Abstract
Background
Longitudinal migraine studies have rarely assessed headache frequency and disability variation over a year.
Methods
The Chronic Migraine Epidemiology and Outcomes (CaMEO) Study is a cross-sectional and longitudinal Internet study designed to characterize the course of episodic migraine (EM) and chronic migraine (CM). Participants were recruited from a Web-panel using quota sampling in an attempt to obtain a sample demographically similar to the US population. Participants who passed the screener were assessed every three months with the Core (baseline, six, and 12 months) and Snapshot (months three and nine) modules, which assessed headache frequency, headache-related disability, treatments, and treatment satisfaction. The Core also assessed resource use, health-related quality of life, and other features. One-time cross-sectional modules measured family burden, barriers to medical care, and comorbidities/endophenotypes.
Results
Of 489,537 invitees, we obtained 58,418 (11.9%) usable returns including 16,789 individuals who met ICHD-3 beta migraine criteria (EM (<15 headache days/mo):
Conclusions
The CaMEO Study provides cross-sectional and longitudinal data that will contribute to our understanding of the course of migraine over one year and quantify variations in headache frequency, headache-related disability, comorbidities, treatments, and familial impact.
Background
Migraine is a common and often debilitating condition, with a one-year prevalence of 11.7% to 13.2% in studies from the United States (US) (1–3). Migraine is generally divided into two groups based on headache days per month. Episodic migraine (EM) is characterized by <15 headache days per month and chronic migraine (CM) is characterized by ≥15 headache days per month. However, the definition of CM has evolved over the past 25 years (4), most recently with the 2013 release of ICHD-3 beta (5).
EM and CM have been well characterized in population studies from many regions of the world (2,6–14), and results have been summarized in meta-analyses (15,16). These studies found significant headache-related disability associated with CM and EM (2,6–8,11,15,17), although more so with CM (6,9,11,17–19). In comparison with people with EM, those with CM experience substantially greater headache impact on daily activities (11,20), higher direct medical costs (9,21,22), greater health care resource utilization (10,11), reduced health-related quality of life (11,23), and higher rates of comorbidities (9,11,19). Also, many studies have defined risk factors for migraine chronification, which have been summarized in multiple review articles (17,24–28).
Despite previous in-depth population studies, much remains to be discovered about the effects of EM and CM on individuals. Largely uninvestigated areas include 1) the natural history or longitudinal disease course of EM and CM over a single year, 2) barriers to effective diagnosis and treatment of migraine (especially CM), 3) differences between EM and CM in observed impact and burden on families, and 4) comprehensive comorbidity profiling and differential association with EM and CM. The Chronic Migraine Epidemiology and Outcomes (CaMEO) Study was designed to extend knowledge about these and other issues. The longitudinal assessments were designed to enhance understanding of the natural history of and subject-specific variations in attack frequency, disability, psychiatric comorbidities, and treatment. In addition, a series of cross-sectional modules assessed previously unexplored aspects of barriers to care, comorbidities, and family burden of migraine among those with CM and EM. In this first report from the CaMEO Study, we describe methodology and design, explore nonresponse bias, and present preliminary findings on respondent demographic and socioeconomic characteristics and headache-related disability.
Methods
Study design
Overview
The study had a screening and recruiting phase and then longitudinal assessments approximately every three months over one year. In addition, a series of cross-sectional surveys assessed health care use, family burden, and comorbidities/endophenotypes. Data collection occurred over 15 months starting in September 2012 and closing in November 2013. The study was approved by the institutional review board of the Albert Einstein College of Medicine.
Screening and recruiting
The screening and recruiting phases occurred from September 2012 through October 2012. Members of an Internet research panel (Research Now, Plano, TX), which has 2.4 million active US members, provided the sampling frame. Panel members were carefully screened and selected to be broadly representative of the US population. Quota sampling was used in an attempt to recruit a study sample that was representative of US population demography. Email invitations were sent to 489,537 panel members inviting them to complete the Screening Module (Figure 1), which was a brief survey capturing headache information and demographics (median completion time = 8 minutes; Table 1). The Study Population section describes the screener’s operating characteristics and migraine definitions used in the CaMEO Study.
