Abstract
Background
Estimates of proportions of people with migraine who report premonitory symptoms vary greatly among previous studies. Our aims were to establish the proportion of patients reporting premonitory symptoms and its dependency on the enquiry method. Additionally, we investigated the impact of premonitory symptoms on disease burden using Headache Impact Test (HIT-6), Migraine Disability Assessment (MIDAS) and World Health Organization Disability Assessment 2.0 (WHODAS 2.0), whilst investigating how various clinical factors influenced the likelihood of reporting premonitory symptoms.
Methods
In a cross-sectional study, premonitory symptoms were assessed among 632 patients with migraine. Unprompted enquiry was used first, followed by a list of 17 items (prompted). Additionally, we obtained clinical characteristics through a semi-structured interview.
Results
Prompted enquiry resulted in a greater proportion reporting premonitory symptoms than unprompted (69.9% vs. 43.0%; p < 0.001) and with higher symptom counts (medians 2, interquartile range = 0–6 vs. 1, interquartile range = 0–1; p < 0.001). The number of symptoms correlated weakly with HIT-6 (ρ = 0.14; p < 0.001) and WHODAS scores (ρ = 0.09; p = 0.041). Reporting postdromal symptoms or triggers increased the probability of reporting premonitory symptoms, whereas monthly migraine days decreased it.
Conclusions
The use of a standardized and optimized method for assessing premonitory symptoms is necessary to estimate their prevalence and to understand whether and how they contribute to disease burden.
Introduction
Migraine is a disabling neurological disorder characterized not only by headache, but also by a range of non-headache symptoms. One of them is premonitory symptoms, which precede the headache phase of migraine without aura and the aura in migraine with aura when they occur (1,2). Commonly reported premonitory symptoms include fatigue, neck stiffness, mood changes and concentration difficulties, presenting up to 48 hours prior to the headache (3). However, the estimates of the proportion of people with migraine who report these symptoms vary considerably and range from 7.8 to 67.4% in population-based studies (4–7) and from 21.5 to 100% in clinic-based studies (8–10). These prominent differences raise methodological concerns regarding definition, overlap with the headache, postdromal and interictal phases, and differences in assessment methods (3). Questionnaires with open-ended enquiry can lead to under-reporting, whereas those with predefined prompting lists encourage agreement bias (11). Without standardized methods, uncertainty exists over which symptoms are truly premonitory and no assessment is possible of their contribution, if any, to migraine-attributed burden. This knowledge is required to understand migraine pathogenesis better, and possibly to develop and guide preemptive treatments (12,13).
This observational study had four aims. The first was to obtain an estimate of the proportion of patients with migraine in a tertiary headache center who report premonitory symptoms. The second was to assess the influence of means of enquiry on this estimate by comparing two different methods: open-ended questions (unprompted enquiry) and selection from a pre-defined list (prompted enquiry). The third was to examine how premonitory symptoms might contribute to migraine-attributed burden by comparing burden estimates in people with and without premonitory symptoms. The fourth was to determine whether specific factors influenced the likelihood of reporting premonitory symptoms.
Methods
Overview
The data presented in the present study were collected as part of a larger parental study called the Registry for Migraine (REFORM). REFORM is a prospective, single-center study that included participants from a single-arm, open-label, phase IV trial (NCT04265755) at the Danish Headache Center (5). The detailed methodology of REFORM is reported elsewhere (14).
Ethics
The parental study, REFORM, was approved by the Committees on Health Research Ethics in the Capital Region of Denmark and conducted in accordance with the Declaration of Helsinki (15). Additional approval for using data from the interview and questionnaires was not needed according to this committee. All participants provided informed consent prior to inclusion and participation.
Recruitment
Recruitment for this study took place between September 2020 and June 2022. Prospective participants were identified from the outpatient lists of the Danish Headache Center and referrals from neurologists. All were screened for eligibility by phone.
