Abstract
Abstract
Background
The present study investigates the wearing-off effect in adults with chronic migraine treated with erenumab or fremanezumab.
Methods
This real-world observational study was based on pre-collected headache diaries from chronic migraine patients in treatment with either monthly injections of 140 mg of erenumab or 225 mg of fremanezumab. Consistent wearing-off was defined as an increase of ≥2 weekly migraine days in the last week compared to the second week over two consecutive 4-week treatment periods. The primary endpoint was wearing-off in the total population. The secondary endpoints were difference in wearing-off in (i) a subgroup of patients treated with erenumab and fremanezumab and (ii) consistent wearing-off in patients with a ≥30% reduction in monthly migraine days, compared to baseline, in the two consecutive treatment months.
Results
In total, 100 patients (erenumab: n = 60, fremanezumab: n = 40) were included. Sixty-two out of 100 (62%) patients had consistent ≥30% treatment response on antibody therapy in both months (erenumab: n = 36, fremanezumab: n = 26). There was no consistent wearing-off over the two consecutive months from week 2 to week 4 (3.04%, p = 0.558). There was no wearing-off within the erenumab (p = 0.194) or the fremanezumab (p = 0.581) groups. Among the ≥30% treatment responders, there was no consistent wearing-off over the two consecutive months (2.6%, p = 0.573).
Conclusions
There was no wearing-off in treatment responders, which is in alignment with premarketing data from placebo-controlled phase III studies. These data suggest that patients should be informed upfront that no wearing-off effect is expected because anxiety for attacks at the end of the month per se may generate migraine attacks.
Introduction
Clinical trial studies investigating the efficacy of monoclonal antibodies against calcitonin gene-related peptide (CGRP) or its receptor have shown promising results (1–3). The neuropeptide CGRP is deeply involved in the pathophysiology of migraine. Intravenous infusion of CGRP in migraine patients promptly causes eruptions of migraine attacks, whereas blocking CGRP signaling prevents migraine attacks (4). Monoclonal antibodies against CGRP (i.e. fremanezumab, galcanezumab and eptinezumab) or its receptor (i.e. erenumab) are treatment options used to block CGRP signaling (5). Real-world evidence studies of erenumab (6) and fremanezumab (7), administered subcutaneously every 28–30 days, reported efficacy and tolerability in patients with chronic migraine and ≥2 previous preventive medication failures.
In the wake of the increasing implementation of antibody treatment, a question has arisen about whether the effect lasts for one month, or whether a more frequent treatment is necessary. Only sparse or no wearing-off data are available from real-world evidence studies of erenumab and fremanezumab. The present study aimed to investigate consistent wearing-off effect within treatment months in routine clinical use of monoclonal antibodies. We hypothesized no consistent wearing-off effect in chronic migraine patients treated with monthly injections of erenumab or fremanezumab.
Methods
Standard protocol approvals, registrations and patient consent
The study was a single-center, observational study of erenumab or fremanezumab for adults with chronic migraine. The study protocol was approved by the ethics committee of the Capital Region of Denmark (J-19085557) and the Danish Data Protection Agency. This study is a part of the larger parental CGRP Antibody Registry study and registry of adults with migraine undergoing treatment with medications targeting CGRP-signaling in Denmark (6). All enrolled patients provided their written informed consent before any study-related tasks or procedures were performed. The study was conducted in accordance with the principles of the Declaration of Helsinki.
Study population
Adult patients with a diagnosis of chronic migraine according to the International Classification of Headache Disorders treated with monoclonal antibodies against CGRP signaling (i.e. erenumab and fremanezumab) were eligible for inclusion in the present study (8,9).
The local clinical treatment guidelines were: a diagnosis of chronic migraine; age ≥18 years; willingness to keep a headache diary at least one month prior to treatment start and throughout the treatment period; previous lack of efficacy or tolerability to at least one antihypertensive and one anticonvulsant medication approved for migraine prevention. Exclusion criteria were active medication overuse headache and pregnancy (8,9). Because the treatment was fully reimbursed, patients had to record daily migraine and headache entries in a headache diary. Patients without a recorded headache diary were excluded as well. Patients were not started on other preventive treatments during the treatment period.
