Abstract
Background
Some studies have suggested an association between migraine and inflammatory bowel disease. We determined migraine prevalence in a cohort of patients with inflammatory bowel disease.
Methods
Patients with inflammatory bowel disease aged 18–65 years were interviewed using an ad hoc headache questionnaire. Those who admitted a history of headache in the last year answered the three questions of the ID-Migraine questionnaire. Those who answered “yes” to the three of them were classified as “definite” and those who answered “yes” to two were classified as “probable” migraine.
Results
We interviewed 283 patients with inflammatory bowel disease. Of these, 176 (62.2%) had headache. Fifty-nine (20.8%; 95% CI 16.3–26.0%) met migraine criteria either definite (n = 33; 11.7%; 95% CI 8.2–16.0%) or probable (n = 26; 9.2%; 95% CI 6.1–13.2). When divided by gender, 12 men (9.6%; 95% CI 5.1–16.2%) and 47 women (29.8%; 95% CI 22.8–37.5%) met migraine criteria. The prevalence of migraine was increased in inflammatory bowel disease patients from the current cohort (20.8%) versus that reported for our general population for the same age group (12.6%; p < 0.0001). These differences remained significant in female inflammatory bowel disease patients (29.8% versus 17.2% in our general population; p < 0.0001), but not in males (9.6% in inflammatory bowel disease vs 8.0%; p = 0.30). Seventeen patients with inflammatory bowel disease (6.0%; 95% CI 3.54–9.44%) fulfilled chronic migraine criteria. There were no differences in migraine prevalence by inflammatory bowel disease subtypes.
Conclusion
Migraine prevalence, including chronic migraine, seems to be increased in patients with inflammatory bowel disease. The fact that this association was stronger for women suggests an influence of sex-related factors.
Introduction
Migraine is often disabling by itself but also due to the co-occurrence of other chronic diseases both neuropsychiatric, including anxiety-depression, insomnia, epilepsy or stroke, and non-neuropsychiatric, such as heart disease or asthma/allergies (1). Gastrointestinal symptoms (nausea/vomiting, gastroparesis) are classical hallmarks of migraine and some gastrointestinal disorders, such as irritable bowel syndrome or celiac disease, are more frequent in migraineurs as compared to the general population (2,3).
Inflammatory bowel disease (IBD), which includes ulcerative colitis (UC), Crohn’s disease (CD) and undetermined-IBD (U-IBD) is a lifetime, chronic inflammatory disorder, with some epidemiological characteristics similar to those of migraine as it is more likely to be diagnosed in children or young adults and to co-occur with other chronic conditions including depression and anxiety, asthma and some neurological disorders (4,5). Several studies have analyzed a possible comorbidity between migraine and IBD with very heterogeneous results (Table 1). Four studies, mostly coming from big insurance data-bases with incredibly low migraine prevalence, have found no association (6–9). The remaining seven (10–16) did find a numerical difference, but these differences were only significant in three of them (12–14). These three positive studies cannot be interpreted as definitive as either just used one question to diagnose migraine (14), the number of included cases was too low (n = 80) (12) or the percentages found for prevalence of migraine in IBD patients and controls (41% and 21.3%, respectively) were too high (13). Our aim, therefore, was to assess, by means of a validated questionnaire with high sensitivity and specificity also in Spanish language (17,18), a possible coexistence between migraine and IBD in a series of unselected, consecutive patients attending our IBD clinic.
Main results of surveys analyzing migraine prevalence in IBD.

All reports appearing in Pubmed journals and studying migraine prevalence in IBD patients are included here (search terms: Crohn’s disease/inflammatory bowel disease/ulcerative colitis AND migraine). *This study included only patients with Crohn’s disease. **Patients were just asked “if they had migraine or severe headache”. ***Migraine screening questionnaire. NS: non-significant. Significant prevalence numbers appear in bold.
Patients and methods
Study population and design
We performed this cross-sectional study, between 21 November 2022 and 22 February 2023. To be included in this survey, IBD patients had to be between 18 and 65 years and have been diagnosed with IBD in our IBD Unit according to the Montreal Classification (19). After providing informed consent, consecutive patients with a confirmed diagnosis of IBD attending our Unit were interviewed using an ad hoc headache questionnaire (Table 2). Detailed clinical data were available for all the participants. The study received IRB approval by the Ethics Committee of Cantabria (2023/004).
Headache questionnaire used in this study.*

IBD patients with a history of headache in the last year unrelated to head injury, hangover or a viral illness such as flu or COVID-19 were asked to answer the three questions of the ID-Migraine validated questionnaire (17). Those who answered “yes” to the three questions were classified as “definite” migraine and those who answered “yes” to two questions were classified as “probable” migraine. In these migraine patients, we also collected the subjective average frequency of headache days in the previous three months (more or less than 15 days a month). If participants had 15 or more headache days and a definite or probable migraine according to the questionnaire, they were defined as having chronic migraine. For all patients we collected demographic data, IBD diagnostic subtype, disease duration, hospitalizations, surgeries and medical treatments due to IBD.
