Abstract
Introduction
Migraine is a common multifactorial disease (1,2). Several different processes are involved in its pathophysiology, such as alteration of pain and sensory input, increased sensitivity of the cortex leading to aura, central pain facilitation, neurogenic inflammation and brain stem nociceptor sensitization (3,4).
BDNF (brain-derived neurotrophic factor) is important for neuronal growth, development and survival in the central nervous system, and is the most abundant neurotrophin in the brain (5,6). BDNF is expressed in the nociceptive sensory neurons, acting as a central pain modulator at both the spinal and supra-spinal level, contributing to central sensitization and modulating the activation of glutamatergic, N-methyl-d-aspartate (NMDA) receptors (7–9). A significant decrease in the levels of BDNF in platelets was found in migraineurs, showing that it may also have a role in migraine pathophysiology (8). One of the mechanisms involved in migraine is central sensitization, characterized by activity-dependent plastic changes in second-order trigeminal neurons (10). BDNF has been pointed out as a mediator of the trigeminal nociceptive plasticity (9,11). A previous study evaluated several genes that can be expressed after cortical spreading depression (CSD), the mechanism hypothesized to underlie migraine pathophysiology, especially migraine with aura (MA) (10,12).
Several studies showed that neurogenic inflammation is associated with pathogenesis of migraine (13). When neuronal excitability occurs, leading to primary brain dysfunction, the perivascular trigeminal sensory nerve fibers are activated, resulting in the release of vasoactive peptides, such as substance P and calcitonin-gene related peptide (CGRP), from trigeminal fibers (13). CGRP is the most abundant neuropeptide in perivascular sensory trigeminal nerve fibers and one of the mediators of neurogenic inflammation; it is a potent vasodilator (14,15), enhancing the activation of second-order neurons, thus contributing to pain transmission (13,16). CGRP has an active role in the activation and sensitization of nociceptors at the peripheral and central level (17), modulating nociceptive transmission in the trigeminovascular system (15).
The involvement of CGRP in migraine pathophysiology has been supported by the finding that the serum levels of CGRP are increased during migraine attacks and there is a decrease after treatment with triptans, although this was not observed in a more recent study (18–21).
CGRP is co-expressed with BDNF in trigeminal ganglion neurons in rats. In addition, CGRP enhances BDNF release from trigeminal neurons in vitro; this is independent of extracellular calcium and is abolished by a CGRP-receptor antagonist (11).
A case-control study found no evidence of a
Subjects and methods
Subjects
A sample of 188 unrelated migraine patients from the neurology clinic, at (Hospital de Santo António) HSA, Porto, was sequentially enrolled in this study. Patients with familial hemiplegic migraine were excluded. Control subjects (287), with no personal history of migraine, were ascertained among healthy blood donors and from the Obstetrics and Gynecology Department of HSA. Controls were from the same ethnic and geographical origin (north of Portugal) as cases, and were age-matched to these. A diagnostic interview was performed both in cases and controls, based on the operational criteria of the International Headache Society (IHS), using the same structured questionnaire. The first edition of these criteria (ICHD-I) (23) was used before 2004; revising the diagnosis using the second edition (ICHD-II) (24) showed no differences in patients’ diagnosis (data not shown). Participants gave their written informed consent and the project was approved by the Ethics Committee of HSA.
Genotyping
Genomic DNA was extracted from peripheral blood leucocytes, using a standard salting-out method (25); or from saliva using ORAGENE kits and DNA extraction according to the manufacturer’s instructions (DNA Genotek, Kanata, Ontario, Canada).
SNPs were selected based on a data dump from the International HapMap Project (Release 24, November 2008, on NCBI B36 assembly, dbSNP build 126) (www.hapmap.org) and tagging SNPs were selected using Haploview 4.1 (Broad Institute, Cambridge, MA, USA) (26), capturing all the variation for both genes studied at an r2 threshold of 0.80 and with a minor allele frequency (MAF) of 0.10, by an aggressive tagging approach. For
Allelic discrimination was performed using Real-Time PCR (iQ5 Real-Time PCR Detection System, Bio-Rad Laboratories). Primers and molecular beacons were designed using Beacon Designer 6.0.
