Abstract
Background
Drugs for migraine attacks include triptans and NSAIDs; their combination could provide greater symptom relief.
Methods
A total of 314 subjects with history of migraine, with or without aura, were randomized to frovatriptan 2.5 mg alone (Frova), frovatriptan 2.5 mg + dexketoprofen 25 mg (FroDex25) or frovatriptan 2.5 mg + dexketoprofen 37.5 mg (FroDex37.5) and treated at least one migraine attack. This was a multicenter, randomized, double-blind, parallel-group study. The primary end point was the proportion of pain free (PF) at two hours. Secondary end points were PF at one and four hours, pain relief (PR) at one, two, four hours, sustained PF (SPF) at 24 and 48 hours, recurrence at 48 hours, resolution of nausea, photophobia and phonophobia at two and four hours, the use of rescue medication and the judgment of the treatment.
Results
The results were assessed in the full analysis set (FAS) population, which included all subjects randomized and treated for whom at least one post-dose intensity of headache was recorded.
The proportions of subjects PF at two hours (primary end point) were 29% (27/93) with Frova compared with 51% (48/95 FroDex25 and 46/91 FroDex37.5) with each combination therapies (p < 0.05). Proportions of SPF at 24 hours were 24% (22/93) for Frova, 43% (41/95) for FroDex25 (p < 0.001) and 42% (38/91) for FroDex37.5 (p < 0.05). SPF at 48 hours was 23% (21/93) with Frova, 36% (34/95) with FroDex25 and 33% (30/91) with FroDex37.5 (p = NS). Recurrence was similar for Frova (22%, 6/27), FroDex25 (29%, 14/48) and FroDex37.5 (28%, 13/46) (p = NS), meaning a lack of improvement with the combination therapy.
Statistical adjustment for multiple comparisons was not performed.
No statistically significant differences were reported in the occurrence of total and drug-related adverse events.
FroDex25 and FroDex37.5 showed a similar efficacy both for primary and secondary end points. There did not seem to be a dose response curve for the addition of dexketoprofen.
Conclusion
FroDex improved initial efficacy at two hours compared to Frova whilst maintaining efficacy at 48 hours in this study. Tolerability profiles were comparable. Intrinsic pharmacokinetic properties of the two single drugs contribute to this improved efficacy profile.
Introduction
Migraine affects approximately 18% of women and 6% of men in Western countries (1). The prevalence peaks in subjects between 25 and 55 years old. The burden of migraine is enormous; in the United States (US) migraine sufferers spend a total of 112 million days bedridden and the annual cost of missed work or reduced productivity is $5.6 billion to $17.2 billion (1,2).
The International Headache Society (IHS) criteria for diagnosing migraine (3) are the most widely used diagnostic criteria around the world. Drugs effective for treating migraine attacks include triptans, analgesics (such as nonsteroidal inflammatory drugs (NSAIDs)), ergot derivatives and antiemetics (4). Triptans, marketed in the early 1990s, were a major advance in the treatment of moderate-severe acute migraine attacks (4). They are selective 5-HT1B/D agonists. Seven triptans are available, being, theoretically, similar, but patients note differences in effectiveness and in tolerance (5).
Frovatriptan is one of the newest triptans and its distinct pharmacokinetic and pharmacodynamics profile means that it has the clinical potential for a long duration of action and a low likelihood of side effects and drug interactions (6). The elimination half-life of frovatriptan is five times that of other triptans, while the time to maximum concentration (tmax) is similar to other triptans when given orally (5,7–10).
NSAIDs and other simple analgesics are indicated for the treatment of mild or moderate migraine attacks (4). Dexketoprofen works by blocking the action of cyclooxygenase (COX), which is involved in the production of prostaglandins, and thereby reduces inflammation and pain (11,12). Dexketoprofen maximum plasma concentrations are observed around 30 minutes after an oral dose. Its elimination half-life, however, is quite short (13). Dexketoprofen proved to be effective and very fast in reducing pain intensity and accompanying symptoms in 42 women affected by migraine with or without aura (14).
