Abstract
The migraine treatment landscape has seen significant advancements in recent years, including the introduction of novel preventive agents specifically targeting the disease. These new treatments offer improved efficacy and tolerability, potentially addressing the issue of poor treatment adherence commonly observed with conventional preventatives. In this context, pragmatic trials emerge as a critical tool for advancing migraine care, offering a real-world approach to evaluating open clinical questions at the same time as avoiding the biases of real-world observational evidence. By prioritizing external validity and patient-centered outcomes, pragmatic trials provide valuable insights into the advantages of new treatments in improving migraine care. Possible applications of pragmatic trials in migraine research include head-to-head comparisons, evaluation of combination therapies, assessment of treatment sequences and switch, testing the added value of patient-reported outcomes, investigation of long-term effectiveness and on optimal treatment duration, understanding the role of preventive treatments in altering the course of migraine and preventing progression, and cost-effectiveness analyses. Pragmatic trials allow for the assessment of interventions in diverse patient populations and healthcare settings, enhancing the generalizability of findings and informing evidence-based clinical practice. As such, pragmatic trials represent an excellent tool to bridge the gap between placebo-controlled trials and real-world practice and should receive consideration for funding, especially by public institutions such as universities, national health services, and charities.
This is a visual representation of the abstract.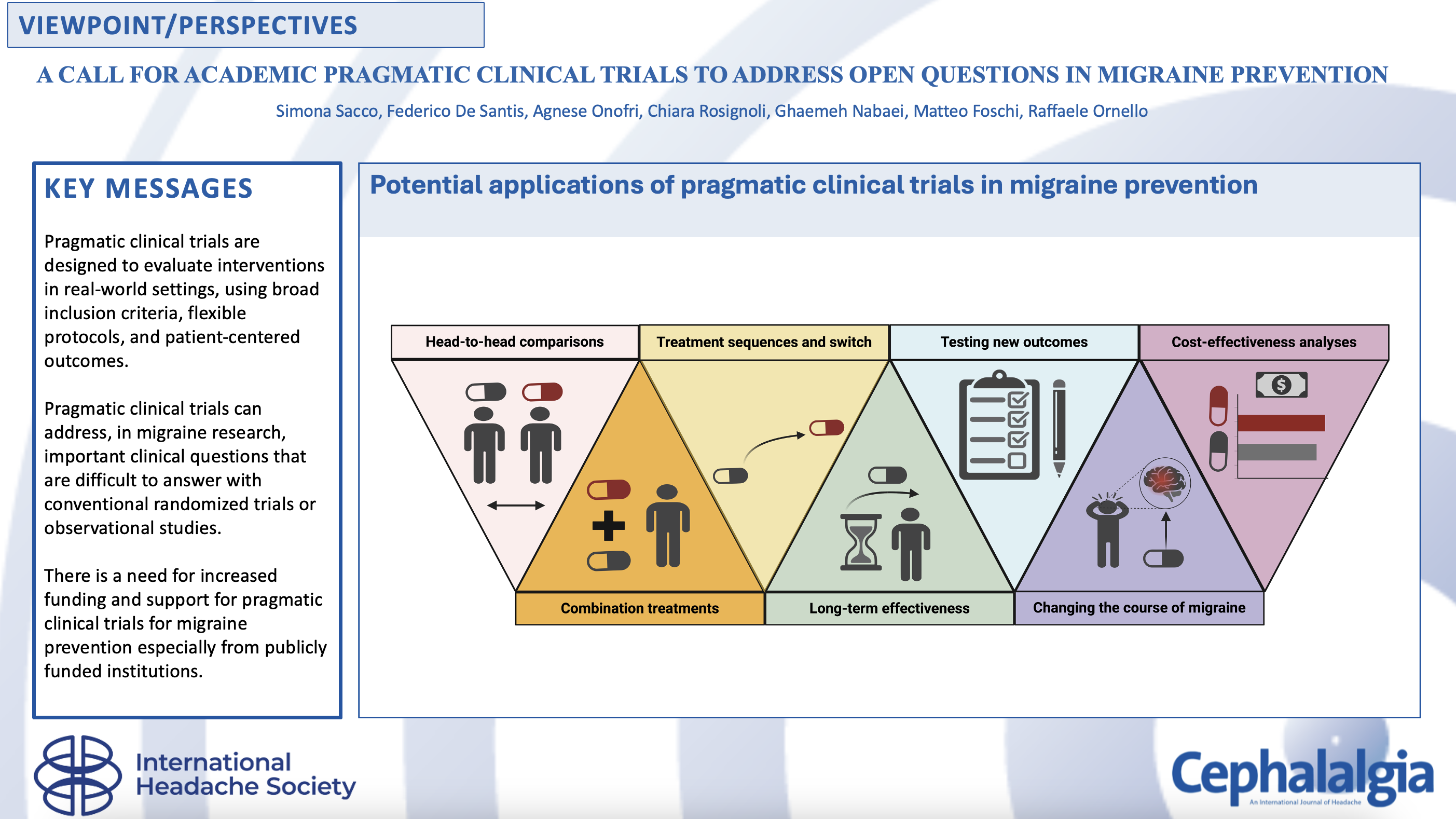
Keywords
Introduction
Recent years have witnessed remarkable advancements in migraine treatment (1). Treatments targeting the calcitonin gene-related peptide (CGRP) pathway, including monoclonal antibodies directed toward CGRP or its receptor and oral agents binding the CGRP receptor (gepants), have demonstrated efficacy and excellent tolerability (2). These novel CGRP-targeting drugs are revolutionizing migraine treatment, opening new therapeutic avenues together with new clinical questions in daily practice. Some of these questions have been explored in real-world studies that are highly relevant to clinical practice and may offer valuable information, but they may lack the methodological rigor needed to answer specific research questions.
Here, we discuss how pragmatic clinical trials may offer a valuable avenue to address several open issues within the migraine preventive treatment landscape.
What is a pragmatic trial?