The inclusion criteria of the parental study were age ≥18 years, a history of migraine with or without aura (as defined by the International Classification of Headache Disorders, version 3; (ICHD-3)) (2) for at least one year and experiencing ≥4 monthly migraine days on average over the last three months. The key exclusion criteria were migraine onset after the age of 50 years, a history of cluster headache or of hemiplegic migraine, and inability to differentiate migraine from other headaches. Participants with unremitting daily headaches (i.e. no headache-free periods) were also excluded from the study population of this study. The full list of inclusion and exclusion criteria for REFORM is provided in supplementary Table 1.
Definition
Premonitory symptoms were defined, as by ICHD-2 and ICDH-3β (16,17), as occurring 2–48 hours before the headache phase in migraine without aura or before the aura phase in migraine with aura. The lower limit of two hours was set to distinguish these symptoms clearly from those of the aura or headache phase.
Procedures
Eligible participants were invited to an on-site semi-structured interview, and medical examination to exclude any participant with findings that might need further investigation. The interview gathered information on demographics, medical history and characteristics of migraine (with or without aura, episodic or chronic, with or without acute medication overuse, current use of preventative medication). Other chronic pain conditions (such as neck pain and back pain), depression and/or anxiety were assessed as present if diagnoses given by a physician were reported by the participant, with validation from medical health records when possible but otherwise accepted.
Participants were then asked to report any premonitory symptoms, as defined. If they had difficulty understanding this task, the question was reformulated, or illustrative examples (without mention of any specific symptom) provided a timeline of when such symptoms occurred relative to headache or aura (Figure 1). The enquiry next proceeded through a 17-item list of possible symptoms. A consensus among our research group led to the selection of symptoms based on their high prevalence in previous studies (18–20). Postdromal symptoms were assessed in a similar manner with prompted and unprompted enquiry, whereas trigger factors were assessed through a prompted enquiry with option to add additional factors.
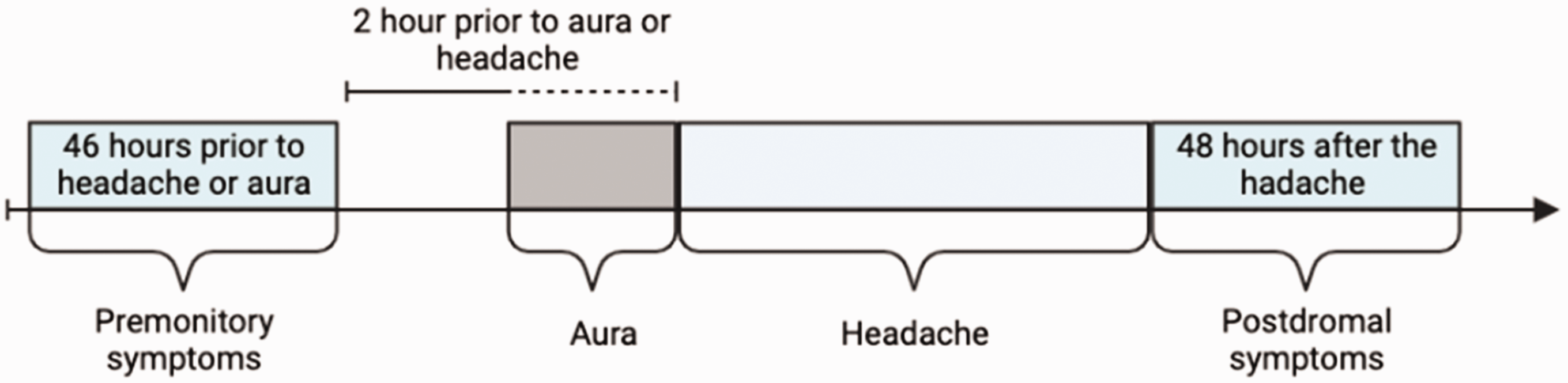
Example of illustration used to explain premonitory symptoms.