The complexity of the pathophysiology of migraine partially explains why none of the currently available monotherapies provides broad coverage of the multiple pathogenic processes in migraine (15–25).
A therapy that targets multiple mechanisms may confer advantages over monotherapy, and triptans and NSAIDs target distinct aspects of the vascular and inflammatory processes hypothesized to underlie migraine. This possibility is supported by the results of several clinical studies, including those on the concomitant use of sumatriptan succinate with naproxen sodium and rizatriptan with rofecoxib (26–30).
The rationale for combining dexketoprofen with frovatriptan is linked to the intrinsic pharmacokinetic properties of the two drugs; dexketoprofen is absorbed rapidly and contributes to the early efficacy of the combination, whereas frovatriptan persists longer and so provides sustained efficacy with less recurrence.
The aims of this study were to evaluate the efficacy and tolerability of the combination therapy frovatriptan plus dexketoprofen over frovatriptan alone in the acute treatment of migraine attacks.
Methods
Study population
The study recruited subjects of male or female gender, 18–65 years old, with a current history of migraine with or without aura, according to IHS 2004 criteria, and with at least one but no more than six migraine attacks per month for six months prior to entering the study. Figure 1 shows the study flowchart. Flow diagram of participants throughout the study.
Patients could not be enrolled in the study if they: (a) had coronary artery or cerebrovascular disease; (b) had uncontrolled hypertension; (c) had other types of headache in addition to migraine with or without aura; (d) were taking ergotamine (or its derivatives), St John’s Wort (Hypericum perforatum), monoamine oxidase (MAO)-inhibitors, NSAIDs (COX-2 inhibitors), oral corticosteroids, warfarin or other coumarins, selective serotonin reuptake inhibitors, or anti-aggregant agents such as aspirin, heparin, lithium, methotrexate, hydantoins and sulphonamides; (e) had severe liver or renal impairment; (f) had a history of alcohol, analgesic or psychotropic drug abuse; (g) had known hypersensitivity to either of the study drugs or to any other NSAIDs; (h) had active peptic ulcer/bleeding or a history of gastrointestinal bleeding or perforation related to NSAID therapies; (i) had bronchial asthma; (j) were pregnant or breast feeding.
Written informed consent was obtained from all patients prior to their inclusion in the study. The study was approved by the independent institutional review boards of the centers in which the study was carried out. The study was conducted according to the Declaration of Helsinki and Good Clinical Practice and was registered internationally (Eudract number 2009-011577-32).
Study design
This was a multicenter, randomized, double-blind, active-controlled, three parallel-group, phase 3 study performed in 25 Italian centers from July 2009 to April 2010. Over-encapsulation was used to ensure blinding.
This active treatment dose-comparison concurrent control study compared frovatriptan 2.5 mg + dexketoprofen 25 mg (FroDex25) or frovatriptan 2.5 mg + dexketoprofen 37.5 mg (FroDex37.5) to frovatriptan 2.5 mg + placebo (Frova), in acute treatment of migraine with or without aura attacks in three parallel groups of 100 subjects each. The study protocol also foresaw a comparison between FroDex25 and FroDex37.5.
After the screening visit, subjects were assigned by the investigators to one of the three treatment groups using randomization lists prepared by InnoPharma s.r.l. generated using three-subject blocks to balance the treatments. The lists were provided to all sites before the start of the study. Code-break envelopes were provided to the sites and could be opened in the case of an emergency. To keep the patients and study investigators blind to the treatment, the study drugs were over-encapsulated.
Frovatriptan, dexketoprofen and placebo tablets were over-encapsulated in capsules suitable to keep the blinding. Two colors of capsules were used: white and red. The extemporary combination of frovatriptan 2.5 mg + dexketoprofen 37.5 mg, frovatriptan 2.5 mg + dexketoprofen 25 mg, frovatriptan 2.5 mg + placebo was provided in indistinguishable carton boxes identified by the randomization numbers, each containing one treatment dose blister with two capsules: a red and a white one.
All study end point parameters were recorded in a personal diary and, for the intensity of the headache attack, a four-point rating scale recommended by the IHS (0 = no headache, 1 = mild headache, 2 = moderate headache, 3 = severe headache) was used. Similar scales were used for assessment of the associated symptoms (nausea, photophobia and phonophobia).
