Abstract
Introduction
Headache is a frequent symptom of infections. We aimed to characterize the clinical phenotype and duration of headache attributed to influenza infection.
Methods
Prospective cohort study done in 53 primary care centers between January and April 2023. Patients were included if they had a confirmed influenza diagnosis, were older than 15 years and had a new-onset headache. Patients’ demographics, prior medical history, headache phenotype and duration, associated symptoms and patients’ outcomes were assessed. The International Classification of Headache Disorders criteria for headache attributed to a systemic viral infection, migraine and tension-type headache were assessed.
Results
Of the 478 patients 75 fulfilled eligibility criteria. The mean age was 43, 56% were men, and 27% had a prior headache history. The headache phenotype was a bilateral headache (52%), with frontal topography (48%), pressing quality (61%), moderate intensity, rhinorrhea (79%), nasal congestion (76%), and photophobia (59%). All patients fulfilled headache attributed to acute systemic viral infection criteria, 43% fulfilled migraine criteria and 31% tension-type headache criteria. The median duration of the headache was four (Inter-quartile range: two-six) days.
Conclusion
The clinical phenotype of headache attributed to influenza infection was similar to other infections, with more pronounced cranial autonomic symptoms. The headache was an early symptom and was self-limited within a few days.
Keywords
Introduction
Acute respiratory infections (ARI) account for 46.6% of all incident cases of disease, according to the Global Burden of Disease 2017 data (1). Their global incidence increased by 37% between 1990-2019 (2). The responsible agents include rhinoviruses, coronaviruses, influenza virus and respiratory syncytial virus (3–5). Besides the respiratory symptoms, general symptoms are common, due to the systemic immune response and the rapid spread of the virus across the body (3–5). Headache is one of the most common symptoms, reported by up to 60% of patients (6,7). Indeed, in 2008, the diagnostic criteria of influenza-like illness were modified, and headache became one of the defining general symptoms, together with fever, malaise and/or myalgia (8).
Despite its major importance and high incidence, the clinical phenotype of headache in patients with influenza has not been reported (9). The objectives of the present study were: to characterize the clinical phenotype of headache attributed to influenza infection; to discover the presence of specific and distinctive features; to note its differentiation from other headache disorders based on the presence of red flags; to determine the duration of headache in patients who suffer it as a symptom of influenza infection and; to evaluate the possible predictors of longer duration of headache during the course of influenza infection.
Methods
Study design
The influenCEF study is a large multipurpose study that aims to evaluate 1) the prevalence of headache as a symptom of influenza, 2) the predictors of headache as an influenza symptom, 3) the impact of headache in the prognosis of influenza, 4) the clinical phenotype of the headache, and 5) the duration of the headache during the course of influenza. In the present study, objectives #4 and #5 were addressed. Figure 1 summarizes the study design and the study procedures conducted in the evaluation, first and follow-up visits.

Study design and procedures.
This was an observational analytic study with a prospective cohort design. The full study protocol was registered in ClinicalTrials.gov (NCT: 05704335). The study was reported following the Strengthening in the Reporting in Observational Studies in Epidemiology checklist (10).
Ethical aspects
The study was approved by East Valladolid Ethics Review Board (PI-GR-22-2840). All study participants signed an informed consent form. The study was done in accordance with the national Personal Data Protection Law (3/2018), and in accordance with the principles of the Declaration of Helsinki. All databases were created in a digital format to minimize the carbon print of the study, except for the informed consent forms.
Study setting
The study was carried out in 53 primary care centers in Castile and Leon, Spain, and in the Headache Unit of the Hospital Clínico Universitario de Valladolid, a third-level, university, public, hospital.
Study participants
The study population were patients with headache during the course of influenza infection.