Diaries were presented at follow-up visits every three months to demonstrate a ≥30% reduction in monthly migraine days from baseline in the larger parental study. According to local practice guidelines, patients were excluded from treatment if they did not achieve a ≥30% reduction in monthly migraine days at each assessment period.
Data extraction
We reviewed the medical records of 350 consecutive patients in the parental study database to include a total of 100 patients with available pre-recorded headache diaries. All diaries were electronically available except from 16 patients. These were retrieved by contacting the patients.
Diary entries for two available months without missing data were extracted for analysis. Two months were selected to investigate consistent wearing-off effect. Consistent wearing-off effect was defined as increase in migraine days in the last week of the month (week 4) compared to the second week of the same month (week 2) over two consecutive treatment months. A month was defined as 28 days. Treatment response was defined as a ≥30% reduction in the mean monthly migraine days over the two study months compared to baseline (one month before CGRP treatment) based on clinical guidelines at the center (6). A clinically meaningful difference in migraine days was defined ≥2 days absolute difference in migraine days.
Outcome measures
Primary outcome was defined as the change in weekly migraine days between week 2 and week 4 over two consecutive months. Secondary outcomes were (i) change in migraine days between week 2 and week 4 in the subgroup of patients treated with erenumab and fremanezumab and (ii) change in migraine days between week 2 and week 4 in the subgroup of patients who had ≥30% reduction in monthly migraine days compared to baseline. Explorative analyses were (i) change in migraine days between the average of weeks 1–3 and week 4 and (ii) subgroup analyses in those who were treated with anti-CGRP treatment for at least three months. In further explorative analyses, we assessed the correlation between change in migraine days and response rate to anti-CGRP treatment.
Statistical analysis
Data are reported as the mean ± SEM, unless stated otherwise. Linear mixed models were used for primary and secondary outcome analyses as well as for explorative analyses. Fixed effects were weekly migraine days (week 2 and week 4 or average weeks 1–3 and week 4) and drug (erenumab and fremanezumab) with patient identification nested in months as random effect. A linear mixed model was used to explore the association between change in weekly migraine days and response rate.
Statistical analyses were performed using R, version 4.3.1 (R Foundation, Vienna, Austria). p values were reported as two-tailed with a level of significance of 5%.
Results
In total, 100 chronic migraine patients were included. Fifty-nine patients received monthly subcutaneous injection with erenumab 140 mg, one patient received erenumab 70 mg and 40 patients received fremanezumab 225 mg. Demographic data and baseline characteristics of the study population before anti-CGRP treatment are reported in Table 1. Average days of treatment was 177.4 days (SD: 96; interquartile range: 92–252) at the time of inclusion in this study. Eighty-one patients out of 100 (81%) were treated with erenumab or fremanezumab for ≥3 months at the time of inclusion in the study. The remaining 19 participants were treated for one month (n = 8, all fremanezumab), two months (n = 3, all fremanezumab) or between two and three months (n = 8, all erenumab).
Demographics and baseline characteristics.
