Abstract
Abstract
Objectives
Grading of Recommendations, Assessment Development and Evaluation (GRADE) tables were created using a standardized and independent assessment of the efficacy and side effects of treatments with monoclonal antibodies (mAb) against calcitonin gene-related peptide (CGRP) or the CGRP receptor for the prevention of migraine. We hope to provide support for author groups writing national or regional treatment or management guidelines for migraine prevention.
Methods
We formulated patient/population, intervention, comparison and outcomes (PICO) questions for the efficacy and safety of mAb against CGRP or the CGRP-receptor for the prevention of migraine attacks. We performed a systematic literature research for randomized studies with eptinezumab, erenumab, fremanezumab and galcanezumab and a pooled analysis was done, using RevMan 5.4 software. For dichotomous outcomes we used risk ratio, and for continuous outcomes we used the mean difference to compare and summarize the evidence between groups. The evidence across studies, for each outcome, except serious adverse events, was assessed using GRADE evidence tables. Additionally, we report the serious adverse effects in the tables of the characteristics of the studies.
Results
All mAb are superior to placebo for the reduction in monthly migraine days (days in which a headache consistent with migraine occurred) in participants with episodic and chronic migraine. There are no major differences between the mAb.
Conclusions
The GRADE evidence summary tables provided will support author groups to write treatment guidelines for the prevention of migraine with mAb.
Introduction
In the last 20 years there have been significant advances in the treatment of acute attacks of migraine and pharmaceutical prevention of migraine attacks. This means that existing national or regional guidelines for the treatment of migraine must be regularly updated. Guidelines’ panels must aim to make recommendations that are evidence based. The scientific evidence and the strength of the recommendations should be based on the Grading of Recommendations, Assessment Development and Evaluation (GRADE) system (1). The GRADE system enables analysis of evidence and assesses its quality across studies in an unbiased and transparent manner. The results are then used to make statements regarding the certainty (confidence) of the evidence. In guideline production, based on the certainty of the evidence, panels can issue recommendations that may be strong or weak (1).
The International Headache Society (IHS) would like to support authors who draft guidelines for the acute treatment of attacks of migraine and those for the prevention of attacks and associated features of migraine by providing GRADE evidence tables. To begin, the working group performed a systematic literature search and reviewed the scientific evidence on the efficacy, tolerability and safety of monoclonal antibodies (mAb) against calcitonin gene-related peptide (CGRP) or the CGRP-receptor for the prevention of migraine. This report differs from recent meta-analyses (2,3) by providing GRADE tables. In this publication, the corresponding GRADE tables are presented. They will also be made available on the IHS website.
The working group will create further tables for the prevention of attacks of migraine and the treatment of attacks of migraine and make them available to author groups via the website of the International Headache Society.
Methods
Formulation of clinical questions
We formulated clinical questions using the Patient Intervention Comparator and Outcome (PICO) format during several online meetings and discussion with the authors. We compared each treatment against placebo.
For each treatment, we had two PICO questions, aiming at episodic or chronic migraine participants (P). Chronic Migraine (CM) is defined by ICHD-3 (4), while Episodic Migraine (EM) is defined by default as a migraine whose frequency is less than 15 days/month (5). EM is not defined specifically in ICHD-3, as the attacks of migraine by definition are episodic. The nomenclature of EM and CM was used for all the CGRP mAb studies, whereas older studies did not make this differentiation. In order to assist with comparisons, we used the same outcomes for each question, which were selected upon a discussion about their clinical importance with the task force. Given this, the PICO questions follow the format reported in Table 1.
Format of the PICO questions.

The PICO questions were as follows: PICO 1: In participants with EM, is the treatment with the mAb against CGRP eptinezumab more effective than placebo for prevention of attacks of migraine? PICO 2: In participants with CM, is the treatment with a mAb against CGRP eptinezumab more effective than placebo for prevention of attacks of migraine? PICO 3: In participants with EM, is the treatment with a mAb against CGRP fremanezumab more effective than placebo for prevention of attacks of migraine? PICO 4: In participants with CM, is the treatment with a mAb against CGRP fremanezumab more effective than placebo for prevention of attacks of migraine? PICO 5: In participants with EM, is the treatment with a mAb against CGRP galcanezumab more effective than placebo for prevention of attacks of migraine? PICO 6: In participants with CM, is the treatment with a mAb against CGRP galcanezumab more effective than placebo for prevention of attacks of migraine? PICO 7: In participants with EM, is the treatment with a mAb against the CGRP receptor erenumab more effective than placebo for prevention of attacks of migraine? PICO 8: In participants with CM, is the treatment with a mAb against the CGRP receptor erenumab more effective than placebo for prevention of attacks of migraine?