Patients were included if they: 1) Fulfilled the European Parliament and European Council 2008 definition of influenza-like illness criteria (8): Sudden onset of symptoms and at least one of the following four systemic symptoms: fever or feverishness, malaise, headache or myalgia; and at least one of the following three respiratory symptoms: cough, sore throat, shortness of breath. 2) Had a confirmed diagnosis of influenza infection by a real-time polymerase chain reaction test from a nasopharyngeal swab test. 3) Were older than 15 years. This age threshold was pre-specified and pre-existing in the VIGIRA sentinel network and differed from the initially anticipated in the influence protocol (older than 18). 4) Signed informed consent form (ICF) and agreed to participate in the study, by both parents and participants if they were younger than 18 years old. 5) Had a new-onset headache presented during the course of the infection, perceived by the patients as not their usual headache. In the case of patients younger than 18 years, both the participant and the relative signed the ICF.
Patients were excluded if they: 1) Had speech or language disturbance that made them unable to collaborate in the evaluation. 2) Died during the course of the disease. 3) Were not able to participate due to a severe or unstable medical situation. 4) Had cognitive or psychiatric diseases that made clinical evaluation difficult. 5) Declined to participate. 6) Were unreachable.
Selection of participants
Since the season 1996-1997, the Health Sentinel Network of Castile and León performs active influenza surveillance, in collaboration with the National Influenza Surveillance System of Spain, integrated in the European Influenza Surveillance Network (EISN). The Acute Respiratory Infections Comprehensive Surveillance Program (Programa de Vigilancia Integrada de las Infecciones Respiratorias Agudas, VIGIRA), previously known as Influenza Comprehensive Surveillance Program (Programa de Vigilancia Integrada de la Gripe, PVIG) gathers around 120 healthcare providers, including physicians, pediatricians and nurses, to actively monitor the 60,000 population in this area (11).
A sample of patients who visited their primary care setting in one of the 66 sentinel general practitioners or pediatricians due to suspected ARI were tested for influenza. Patients were informed of the study and invited to participate by the VIGIRA healthcare providers. Patients signed the informed consent form prior to any study procedures. Confirmed cases were contacted afterwards by the study physician, and were invited to participate in the study again. For patients who still agreed to participate, the subsequent evaluation was done within 20 days after the start of the symptoms, to confirm the etiology of the infection, allow the responsible primary care physician to communicate the diagnosis and the therapeutic measures to be adopted, and minimize the memory bias that would accompany a more delayed evaluation. The evaluation was done by a trained physician by phone. Afterwards, patients were contacted biweekly by phone until the complete resolution of the symptoms.
Study variables
A structured questionnaire was administered, based on previous studies (12,13). A series of variables were collected, related to patients’ demographics, patients’ prior medical history, headache phenotype, other symptoms, and patients’ outcome.
A. Demographic data: Age in the moment of the infection, sex, ethnicity. Ethnicity was classified according to the Updated Guidance on the Reporting of Race and Ethnicity in Medical and Science Journals (14). Reporting race and ethnicity was mandated by the Regional Health Administration. Individuals participating in the study were categorized as Black, Hispanic or Latino, Caucasian and North African with Arab ancestry. B. Prior medical history: Defined as risk factors or disorders that were present before the influenza infection and still active by the time the patient was infected. These included vascular risk factors, such as hypertension, hyperlipidemia, diabetes, obesity (body mass index (BMI) >30), overweight (BMI 25-30), smoking, alcohol misuse (defined as more than 2/1 drinks per day on average in men/women); medical comorbidities, including cardiological disorders, respiratory, immunological, oncological, or neurological disorders. And prior history of headache disorders, which included primary headache disorders, prior headache in the context of other infections, and family history of headache. In patients with prior history of primary headache disorders, the similarity between the prior headache and the headache experienced during the influenza infection was subjectively rated in a 0–100% scale (0: completely different, 100%: the same). Vaccination status to prevent influenza was also assessed. C. Headache phenotype: The phenotypic characterization evaluated a series of features that included the main characteristics of other primary headache disorders, according to the International Classification of Headache Disorders, 3rd version (ICHD-3) (15), and headache-specific red flags, according to the International Headache Society (IHS) Secondary Headache Special Interest Group (16). In