Finally, participants were provided with burden assessments in the form of electronic patient-reported outcome measures (PROMs), using the Research Electronic Data Capture Software (REDCap) (https://www.project-redcap.org). They were instructed to complete these PROMs within 24 hours after the visit.
Outcomes and measures
The primary measure was the proportion of patients reporting one or more premonitory symptoms.
Three PROMs were used as secondary measures: Migraine Disability Assessment (MIDAS) (21), Headache Impact Test (HIT-6) (22) and a shortened version of the World Health Organization Disability Assessment 2.0 (WHODAS 2.0) (23). MIDAS scores (the sum of missed days per three months because of headache) were categorized as “little or no disability” (0–5), “mild disability” (6–10), “moderate disability” (11–20) or “severe disability” (≥21) (21). HIT-6 scores in the range 36–78 were categorized as “little or no impact” (≤49), “some impact” (50–59), “substantial impact” (56–59) or “severe impact” (60–78) (22). WHODAS-12 scores, in the range 12–60, assessed daily difficulties related to health, with higher scores indicating greater difficulties (23).
Data from the semi-structured interviews were used to investigate whether clinical factors (i.e. age, female sex, migraine with aura, chronic migraine, medication-overuse headache, monthly migraine days, current use of preventive treatment, reporting postdromal symptoms or trigger factors) influenced the likelihood of reporting premonitory symptoms.
Statistical analysis
We used descriptive statistics to present the characteristics of participants. Normality of outcome data was tested using Kolmogorov–Smirnov normality test. We used the mean ± SD to present normally distributed continuous data; medians and interquartile ranges (IQRs) to present non-normally distributed data; and proportions (n and %) to present categorical data.
We made comparisons between participants with and without premonitory symptoms and, in the former, correlated outcome measures with the number of premonitory symptoms reported. Paired nominal data were compared using McNemar's test. Non-parametric continuous data and grouped, ordinal data were compared using the Mann–Whitney U-test. Correlation between variables was assessed using Spearman correlation and reported with the Spearman's rank correlation coefficient (ρ) and corresponding p values.
A two-step approach was used to evaluate risk assessment. In the first step, a simple logistic regression was conducted with potential variables. Significant variables (p < 0.05) were then included in a binomial logistic regression for further risk assessment. Odds ratios (ORs), 95% confidence intervals (CIs) and p values were reported. p < 0.05 was considered statistically significant. Bonferroni correction was applied to adjust for multiple comparisons following the binomial logistic regression analysis. All statistical analyses were performed using R, version 4.2.0 (R Foundation, Vienna, Austria).
Results
Participants
In total, 633 participants fulfilled the study criteria, although one was later excluded because of missing data on premonitory symptoms. The final analysis included 632 participants (563 females, 89%; 69 male, 11%; mean ± SD age: 44.6 ± 12.0 years). Most participants (621; 98.3%) had migraine without aura, 199 (31.5%) had migraine with aura and 381 (60.3%) had chronic migraine. Additional demographic and clinical characteristics are provided in Table 1.
Demographics and clinical characteristics.

Premonitory symptoms
In total, 448 participants (70.9%) reported ≥1 premonitory symptoms, 272 (43.0%) unprompted and 442 (69.9%) when prompted (six reported symptoms unprompted but not when prompted). Participants reported more premonitory symptoms when prompted (mean 3.5; median 2; IQR 0–6) than when unprompted (mean 0.81; median 1; IQR 0–1]; p < 0.001). All individual premonitory symptoms were reported more frequently with prompting than without (p < 0.001) (Table 2). Symptoms categorized as “other” were unspecific, such as hot flushes, general discomfort (not involving the head), trembling sensation and phantosmia, among others.
Relative frequency of reporting premonitory symptoms unprompted or prompted.

All values are reported as n (%), unless otherwise specified.
The three most frequently reported premonitory symptoms without prompting were tiredness (reported by 88 participants; 13.9%), neck pain (reported by 47; 7.4%) and nausea or vomiting (reported by 34; 5.4%). The three most frequently reported symptoms with prompting were tiredness (reported by 252; 39.9%), difficulty concentrating (reported by 221; 35.0%) and neck pain (reported by 210; 33.2%).