A pragmatic trial is a type of clinical trial designed to inform decision makers, including participants, clinicians and policymakers, and to enroll a population representative of the individuals and clinical settings for whom the decision is relevant (3). In pragmatic trials, participants are randomized to different interventions or treatment strategies that are all suitable for their condition, where the comparative advantages and disadvantages are still unclear. Unlike randomized controlled trials (RCTs) with an explanatory design, which focus on establishing efficacy under ideal conditions with strict protocol adherence, pragmatic trials prioritize external validity and aim to evaluate interventions as they would be used in routine practice (3,4). They can inform clinicians and policymakers about the most beneficial and cost-effective interventions for diverse populations. Tools such as the PRECIS-2 (5) provide a framework to assess the characteristics of pragmatic trials, which include the following:
Broad inclusion criteria: pragmatic trials include a broad and diverse population, reflecting the demographics and clinical characteristics of individuals encountered in everyday clinical practice (6,7). This approach enhances the generalizability of trial results to real-world settings. Recruitment via usual clinical appointments at a diverse range of clinical centers. This approach ensures that individuals are recruited from a setting as close as possible to everyday clinical practice. Flexible setting, representing the same geography, healthcare system, country, and the socioeconomic and ethnic mix of the population in which the interventions should be applied. Incorporation into the usual clinical organization of care and workflow to allow the use of the existing staffs for the intervention. Flexible protocols: although pragmatic trials maintain standards of rigor and safety, they allow for flexibility in treatment delivery and management (6). This flexibility mimics the variability seen in routine clinical practice and enables investigators to assess the intervention's effectiveness in different contexts and populations. The possibility of keeping into the trial those individuals who have scarce adherence to the intervention protocol, in order to assess how participants will engage with the intervention. Patient-centered outcomes: pragmatic trials typically focus on patient-centered outcomes that are relevant to stakeholders, such as improvements in quality of life, symptom relief, functional status and long-term health outcomes (3). By prioritizing outcomes that matter most to patients, pragmatic trials provide valuable insights into the intervention's real-world impact (3,6). Primary analysis including all the available data according to an intention-to-treat analysis, without flexibility in protocol adherence or confrontation with the variability of clinical practice. This would include a more thorough assessment of outcomes, thus enhancing the applicability of trial outcomes to clinical practice.