addition, the headache phenotype was classified according to the ICHD-3 (15). The evaluated features included the number of days from symptom onset to headache onset, duration of headache, circadian pattern, association of headache with fever, location (holocranial, unilateral), topography (frontal, temporal, parietal, occipital, vertex, facial, cervical and/or periocular), quality (pressing, stabbing, throbbing, burning, electric (brief electric shock-like), or others), intensity (verbal analogic scale 0–10, 0: no pain, 10: worst possible pain), duration (in days), associated symptoms and features (photophobia, phonophobia, osmophobia, nausea, vomiting, worsening with physical activity, clinophilia, cranial autonomic symptoms). The presence of transient neurological symptoms, such as visual symptoms, sensory symptoms, speech disturbances, weakness or hypoesthesia) was assessed. Change of the headache with orthostatic and postural changes were evaluated. Patients rated subjectively the disability caused by the headache in a 0–100 scale, as the proportion of daily activities that could not be performed, 0 being none and 100 all activities). Presence of headache-related red flags was systematically addressed, including wake-up headache, progressive worsening, resistance to treatment, worst headache ever experienced, sudden onset of the headache, confusion, or impaired awareness (16). The need, type and outcome of acute treatment was evaluated and rated. Response to acute treatment was defined, according to the Guidelines of the IHS for controlled trials of acute treatment of migraine attacks in adults at two hours post-administration. The response was coded as optimal if pain freedom occurred within two hours in more than 75% of uses, positive if it occurred in 50-74% of uses, partial if it occurred in 30-49% and no-response if it occurred in less than 30% of uses (17). D. Patients’ outcome: presence of pneumonia, need for oxygen therapy, and need of hospitalization were assessed.
Data sources/measurement
The necessary information was collected by using a systematic questionnaire, administered by a trained physician. Patients’ electronic health records were also reviewed. The information was anonymously collected in RedCap.
Study period
Patients’ evaluation in primary care and sample collection took place between 3 January (first patient-first visit) and 16 April 2023 (last patient – first visit). Data was collected between 23 January and 4 May 2023 (last patient – last visit).
Bias
Selection bias was considered, and patients were selected from a representative sample of 60,000 people out of the 2,315,572 inhabitants of Castile and Leon region attending the healthcare public system, including both urban and rural areas. Outcome bias was minimized by a systematic and consistent evaluation of all the study participants, done by LSL, with a structured questionnaire. In addition, follow-up was done by a telephonic evaluation, checking whether symptoms persisted or were resolved. The clinical assessment was long enough for the outcome to resolve.
To evaluate whether the outcome of interest was present at the study start, patients’ prior history of headache was systematically assessed and characterized. The ICHD-3 criteria of secondary headache were considered, as a twofold worsening of the frequency and/or intensity of the pre-existing headache, whenever these existed (14).
To minimize memory bias, patients were informed about the study upon diagnosis and were interviewed shortly after the symptom onset. The days elapsed between the symptom onset and the evaluation were controlled and registered.
We anticipated that prior history of headache could act as a confounder, and a comparison between patients with and without prior history of headache was conducted.
Statistical analysis and sample size
Qualitative variables and ordinal variables are reported as frequencies and percentages. Quantitative continuous variables are reported as mean and standard deviation (SD) or median and inter-quartile ranges [IQR] based on the type of distribution. Normality of these variables was assessed with the Kolmogorov-Smirnov test. The duration of headache was described with a Kaplan-Meier survival curve. In the analysis of the treatment response, all non-steroidal anti-inflammatory drugs (NSAIDs) were grouped under the same category, and no specific analysis was done based on the different dose, however, in Spain, only ibuprofen provides two different doses (400 mg vs. 600 mg).
In the hypothesis testing, two-tailed chi-squared tests were used in the contrast of hypotheses between qualitative variables. To evaluate whether age was associated with a different probability of fulfilling ICHD-3 phenotypic criteria for tension type headache (TTH) and/or migraine, a logistic regression was conducted, with ICHD-3 criteria as dependent variable. Odds ratio (OR) and 95% confidence intervals (CI) are presented. In the evaluation of the variables associated with a longer duration of the headache, a cox-regression was done. First, a univariate analysis was done, and all the variables with a P-value lower than 0.2 were included in a multivariate analysis. Hazard ratios (HR) and its 95% CI are presented.