Endpoints
The primary endpoint was to determine the prevalence of migraine in a cohort of unselected IBD patients versus that reported in our general population (20). The secondary endpoints were to analyze the prevalence of migraine in IBD patients according to sex, IBD diagnostic subtype and severity and treatment of the IBD.
Statistical analysis
Based on a reported migraine prevalence of 12.6% for our local population at 18–65 years of age (20) and an average prevalence of migraine in patients with IBD of 19% described in a recently published meta-analysis (21) we calculated that with alpha equal to 0.05 and a power of 80%, we had to include a minimum of 235 individuals.
Data are expressed as percentages with 95% confidence intervals (95% CI) calculated using the Clopper-Pearson method unless stated differently in text. Comparisons of prevalence between our IBD patients and the general population were performed using the binomial test, inferring the hypothesized values from the reported local migraine prevalence data at 18–65 years of age (20). Comparisons of proportions found in our IBD sub-groups were carried out using Fischer’s exact test.
Results
We interviewed a total of 283 consecutive IBD patients aged 18–65 years . There were 158 (55.8%) women with a mean age of 48.3 years (median 48.5 years, range 22–65 years) and 125 men (median 49 years, range 20–65 years). A total of 140 (49.1%; mean age 49.2, median 50, range 22–65 years) patients met UC criteria, 131 (46.7%; mean age 46, median 47, range 20–65 years) had CD and the remaining 12 (4.2%; mean age 46.2, median 47, range 27–62 years) were diagnosed as U-IBD. Main characteristics of this population are described in Table 3.
Main characteristics of total IBD population, and stratified by headache/no headache.

SD: standard deviation, IBD: inflammatory bowel disease, UC: ulcerative colitis, CD: Crohn’s disease, U-IBD undefined inflammatory bowel disease.
A total of 176 (62.2%) admitted a history of headache unrelated to head injury, hangover or a viral illness. Of those, 59 (20.85%; 95% CI 16.26–26.05%) IBD patients met migraine criteria either definite (n = 33; 11.66%; 95% CI 8.16–15.99%) or probable (n = 26; 9.18%; 95% CI 6.09–13.17) (Figure 1). When divided by gender, 12 men (prevalence 9.6%; 95% CI 5.06–16.17) and 47 women (prevalence 29.75%; 95% CI 22.75–37.53%) met migraine criteria. There were 17 IBD patients (6.0%; 95% CI 3.54–9.44%) who met chronic migraine criteria (11 women, eight definite and three probable migraine; prevalence 7% and six men, three probable and three definite; prevalence 4.7%) (Table 4).

Flow chart of IBD patient selection and distribution.