Throughout the trial process, pragmatic trials emphasize considerations related to intervention implementation, scalability and sustainability in real-world settings (8). This focus helps bridge the gap between research evidence and clinical practice, facilitating the adoption of effective interventions into routine care.
The role of pragmatic trials in the hierarchy of evidence for migraine prevention
In line with the principles of evidence-based medicine, exploratory RCTs are designed to establish the efficacy and safety of migraine preventive treatments (9,10). These studies prioritize demonstrating the efficacy of interventions, by including highly selected populations adhering to rigid protocols with close outcome monitoring.
Conversely, real-world observational studies align closely with clinical practice, addressing questions related to the effects of interventions in broader populations with different treatment durations. However, they often lack methodological rigor and carry a high risk of bias (11), especially when they are trying to address questions that would need a comparator arm to exclude confounders. The advent of injectable treatments for migraine treatments has led to a proliferation of real-world observational studies in recent years (12,13). Although RCTs and real-world observational studies have significantly advanced our understanding of migraine prevention, they cannot adequately address certain critical questions related to optimizing preventive care for individuals with migraine.
Pragmatic clinical trials can bridge the gap between RCTs and real-world observational studies by providing high-quality evidence to inform clinical decisions. These trials maintain the randomization procedure and rigorous design of RCTs at the same time as incorporating real-world study elements in terms of participant characteristics and