Participant flow diagram. Screening, core, and snapshot module construct areas and validated instruments. DSM-IV: Diagnostic and Statistical Manual for Mental Disorders, 4th ed.; ICHD-3 beta: International Classification of Headache Disorders, 3rd edn (beta version) (5); NRS: numerical rating schedule; OTC: over the counter; PHQ: Patient Health Questionnaire; Rx: prescription. Validated instrument. Adapted from Horowitz M, Schaefer C, Hiroto D, et al. Life event questionnaires for measuring presumptive stress. Longitudinal return and attrition rates. For wave 1 no cases could attrit, the total returns screening positive for migraine were Percentages in these cells denominated by baseline total ( Percentages in these cells denominated by wave-specific returns. See Figure 2 for how we arrive at the The drop in outgo at wave 4 is reviewed in detail in the Discussion under the section on “Benefits/risks of conducting large scale epidemiologic studies using national online sampling panels.”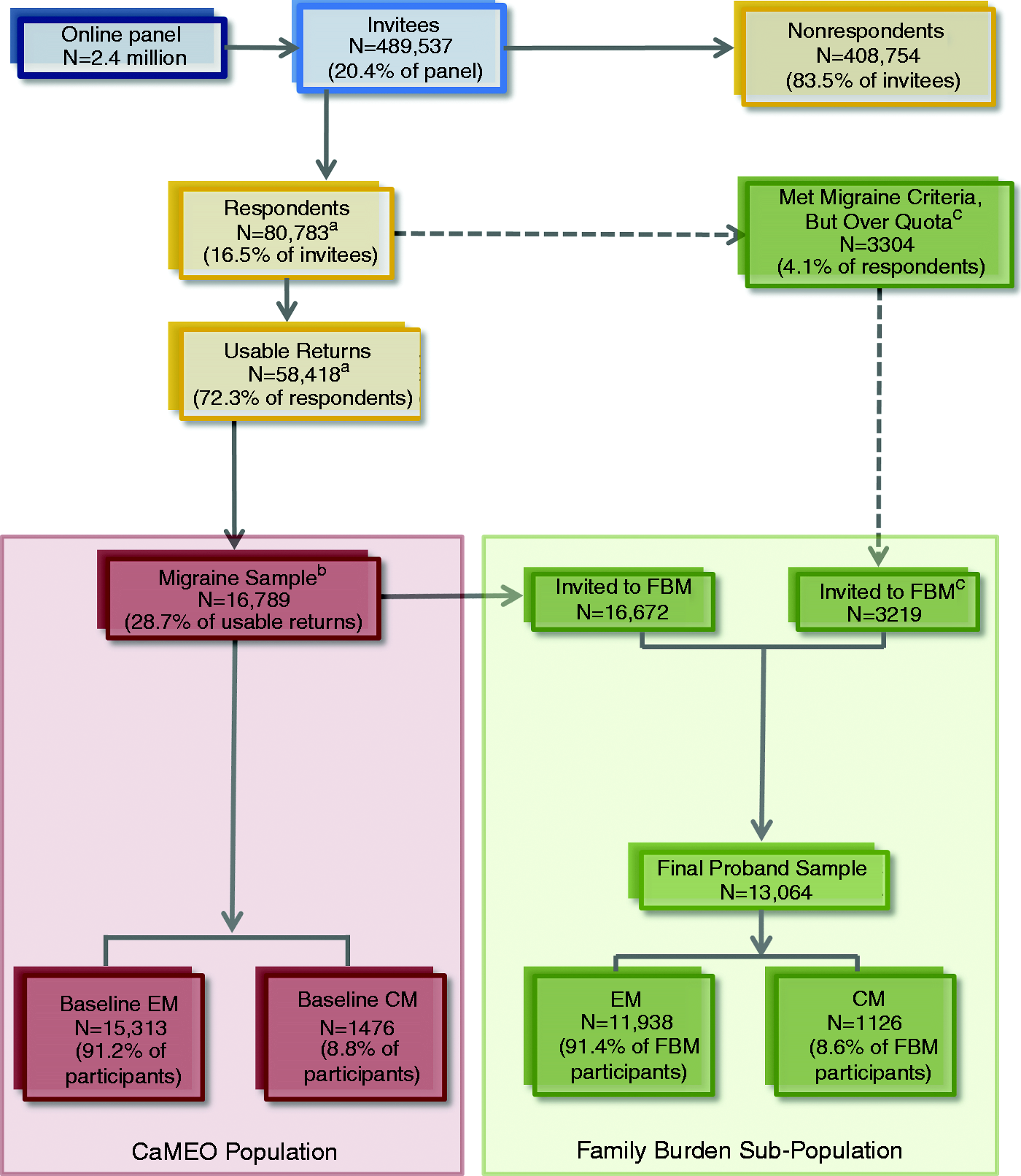


Longitudinal modules
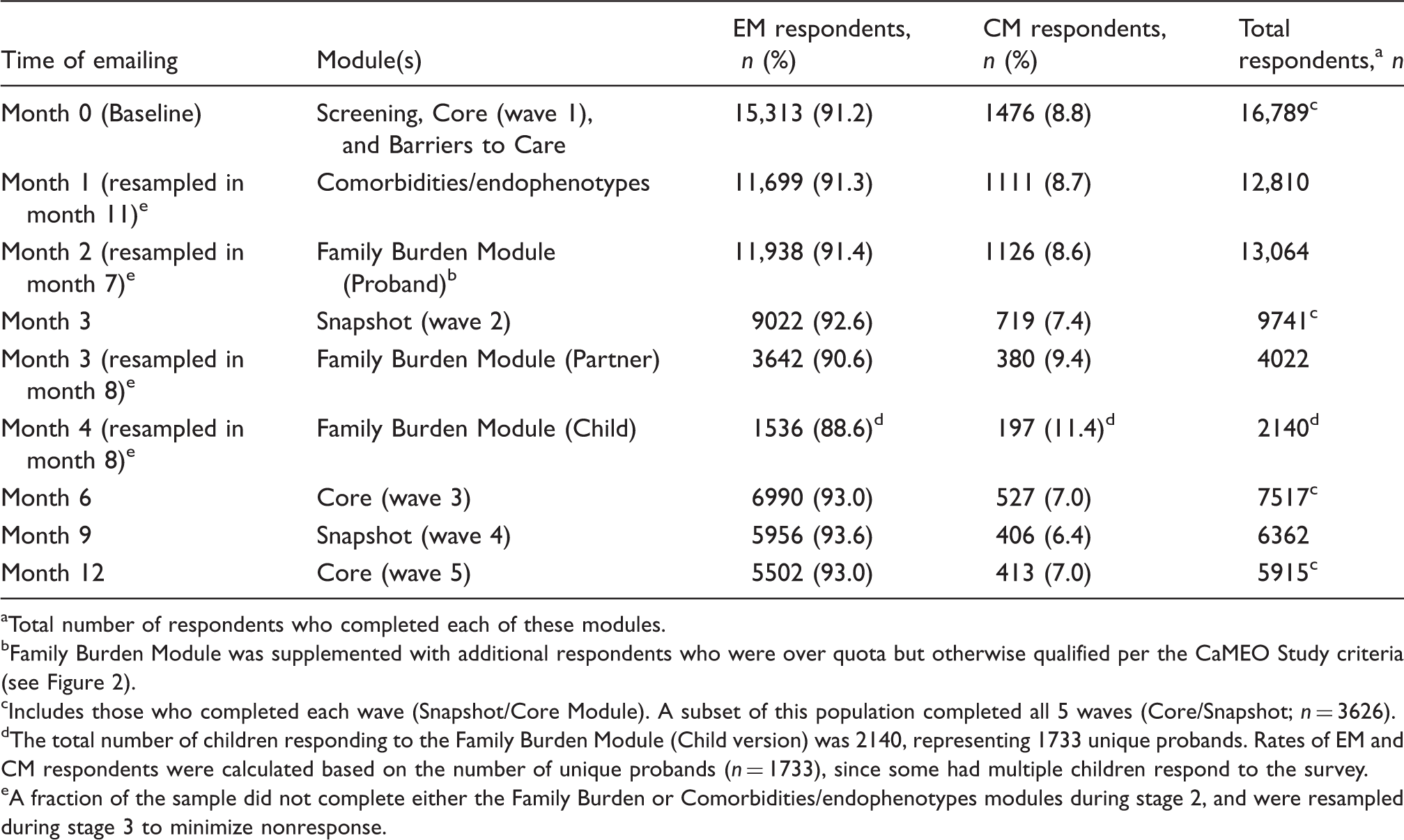
Qualified participants received emails at approximately three-month intervals (three, six, nine, and 12 months after baseline was sent) inviting them to complete multi-component Internet surveys (Figure 2). The longitudinal assessments were of two kinds: 1) the Core Module and 2) the Snapshot Module. The Core was offered three times (baseline, six, and 12 months) and the Snapshot was offered twice (three and nine months). The Core Module (median completion time = 10 minutes) consisted of 10 construct areas, most of which used validated instruments measuring headache frequency, depression, anxiety, interictal burden, headache-related