A post-hoc analysis revealed no discernible differences in estimated proportion of premonitory symptoms between groups categorized by headache frequency (Table 3).
Proportions reporting premonitory symptoms categorized by headache frequency

CI, confidence interval.
Burden measures
The MIDAS questionnaire was completed by 412 (92.0%) of the 448 participants reporting premonitory symptoms and by 167 (90.8%) of the 184 without. There was no difference between these groups in MIDAS grades (p = 0.59) or MIDAS scores (53, IQR 29.0–95.0 vs. 65, IQR 26.5–120.0, respectively; p = 0.10) (Table 4). There was still no significant association (p = 0.63) after adjusting for monthly migraine days (p < 0.001) and the presence of other chronic pain (p = 0.012). MIDAS scores did not correlate with the number of reported premonitory symptoms, whether unprompted (ρ = –0.05; p = 0.24) or prompted (ρ = 0.04; p = 0.40).
Premonitory symptoms and migraine-attributed burden.
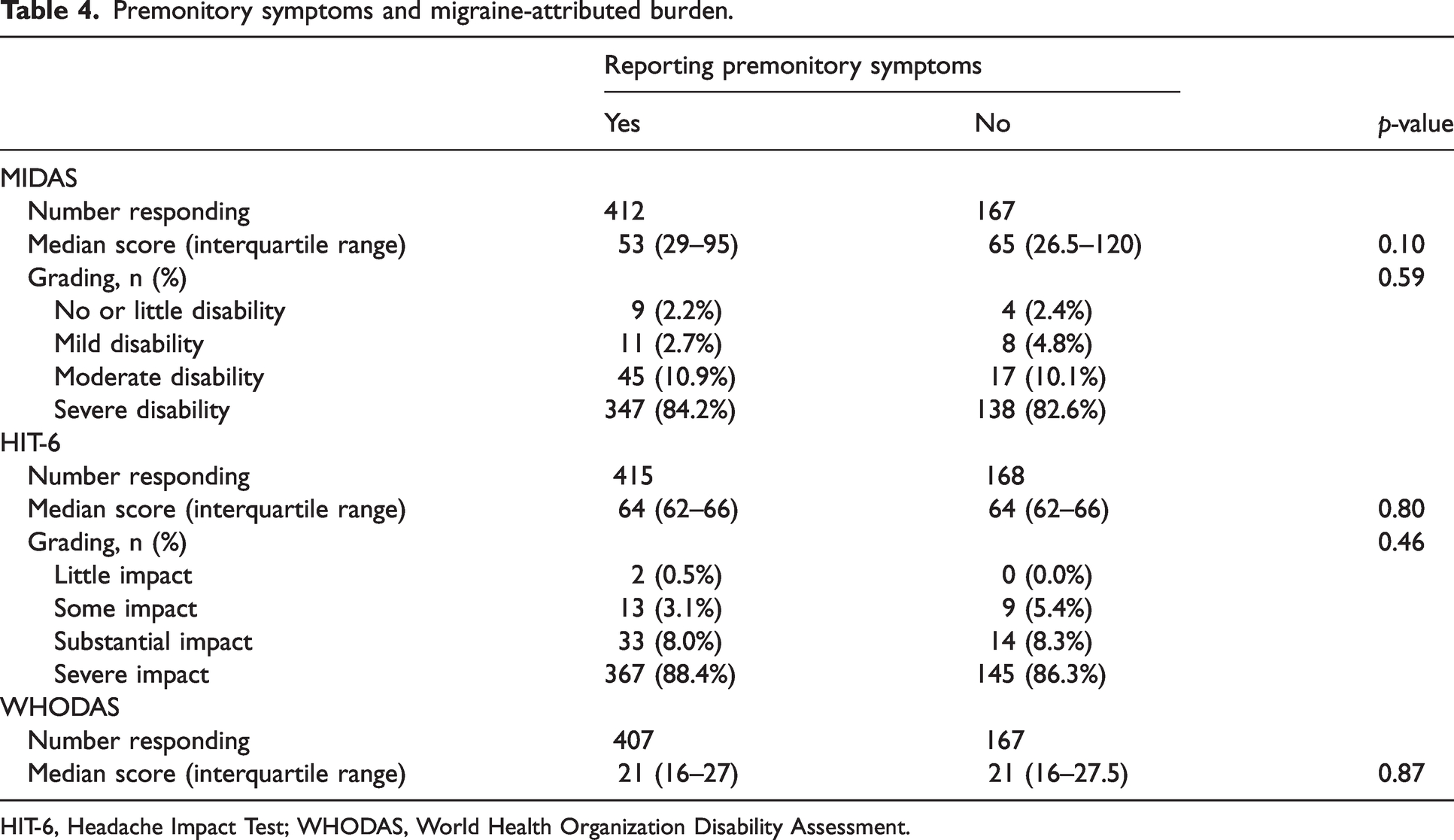
HIT-6, Headache Impact Test; WHODAS, World Health Organization Disability Assessment.
The HIT-6 questionnaire was completed by 415 [92.6%] of the 448 participants with premonitory symptoms and by 168 (91.3%) of the 184 without. There were no differences in mean HIT-6 scores (64, IQR 62–66 vs. 64, IQR 62–66, respectively; p = 0.80) or grades (p = 0.46) (Table 4) between those reporting premonitory symptoms and those who did not. This remained the case (p = 0.22) after adjusting for monthly headache days (p < 0.001) and other chronic pain conditions (p = 0.51). HIT-6 scores were very weakly correlated with the number of reported premonitory symptoms when prompted (ρ = 0.14; p < 0.001) but not when unprompted (ρ = 0.01; p = 0.81).