Literature search and data extraction
We searched Pubmed, Scopus and the Cochrane database for the following search strategy:
Terms dedicated to the condition in question (migraine) Terms dedicated to prevention of attacks of migraine Terms dedicated to the corresponding intervention for each PICO
The exact search terms for each PICO are presented in Online Supplementary Material 2.
Inclusion criteria were randomized controlled studies, meta-analyses or systematic reviews including participants with EM or CM (according to each corresponding PICO), and comparing the effect of the corresponding treatments for each PICO vs placebo on any of the outcomes reported above. There were no limitations on the language or the study period. We analyzed the dosages of the drugs that are approved by the Food and Drug Administration (FDA) and European Medicines Agency (EMA).
Exclusion criteria were open label studies, studies including secondary analyses of randomized controlled trials (RCTs) or post hoc analysis, observational studies, studies on headaches other than migraine, studies including only non-approved dosages of the drugs, narrative reviews, conference abstracts and position papers.
The literature search was done for each treatment by two authors independently and if disagreements were met, the final decision was made by a discussion with a third reviewer.
Data extraction and analysis or the evidence
The following data were extracted from the papers, using a pre-defined excel sheet: author, year of publication, country, total number of participants, female percentage, mean age, type of migraine, dose of the drugs, follow-up time, mean number of day with attacks consistent with migraine (“mean migraine days” [MMD]) at baseline, change of MMD after treatment, serious adverse events, other adverse events (Online Supplementary Material 1). The term MMD was used in most studies to refer to a day in which an attack occurred that met ICHD criteria for an attack consistent with migraine. There were some reports of “mean headache days” in which any day in which an attack of headache occurred in a participant with a diagnosis of migraine was assessed.
The risk of bias of the eligible studies was assessed using the Cochrane risk of bias tool for randomized controlled studies (ROB2) (6) by two authors independently. After each assessment, the results of each author were compared and if incongruences were met, they were resolved by a discussion among the authors. The results of the risk of bias assessment were reported in dedicated tables (Online Supplementary Figures 1–7).
Analysis of the evidence
For the PICOs in which more than one study was available, and if it was considered appropriate, a pooled analysis was done, using the RevMan 5.4 software. For the dichotomous outcomes we used the risk ratio (RR) and for continuous outcomes the standardized mean difference (SMD) to compare and summarize the evidence between groups. The confidence intervals (CIs) were reported for each outcome and the heterogeneity between studies was assessed with I2.
Furthermore, the evidence across studies for each outcome, except serious adverse events, was assessed using GRADE evidence tables. Additionally, we report the serious adverse effects in the tables of the characteristics of the studies.
Results
The literature search for all PICOs was done between May and July 2021, and an update was done in October 2022. This included all primary studies in adults for the CGRP mAb and no further studies have been reported.
PICO 1 and 2
We found recent, good quality systematic reviews (7–10) with similar aims and search strategies as ours, and no additional studies were found after the last search dates in those papers. From these reviews, three studies were eligible for inclusion in our analysis (11–13). The characteristics of the included studies are summarized in the Online Supplementary Material 1.
A pooled analysis was not possible for PICO 1 because there was only one study available, and for PICO 2, a pooled analysis was possible only for the 100 mg dose. The meta-analysis showed a statistically significant difference between eptinezumab and placebo for the change of the mean monthly days of attacks with migraine from baseline (SMD: −0.36 CI: −0.49, −0.24) (Online Supplementary Figure 1).
The outcome parameter MMD per month or three months is presented in the publications and the GRADE evidence assessments for participants with EM and CM are presented in Tables 2 and 3 accordingly. The evidence for both PICOs showed statistically significant change in mean monthly migraine days, compared to placebo (adjusted for baseline values), but the certainty of the evidence is low, due to imprecision because of the limited number of participants and possible publication bias, as well as ambiguity of the outcome for PICO 2 (Table 2 and 3).
Eptinezumab compared to placebo for episodic migraine.