disability, health-related quality of life, major life events, treatment/treatment satisfaction, and health care resource utilization (Table 1). The Snapshot Module (median completion time = 6 minutes) was an abbreviated version of the Core and assessed headache day frequency, headache-related disability, and any changes in headache treatments or treatment satisfaction in the past three months (Table 1). The components common to the Core and Snapshot modules will be used to assess longitudinal variation in headache frequency and disability (Figure 2).
Study design.
Cross-sectional modules
In addition to the longitudinal components, several cross-sectional modules were administered one time each (Figure 2). These modules assessed barriers to care, comorbidities/endophenotypes, and family burden.
The order of module administration was embedded in the longitudinal design (Figure 2) and may be described as having three stages. In stage 1, baseline assessments consisted of the Screener, first Core Module, and Barriers to Care Module in quick succession from September 2012 to October 2012. This was followed by stage 2, during which the Comorbidities/Endophenotypes Module and Family Burden Module were administered from October 2012 through February 2013. Stage 3 collected the remaining four waves of longitudinal data, with the final surveys emailed in September 2013 and closed in November 2013. Receipt and initiation of modules were staggered based on subject-specific completion; thus, while Figure 2 would suggest stages 2 and 3 overlapped, at the respondent level there was no overlap, though an individual could be completing a stage 2 module at the time that another was initiating a stage 3 module.