WHODAS was completed by 407 (90.8%) of the 448 participants with premonitory symptoms and by 167 (90.8%) of the 184 without. There was no difference in mean scores between those with and without symptoms (21, IQR 16–27 vs. 21, IQR 16–27.5, respectively; p = 0.87) (Table 4). Again, this remained so (p = 0.34) after controlling for monthly headache days (p < 0.001), other chronic pain conditions (p < 0.001), ongoing anxiety (p = 0.18) and ongoing depression (p < 0.001). WHODAS scores were very weakly correlated with the number of premonitory symptoms reported when prompted (ρ = 0.09; p = 0.041), but not unprompted (ρ = –0.032; p = 0.44).
Associated factors
In a simple logistic regression, both the reporting of postdromal symptoms (OR = 10.66, 95% CI = 6.83–16.97; p < 0.001) and the reporting of trigger factors (OR = 3.27, 95% CI = 1.46–7.51; p = 0.004) were associated with increased likelihood of reporting premonitory symptoms (Table 5). However, this likelihood decreased with increasing monthly migraine days (OR = 0.93, 95% CI = 0.91–0.95; p < 0.001). Other factors such as age, gender, migraine type, presence of medication overuse and current use of preventative treatment showed no associations with premonitory symptoms (p > 0.05).
Reporting of premonitory symptoms and associated factors.

Significant following Bonferroni correction. CI, confidence interval.
Binominal logistic regression analysis confirmed that participants reporting postdromal symptoms (OR = 9.14, 95% CI = 5.77–14.74; p < 0.001) or trigger factors (OR = 3.73, 95% CI = 1.48–9.34; p = 0.005) had increased likelihood of reporting premonitory symptoms (Figure 2 and Table 5), and that likelihood decreased with increasing monthly migraine days (OR = 0.95, 95% CI = 0.92–0.98; p < 0.001). These factors remained significant after Bonferroni correction.

Associated factors that impact the odds of experiencing premonitory symptoms.