procedures. They are particularly suited for testing alternate interventions that are plausible for a given population (e.g. combination treatment versus single treatment) or exploring new hypotheses (e.g. preventing migraine complications by comparing different treatment options). It is important to note that the International Recommendations for conducting trials in the migraine field (9,10) apply to pragmatic trials as well as to RCTs.
Figure 1 illustrates the characteristics of RCTs, pragmatic trials and real-world observational studies, emphasizing the role of pragmatic trials as a link between controlled research settings and clinical practice.
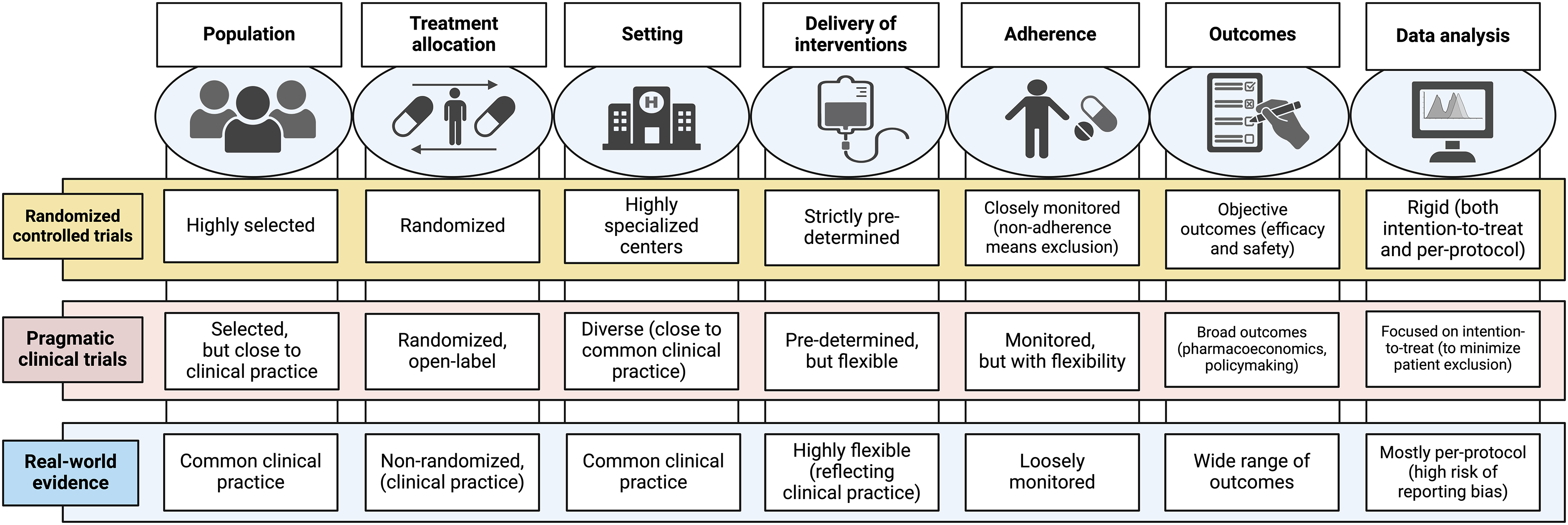
The main features of pragmatic trials compared with randomized controlled trials and real-world studies. Created with BioRender.com
Potential applications of pragmatic trials to migraine prevention
Below, we deal with some of the unresolved questions in the realm of migraine prevention that in our opinion can be explored through a pragmatic trial approach. Those questions are summarized in Figure 2.
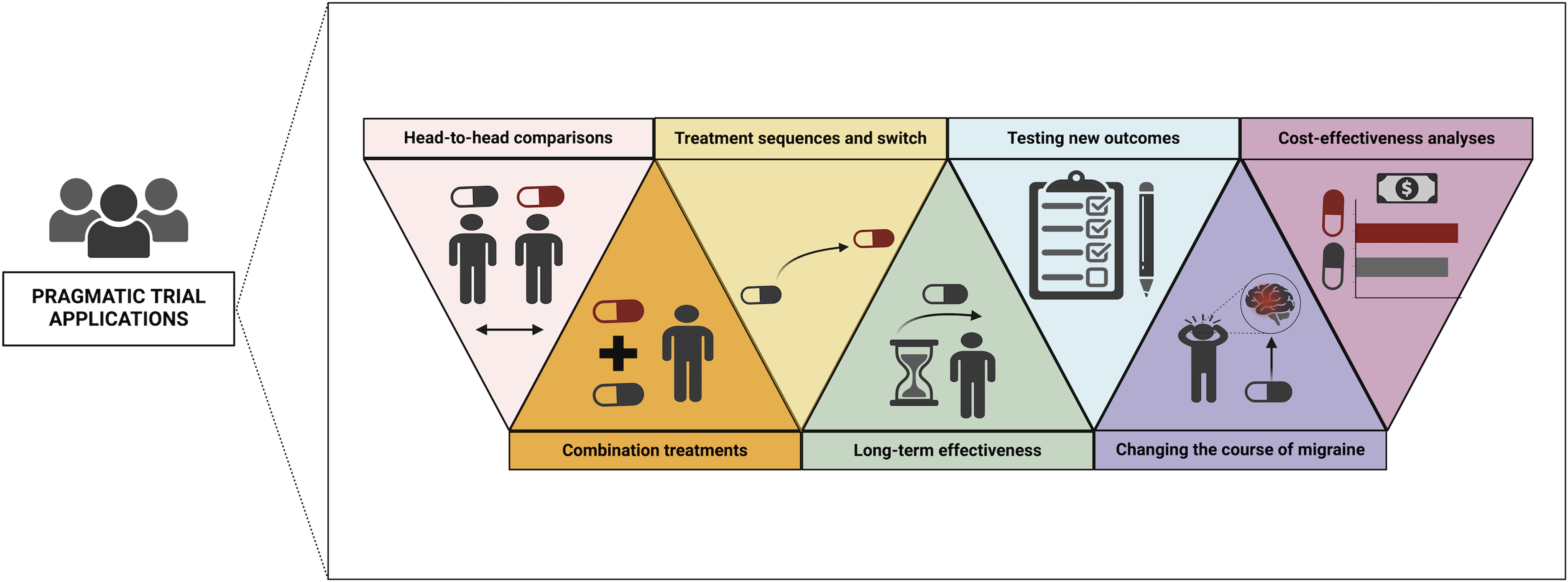
Potential areas of application of pragmatic clinical trials for preventive migraine treatment. Created with BioRender.com
Head-to-head comparisons
Although new CGRP-targeting medications have expanded migraine prevention options, the evidence base from RCTs comparing their benefits to conventional preventatives is limited. The HER-MES study stands out as it provided a high-quality direct comparison between erenumab and topiramate, demonstrating that erenumab has a higher rate of ≥50% reduction in monthly migraine days and a lower discontinuation rate compared to topiramate (14). Apart from the HER-MES trial, most of the available investigations in this area are network meta-analyses reporting indirect comparisons (15,16).