Assessments
Questionnaire development
The CaMEO Study modules included validated instruments and surveys as well as original items developed based on the results of focus groups, expert opinion, and pretesting among target respondents.
Modules
Other modules.

GERD-Q: Gastroesophageal Reflux Disease Questionnaire; HCP: health care professional; IBS-1: Irritable Bowel Syndrome Module 1, validated, uses Rome III criteria; MOS: Medical Outcomes Study; PC-PTSD: Primary Care Post-Traumatic Stress Disorder; PCL-C: Post-Traumatic Stress Disorder Checklist-Civilian Version.
Doctor was defined for the respondent as an HCP who is licensed to prescribe medications and included a medical doctor (MD/DO), nurse practitioner, physician assistant, or a dentist (DDS). A complimentary or alternative HCP (non-prescribing) was defined as a chiropractor, psychologist, massage therapist, acupuncturist, physical therapist, naturopath, natural health consultant, or any other type of alternative HCP.
Domains based on Proband Module, but Partner and Child modules had a similar structure.
In stage 2, the Comorbidities/Endophenotypes Module (median completion time = 19 minutes) collected data on a broad set of indicators (e.g. migraine features, allergies and respiratory disorders, chronic pain, chronic fatigue, sleep problems, autonomic disorders, psychiatric disorders, and internal disorders; Table 3), in an attempt to discover new empirically supported subclasses of migraine and increase taxonomic classification accuracy within the migraine spectrum.
The Family Burden Module assessed headache-related burden from the perspective of the migraineur (proband), their partners (i.e. spouses/significant others), and their children. The Proband Family Burden Module assessed the proband’s perspective on the impact of their headaches on family well-being, interpersonal relationships, and family and social interactions and activities as well as the impact of migraine on family finances, career, and health-related quality of life (Table 3). Partners and children were identified by the proband; participating partners and children reported their perspectives on the effect of the proband’s migraines on family life.
Study population
Migraine respondents
The migraine Screening Module (see Table 1), based on the American Migraine Study (AMS)/the American Migraine Prevalence and Prevention (AMPP) Study diagnostic module (7,8), was used for migraine classification. This screener has a sensitivity of 100% and specificity of 82% for migraine diagnosis (30), and sensitivity of 91% and specificity of 80% for CM diagnosis (31). The screener was based on lifetime recall of migraine symptoms associated with the respondents’ most severe headache (e.g. unilateral, pulsating, moderate/severe pain intensity, routine activity exacerbation of headache pain, nausea, phonophobia, photophobia), and was used to make modified ICHD-3 beta migraine classifications (5). Although the AMS/AMPP diagnostic module was based on ICHD-2 migraine criteria, no significant changes occurred between ICHD-2 and ICHD-3 beta that are related to the criteria used in this study. The AMS/AMPP diagnostic module classification criteria were considered modifications of ICHD-3 beta migraine criteria because two criteria were not confirmed: ≥5 lifetime migraine events (criterion A) and duration of attack untreated from four to 72 hours (criterion B).
In addition, this module assessed baseline headache frequency with a single open-response item on three-month recall of headache days, which was averaged to obtain a one-month headache day rate to classify respondents as having EM or CM using modified Silberstein-Lipton criteria (headache frequency of ≥15 (CM) or <15 (EM) days per month over the preceding three months) (32,33). The modified Silberstein-Lipton CM criteria employed in the CaMEO Study align to ICHD-3 beta criteria for CM, except for the requirement that ≥8 of the headache days per month be migraine (criterion C from the ICHD-3 beta CM criteria). Criterion C was not employed because it is difficult to assess in a large, self-report data-collection paradigm and requires the use of a diary and physician interview to accurately assess.
Only reliable respondents were included in the final sample (defined as those who completed baseline surveys in a reasonable time to read and respond to items (≥10 minutes), and reported consistent age and sex). Respondents eligible for inclusion were reliable active panel members, aged ≥18 years, met migraine criteria, and had experienced ≥1 headache within the past 12 months.
Family members
For the proband, partners were defined as “currently in a relationship with a spouse, partner, or significant other.” Partners were eligible to participate if they had cohabitated with the proband for ≥2 months. Eligible child respondents included adolescent/adult children, grandchildren, and stepchildren aged 13 to 29 years, cohabitating with the proband for ≥2 months. Because of the risk of potentially low partner and child Family Burden Module response rates, baseline respondents who were considered to be over-quota, but otherwise qualified for the CaMEO Study, were resampled for the Family Burden Module (Figure 1). Data from these over-quota respondents were not used for any other module.
