academy of Neurology, American Headache Society, Diamond Headache Clinic Research and Educational Foundation, MedLink Neurology, MJH Life Sciences, NACCME, Neurology Learning Network, Springer, WebMD/Medscape, and Wolters-Kluwer. J.A. has served as a consultant for AbbVie, Aeon, Amgen, Biohaven, Eli Lilly, GlaxoSmithKline, Gore, Impel, Linpharma, Lundbeck, Miravo, Nesos, Neurolief, Pfizer, Satsuma, Teva, and Theranica; received stock options from Ctrl M; provided editorial services to Current Pain and Headache Reports, SELF, and Medscape; and received clinical trial support from AbbVie, Biohaven, Eli Lilly, Rehaler, Satsuma, and Ipsen.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: AbbVie funded this study and contributed to the study design, the collection, analysis, and interpretation of data, and the review and approval of the final version for publication. No honoraria or payments were made for authorship. Medical writing support was provided to the authors by Peloton Advantage, LLC, an OPEN Health company, and was funded by AbbVie.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