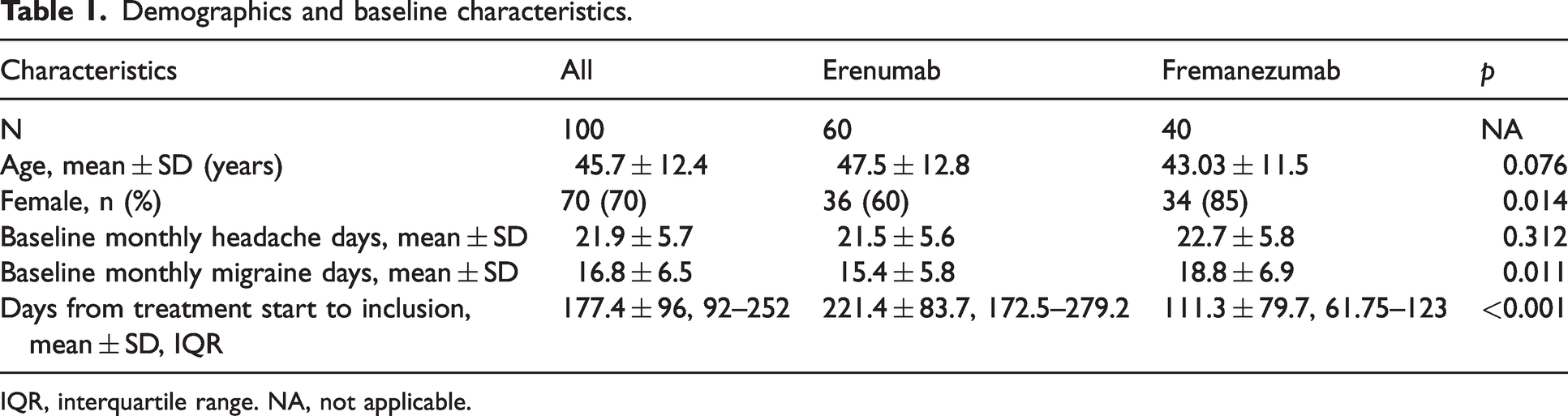
IQR, interquartile range. NA, not applicable.
The average monthly migraine days in the first month after inclusion was 9.03 days (SD: 7.5; interquartile range: 4–11) and in the second month 9.6 days (SD: 7.7; interquartile range: 4–12) with no difference between erenumab and fremanezumab groups (month 1: p = 0.071; month 2: p = 0.105).
Sixty-two out of 100 (62%) patients had consistent ≥30% response on antibody therapy (erenumab: n = 36, fremanezumab: n = 26) in both months.
Wearing-off effect from week 2 to week 4
There was no difference in migraine days between week 2 (2.30 ± 0.15) and week 4 (2.37 ± 0.12) over two consecutive treatment months (3.04%, p = 0.558) (Figure 1). There was no difference in migraine days between week 2 and week 4 in the erenumab (p = 0.194), nor the fremanezumab (p = 0.581) groups. There was no difference between treatment groups (p = 0.218). Explorative analysis showed no association between wearing-off and response rate (i.e. reduction in monthly migraine days) in the total group (p = 0.935) or within the erenumab (p = 0.811) and fremanezumab (p = 0.981) groups.
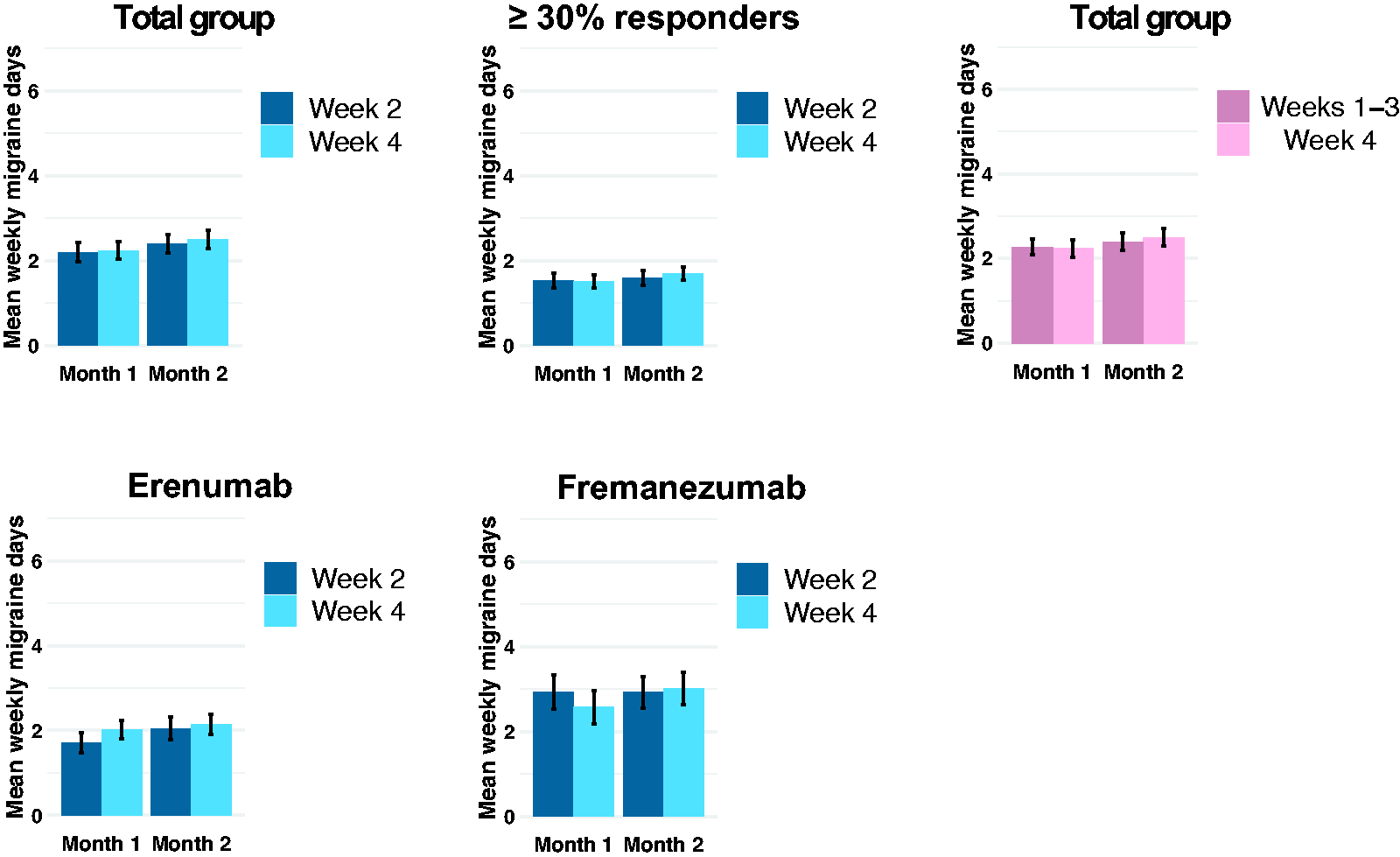