Pragmatic trials not only enable a head-to-head comparison between novel and non-specific migraine treatments in everyday practice, but also offer a cost-effective way to update the evidence base for even the oldest and cheapest conventional drugs, some of which may have outdated data due to lack of investment. Additionally, trials of oral preventive medication for migraine are mostly old and were performed with outdated methods; therefore, it might be valuable to provide high-quality data with up-to-date standards of research for those drugs.
In this approach, participants from real-world settings would be randomly assigned to receive either medications targeting the CGRP pathway or conventional migraine treatments. To prevent detection bias (17), blind endpoint assessment, in which the endpoints are adjudicated by personnel that is unaware of participants’ clinical data, would be preferable over an endpoint assessment performed by the study investigators. Effectiveness and adherence would serve as primary outcomes with the treatment duration typically aligning with pivotal RCTs typically lasting 12–24 weeks (18–20). Studies should clearly define their primary endpoint and whether they aim to assess superiority, non-inferiority, or equivalence. They should also ensure that the sample size is sufficiently large to detect differences between groups. In pragmatic trials, broader inclusion criteria reduce selection bias from restricted randomization and decrease participants’ allocation to more likely treatments, which often affects observational studies (21). Being open-label, head-to-head pragmatic trials imply that double-blind or double-dummy methodologies cannot be used.
Combination treatments
Combination therapies are commonly used in headache clinics as an additive effect on migraine prevention is expected. This approach might be required even for medications targeting the CGRP pathway because some individuals treated with those drugs has a high residual burden of migraine (22). However, there are not many studies available on the combination between different preventive treatments for migraine. Although existing research has primarily focused on the combination of oral medications (23–26), there is a lack of robust studies investigating the concurrent use of oral medications along with injectable therapies such as onabotulinumtoxinA and medications targeting the CGRP pathway. Combination treatments might expand in the near future given the advent of migraine-specific preventive medications that do not interact with other treatments and have minimal tolerability issues (27). Additionally, oral treatments can be integrated with non-pharmacological interventions such as cognitive–behavioral therapies or neuromodulation (28). RCTs are traditionally structured to evaluate the efficacy and safety of individual treatments, making pragmatic trials essential to assess the effectiveness of these combined therapeutic interventions. Treatment doses and dosing schedules should be standardized and comparable between the treatment arms. Additionally, when assessing drug combinations, a study should be able to show the effect of each single drug.