Main characteristics of IBD population, stratified by headache without migraine, total migraine, probable migraine and definite migraine.
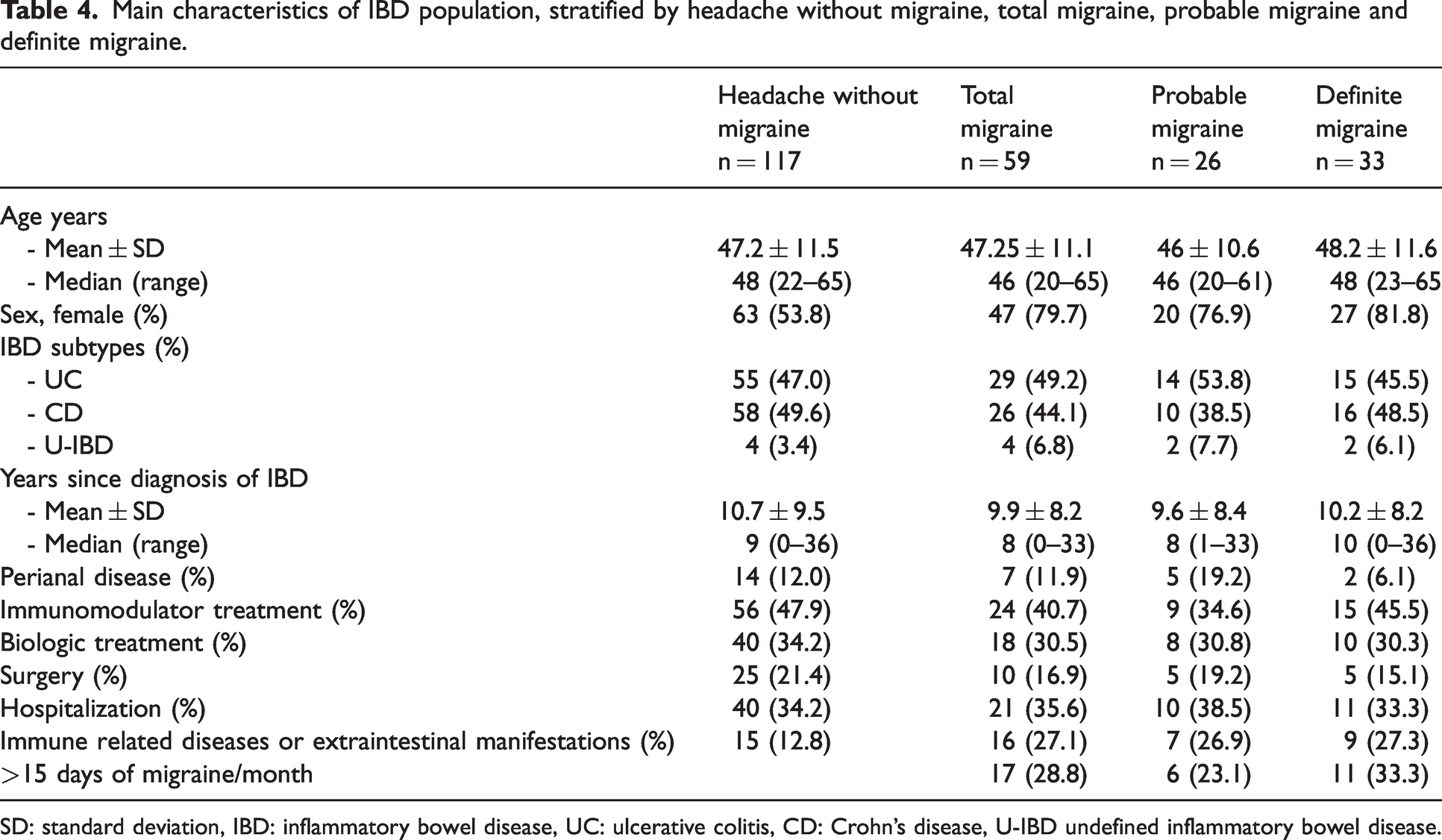
SD: standard deviation, IBD: inflammatory bowel disease, UC: ulcerative colitis, CD: Crohn’s disease, U-IBD undefined inflammatory bowel disease.
We compared these prevalence data with those reported on migraine prevalence in our country in the same age group (20). The prevalence of migraine was significantly increased in IBD patients versus that reported for our general population (12.56%; 95% CI 11.6–13.6%; p < 0.0001). The prevalences of definite and probable migraine are also increased in IBD (p < 0.05 and p < 0.001, respectively) when compared with the general population (definite: 8.40%; 95% CI 7.69–9.15%; probable:4.16%; 95% CI 3.7–4.7%). Stratified by sex, differences in total migraine prevalence remained significant in women (general population: 17.17%; 95% CI 15.82–18.61%; p < 0.0001), but not in men (general population: 8.04%; 95% CI 7.10–9–10%; p = 0.30) (Figure 2).

Migraine prevalence in our IBD patients versus the general population. Notice that prevalence of total, probable and definite migraine in IBD patients was significantly higher than those of the general population, but only for females. General population data taken from reference 20. ns: non-significant; *p < 0.05; **p < 0.01: ***p < 0.001; **** p < 0.0001.
By IBD diagnostic subtypes, 29 (20.7%; 95% CI 14.33–28.38%; 14 probable and 15 definite) of UC, 26 (19.8%; 95% CI 13.39–27.71%; 10 probable and 16 definite) of CD and four (33.3%; 95% CI 6.81–49.90%; two probable and two definitive) of U-IBD patients met migraine diagnostic criteria. There were no significant differences in migraine prevalence between CD and UC (p = 0.88) (Figure 3).

Prevalence of migraine in IBD and main IBD subtypes (UC and CD) versus our general population showing a significant, uniform increase in migraine prevalence for IBD, CD and UC. General population data taken from reference 20. ns: non-significant; ****: p < 0.0001.
Regarding IBD characteristics and migraine, men with total migraine had a higher rate of use of biologic treatment (75% vs 40.7% p < 0.05) and of immune mediated inflammatory diseases or extraintestinal manifestations of IBD (33.3% vs 9.6% p < 0.05). These associations were not significant for women, nor for the overall population (p>0.05). Additionally, we did not find any significant influence in the prevalence of migraine with/without the use of mesalazine or immunomodulators, previous surgeries or hospitalizations due to IBD, neither in total number of patients nor stratified by sex.