Missing data was prevented by a systematic evaluation of all the study variables. In case that a variable was not available, was considered as absent. There were no cases of loss of follow-up.
There was no former sample size calculation. The analyses were done based on the available sample. Based on the existing literature on headache attributed to coronavirus disease 2019 (COVID-19), a sample size larger than 60 patients was deemed adequate.
Results
During the study period, 478 patients were screened for eligibility and 75 enrolled patients completed the study. Figure 2 shows the flow-diagram of the study. All patients were managed in an outpatient setting. No patients needed oxygen therapy or hospitalization.

Flow diagram of screened, included, excluded and enrolled patients.
A) Demographic data
Patients’ mean age was 43.0 (SD: 15.9), with a range between 15 and 80 years. Forty-two (56%) patients were male. Regarding ethnicity, 66 (88%) participants were Caucasian, six (8%) were North African with Arab ancestry, two (2.6%) participants were Hispanic, or Latino and one (1.3%) participant was Black.
B) Prior medical history
Table 1 shows the frequency of prior diseases. Twenty (26.7%) patients reported prior history of headache and rated the similarity between headache during the course of influenza and their usual headache 65.2% (SD: 24.8%) similar.
Prior history of patients.

No patients with migraine fulfilled criteria for 1.3 Chronic migraine. One patient with tension-type headache fulfilled criteria for 2.3.2 Chronic tension-type headache not associated with pericranial tenderness, with 15 headache days per month, on average, in the preceding three months.
C) Headache phenotype
The most common phenotype was a bilateral headache, with frontal topography, moderate intensity and commonly accompanied by associated symptoms (Table 2). Patients with prior history of headache had photophobia more frequently (80% vs 50.9%, p = 0.033). There were no other statistically significant differences between patients with and without prior history of headache.
Headache phenotype and differences between patients with and without prior history of headache.

IQR: inter-quartile range.
ICHD-3 criteria
Regarding ICHD-3 criteria of 9.2.2 headache attributed to systemic viral infection, due to the eligibility criteria of the study, all patients had a microbiologically confirmed influenza infection, and in all cases, headache was developed in temporal relation to the onset of the infection and improved or resolved in parallel with the infection. About the criterion C.4: 11 (14.7%) patients reported diffuse pain and 73 (97.3%) patients had a moderate-to-severe headache. In the case of the phenotypic criteria for migraine or tension-type headache, these were fulfilled by 42.7% and 30.7% of patients, respectively (Table 3). Migraine criteria were more frequently fulfilled by women (Figure 3). Older age was associated with a higher frequency of fulfilling ICHD-3 phenotypic TTH criteria (OR: 1.042; 95% CI: 1.007–1.077; p = 0.017). Age was not associated with a different odds of fulfilling ICHD-3 phenotypic migraine criteria (OR: 0.983; 95%CI: 0.955–1.013; p = 0.266).
Proportion of patients who fulfilled the phenotypic criteria of migraine or tension-type headache.


comparison of ICHD-3 criteria for migraine and tension-type headache between male and female participants.
Frequency and type of red flags
Regarding headache-related red-flags, these were detected in all patients, with systemic symptoms being present in all patients, pattern change in 73 (97.3%), age over 50 years in 25 (33.3%), including acute medication resistance in 23 (30.7%), wake up-headache in 23 (30.7%), neurological deficit or dysfunction in 20 (26.7%), worst headache ever experienced in 15 (20%), progressive worsening in six (8%), confusion in one (1.3%). No patients reported sudden onset or impaired awareness.
Acute treatment
Acute treatment was needed by all patients, including paracetamol in 65 (86.7%), non-steroidal anti-inflammatory drugs in 40 (53.3%), metamizol in six (8.0%), and triptans in one (1.3%). No patients used ergot derivates, opioids or barbiturates. Figure 4 shows the response to acute medication.

Response to acute medications.
Duration of the headache
The median duration of the headache was 96 [IQR: 48–144] hours, with a range between 0 and 1479 (Figure 5). Patients with prior history of headache 96 [IQR: 49–156] hours versus patients without no prior history of headache 95 [IQR: 48–144] hours.