Treatment sequences and switch
Current migraine preventive treatment practice typically employs oral preventives as first-line therapy, reserving migraine-specific medications for selected patients. However, this approach is largely based on reimbursement criteria rather than clinical evidence or pharmacological considerations. The European Headache Federation recommends monoclonal antibodies targeting the CGRP pathway as first-line treatment options for migraine, citing their high efficacy and excellent tolerability (29). The American Headache Society has subsequently endorsed this approach (30). Pragmatic clinical trials can provide robust evidence on the efficacy, tolerability, adherence, and costs of earlier implementation of migraine-specific preventatives, which can influence the decisions of regulatory agencies. More generally, pragmatic trials would be helpful in determining the best timing to switch from a preventive treatment to another. Additionally, they would help solving clinical problems such as the opportunity of switching between two treatments with similar mechanisms of action. As an example, some small case series have suggested potential benefits from switching between two CGRP monoclonal antibodies (31–33); however, this suggestion needs to be confirmed with stronger studies controlling for non-pharmacological effects and spontaneous migraine fluctuations.
Testing new outcomes
Patient-reported outcomes (PROs) are important to tackle the individual migraine burden and are extensively utilized in migraine clinical trials. Although the reduction in monthly migraine or headache days is recommended as the primary efficacy endpoint for RCTs of preventive treatments (9,10), it might not fully capture the impact of migraine on individuals’ lives. The available RCTs presented a vast repertoire of PROs in the absence of a thorough assessment of their value for participants’ lives. On the other hand, PROs assessed by real-world studies are prone to the biases that are typical of those studies. Pragmatic trials can focus on assessing headache-related physical and psychological disability as outcomes at the same time as maintaining a robust design. This approach can assist clinicians in selecting the most appropriate treatment based on individual variations in the impact of the disease. Ideally, PROs should be validated and standardized to ensure comparability among studies.
Long-term effectiveness
Migraine is a life-long condition that may require long-term management to mitigate its debilitating effects. However, determining the ideal duration of preventive treatment remains a critical challenge in clinical practice (34–36). Because of restrictions in funding and to the necessity of careful monitoring, placebo-controlled trials are generally limited to 12–24 weeks. Open-label extensions of those trials provide data on efficacy and safety over longer periods; however, they maintain a controlled setting different from clinical practice. Pragmatic trials allow the assessment of long-term treatment effectiveness and tolerability in the diversity of real-world scenarios. Additionally, pragmatic trials might give unbiased insights on the ideal treatment duration and also explore factors influencing treatment duration, including the migraine burden, comorbidities, and treatment adherence patterns, thus contributing to treatment personalization. Finally, pragmatic trials could evaluate the persistence of benefit after treatment discontinuation. Notably, survivor bias should be avoided by including in the analyses all participants included at the beginning of the study, and not only those reporting a benefit from the drug.
Understanding the role of preventive treatments in preventing migraine complications
It is well known that migraine may progress from a low-burden and easy-to-manage condition into a high-frequency form or into chronic migraine; subjects can also develop medication overuse and resistance to treatments (37). Poorly effective preventive medication might have a role in determining migraine progression and complications; however, this role had not been confirmed yet. Additionally, when migraine has progressed to difficult-to-treat forms, long preventive treatments might be required to achieve migraine control, with potential negative impact on tolerability. Through the evaluation of various treatment approaches over an extended duration, pragmatic trials might reveal the potential of both new and conventional drugs to alter the course of migraine and to prevent the progression and emergence of resistance to treatments.
Cost-effectiveness analyses of migraine preventive treatments
The cost-effectiveness of migraine treatments is crucial in evaluating their overall value in healthcare (38). Pragmatic trials offer a unique opportunity to comprehensively evaluate the cost-effectiveness of these treatments in real-world clinical settings (39). Despite some analyses (40–44), it remains mostly unclear whether new migraine preventive treatments, despite their higher direct costs compared to older treatments, offer benefits from a pharmacoeconomic perspective by reducing healthcare resource utilization and indirect costs of the disease, such as absenteeism and loss of productivity. Pragmatic trials can comprehensively evaluate the cost-effectiveness of these treatments in real-world scenarios by collecting outcomes related to cost in individuals representing the real-world population. By randomly assigning participants to the treatments of interest and analyzing the ensuing costs, these trials can provide unbiased information for clinicians and policymakers to make informed decisions regarding treatment selection and resource allocation.