Consistent wearing-off in chronic migraine patients in the total study population (n = 100), erenumab (n = 60) and fremanezumab (n = 40) subgroups as well as patients with ≥30% reduction in monthly migraine days (n = 62). There was no difference in consistent wearing-off in weekly migraine days over two consecutive 4-week treatment periods in all analyses. Error bars denote the SEM.
Seventeen out of 100 (17%) patients reported ≥2 migraine days in week 4 compared to week 2 in the first month, whereas 13 patients (13%) reported ≥2 migraine days in week 4 compared to week 2 in the second month. Three patients (3%) (erenumab: n = 1, fremanezumab: n = 2) reported ≥2 migraine days in week 4 compared to week 2 in both months.
Wearing-off effect from week 2 to week 4 in ≥30% responders
There was no difference in migraine days between week 2 (1.43 ± 0.12) and week 4 (1.52 ± 0.16) over two consecutive treatment months in participants with consistent ≥30% reduction in migraine days in both months (2.6%, p = 0.573). There was no difference in migraine days between week 2 and week 4 within the erenumab (p = 0.102) and fremanezumab (p = 0.612) groups or between the treatment groups (p = 0.190).
Eleven patients of the ≥30% responders (17.7%) reported ≥2 migraine days in week 4 compared to week 2 in the first month and seven patients (11.3%) in the second month. One patient (1.6%) reported ≥2 migraine days in week 4 compared to week 2 in both months and were on fremanezumab.