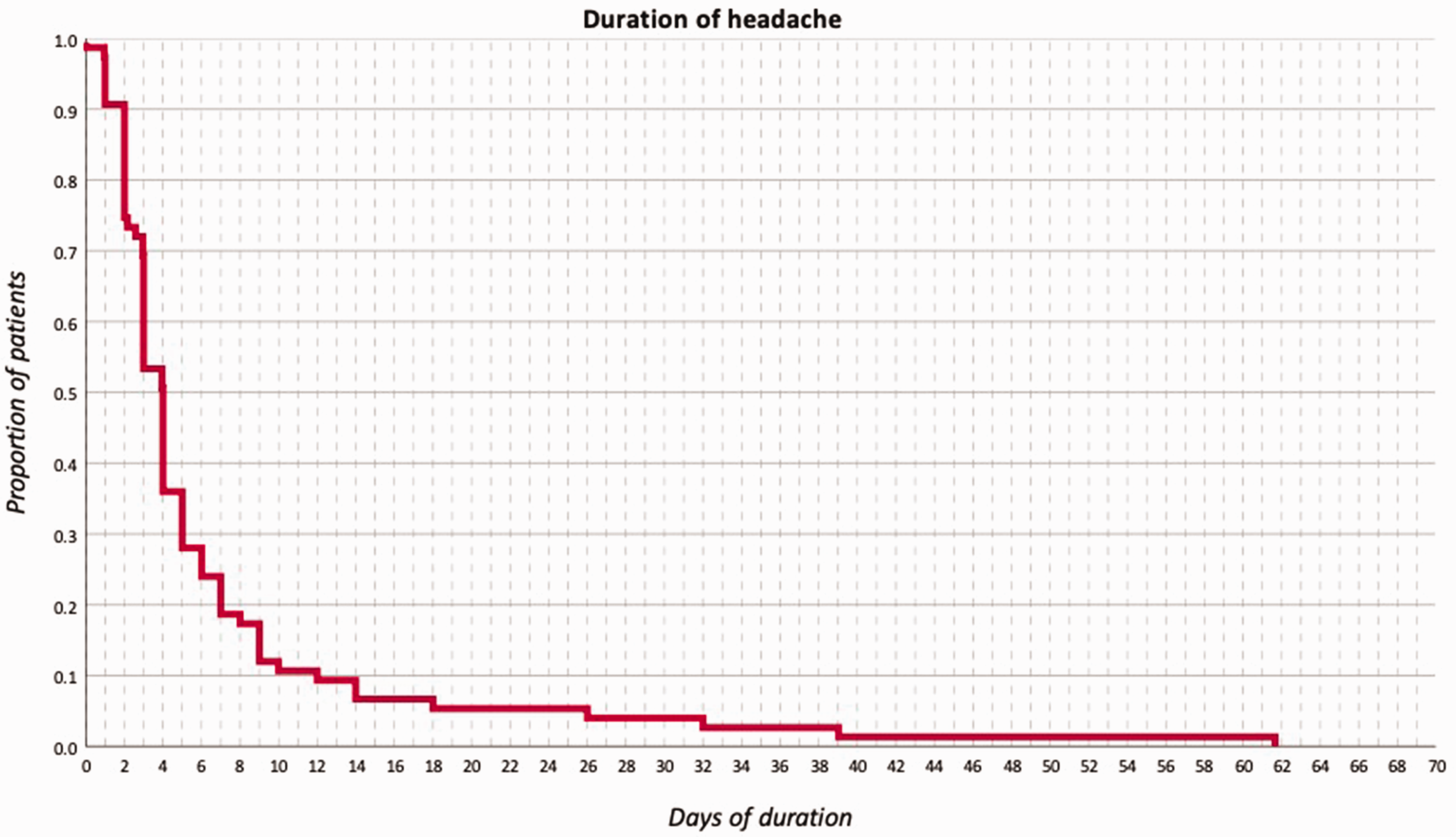
Kaplan-Meier curve depicting the duration of the headache, in days.