Notes: a, only one study; b, low number of patients, one study; c, only one, industry sponsored study available. CI, confidence interval; MD, mean difference.
Eptinezumab compared to placebo for chronic migraine.

In EM patients, 11 participants from the treatment group and six from the placebo group reported serious adverse events (SAEs), but none was considered treatment-related (12). In CM in one of the studies, one SAE was reported in the placebo group and five in the treatment group; none considered related to the study drug (11) and in the other study three participants that received placebo and seven that received eptinezumab had SAEs, one of which was considered drug-related (worsening of the visual symptoms in a patient receiving 300 mg eptinezumab) (13).
In conclusion, eptinezumab was superior to placebo for the prevention of migraine in episodic and chronic migraine. The certainty of evidence was low.
PICO 3 and 4
The results of the literature search for PICO 3 and 4 on fremanezumab are summarized in Online Supplementary Material 1. From a total of 426 studies initially screened, six were considered eligible for inclusion. Four of those included EM participants (PICO 3) and three of them CM participants (PICO 4) (14–19). The characteristics of the individual studies included are presented in Online Supplementary Material 1.
The pooled analysis of the studies, for the outcome “mean monthly migraine days”, showed high heterogeneity (I^2 > 80%), which was resolved upon dividing the effect estimates by the mode of the administration of the drug into monthly and quarterly drug administration, for both PICOs, and excluding one study that recruited refractory migraine participants (17) (Online Supplementary Figures 2.1–2.3). The pooled SMD for monthly and quarterly fremanezumab for EM was −0.56 (CI −0.80, −0.32) and −0.51 (CI −0.79, −0.23), respectively; and for monthly and quarterly fremanezumab for CM −0.44 (CI −0.56, −0.32) and −0.36 (CI −0.52, −0.20), respectively, with unexplained moderate heterogeneity for EM. All estimates were adjusted for baseline mean monthly migraine days.
Again, only the outcome of MMD was presented in the publications. The GRADE evidence assessments for EM and CM participants are presented in Tables 4 and 5, accordingly. The certainty of the evidence is very low for EM and low for CM, due to the variability of the effect estimates among studies (for EM) and indirectness due to the short follow-up and possible publication bias (for both migraine types). (Table 3 and 4)
Fremanezumab compared to placebo for episodic migraine.

Notes: a, variability in the SMD among the studies; b, length of follow up in all studies was 12 weeks and a longer follow-up could provide more relevant results; c, only sponsored studies available. CI: confidence interval; SMD: standardized mean difference.
Fremanezumab compared to placebo for chronic migraine.
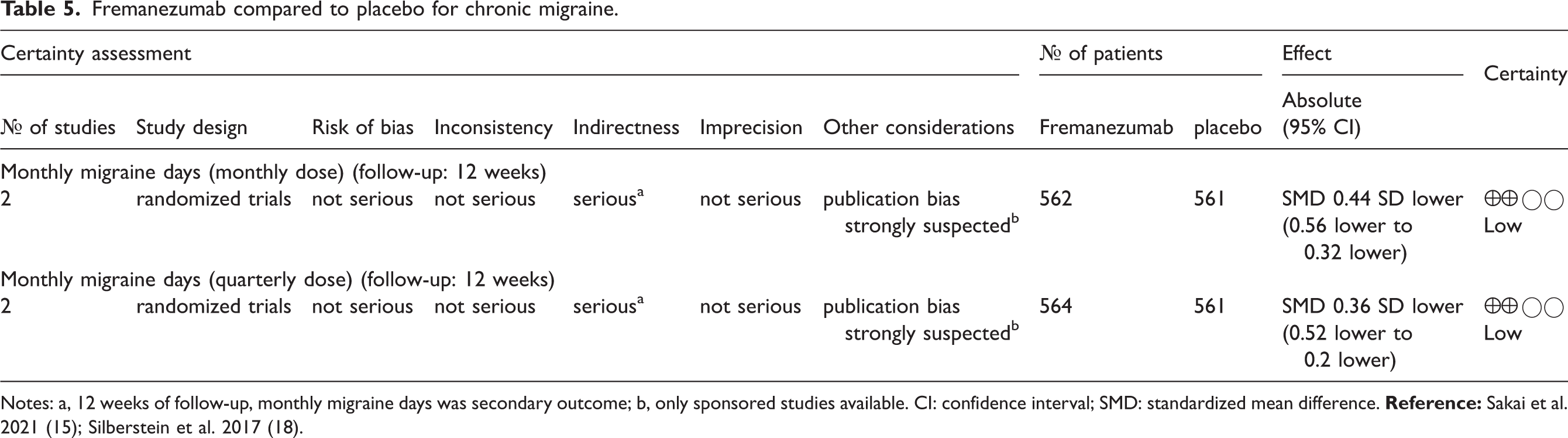
The SAE are reported along with the characteristics of the included studies (Online Supplementary Material 1). Overall, no important differences between placebo and intervention groups were noted.