Wearing-off effect from weeks 1–3 to week 4
There was no difference in weekly migraine days from week 1 to week 3 (2.32 ± 0.14) compared to week 4 (2.37 ± 0.9) (2.2%, p = 0.582). There was no difference within the erenumab (p = 0.485) nor the fremanezumab (p = 0.978) groups. There was no difference between the treatment groups (p = 0.680). There was no difference in weekly migraine days from week 1 to week 3 (1.39 ± 0.10) compared to week 4 (1.52 ± 0.12) in those who were ≥30% responders (9.4%, p = 0.264). Explorative analysis showed no association between response rate and wearing-off effect from week 1 to week 3 compared to week 4 in the total group (p = 0.626) or within the erenumab (p = 0.634) and fremanezumab (p = 0.902) groups.
Nine of 100 (9%) patients reported ≥2 migraine days in week 4 compared to weeks 1–3 in the first month, whereas six patients (6%) reported ≥2 migraine days in week 4 compared to weeks 1–3 in the second month. One patient (1%) on fremanezumab reported ≥2 migraine days in week 4 compared to weeks 1–3 in both months.
Five patients of the consistent ≥30% responders (8.1%) reported ≥2 weekly migraine days in week 4 compared to weeks 1–3 in the first month and two patients (3.2%) in the second month, whereas no patient reported ≥2 weekly migraine days reduction in both months.
Wearing-off effect in patients with ≥3 months treatment
There was no difference in migraine days between week 2 (1.91 ± 0.14) and week 4 (2.06 ± 0.14) (7.9%, p = 0.271) or within the erenumab (p = 0.107) and fremanezumab (p = 0.951) groups. There was no difference in migraine days from weeks 1–3 (1.93 ± 0.13) to week 4 (2.06 ± 0.10) (6.7%, p = 0.191) in the subgroup or within the erenumab (p = 0.237) and fremanezumab (p = 0.534) groups. There was no association of response rate and wearing-off between week 2 and week 4 (p = 0.610) or from weeks 1–3 to week 4 (p = 0.882). In patients with ≥30% reduction in monthly migraine days (n = 57), there was no difference between week 2 (1.51 ± 0.13) and week 4 (p = 1.59 ± 0.17) (5.3%, p = 0.640) or from weeks 1–3 (1.47 ± 0.11) to week 4 (1.59 ± 0.12) (8.2%, p = 0.317).
Discussion
We present a real-world evidence study investigating the wearing-off effect in chronic migraine patients treated with monthly administrations of subcutaneous erenumab or fremanezumab. The main finding of this study was that we found no consistent wearing-off effect of treatment with erenumab or fremanezumab in patients with chronic migraine. There was no difference between erenumab and fremanezumab.
Single-month versus consistent wearing-off
Consistency is important when examining wearing-off effects in patients. Increase in migraine days in the last week of the treatment month may be a coincidence or attributed to random causes (e.g. common upper respiratory tract infection, sleep disturbance or menstruation). However, if the patient experiences an increase in migraine days in the last week of the month in two or more consecutive treatment periods, random causes are less likely, and the increase may represent a true wearing-off effect. Consequently, we included two treatment months in this study.
Agreement between present real-world and previous phase III study data
In the present sample of real-world patients, we found no difference in weekly migraine days between week 2 and week 4, nor between the average of weeks 1–3 and week 4. Our data are in alignment with the most recently published post-hoc analysis data from two premarketing phase III studies of treatment with erenumab (10) and fremanezumab (11). The phase III erenumab study showed no persistent wearing-off from weeks 1–3 to week 4 compared to placebo across at least two months treatment period (in consistent responders to erenumab 140 mg). The wearing-off was assessed as ≥2 migraine days in the last week compared to the average weekly migraine days over the first three weeks of the month. The study showed that merely 4.3% (n = 2) of chronic migraine patients, who were consistent responders (n = 46), reported persistent wearing-off after erenumab in at least two months compared to 3.3% (n = 1) in the placebo group (10). In the present study, one ≥30% responder reported a clinically meaningful wearing-off (≥2 days difference) in week 4 compared to week 2, whereas none were reported in week 4 compared to average of weeks 1–3.
The previous post hoc analysis data from the phase III fremanezumab study also reported no wearing-off at the end of dosing when the average of the first three weeks was compared with week 4 in 223 chronic migraine patients followed over 15 months (11).