The active controlled design was chosen in accordance with guidelines on the choice of control groups in studies aimed at demonstrating superiority of a new treatment when an established treatment already exists (3,4). The two doses of 25 mg and 37.5 mg of dexketoprofen were chosen based on available literature data, and on the results from previous pre-clinical and clinical studies (12). The frovatriptan dose of 2.5 mg is the dose recommended in the SmPC (31).
During the randomization visit, after signing written informed consent, subjects provided their clinical, medication and migraine history. Physical and neurological examinations and a pregnancy test (if appropriate) were performed. Blood pressure and heart rate were measured for all subjects. The degree of migraine-associated disability was also determined, using the Migraine Disability Assessment (MIDAS) questionnaire. At the end of the visit a headache diary was dispensed with the study medication in order to document the characteristics of the headache pain and associated symptoms, and subjects were instructed to take the study medication and complete the diary for the first migraine attack that occurred during the study period (i.e. within one month from randomization).
The study treatment was self-administered orally in single administration as early as possible after the onset of the migraine headache. During the study, three visits to the clinical center were scheduled: one at the screening, one at the randomization visit and a follow-up visit between 72 hours and 14 days (±2) after the migraine attack.
Data analysis
The primary efficacy variable to assess the possible superiority of frovatriptan plus dexketoprofen (high and low doses) over frovatriptan alone was the percentage of subjects pain free at two hours before any rescue medication. A minimum difference of 20% in the proportion of patients who were pain free was considered evidence of the clinical superiority of frovatriptan plus dexketoprofen (high dose) over frovatriptan alone.
The 20% difference for the power was pre-specified in the study protocol and was determined by taking into account that there were not published or unpublished data of FroDex combination therapy versus Frova monotherapy; there were not specific requests coming from migraine guidelines for superiority trials (32). The only available data published of a combination therapy (SumaNapro) versus monotherapy (Suma) showed a mean difference within the range of 9% and 15%. For these reasons we considered a 20% difference a reasonable clinically important difference to reach (25–27,35). The study’s primary analysis population was the full analysis set (FAS) population that included all subjects randomized and treated for whom at least one post-dose intensification of headache was recorded.
The per-protocol (PP) population consisted of all patients who treated a migraine attack without protocol violations. This population was identified prior to database lock and was used as a confirmatory analysis.
Secondary study end points were: Proportion of pain-free subjects at one hour and four hours, defined as subjects free of pain one hour and four hours before any rescue medication, according to IHS guidelines (3); Proportion of patients with headache relief at one, two and four hours, defined as the percentage of subjects with a decrease in headache from severe or moderate to mild or none within one hour, two hours and four hours, according to IHS guidelines (3); Sustained pain free, defined as the percentages of subjects who were pain free within two hours with no use of rescue medication or recurrence within 24 hours and 48 hours, according to IHS guidelines (3); Recurrence, defined according to IHS guidelines as a headache of any severity returning within 48 hours, in a subject who was pain free at two hours and who had not taken any rescue medication (3); Proportion of subjects with resolution of nausea, vomiting, photophobia and phonophobia; Proportion of subjects taking rescue medication; Time to meaningful relief, defined as a reduction of at least two points on the four-point scale (i.e. from severe pain score of 3 at baseline to mild or no pain of score 1/0); Speed of onset at 60, 90, 120 and 240 minutes, defined as a decrease of one point in pain intensity; Subjects’ preference for treatments.
Main and secondary variables were analyzed at a two-sided significance level of 0.05.
The tolerability analysis was conducted on all randomized patients and included the incidence of adverse events and changes in vital signs during the study. Continuous variables were summarized by computing mean values and their standard deviation (SD), while categorical variables were summarized by computing their absolute values and frequencies (as percentages).
The efficacy assessments were made by the patient at the time of dosing (baseline) and at two, four, 24 and 48 hours after dosing. The data were analyzed using SAS® version 9.2.