Statistical methods
Sample size determinants
Because we planned to compare people with CM and EM in multiple ways, no formal sample size calculations were performed. We targeted a sample of ≥315 individuals with CM who would complete all study assessments over the course of a year. We assumed that prevalence of migraine in this US panel would be 12%, and that of those people with migraine 7% would have CM. In order to achieve a sample of
Nonresponse bias
Nonresponse bias was explored through a variety of methods and contrasts; details of these methods and results are provided in the online Supplementary Appendix.
Statistical analyses
Descriptive and inferential statistics were used to compare the EM and CM samples on baseline survey demographics and outcomes. Continuous and count outcomes were described using means and standard deviations; inferential contrasts for continuous outcomes were based on analysis of variance (ANOVA) mean differences and 95% confidence intervals (CI), while rate ratios (RR; 95% CI) derived from negative binomial regression models were employed for count outcomes. Binary and multinomial outcomes were described using the number and percentage endorsing. Inferential contrasts for binary outcomes were based on odds ratios (OR; 95% CI) obtained from logistic regression models. For ordered multinomial category outcomes, cumulative ORs (95% CI) were obtained from cumulative logistic regression models. All statistical tests were two-tailed (significance level, 0.05) and were not adjusted for multiple comparisons. Imputation methods for missing values were not conducted. All analyses were conducted using SAS statistical software, version 9.2 (SAS Institute Inc., Cary, NC).
Results
CaMEO sample and baseline response rates
Distribution of respondents with episodic (EM) and chronic migraine (CM) by study module.
Total number of respondents who completed each of these modules.
Family Burden Module was supplemented with additional respondents who were over quota but otherwise qualified per the CaMEO Study criteria (see Figure 2).
Includes those who completed each wave (Snapshot/Core Module). A subset of this population completed all 5 waves (Core/Snapshot;
The total number of children responding to the Family Burden Module (Child version) was 2140, representing 1733 unique probands. Rates of EM and CM respondents were calculated based on the number of unique probands (
A fraction of the sample did not complete either the Family Burden or Comorbidities/endophenotypes modules during stage 2, and were resampled during stage 3 to minimize nonresponse.
Stages 1 and 2: Cross-sectional module response rates
Barriers to Care, Comorbidities/Endophenotypes, Family Burden modules
All qualified respondents (
Stage 3: Longitudinal module response rates
Core and Snapshot modules
Longitudinal response patterns. a

Over the five waves of longitudinal data collection, respondents had to participate at baseline, but were free to respond or not respond at any wave thereafter. Response patterns for one wave and five waves can occur only a single way, but a variety of response patterns could be observed for two to four waves of participation.
Respondent demographic and comorbidity contrasts
Demographic, socioeconomic, and headache characteristics of participants with episodic and chronic migraine.
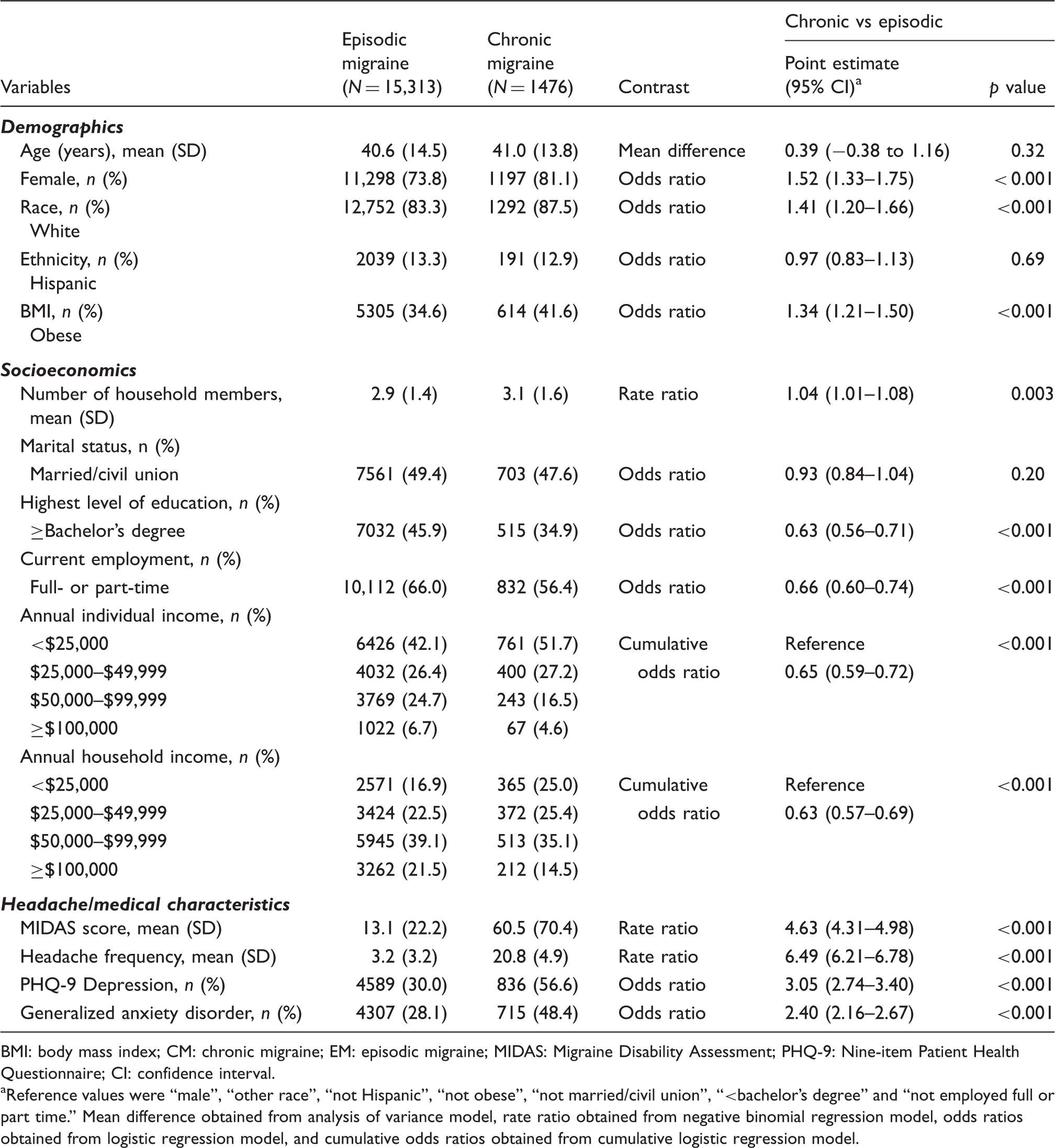