Discussion
The present study found that a high proportion (43–70%) of tertiary clinic patients with migraine reported premonitory symptoms, at the same time as revealing a strong influence of method of enquiry: prompting increased reporting likelihood by almost two-thirds. The findings align well both with previous research on influence of methodology in data collection (24,25) and with a recent meta-analysis (3) in which, in clinic-based studies utilizing predefined lists of 7–29 symptoms and varying assessment methods (8,9,18,19,26–30), the pooled probability of reported premonitory symptoms was 66% (95% CI = 45%–82%) (3).
Methodological issues
What, then, is the optimal method for identifying premonitory symptoms? The meta-analysis suggested using a predefined list of common symptoms, along with dummy questions, to supplement unprompted enquiry (3). Predefined lists encourage acquiescence bias (false positives), at the same time as possibly discouraging the reporting of true symptoms not included in the lists (false negatives) (11). Conversely, in an unprompted enquiry, respondents may neglect symptoms through lack of understanding or awareness (false negatives). Although a combination of methods appears preferable, it is not clear how discrepancies between responses to unprompted and prompted enquiries should be resolved.
An important methodological issue is that of the definition of premonitory phase. In the present study, we used the definition from ICHD-2 and ICHD-3β, which specifies occurrence 2–48 hours before the onset of aura in migraine with aura or of headache in migraine without aura (16,17). The later definition from ICHD-3 (2) omits the lower limit of two hours. This limit, although perhaps arbitrary, appears necessary to distinguish premonitory from ictal symptoms because nausea, photophobia and phonophobia (prominent as ictal symptoms) are among those reported, perhaps questionably, as premonitory. Equally necessary is distinction between premonitory symptoms and postdromal in individuals with high-frequency headache, in whom the latter can run into the former. This might explain why no evident trend in the proportions reporting premonitory symptoms were seen among our participants across the range of headache frequencies (Table 3). Thus, our findings show empirically that ICHD-3 definitions, allowing up to 48 hours in duration for both premonitory and postdromal symptoms, are problematic when migraine attacks are frequent: neither can be reliably assessed when there are fewer than five headache-free days between succeeding episodes. These definitions favour sensitivity over specificity, in contrast to the approach that ICHD adopts generally in the formulation of diagnostic criteria (2). They need to be reconsidered, but better empirical evidence is required before this is carried out.
Premonitory symptoms and disease-attributed burden
We found that the mere presence of premonitory symptoms had no significant impact on MIDAS score or grade, HIT-6 score or grade or WHODAS score. However, the number of prompted premonitory symptoms correlated positively but weakly with both HIT-6 and WHODAS scores, but not MIDAS scores. Although these findings may suggest that a multiplicity of premonitory symptoms can increase migraine-attributed burden, they are not convincing.
Few previous studies have investigated potential associations between premonitory symptoms and patient-reported burden measures. One prospective study found, as we did, no clear association between number of symptoms and MIDAS grades (27). A retrospective cohort study did, however, report a weak correlation between number of symptoms and MIDAS scores (18). Additionally, a recent cross-sectional, web-based survey reported that the presence of premonitory symptoms is related to higher rates of moderate-to-severe disability based on MIDAS (31). These findings are not comparable to the present study as a result of significant differences in the definition of premonitory symptoms, study design and study population.
More detailed studies are needed that take better account of the nature and severity of individual specific symptoms because some are more likely to be considered burdensome than others. An urge to yawn or food cravings may be no more than irritating, whereas feeling tired or difficulty concentrating may impair abilities. However, for most patients, we suspect ictal symptoms wholly overshadow premonitory symptoms.