The planned sample size of 300 randomized subjects (100 per group) (allowing for a drop-out rate of at least 20%) was chosen to provide the study with the statistical power to detect the possible superiority of the test drug (FroDex37.5) with respect to the reference treatment (Frova alone), on the percentage of subjects pain free at two hours before any rescue medication, as the primary variable, and assuming 20% as the margin of superiority and a power of almost 90% (88%). Thus, the test drug was to be considered superior to the reference one if the difference (test minus reference) in the primary variable was >20%.
The primary variable was assessed by the Fisher-Freeman-Halton Exact test statistic using a 3 × 2 contingency table for tests of association and a 2 × 2 contingency table for comparisons between treatments. All other secondary parameters were assessed in the same way as the primary variable. The level of significance was set at 0.05 for all analyses. The primary and all secondary parameters were assessed in the FAS population, which included all subjects randomized and treated for whom at least one post-dose intensification of headache was recorded.
All p values for secondary end points are reported without adjustment for multiplicity.
Results
Demographic and clinical data of the 279 patients in the FAS population at the time of randomization. Data are shown as mean (±SD), or absolute (n) and relative frequency (%).
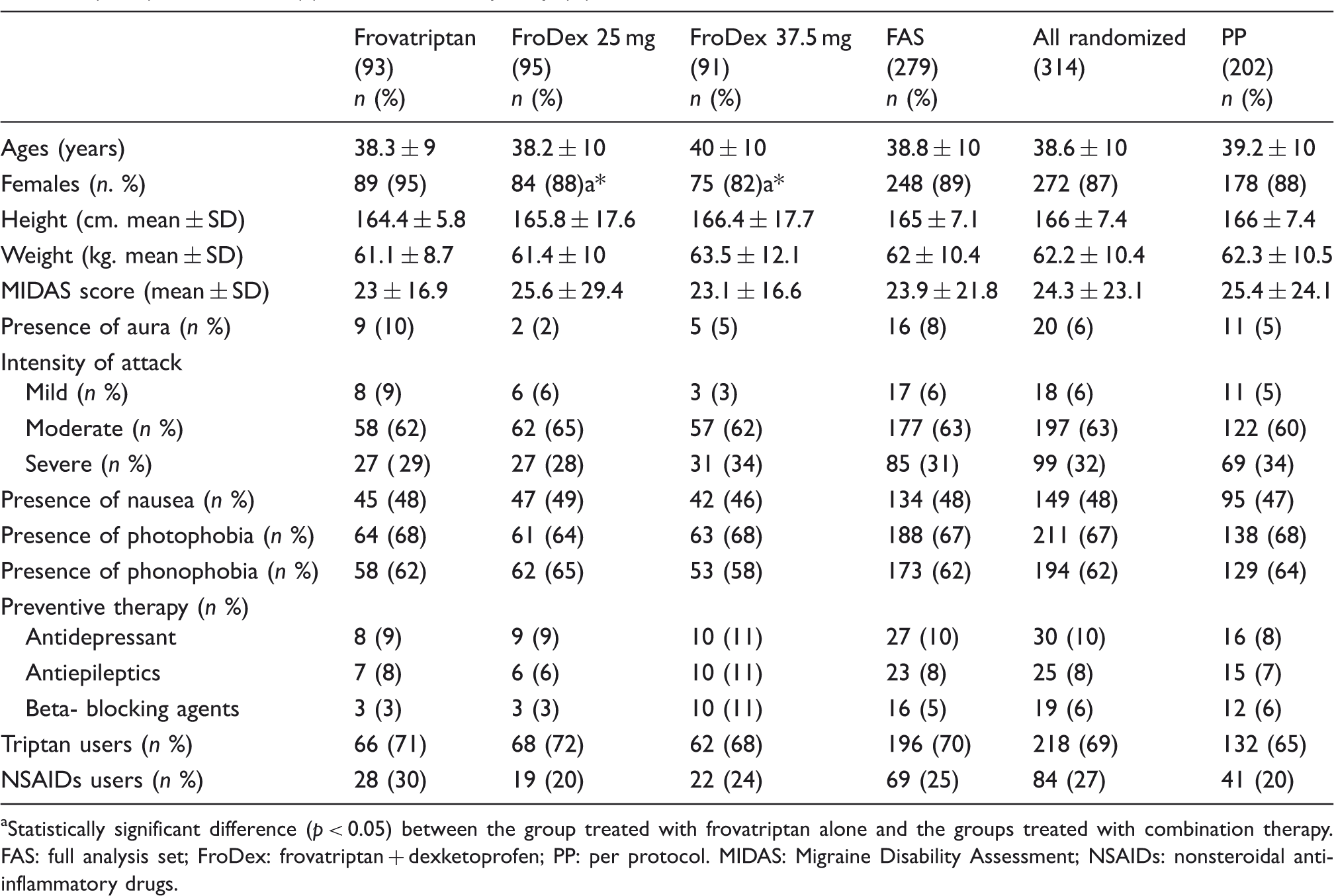
Statistically significant difference (p < 0.05) between the group treated with frovatriptan alone and the groups treated with combination therapy. FAS: full analysis set; FroDex: frovatriptan + dexketoprofen; PP: per protocol. MIDAS: Migraine Disability Assessment; NSAIDs: nonsteroidal anti-inflammatory drugs.