For
Primer and probe sequences are available upon request. Cycling conditions used were: an initial denaturing at 95°C, for 3 minutes and 30 seconds, followed by 35 cycles of 30 seconds, at 95°C, 60 seconds at 60°C (for annealing, and at this point fluorescence was measured), and 30 seconds at 72°C. Genotypes were determined automatically by the signal processing algorithms of the iQ5 Optical System Software, version 2.0 (Bio-Rad Laboratories); results were compared with the genotypes visually scored, based on the fluorescent emission data depicted (relative fluorescent units [RFUs]). To validate the Real-Time PCR reaction, we included three individuals previously genotyped by sequencing (a heterozygote, a homozygote for variant 1 and a homozygote for variant 2). If genotypes were uncertain, they were additionally sequenced, using the Big Dye Terminator Cycle Sequencing v1.1 Ready Reaction (Applied Biosystems), according to the manufacturer’s instructions, and an ABI-PRISM 3130 XL genetic analyzer (Applied Biosystems).
Statistical analysis
The Genetic Power Calculator (http://pngu.mgh.harvard.edu/∼purcell/gpc/) was used, assuming a high-risk allele frequency of 0.1, a relative risk for a homozygous genotype of 2.25 and 1.5 in heterozygosity. Hardy-Weinberg equilibrium was tested, using HWE software (http://linkage.rockefeller.edu/ott/linkutil.htm). Demographic data of patients and controls were compared using a chi-square test for qualitative variables. To compare allele frequencies between cases and controls, a chi-square test was used and odds ratios (OR) were estimated, with 95% confidence intervals (CIs). Our initial significance level was set at α = 0.01.
A multivariable-logistic regression was performed (with the most frequent homozygote as the reference), to evaluate association between SNPs and migraine, including the four SNPs of
Gene-gene interactions were also evaluated using multifactor dimensionality reduction (MDR) software (version 2.0) (27,28). MDR is a non-parametric and genetic model-free approach that can identify combination of SNPs involved in disease susceptibility (27). We performed a MDR analysis of the rs2049046*rs1553005 interaction using a forced approach. By using this forced analysis, we evaluated a specific set of attributes and we obtained an unbiased estimate of the testing accuracy. We used a tenfold cross-validation to avoid false-positives (29). The significant results obtained were corrected for multiple testing using the permutation test implemented on the MDR Permutation Tool (version 1.0) (30).
Haplotype frequencies were compared between cases and controls, for the
Results
Demographic and clinical data of migraine patients and controls

SD = standard deviation.
A migraine prevalence of 16% has been estimated in a Portuguese population (31,32). We found that we had a power of 64% to detect an association with our sample (for a nominal significance level of 0.05).
Both case and control groups were in Hardy-Weinberg (H-W) equilibrium for the four SNPs of the
Allele frequencies of SNPs studied in migraine patients and controls

SNP = single nucleotide polymorphism.
Results from the multivariable logistic regression, gender-adjusted

(ref) = reference category. OR = odds ratio. CI = confidence interval.
Interaction results found between
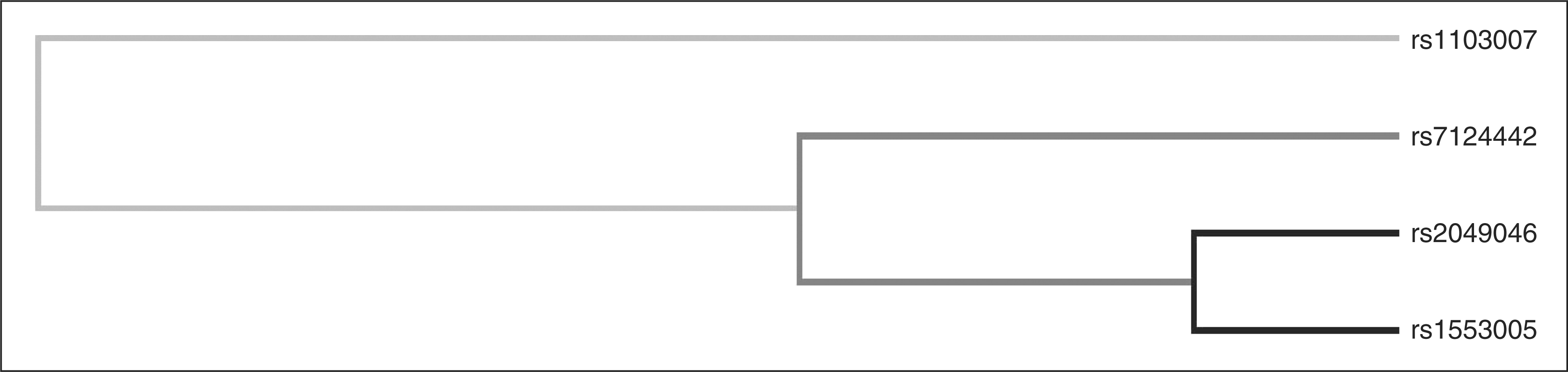
With the MDR analysis, we found that the best model for the interaction between rs2049046 and rs1553005 showed a testing balanced accuracy of 0.53 and a cross-validation consistency of 10/10 (Table 5). After permutation testing, this model was still significant ( Interaction graphic for rs2049046 and rs1553005. High-risk combinations are depicted as dark-shaded cells, and low-risk combinations as light-shaded cells. For each cell the left bar indicates the cases and the right bar indicate the controls. Interaction dendogram for the SNPs genotyped. The dendogram clearly shows a strong interaction effect between rs2049046 and rs1553005 (darker lines suggest that there is a synergistic relationship: the shorter the lines, the stronger the interaction). SNPs = single nucleotide polymorphisms. Interaction results found between


For
Discussion
The aim of this study was to assess the possible involvement of
For
For
We have also assessed a possible interaction between
A significant interaction between rs2049046 and rs1553005 was found, showing an increased risk for the double heterozygotes AT and GC (OR = 1.88, 95% CI: 1.20–2.93) when compared with the most frequent double homozygotes (AA of rs2049046 and GG of rs1553005).