Premonitory symptoms and associated factors
In the present study, reporting of postdromal symptoms and of trigger factors were each associated with increased likelihood of reporting premonitory symptoms, whereas a higher number of monthly migraine days showed a negative association. Premonitory symptoms, postdromal symptoms and trigger factors were all assessed in similar manner, and so confounders are possible. Common susceptibility may be present, but there is also a behavioral element in reporting that may have similar influence across symptom types and trigger factors. Previous studies have reported co-occurrences of premonitory and postdromal symptoms (20,32,33). It has been suggested that premonitory symptoms could persist through the headache phase and be reported also as postdromal symptoms (20,33), which, perhaps percipiently, throws into question the definitions and mechanistic bases of each. Other studies have reported associations between trigger factors and premonitory symptoms (18,32,34). Caution is needed in interpreting these findings: the co-occurrence of some symptoms and triggers may be the result of a lower threshold or increased sensitivity for a given trigger during the premonitory phase (28,32). However, it has been suggested that triggers are not actual triggers, but instead are symptoms caused by abnormal brain activity, leading to the perception of triggers or premonitory symptoms (1). The negative correlation between monthly migraine days and presence of premonitory symptoms does not appear to be unexpected: more migraine days, with more time in the ictal state, lessen the opportunity to experience, recognize and attach importance to premonitory (and postdromal) symptoms. Conflicting data have been suggested among children, although no correlation was found between placebo-triggered premonitory symptoms and baseline headache days in another study (35,36). Differences in study population and setting may be the cause of the inconsistent findings, although migraine days may affect premonitory symptoms differently compared to headache days (35).
Limitations
There were several limitations to the present study. Among the most important was its dependence on retrospective assessment of premonitory symptoms, with potential recall error. One study reported that participants retrospectively estimated that 90% of attacks were preceded by premonitory symptoms, but, prospectively, only 72.1% were (this was not statistically tested) (9). Similarly important was the inclusion of many participants (over 60%) with high-frequency headache, introducing the problems that have been discussed. This was a consequence of recruiting predominantly from a tertiary care center, with patients at the high end of the migraine-severity spectrum, itself a limitation because the findings are not generalizable to patients more broadly, let alone the general population of people with migraine. The use of preventive treatment might also have impacted the reporting of premonitory symptoms (37). Our prompting list of premonitory symptoms did not include any “dummy” responses, which might have aided recognition of acquiescence bias. Our list of symptoms consisted of 16 individual symptoms, even though 96 individual symptoms have been reported (3), which results in missing symptoms and lower response. We did not use the most recent (ICHD-3) definition of premonitory symptoms (2), although, for reasons already discussed, this was not necessarily a limitation since it better enabled distinction of premonitory symptoms from those of the headache and aura phases. However, it did mean that symptoms appearing within two hours before aura or headache onset (if these should be regarded as premonitory) would not have been registered. Lastly, we did not take any account of time of symptom from last headache, with the possibility (especially among those with frequent headache) that postdromal symptoms were reported as premonitory.
Future directions
The present study highlights the importance of standardized methods in assessing premonitory symptoms. We recommend combining methods (unprompted and prompted enquiries). Prospective, electronic diary- and population-based studies, with time-stamped entries, may eliminate recall error and acquiescence bias. Collecting prospective data will elucidate the predictability of premonitory symptoms (38,39), which might be relevant to use in the definition of premonitory symptoms. Future studies assessing both triggers, premonitory and postdromal symptoms in the same participants, as well as into symptom persistence throughout an attack, may provide insights into the underlying mechanisms and the brain structures involved during the different phases. However, the definition of premonitory symptoms may first need review, although future research taking account of number of migraine days and other time factors may necessitate its further reconsideration. If lists are to be used, once should also consider that some symptoms may be related such as yawning and tiredness or irritability and being emotional.
Because this study suggested (albeit very tentatively) a possible contribution of premonitory symptoms to disease-attributed burden, future studies (ideally prospective and population-based) should specifically enquire into this, taking account of nature, severity and duration of specific premonitory symptoms.
Conclusions
Premonitory symptoms, reported by a high proportion of tertiary-clinic patients with migraine, may be a common feature of migraine, although only population-based studies can confirm this. The method of enquiry has a strong influence on reporting probability, so standardized methods, as well as a clear definition, are required for future studies. These should look further into whether premonitory symptoms can contribute to migraine-attributed burden, taking account of number of symptoms and their individual nature and severity. Future studies should also consider how the probability of reporting premonitory symptoms varies with (and may be influenced by) the simultaneous reporting of postdromal symptoms or trigger factors.