BMI: body mass index; CM: chronic migraine; EM: episodic migraine; MIDAS: Migraine Disability Assessment; PHQ-9: Nine-item Patient Health Questionnaire; CI: confidence interval.
Reference values were “male”, “other race”, “not Hispanic”, “not obese”, “not married/civil union”, “<bachelor’s degree” and “not employed full or part time.” Mean difference obtained from analysis of variance model, rate ratio obtained from negative binomial regression model, odds ratios obtained from logistic regression model, and cumulative odds ratios obtained from cumulative logistic regression model.
Discussion
Characteristics of other large-scale epidemiologic migraine studies that assessed chronic migraine and episodic migraine in the United States.

AMS: American Migraine Study; AMPP: American Migraine Prevalence and Prevention Study; CaMEO: Chronic Migraine Epidemiology and Outcomes Study; CM: chronic migraine; EM: episodic migraine; IBMS: International Burden of Migraine Study; ICHD-2: International Classification of Headache Disorders, 2nd edn.
Australia, Canada, France, Germany, Italy, Spain, United Kingdom, Taiwan, United States (US).
Met the study case definition for migraine, which was based on (but not strictly the same as) 1988 International Headache Society (IHS) diagnostic criteria. Data collection was not stratified by episodic or chronic forms of migraine, because this distinction was not made until 2004, with the release of ICHD-2.
Previous studies characterized migraine prevalence in the US. CaMEO was not designed to assess overall migraine prevalence and used quota sampling to provide a population construct in an attempt to represent the US population demography.
The CaMEO Study replicated many well-known findings regarding EM and CM. In CaMEO, chronic migraineurs were 52% more likely to be female, 41% more likely to be white, 34% more likely to be obese, 205% more likely to have depression, and 140% more likely to have anxiety than episodic migraineurs (Table 6). Results from CaMEO and AMPP Studies and IBMS were similar in that those with CM were primarily female and significantly more likely than those with EM to be obese, white, and have psychiatric comorbidities (9,36). Within IBMS, the percentage of females was not significantly different between the EM and CM groups, but the proportion of females was still higher in the CM group versus the EM group (like CaMEO); significantly different ages were noted in IBMS (unlike CaMEO), but the mean actual age was almost identical between the two studies, with chronic migraineurs being slightly older than episodic migraineurs (9,10). In a single-center study (19) of consecutive outpatients with EM and CM (with probable medication overuse (revised ICHD-2R) (37)), the CM group had a greater proportion of females and was older. Study design and population differences, including international versus US participation, may result in inconsistent demographic data between studies.
The CaMEO Study found that the rate of disability days per month (using MIDAS) was 3.63 times greater for those with CM than for those with EM. This is consistent with previous studies (6,9,18,38). Thus, chronic migraineurs are less able to perform work and leisure activities than those with EM, in line with previous findings of a direct correlation between attack frequency and headache-related disability for people with migraine (38).