In the univariate cox-regression analysis, the variables that were associated with a more prolonged duration of the headache were frontal topography (HR: 0.617; 95%CI: 0.383–0.994; p = 0.047), photophobia (HR: 0.616; 95% CI: 0.382–0.994; p = 0.047) and confusion (HR: 12.070; 95%CI: 1.453–100.261; p = 0.021) (Online Supplementary Table 1). In the multivariate cox-regression analysis, after adjusting for multiple comparisons, restlessness (HR: 1.672; 95% CI: 1.017–2.750; p = 0.043; smoking habit (HR: 0.106; 95% CI: 0.012–0.903, p = 0.043) and confusion (HR: 16.764; 95%CI: 1.790–157.006; p = 0.042) remained statistically significant.
Discussion
In this prospective cohort study, the clinical phenotype and the duration of headache in patients with influenza infection was characterized. Despite the high incidence of influenza (1), the phenotype of this headache had not been assessed so far (9). The search for specific features may be relevant in the differentiation of other primary or secondary headache disorders (15). Furthermore, some secondary headache disorders are a valuable opportunity to better understand the pathophysiology of some primary headache disorders.
The predominant headache phenotype was a bilateral headache (52%), with frontal (48%) or temporal (39%) topography, pressing in quality (61%), moderate-to-severe intensity, and commonly associated with photophobia (59%) or phonophobia (51%). This phenotype was similar to other secondary headache disorders, such as headache attributed to COVID-19 or alcohol-induced headache (12,13,18), however, there were remarkable differences. First, the frequency of cranial autonomic symptoms was higher than in other disorders, especially rhinorrhea (79%), nasal congestion (76%), sweating (53%) or conjunctival injection (24%). This could be related to the activation of trigeminal nerve within the cranial sympathetic and parasympathetic system (19), caused by the cytotoxic effect of the virus and/or the local immune response (9,20,21).
Second, another surprising finding was the short-lasting nature of the headache. This could be related with the high prevalence of prior influenza infection (31%), the prior occurrence of headache in the context of other infections (61%), and the proportion of patients who had been previously vaccinated (21%), as it has been observed in patients with SARS-CoV-2 (22,23). This points toward the immune response as a likely generator of the headache, suggesting that treatments should minimize the negative consequences of the immune activation, without preventing its physiologic function (23).
Indeed, all patients required acute medication, and in most cases, drugs that can be obtained over the counter were used, mostly paracetamol or non-steroidal anti-inflammatory drugs. Self-treatment of common cold symptoms in common, despite the existing evidence of most therapies is limited (24). Its use is negatively correlated with the healthcare resource utilization (25), despite the annual cost being estimated at >USD$100 billion (26). In the case of the specific treatment response, the proportion of patients who achieved at least a 50% response was higher in patients using NSAIDs that in patients treated with paracetamol, which could suggest that part of the headache could be caused by the release of prostaglandins, thromboxane and prostacyclin (27).
Secondary headache disorders may increase our knowledge about some primary headaches. It could be argued that some patients experienced their pre-existing headache, perhaps triggered by the infection. However, this item was anticipated in our study, and all but two patients described this as a different headache (12). Indeed, the ICHD-3 states that a two-fold worsening in terms of intensity and/or frequency should be used in the classification of secondary headaches in patients with primary headache disorders (15).
The ICHD-3 criteria for headache attributed to a systemic viral infection are generally unspecific, with only two items related to the headache itself: diffuse location and moderate-to-severe intensity (15). In our sample, the diffuse topography was uncommon, with the headache being typically located in the anterior pole of the head and periocular. In addition, there was a substantial heterogeneity within patients, which could be associated with the individual predisposition and a personal biology more prone to exhibit typical migraine features (23). In our opinion, the activation of some intracranial structures within the migraine attack, or some secondary headaches, are responsible for the phenotypic manifestations (28). Unfortunately, evidence in this regard, and the precise association between the involved structures is still limited (29). Due to the scarcity of studies addressing headache attributed to systemic infections, future studies should test the ICHD-3 criteria and seek specific phenotypic or paraclinical biomarkers.