Clinical implications
The estimated proportion of patients reporting premonitory symptoms depends on the assessment method, and therefore standardized methods are needed. The impact of premonitory symptoms on migraine-attributed burden has previously been overlooked, although our findings suggest it is of less importance, relative to the impact of headache, both in clinical practice and to public health.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231223979 - Supplemental material for Premonitory symptoms in migraine: A REFORM Study
Supplemental material, sj-pdf-1-cep-10.1177_03331024231223979 for Premonitory symptoms in migraine: A REFORM Study by Janu Thuraiaiyah, Håkan Ashina, Rune H Christensen, Haidar M Al-Khazali, Astrid Wiggers, Faisal Mohammad Amin, Timothy J Steiner and Messoud Ashina in Cephalalgia
Footnotes
Acknowledgments
We express our gratitude to the following investigators and staff members of the Danish Headache Center for their invaluable contributions to this study: Sub-investigators: Andreas Vinther Thomsen, Betel Tesfay, Christopher Kjær Cullum, Lili Kokoti, Nadja Bredo Rasmussen, Navid Noory, Rogelio Domínguez-Moreno, Thien Phu Do, William Kristian Karlsson and Zixuan Alice Zhuang. Clinical research nurses: Anne Mette Autzen, Susanne Leed and Marianne Hestad. Medical secretaries: Ane Lundgaard Dahl, Dianna Bartolin Christiansen and Pia Frydendall. Research administrator: Kateryna Kolkova. Medical students: Amanda Poulsen, Amenah Ayyoub, Amir Al-Saoudi, Emil Gazolov, Johanne Gry Larsen, Kathrine Rose, Mikkel Johannes Henningsen, Mohammed Bakir Ahmad Lafta, Sarra Al-Khazali, Sarah Hugger and Shan Elahi Goandal. Biostatistician: Yi Xiao (Affiliation: Department of Populations Sciences, City of Hope National Medical Center, Duarte, CA, USA). We greatly appreciate their dedicated efforts and expertise, which were instrumental to the success of this study.
Author contributions
MA, FMA and HA were responsible for the study concept and design. JT, RHC, HMA, AW, HA, FMA, TJS and MA were responsible for acquisition, analysis or interpretation of data. JT, RHC, HMA, AW, HA and MA were responsible for drafting the manuscript. JT, HMA, AW, RHC, HA, FMA, TJS and MA were responsible for critical revision of the manuscript for important intellectual content. JT was responsible for statistical analysis. MA, FMA and HA were responsible for administrative, technical or material support. MA, FMA and HA were responsible for supervision.
Data availability
The dataset used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
JT, RHC, and AW report no conflict of interest. HA reports personal fees from Teva and Lundbeck, outside of the submitted work. HMA reports personal fees from Pfizer, outside of the submitted work. FMA has received personal fees from Pfizer, Teva, Lundbeck, Novartis, Eli Lilly, outside of the submitted work. TJS is co-editor of The Journal of Headache and Pain. MA reports receiving personal fees from AbbVie, Amgen, Eli Lilly, Lundbeck, Novartis, Pfizer and Teva Pharmaceuticals outside of the submitted work. MA has received institutional grants from Lundbeck Foundation, Novo Nordisk Foundation and Novartis. MA reports serving as associate editor of Cephalalgia, associate editor of The Journal of Headache and Pain, and associate editor of Brain.
Ethical statement
The parental study, REFORM, was approved by the Committees on Health Research Ethics in the Capital Region of Denmark and conducted in accordance with the Declaration of Helsinki. Additional approval for using data from the interview and questionnaires was not needed according to this committee. All participants provided informed consent prior to inclusion and participation.
Funding
This study received funding from the Lundbeck Foundation professor grant (R310-2018-3711).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