In the CaMEO Study, socioeconomic status was reduced in those with CM compared with those with EM, as chronic migraineurs were found to have less education, less full- or part-time employment, and were more likely to have lower annual income. This finding is generally consistent with previous studies (17,19,36,39); however, IBMS and the Taiwanese clinic study found similar annual household income in EM and CM groups (9,10,39). This discrepancy with the CaMEO Study may be caused by potentially different income sources internationally (e.g. work, government-issued disability payments). A single-center outpatient study (19) found the CM group had less education, and was more likely to be married than those with EM.
Assessment of potential bias
All surveys are prone to selection bias, which can include under-coverage, voluntary response bias, and nonresponse bias. Under-coverage occurs when some members of the population are inadequately represented in the sample. The CaMEO Study addressed this by balancing the target sample on key demographics and by adjusting for demographic characteristics in the analyses. Voluntary response bias occurs when respondents are self-selected volunteers and may represent those with the strongest opinions or most severe disease. Although not directly assessed in the CaMEO Study, we suspect that this effect may work in both directions; more severe migraine cases may be both unable to respond and more interested in responding, which may negate this potential source of bias. Beyond the rates of CM, the AMPP Study, US IBMS, AMS, and CaMEO Study profiles are similar (2,6–8,10), which suggests that biases in CaMEO are not very different from these earlier studies.
The CaMEO Study was not designed to be a prevalence study. We acknowledged at inception that use of online panel sampling would limit generalizability in a way that mailed surveys do not, thus precluding explorations of prevalence. But prevalence has been well addressed (6,15,16) and was not of central interest here. Though we used probability sampling of quotas from a nationally distributed online panel to obtain the CaMEO migraine sample, response rates were low. Only 16.5% of invitees responded, 11.9% of whom provided useable data.
Analyses of nonresponse are imperative to understand the implications of survey results, as characteristics of nonrespondents may differ from those of respondents (40). Nonresponse bias occurs when individuals chosen for the sample are unwilling or unable to participate in the survey, causing respondents to differ in meaningful ways from nonrespondents. To assess this potential source of bias in the CaMEO Study, we sent a follow-up survey to nonrespondents to obtain demographics and disease severity (see the online Supplementary Appendix). Comparison of respondent and nonrespondent demographic data found that large sample sizes exaggerated the significance of small differences (online Supplementary Table 1). Respondents to the Nonrespondent Survey and the initial survey are similar; however, the low rate of participation in the Nonrespondent Survey leaves open the possibility of nonresponse bias.
This study has several limitations. All data were collected via self-report, and no supporting documentation or medical or pharmacy records were collected for verification, as this would be impractical given the large study size. As noted above, despite quota sampling, baseline demographic characteristics differed between the CaMEO Study respondents and nonrespondents (online Supplementary Tables 1 and 2). Consistent with other epidemiologic studies (6,9,36), the large sample sizes in CaMEO may have made minor demographic differences become significant. The migraine-positive case rate in the overall Nonrespondent Survey was generally similar to that of the respondents, suggesting disease state did not present a sampling bias. However, data for only 2.0% of nonrespondents (8225/408,754) were available for these analyses, and thus may not be representative of the entire set of nonrespondents. Other analyses of nonrespondents have found that nonresponse was associated with low socioeconomic status, but not health status (40). Because the variable of interest in our study is health status (i.e. migraine), it is possible that any differences that may have been caused by nonresponse may not have a great effect (40).
Benefits/risks of conducting large-scale epidemiologic studies in national online sampling panels
Internet research panels provide the advantage of enabling access to large numbers of willing survey participants who can be sampled and stratified according to pre-identified demographic and disease/symptom-based criteria. In CaMEO, over the course of a year, we succeeded in screening and following 16,789 migraineurs, replicated many of the findings of prior studies like the AMPP Study during the data processing and verification phase, and succeeded in obtaining our target sample proportions. We succeeded in guiding a total of 3626 migraineurs through five complete waves of longitudinal assessments and 3513 migraineurs with complete data through all nine study modules. However, Internet data capture, by virtue of its electronic and nearly automatic nature, is not faultless and the risk remains that some groups of interest may be over- or under-sampled because of issues of Internet access (41–44) and a lack of rigor in the management of Internet panel members.
Internet sampling panels often have limits on the number of studies in which members can participate. These limits are set by the panelist and are designed to minimize panelist burden and optimize participation. Our sampling protocol required panelist limits to be overridden so that baseline participants would receive invitations to all modules and every phase of the quarterly longitudinal assessments. This occurred in nearly all cases, except in wave 4 of the longitudinal assessment and a few cases spread across other modules.