In conclusion, fremanezumab was superior to placebo for the prevention of migraine in episodic and chronic migraine. The certainty of evidence is low.
PICO 5 and 6
The results of the literature search for PICO 5 and 6 for galcanezumab are summarized in Online Supplementary Material 2. From a total of 306 studies initially screened, seven studies were considered eligible for inclusion, six of those for EM (PICO 5) and two of them for CM (PICO 6). One study was subsequently excluded from the analysis because it did not include a loading dose of the drug (32). The characteristics of the individual studies included are presented in Online Supplementary Material 1.
The pooled analysis of the studies for both PICOs showed moderate to high heterogeneity, which could not be resolved upon adjusting the analysis by dosage of the drug and follow-up time (Online Supplementary Figures 6.1–6.3 and 7.1 and 7.2). The results for EM, for three-month follow-up and 120 mg dose, were −0.60 [−0.85, −0.35] (SMD [CI]), with moderate heterogeneity, but the heterogeneity was high among the studies of six-month follow-up and 240 mg (Online Supplementary Figures 6.2 and 6.3). All estimates were adjusted for baseline mean monthly migraine days.
Again, only the outcome MMD was included in the publications and the GRADE evidence assessments for EM and CM participants are presented in Tables 6 and 7 accordingly. The certainty of the evidence is low for both PICOs due to inconsistency, the variability of the effect estimates among studies and possible publication bias (Tables 6 and 7).
Galcanezumab compared to placebo for episodic migraine.

Notes: a, unexplained moderate heterogeneity among studies, that did not change substantially after dividing the studies by follow-up time; b, possible publication bias (recent drug, mainly industry-sponsored studies available); c, substantial heterogeneity among studies (I^2: 76%). CI: confidence interval; SMD: standardized mean difference.
Galcanezumab compared to placebo for chronic migraine.

Notes: a, substantial heterogeneity (I^2: 77%); b, only two industry-sponsored studies available; c, only one study; d, only one study, lower point of CI close to 0; e, only industry study available. CI: confidence interval; SMD: standardized mean difference.
Serious adverse effects are presented in the table of the study characteristics (Online Supplementary Material 1).
In conclusion, galcanezumab was superior to placebo for the prevention of migraine in episodic and chronic migraine. The certainty of evidence is low.
PICO 7 and 8
The results of the literature search for PICO 7 and 8 for erenumab are summarized in Online Supplementary Material 2. From a total of 676 studies initially screened, eight were considered eligible for inclusion, six of those for EM (PICO 7) and two of them for CM (PICO 8) (20–27). The characteristics of the individual studies included are presented in Online Supplementary Material 1.
The pooled analysis of the studies for PICO 7 showed high heterogeneity, which was partially resolved upon adjusting the analysis by dosage of the drug and follow-up time (Online Supplementary Figures 4.1–4.3 and 5.1–5.3). The meta-analysis showed statistically significant decrease in mean monthly attacks of migraine for EM participants for both 70 and 140 mg and 12 and 24 weeks, but the results for 140 mg should be taken with caution because of the unexplained heterogeneity in the longer follow-up groups (Online Supplementary Figures 4.1–4.3 and 5.1–5.3). All estimates were adjusted for baseline mean monthly migraine days.
Again, only the outcome MMD was presented in the papers and the GRADE evidence assessments for episodic and CM participants are presented in Tables 7–9. The certainty of the evidence for EM participants is medium for the 70 mg and 24 weeks follow-up group, with a possible publication bias because only two industry sponsored trials were available (Table 7). The certainty of the evidence for the 140 mg dose was low and very low for the 12 and 24 weeks follow-up, accordingly, because of the high heterogeneity between studies, imprecision, and possible publication bias (Table 8).