Primary end point
Overall comparison among treatments showed a statistically significant difference (p = 0.003, Fisher-Freeman-Halton Exact test). In the FAS population, the proportion of patients who were pain free at two hours was 29% (27/93) with Frova, 51% (48/95) for FroDex25 (odds ratio (OR) = 2.5 (1.37–4.5), p < 0.05) and also 51% (46/91) for FroDex37.5 (OR = 2.5 (1.36–4.6), p < 0.05) (Figure 2). There was not a statistically significant difference in the comparison between FroDex25 and FroDex37.5 (OR = 1 (0.56–1.78), p = 1). Proportion (%) of pain free at two hours after administration of Frova, FroDex25 and FroDex37.5 in the 279 patients of the FAS population. Asterisks indicate a statistically significant difference (p < 0.05) between the group treated with frovatriptan alone and the groups treated with combination therapy.
Secondary end points
Results for the secondary end points of the study.

Analyses were conducted with Fisher’s exact test, except Time to meaningful relief (Log rank, Mantel-Cox). There has been no correction for multiple comparisons. aPain relief at one, two and four hours was calculated only on moderate-severe migraine, 85, 89, 88 patients in the Frova, FroDex25 and FroDex37.5 group, respectively. bRecurrence was calculated on Pain free at two hours. FAS: full analysis set; FroDex: frovatriptan + dexketoprofen. All end points were calculated on the FAS population.
There was not a statistically significant difference in the comparison between FroDex25 and FroDex37.5 in all the secondary end points.
FroDex 25 was better than Frova in nearly all of the secondary end points. In particular the proportions of pain free at four hours and of pain relief at one and two hours were significantly better (p < 0.05). Also in terms of sustained effect, more so at 24 than at 48 hours, FroDex25 was better than Frova. With respect to recurrence, however, the results are similar between the Frova and FroDex25 groups.
FroDex37.5 was significantly better than Frova in nearly all of the secondary end points, including pain free at four hours and pain relief at one and two hours. There was also a significant increase in the proportion of patients who were sustained pain free at 24 hours, but a nonsignificant increase (from 23% to 33%) in sustained pain free at 48 hours.
Also in this comparison, the proportion of recurrence was similar between Frova and the combination therapy. The percentages of subjects with resolution of nausea, photophobia and phonophobia at one and two hours were higher among patients taking the combination therapy than among those receiving Frova monotherapy, reaching statistically significant difference in most of them, as summarized in Table 2.
Mean time to meaningful relief was 15.43 ± 16.06 for Frova, 9.80 ± 13.37 for FroDex25 and 9.70 ± 12.35 hours for FroDex37.5. This difference reached a statistical significance for FroDex25 versus Frova.
The speed of onset was calculated at 60, 90, 120 and 240 minutes. In all time points, the combination therapy was better than monotherapy, reaching a statistically significant difference at 60, 90 and 120 minutes.