The previous post-hoc phase III study of erenumab assessed wearing-off effects in the first three months of treatment while reporting no wearing-off (10). In the present study, we showed that the wearing-off effect is also absent in those treated beyond three months with erenumab or fremanezumab. Although treatment duration was longer in the erenumab than the fremanezumab group, there was no difference in wearing-off in patients who received treatment for at least three months and with no difference between the erenumab and fremanezumab groups.
Patients in the fremamezumab group had a higher number of monthly migraine days at baseline before receiving the treatment compared to the erenumab group. However, there was no difference in mean monthly migraine days between erenumab and fremanezumab groups in the treatment month 1 or month 2 after inclusion in the study. Moreover, the present study showed that there was no association between response rate and wearing-off.
In the current real-world data study, there was no placebo group due to the nature of the study. Previous phase III trial post-hoc erenumab studies showed no difference between wearing-off effects in treatment groups compared to placebo (10). Moreover, variation as a result of inter-subject variability and variability between each treatment month within-subject was accounted for in the analyses. Post-hoc power analysis, based on the within-subject SD from the primary analysis, showed that, in our sample of 100 patients, it would be possible to detect at least a 0.47 absolute difference in weekly migraine days with 95% power and a significance level of 5%. This demonstrates that the applied sample size is sufficient to detect clinically meaningful difference in weekly migraine days, which was defined as two days. It was not possible to collect data for more consecutive months in all patients because the clinic only asked patients to provide headache diaries for the last month prior to a follow-up visit. Despite this, we consider our real-world data to be representative and generalizable because of an excellent alignment with premarketing placebo-controlled data with longer follow-up periods (6).
How to address wearing-off in the clinic
The wearing-off effect is more than an academic interest as patients may ask their clinician about the risk of wearing-off or for more frequent injections. The latter may be the result of a fear of having migraine attacks at the end of the treatment period, especially in those patients who experience a good effect during the treatment period. This anxiety (i.e. nocebo) can by itself generate migraine days in some patients. Therefore, both placebo-controlled and open-label real-world evidence data are important to consistently confirm that there is no reason to suspect wearing-off in patients treated with monthly injections of 140 mg of erenumab or 225 mg of fremanezumab. Patients should be informed about this fact based on available evidence rather than experimenting with shorter injection intervals. Furthermore, it is uncertain whether more frequent treatment will produce the same tolerability.
Conclusions
Present real-world evidence data demonstrate no wearing-off effect in chronic migraine patients treated with monthly injections of erenumab or fremanezumab. These results are in alignment with premarketing placebo-controlled data on erenumab and fremanezumab. All patients should upfront be informed about the no wearing-off effect to avoid potential migraine attacks generated by the nocebo effect.
Clinical implications
Real-world data showed that less than 5% of treatment responders experienced a clinically meaningful and consistent wearing-off effect. There was no difference between erenumab and fremanezumab.
Footnotes
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
AMF, LVL, SY, CKC and BAC have no conflicts of interests to report. TPD reports personal fees from Teva, outside of the submitted work. FMA has received Honoria or personal fees from Pfizer, Teva, Novartis, Lundbeck, AbbVie and Eli Lilly for lecturing or participating in advisory boards; is principal investigator for phase IV trials sponsored by Novartis and by Teva; serves as president of Danish Headache Society and board member of the European Headache Federation; serves as associate editor for Acta Neurologica Scandinavica, Frontiers in Neurology, Frontiers in Pain Research and Headache Medicine; serves as junior associate editor for Cephalalgia and Cephalalgia Reports; and is a member of the editorial board of The Journal of Headache and Pain.
Ethical statement
The study protocol was approved by the ethics committee of the Capital Region of Denmark (J-19085557) and the Danish Data Protection Agency. All enrolled patients provided written informed consent before any study-related tasks or procedures were performed. The study was conducted in accordance with the principles of the Declaration of Helsinki.
Funding
The study was supported by Lundbeck Foundation (R396-2022-301). Funding had no influence on study design, patient inclusion or data interpretation.