Conclusion: a call for funding pragmatic trials
RCTs are essential for drug development, providing large-scale evidence of efficacy and safety for migraine preventive drugs. These trials, primarily funded by pharmaceutical companies, have complex methodologies to meet quality criteria that translate into high costs. Conversely, the guidance for clinical applicability of migraine preventive treatments largely relies on real-world studies, which are typically conducted with limited resources and are susceptible to various biases mostly as a result of their observational design. Although many of these studies are performed in academic settings, their design often fails to adequately address clinically relevant questions in migraine management.
To raise the standards of clinical research in migraine, pragmatic trials are essential because they can provide unbiased, clinically relevant information at the same time as avoiding both the rigidity of industry-funded trials and the methodological limitations of real-world observational studies. By bridging this gap, pragmatic trials offer a balanced approach to generating evidence that is both scientifically robust and clinically applicable. Such trials should be informed by the highest possible methodological standards, including the definitions, outcomes and time points of evaluations suggested by International guidelines (9,10).
As is the case for RCTs, running a pragmatic trial involves costs that can vary widely depending on the trial size, duration, complexity and geographic location. Pharmaceutical companies are unlikely to fund pragmatic trials because the clinical questions referring to implementation of migraine preventive treatments in everyday practice often do not match commercial interests. Publicly funded institutions with access to independent research grants, such as universities, national health services, the National Institutes of Health in the USA, the European Union or other charities, have the expertise and resources to lead pragmatic trials, for which feasibility has been demonstrated in other areas of medicine and neuroscience (45,46). The importance of academic-led, publicly funded pragmatic trials cannot be overstated because they contribute to the integrity, transparency and independence of, thus fostering a constructive collaboration among multidisciplinary research teams, healthcare providers, policymakers and patient advocacy groups. In our opinion, there is a strong need to support and fund pragmatic trials of migraine prevention to inform high-standard, evidence-based clinical practice and healthcare policy.
Clinical implications
In migraine prevention research, pragmatic clinical trials have the potential to bridge the gap between traditional randomized controlled trials and real-world observational studies. Pragmatic trials are designed to evaluate interventions in real-world settings, using broad inclusion criteria, flexible protocols, and patient-centered outcomes. They can address important clinical questions that are difficult to answer with conventional randomized trials or observational studies. The potential applications for pragmatic trials in migraine prevention include head-to-head comparisons of treatments, evaluation of combination therapies, assessment of treatment sequences and switching, testing new patient-reported outcomes, studying long-term effectiveness, understanding the role of preventive treatments in avoiding migraine complications, and conducting cost-effectiveness analyses. There is a need for increased funding and support for pragmatic trials in migraine research to inform treatment decisions and healthcare policy.
Footnotes
Acknowledgments
Part of the text was edited with ChatGPT, version 3.5, to improve clarity.
Declaration of conflicting interests
SS declares personal fees as speaker or advisor from Abbott, Allergan-Abbvie, AstraZeneca, Boheringer, Eli Lilly, Lundbeck, Novartis, NovoNordisk, Pfizer and Teva; she reports having received research grants from Novartis and Uriach; she is president of the European Stroke Organisation, Editor-in-chief of Cephalalgia and Cephalalgia Reports and Assistant Editor for Stroke. RO reports personal fees as speaker or advisor from AbbVie, Eli Lilly, Novartis, Organon, Pfizer, and Teva, as well as non-financial support from AbbVie, Eli Lilly, Novartis and Teva; he is Associate Editor of the Headache and Neurogenic Pain section for Frontiers in Neurology and Junior Editorial Board Member for The Journal of Headache and Pain. All of the other authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The publication of this review was funded by intramural DISCAB GRANT 2024 (project code 07_DG_2024_09) awarded by the Department of Biotechnological and Applied Clinical Sciences, University of L'Aquila.