Finally, the treatment was judged excellent or good by 56% of patients overall (by 44% in the Frova group, 60% in the FroDex25 group and 64% in the FroDex37.5 group, p < 0.05 for both comparisons).
Tolerability
The tolerability analysis was carried out in 281 patients (FAS population, 279 patients, plus two patients who didn’t take the drugs in the correct way but were, however, counted in the tolerability analysis). Total adverse events were reported by 9 patients during treatment with Frova, by 18 during treatment with FroDex25 and by 15 patients during treatment with FroDex37.5 (10%, 19% and 16%, respectively, p = NS). The number of total adverse events was 59, (19 with Frova, 22 for FroDex25 and 18 with FroDex37.5, p = NS), most of which were of mild or moderate intensity. There were two serious adverse events (one case of vestibular neuronitis in the FroDex25 group and one case of loss of consciousness in the Frodex37.5 group), neither of which was considered related to the study treatments.
Drug-related adverse events were reported by 3 patients during treatment with Frova, by 10 during treatment with FroDex25 and by 5 patients during treatment with FroDex37.5 (3%, 11% and 16%, respectively, p = NS).
Distribution of absolute numbers of all adverse events and drug-related adverse events between the three treatment groups in the 281 patients included in the safety analysis.

FroDex: frovatriptan + dexketoprofen; SAE: severe adverse event.
Discussion
In this active treatment dose comparison concurrent control study, the combination of frovatriptan 2.5 mg + dexketoprofen 25 mg or 37.5 mg showed better efficacy than frovatriptan alone across a range of efficacy end points, while maintaining a similar tolerability profile. Our results were consistent both for the FAS and PP populations.
The doses of the two components of the combination were within the respective approved ranges of single daily doses recommended for use by adults in Europe. In fact, the frovatriptan dose is half the maximum allowed daily dose (5 mg), while dexketoprofen 25 mg is a third and dexketoprofen 37.5 mg is half of the maximum allowed daily dose (75 mg) (13,31).
The primary end point of the study was the proportion of subjects who were pain free at two hours without rescue medication. This end point was chosen as primary because it is recommended by many authorities as being the most appropriate for assessing the efficacy of acute migraine treatments in randomized, controlled trials (3,4,33,34).
The most relevant result of our study is that the proportion of patients who were pain free at two hours was significantly higher with combination therapy than with frovatriptan alone and reached more than 50% in the groups treated with combination therapy, meaning that for every two patients treated with combination therapy, one was pain free at two hours. The proportion of pain free obtained with frovatriptan alone in this study is very similar to those obtained in recently published double-blind studies of multiple migraine attacks (7–10) while the results obtained with the combination therapy are consistent with, or even better than, those obtained with sumatriptan + naproxen sodium (pain free at two hours: 30%–34% with combination therapy versus 23%–25% with sumatriptan alone, with a mean difference within the range of 9% and 15%) (25), although direct comparison studies of the two combination treatments are lacking.