In wave 4 panelist limits were not overridden for 1657 active participants. As a result, these 1657 participants never received invitations for wave 4; the number of these cases that would have responded in wave 4 had they been invited is unclear. To estimate the impact of the omission of these cases, we focused on a decomposition of the 34% of cases (556/1657) who had not attrited by wave 3. Our best estimate of those we would have lost has a lower and upper estimate based on responsiveness patterns among these 34% of cases. Our lower bound is based on those who responded sequentially in all waves 1 to 3, but not in wave 5. Our upper bound is based on those who responded in every wave but wave 4, the wave to which they were not invited. Our lower bound total sample is 184 subjects, who represent 2.8% of the 6362 wave 4 respondents. These 184 cases were composed of 173 persons with EM (2.9% of wave 4 EM respondents) and 11 individuals with CM (2.7% of wave 4 CM respondents). Our upper bound total sample was
There were also 199 cases who were eligible for at least one component of the Family Burden Module (either proband or partner), 15 cases who should have been invited to the Comorbidities/Endophenotypes Module, and four cases who should have been invited to the wave 2 Snapshot Module who were not invited because of this restriction limit error.
Despite limitations, the strengths of the CaMEO Study include a large nationwide sample (
Conclusions
Baseline results from the CaMEO Study confirmed previous findings that CM is associated with increased headache-related disability, psychiatric comorbidities, and greater financial and occupational burden than is EM. As more data are analyzed, the CaMEO Study will continue to provide a naturalistic understanding of the longitudinal course and consequences of EM and CM; quantify variations in headache frequency, headache-related disability, comorbidities, medication use, and effect of migraine on the family unit; and contribute a wealth of information to the limited amount of data on CM. The insights gleaned from the CaMEO Study into the characteristics of EM and CM will help to optimize future treatment paradigms to improve outcomes for the many individuals affected by migraine.
Clinical implications
Limited data are available to characterize chronic migraine. The Chronic Migraine Epidemiology and Outcomes (CaMEO) Study was designed with a particular emphasis on understanding the differences between chronic and episodic migraine, and aims to provide a naturalistic understanding of the longitudinal course and consequences of migraine (every three months over 12 months) and quantify variations in headache frequency, headache-related disability, comorbidities, medication use, and familial impact, as a complement to earlier studies designed to assess prevalence and burden of illness.
Footnotes
Funding
This study and analysis were sponsored by Allergan, Inc. (Irvine, CA). The study sponsor was involved in the study design, data collection, data analysis, data interpretation and the writing of the article. AA, MR, VM, DS, KF, DB, and RL were involved in development of the Web-based questionnaire. AA, MR, DS, KF, and RL were involved in development of the statistical analyses plan. DS and KF were responsible for conducting all the statistical analyses in this manuscript. All authors had full access to all of the data. The corresponding author had final responsibility for submission of this paper. All authors met the International Committee of Medical Journal Editors (ICMJE) authorship criteria. Neither honoraria nor payments were made for authorship.
Writing and editorial assistance were provided to the authors by Amanda M. Kelly, MPhil, MSHN, and Kristine W. Schuler, MS, of Complete Healthcare Communications Inc. (Chadds Ford, PA) and funded by Allergan, Inc. (Irvine, CA).
Conflicts of interest
Richard B. Lipton, MD, receives research support from the National Institutes of Health (NIH) (PO1 AG03949 (Program Director, Project and Core Leader), RO1AG025119 (Investigator), RO1AG022374-06A2 (Investigator), RO1AG034119 (Investigator), RO1AG12101 (Investigator), K23AG030857 (Mentor), K23NS05140901A1 (Mentor), and K23NS47256 (Mentor)), the National Headache Foundation, and the Migraine Research Fund; serves on the editorial board of
Dawn C. Buse, PhD, has received grant support and honoraria from Allergan Inc./MAP Pharmaceuticals, Novartis, NuPathe, and Zogenix. She is an employee of Montefiore Medical Center, which has received research support funded by Allergan Inc., Colucid, Endo Pharmaceuticals, GlaxoSmithKline, MAP Pharmaceuticals, Merck & Co. Inc., NuPathe, Novartis, Ortho-McNeil, and Zogenix, via grants to the National Headache Foundation. Aubrey Manack Adams, PhD, is an Allergan Inc. employee and receives stock options. Daniel Serrano, PhD, is an employee of Endpoint Outcomes. This work was completed while serving as a consultant for Vedanta Research, and he has received support funded by Allergan Inc., Colucid, Endo Pharmaceuticals, GlaxoSmithKline, MAP Pharmaceuticals, Merck & Co. Inc., NuPathe, Novartis, and Ortho-McNeil, via grants to the National Headache Foundation. Michael L. Reed, PhD, Valerie Marske, and Kristina M. Fanning, PhD, are employees of Vedanta Research, which has received support funded by Allergan Inc., Colucid, Endo Pharmaceuticals, GlaxoSmithKline, MAP Pharmaceuticals, Merck & Co. Inc., NuPathe, Novartis, and Ortho-McNeil, via grants to the National Headache Foundation.
Clinicaltrials.gov identifier: NCT01648530
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