However, logistic regression parameters may not be completely reliable due to the limited number of subjects in each group (34).Therefore, we employed a MDR analysis. This method combines genotypes into a single dimension with two groups (high or low risk). MDR is also able to detect a high-order interaction in the absence of any statistically significant main effects. The combination of cross-validation and permutation testing minimizes the occurrence of false positive results (27). Furthermore, MDR is able to detect gene-gene interactions even in small sample sizes (35), which gives us confidence in our results. With the MDR analysis we also found a highly significant two-way interaction between those SNPs associated with an increased risk for migraine, confirming the results of the logistic regression analysis. Therefore, an interaction between these variants, or between variants in LD with them, may be involved in migraine susceptibility.
Our sample size is a limitation of our study and does not give us enough power to detect a variant with an OR < 1.5. Also, rare alleles may also be involved in the susceptibility of migraine (36), although we were interested in studying the role of common variants (MAF > 0.10). Therefore, we could not exclude that these genes may be independent risk factors for migraine, although this was not detected in our sample. However, some gene-gene interactions occur even without the presence of a significant main effect (37) and future association studies should take this into account.
Nonetheless, for common variation, we covered the whole gene region with the SNPs studied for both genes. Importantly, our high case:control ratio increases the power to detect any effects in our sample. In addition, cases and controls were matched for age-at-observation and gender, and were from the same population and geographic region; several studies using lineage markers, sensitive to population stratification, have showed there is no substructure within the Portuguese population (38–40). Also, we corrected for multiple testing, to prevent type-I errors, and used logistic regression analyses to incorporate all SNPs in the model and analyze their effects together. Additionally, we set our initial level of significance at
Several findings support a role of CGRP in initiating and sustaining migraine episodes (42). CGRP has been extensively studied regarding its possible role in migraine treatment. Antagonists for CGRP receptors may act at the central level and modulate nociceptive trigeminovascular transmission in the cat, and are arising as a new generation of migraine drugs (42–44). Furthermore, studies in transgenic mice showed that modulation of one of the CGRP receptors (RAMP1) levels may contribute to migraine susceptibility and, in particular, to photophobia (45). More interestingly, a recent study showed that adenosine triphosphate (ATP)-gated P2X3 receptors may be involved in chronic pain (including migraine) and CGRP seems to enhance its transcription, involving also mediation by BDNF (46). BDNF is also a mediator of neuronal survival and plasticity of dopaminergic, cholinergic and serotonergic neurons, and its involvement in migraine seems clear as a mediator of the trigeminal nociceptive plasticity (9,11,47).
Our data support the hypothesis of an interaction between
Footnotes
Acknowledgements
We would like to thank all patients and controls for participating in this study. We thank also Dr Serafim Guimarães and nurses Teresa Gomes and Palmira Gouveia for their assistance with sample collection, Paula Magalhães for technical assistance and Jorge Pinto-Basto and Zoltán Bochdanovits for their helpful suggestions.
This study was supported in part by grants of Fundação para a Ciência e Tecnologia (FCT) (POCTI-034390/99/FCT), Sociedade Portuguesa de Cefaleias (SPC) and the National Headache Foundation (NHF) (USA). CL was the recipient of an FCT fellowship (SFRH/BD/17761/2004). The authors report no conflicts of interest.