Among the secondary end points a sustained pain-free status, the ideal migraine treatment response, is today considered the hardest end point achievable in clinical studies and in our study we used the best definition from recent guidelines (3,4,33). In our study the proportion of sustained pain free patients at 24 hours was significantly higher with both doses of the combination therapy than with frovatriptan monotherapy, 42%–43% versus 24%. The proportion of sustained pain free at 48 hours was also different between the frovatriptan monotherapy and combination therapy groups with around a quarter (23%) achieving this endpoint with Frova compared to about a third with both combination treatments (33% and 36% for the FroDex25 and FroDex37.5 groups, respectively) (Figure 3). These differences were not statistically significant but it should be remembered that the trial was not powered for this particular endpoint. Proportion (%) of sustained pain free at 24 and 48 hours and relapse at 48 hours after administration of Frova, FroDex25 and FroDex37.5 in the 279 patients of the FAS population. Asterisks indicate a statistically significant difference (p < 0.05) between the group treated with frovatriptan alone and the groups treated with combination therapy.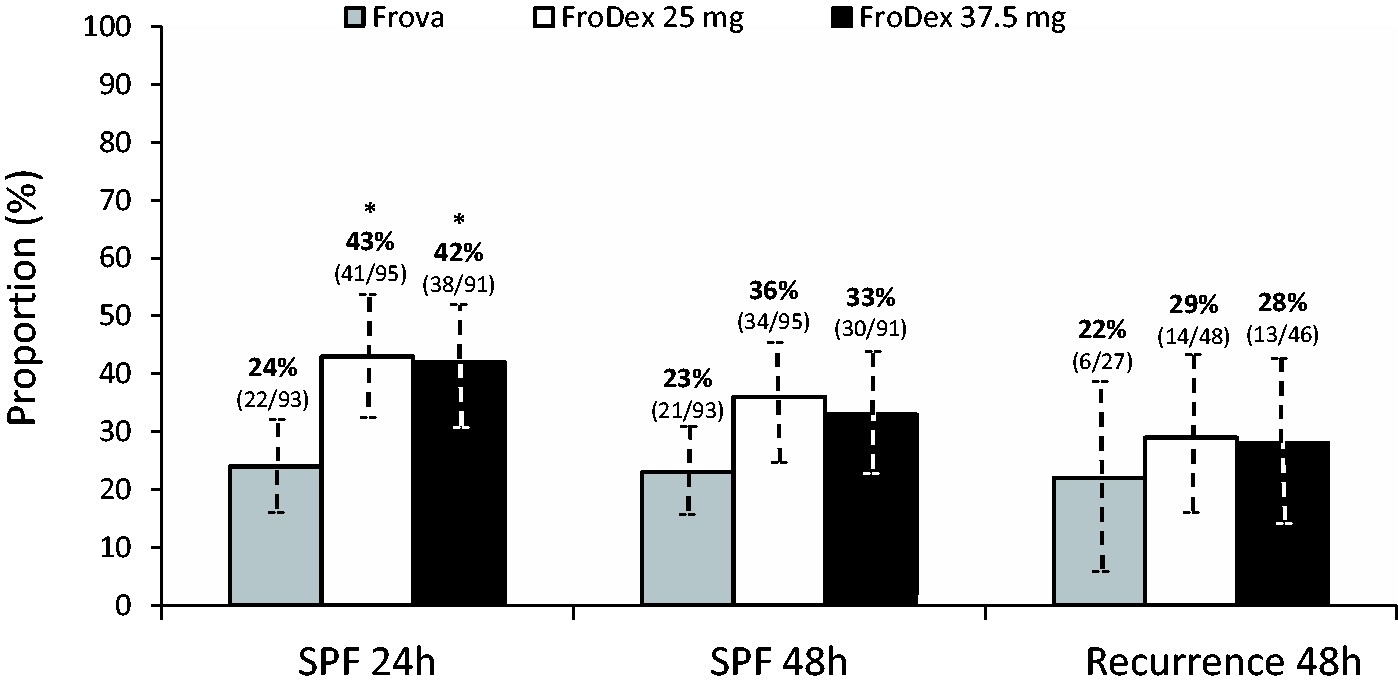
The proportion of sustained pain free with frovatriptan alone is consistent with those obtained in recently published, double-blind studies (7–10) while results obtained with combination therapy at 24 hours are in line with those obtained with sumatriptan + naproxen sodium (sustained pain free at 24 hours: 23%–25% with combination therapy versus 14%–6% with sumatriptan alone) (25).
As indicated by IHS guidelines, recurrence was assessed in the time period up to 48 hours, which is a more stringent end point than recurrence at 24 hours (3). The proportion of recurrence was similar in the three groups (Frova 22%, FroDex25 29%, FroDex37.5 28%, p = 0.80). The recurrence findings with frovatriptan alone in our study were in line with the rates in three recently published head-to-head studies comparing frovatriptan against three different triptans (7–10). The proportion of recurrence among the patients who took the combination therapies was similar to the rate in the group treated with frovatriptan alone. These results are surprising as one of the aims of the study was to demonstrate a superiority of combination therapy versus monotherapy in reducing recurrence. One possible explanation could be that the sustained effect of combination therapy is largely driven by the long half-life of frovatriptan (31–35) (Figure 3).