The key in the differentiation between primary and secondary headache disorders are headache-related red flags. The special interest group of the International Headache Society crated a proposal that summarized the main red flags, called SNNOOP10, which included 15 items (16). In the present study, red flags were common and were observed in all patients. This finding is in line with prior studies that evaluated the prevalence of these in patients with SARS-CoV-2, where all patients also had at least one red flag (30). Some of the red flags were observed by very few – sometimes only one – patient(s), and therefore the results should be interpreted carefully. The very large confidence intervals from the regression analyses assessing which variables are associated with a more prolonged duration of the headache suggest that the accuracy of the variables that remained statistically significant is not optimal and this should be confirmed and validated in future studies.
Regarding the study limitations, the sample size was limited, so the study could be underpowered to detect differences that are not very large between patients with and without headache. The association between headache and other systemic symptoms was not addressed in the study. Differences in the headache phenotype depending on the strain are still to be understood. There was a selection bias towards a non-hospitalized population. In the response to acute medication, due to the limited sample size, all NSAIDs were grouped, and this may bias the results, if not all NSAIDs were equally effective.
Conclusion
The clinical phenotype of headache attributed to influenza infection was a bilateral headache, with frontal or temporal topography, moderate-to-severe intensity, that frequently worsened by physical activity. The only difference that was found between patients with and without prior history of headache was a higher frequency of photophobia within patients with prior headache. The median duration of the headache was four days, and no patients developed a chronic headache. Cranial autonomic symptoms such as rhinorrhea and nasal congestion were prevalent, and other associated symptoms such as photophobia or phonophobia were common. The phenotypic International Classification of Headache Disorders criteria for migraine or tension-type headache were fulfilled by 43% and 31% of patients, respectively. All patients had headache-specific red flags and all patients needed acute medication, with non-steroidal anti-inflammatory drugs showing the higher response rate.
Article highlights
Acute headache attributed to influenza infection seems similar to other acute viral infections. The headache phenotype may resemble that of migraine or tension-type headache, even in patients with no prior history of such headache disorders. Most patients required acute treatment, however, the headache lasted a few days in most cases. The headache pathophysiology seems related to the local innate immune response.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231212900 - Supplemental material for InfluenCEF study: Clinical phenotype and duration of headache attributed to influenza infection
Supplemental material, sj-pdf-1-cep-10.1177_03331024231212900 for InfluenCEF study: Clinical phenotype and duration of headache attributed to influenza infection by David García-Azorín, Laura Santana-López, José Eugenio Lozano-Alonso, Ana Ordax-Díez, Yésica González-Osorio, Silvia Rojo-Rello, José M Eiros, Javier Sánchez-Martínez, Andrea Recio-García, Álvaro Sierra-Mencía, Ivan Sanz-Muñoz and Ángel Luis Guerrero-Peral in Cephalalgia
Footnotes
Acknowledgements
The authors gratefully acknowledge all the members and participants of the Sentinel Network of Castile and Leon (Red Centinela Sanitaria de Castilla y Leon) and the members of the Public Health Headquarters at the Regional Health Administration (Dirección General de Salud Pública de la Consejería de Sanidad de la Junta de Castilla y Leon).
Author contributions
DGA, JELA, AOD, YGO, ISM and ALGP designed and conceptualized the study, LSL, ARG, ASM acquired the data, DGA, JELA, AOD analyzed the data, DGA and LSL drafted the manuscript; JELA, SRR, JEB, JSM, ISM, ALGP revised the manuscript for intellectual content. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DGA received honoraria for lectures/presentations from Abbvie/Allergan, Eli Lilly, Teva, Lundbeck, and Novartis. DGA Participated in clinical trials as the principal investigator for Pfizer, BioHaven and Lundbeck. DGA is junior editor of The Journal of Headache and Pain and Neurological Sciences. DGA received honoraria from the World Health Organization as subject matter expert.
ÁLGP received honoraria for lectures/presentations from Abbvie/Allergan, Eli Lilly, Teva, Lundbeck, and Novartis. ALGP Participated in clinical trials as the principal investigator for Eli Lilly, Teva, Abbvie, Novartis, Amgen and Lundeck.
All other authors report no conflicts of interest.
Fundings
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