Discussion
The main finding of this work is that one out of five (exactly 20.85%) among unselected patients with an established diagnosis of IBD met migraine criteria according to the validated ID-Migraine questionnaire (11.66% and 9.18% definite and probable criteria, respectively). There are two big surveys of migraine prevalence in the general population exactly at the age range of this study in our country (20,22). In both studies patients were interviewed using a structured questionnaire. The first, carried out in 1992 and involving 2232 subjects examined at home by neurologists, used the 1988 IHS migraine criteria and found that exactly 12% of the population (17% women and 8% men) in Spain had migraine (22). The most recent study involved 5668 subjects from the general population aged 18–65 years and found an almost identical 12.6% 1-year migraine prevalence (17.2% in males and 8.0% in females) (20). In this study, 8.4% and 4.2% met definite and probable migraine criteria, respectively. When compared with data coming from Matías-Guiu et al.’s survey (22), global migraine prevalence in our series of IBD patients was numerically and significantly increased. The same can be concluded comparing our results with those from a very recent meta-analysis, which has calculated that 15.2% of the population aged 20–65 years (18.6% of women and 8.7% of men) suffers from active migraine (23). As shown in Table 1, results from 11 previous reports testing this possible association are heterogenous. Although our final numbers are low to draw definitive conclusions and our study does not include a longitudinal follow-up, the proportion of our IBD patients who would meet chronic migraine criteria (6%) are also clearly higher than those reported globally (24) and specifically in our region (2%) (25), which suggests not only an association between IBD and migraine, but that the presence of IBD would predispose to migraine chronicity.
We also show that global migraine prevalence remains higher than that of the general population in, CD and UC, the two main types of IBD, a question unexamined in those studies which had found a significant association between IBD and migraine. What could be the reasons for such migraine and IBD comorbidity? By definition, IBD is a chronic (systemic) inflammatory disease (4,5) and inflammation might play a role in migraine initiation and chronification (26). Migraine has been shown to be associated with other inflammatory disorders and neurogenic inflammation, with increased expression of proinflammatory cytokines, including interleukins 1β, 6 and 8, and tumor necrosis factor alpha, has been linked to migraine pathophysiology (26). This comorbidity between migraine and IBD could be another example of the bidirectional relationship between the gastrointestinal and the central nervous system known as the “gut-brain axis”, which has been involved in the pathophysiology of migraine in other gastrointestinal disorders, such as celiac disease, irritable bowel syndrome or Helicobacter infection (27). The mechanisms of how brain and the gut interact in migraine patients are not entirely clear, but changes in the microbiota and in the biochemistry of neuronal innervation on the gastrointestinal tract could be implicated. Microbiota prevents systemic inflammation and plays a key role in nociception by preparing host adaptation to stress factors (27,28). The neuronal influence on the inflammatory status of the gastrointestinal tract is well-demonstrated involving a variety of neurotransmitters, such as serotonin, and of pain-producing neuropeptides, including calcitonin gene-related peptide (CGRP). CGRP has been shown to exert a key role in both migraine and gut mucosa homeostasis: an excess of CGRP induces migraine pain and diarrhea (29), while lack of CGRP induces colitis and spontaneous lymphoid hyperplasia, which indicates a protective role in bowel inflammation (30).
There are other factors which could contribute to this association. The association between migraine and IBD was only significant for females, which could suggest a role for sex-linked factors, such as oestrogens (31,32) or even genetic predisposition depending on sexual chromosomes (33). On the other hand, non-steroidal anti-inflammatory agents are the most popular acute treatment of migraine and they have been shown to have the potential to trigger the onset or relapse of IBD (34).
Even though all patients with IBD — regardless of their clinical situation — are followed in our IBD Clinic, one limitation of our study is that it was conducted in a specialized clinic in a tertiary-care center and, therefore, their results are not necessarily transposed to the generality of IBD subjects. Although our study contains the highest number of subjects tested in this regard in a specialized IBD clinic (see Table 1), it is true that we should be cautious on interpreting results coming from subgroups with a final low number of subjects, such as males or chronic migraineurs, as definitive. In spite of its high sensitivity (0.92) and specificity (0.82) for migraine diagnosis (18), ID-Migraine questionnaire is a screening tool rather than a diagnosis made by an expert physician. Finally, one further limitation is that there was no simultaneous, matched control group from the general population to compare migraine prevalence, but we think that the very consistent results from the two big prevalence studies in our country (and those of the recent metanalysis of migraine prevalence in Europe) make a fair comparison possible.
Conclusions
This cross-sectional study supports previous surveys reporting an association between IBD and migraine. The fact that this association was stronger for women suggests an influence of sex-related factors.
Article highlights
IBD and migraine are comorbid diseases; the presence of IBD could contribute to migraine chronicity. This comorbidity was significant only for women, which suggests an influence of sex-related factors.
Footnotes
Acknowledgments
We are very grateful to our IBD nurse María Soledad Serrano for her continuous support.
Declarations of conflicting interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been founded by Instituto de Salud Carlos III (ISCII) through the project PI20/01358 and co-funded by Fondos Europeos de Desarrollo Regional (FEDER), “Una manera de hacer Europa”.