Interestingly, the use of rescue medication was lower in the groups treated with combination therapy (FroDex25 33%, FroDex37.5 29%) than in the group treated with Frova (45%). In particular the difference between the Frova and FroDex37.5 groups was statistically significant (p < 0.05): combination therapy demonstrated both a more immediate and a more sustained effect over 24 hours versus monotherapy; this is probably the reason why patients taking combination therapy needed to take less rescue medication. All the study treatments were well tolerated with no safety concerns identified in this study. The number of treatment-related adverse events was low and the type of events reported were similar to those previously described for the two active ingredients. It is important to underline that in this study population the higher dose of the combination therapy (frovatriptan 2.5 mg + dexketoprofen 37.5 mg) showed a similar efficacy, not statistically significant, both for primary and secondary end points. This lack of dose response seen with the two doses of FroDex could be explained by a ceiling effect that occurs when adding an NSAID (dexketoprofen) to a triptan (frovatriptan).
The results of this study should be interpreted in the context of its limitations. First of all, as in the study by Brandes et al. (25), the patients’ most common pre-study migraine medications were NSAIDs and triptans. These are the most common medications taken by migraineurs generally. The degree to which the efficacy data from this study can be extrapolated to NSAID- and triptan-naïve patients and to patients with migraine refractory to these kinds of drugs is unknown (21). A second limitation is that the tablets of frovatriptan, dexketoprofen and placebo were over-encapsulated separately. The over-encapsulation could decrease the speed of absorption of the drug during a migraine attack. Moreover, we did not compare FroDex with frovatriptan and dexketoprofen taken together as a unique tablet, but for patients for whom adherence to taking two pills is an issue, the combination pill would have obvious advantages.
Our study did not include a dexketoprofen alone arm, but we must underline that the purpose of developing a combination therapy is to maximize both the proportion of patients who were pain free at two hours and the proportion who are sustained pain free at 24 and 48 hours; dexketoprofen is well known to have a short half-life and its sustained effect is not relevant.
Our study also did not include a placebo arm; in this way it was not possible to take into account the placebo effect, which is large and highly variable in migraine studies (32).
Finally, the results should be interpreted cautiously as some results may be due to chance, because statistical adjustment for multiple comparisons was not foreseen and performed in the study protocol and statistical analysis plan.
Conclusion
In our study comparison of both combination therapies, on primary and secondary end points showed no statistically significant differences.
Regarding the comparison of combination therapy versus monotherapy, the results from this study are consistent with those of a comparison between sumatriptan alone and sumatriptan plus naproxen sodium: the combination of the NSAID and a triptan resulted in superior clinical benefits over monotherapy (25). The efficacy of triptans plus NSAIDs in general compared with triptan monotherapy might, in part, be explained by the fact that the combination therapy targets multiple pathogenic mechanisms in migraine (25).
Finally, results from this study need to be confirmed by larger clinical trials.
Clinical implications
A randomized controlled trial (RCT) with 314 subjects with history of migraine (with or without aura) using three different treatments for migraine attacks has never been compared before. The primary efficacy end point was pain free at two hours, making the results comparable with other RCTs on migraine. Secondary end points (pain relief, sustained pain free, relapse within 48 hours) are also common to several RCTs on migraine. Demonstration that frovatriptan plus dexketoprofen is, in broad terms, superior to frovatriptan alone.
Footnotes
Funding
This work was supported by Istituto Lusofarmaco d’Italia.
Conflicts of interest
All authors have occasionally served as scientific consultants for manufacturers of frovatriptan. Dario Zava and Deborha Pezzola are employees of the manufacturer of frovatriptan.
Author contributions
Coordinator: Prof G Bussone (Milano). Investigators: GL Mancardi (Genova), MC Tonini (Garbagnate), M Guidotti and M Mauri (Como), R Cerbo (Roma), M Gionco (Torino), G Comi (Milano), G Rapisarda (Catania), M Aguggia (Asti), A Ganga (Sassari), M Bartolini (Ancona), R Sterzi (Milano), B Panascia and R Rapisarda (Catania) and R De Simone (Napoli).