Erenumab 70 mg compared to placebo for episodic migraine.

Notes: a, only 12 weeks follow-up; b, only studies sponsored by pharmaceutical companies available. CI: confidence interval; SMD: standardized mean difference.
Erenumab 140 mg compared to placebo for episodic migraine.

Notes: a, 12 months follow-up; b, industry sponsored studies only available; c, considerable heterogeneity; d, pooled analysis not relevant due to heterogeneity. CI: confidence interval; SMD: standardized mean difference.
In conclusion, erenumab was superior to placebo for the prevention of migraine in episodic and chronic migraine. The certainty of evidence is low.
For PICO 8, a pooled analysis was not possible because of the different follow-up times in the two eligible studies.
One study showed statistically significant decrease of MMD compared to placebo, for both dosages and follow up of 12 weeks, but the other one showed no statistically significant change in MMD, for a follow-up of 24 weeks. However, the certainty of the evidence for all outcomes was low due to the short follow-up (23), small number of participants (26), and possible publication bias because both of the studies were sponsored by the industry (Table 10).
Erenumab (140 mg and 70 mg) compared to placebo for chronic migraine.
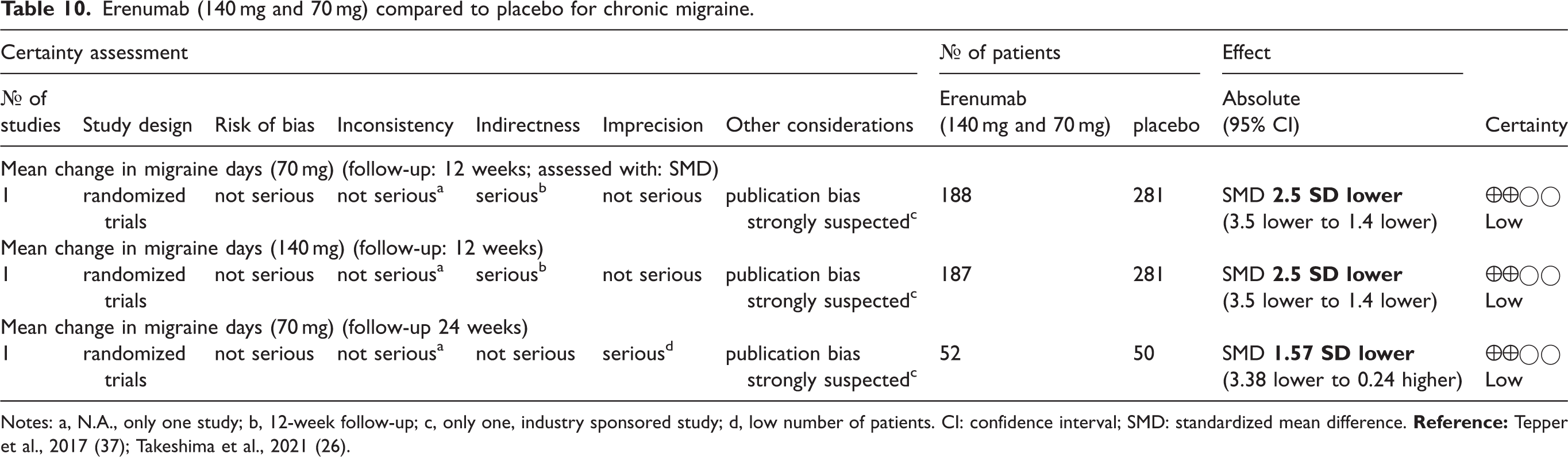
SAEs are reported in the table of the characteristics of the studies (Online Supplementary Material 1).
Discussion
The analysis of the randomized placebo-controlled trials with monoclonal antibodies against CGRP or the CGRP-receptor shows superiority over placebo and a good safety profile. We present GRADE tables that can be used by guideline panels to write treatment guidelines and recommendations for the prevention of EM and CM with mAb against CGRP or CGRP receptor. As new studies are developed and reported, the tables will be updated on a yearly basis in the future. We compared each mAb with placebo given the absence of head-to-head trials. There is only one study which compared a mAb (erenumab) with topiramate and the primary endpoint was not the reduction in monthly migraine days (28). A network meta-analysis was done independently by another working group of IHS and was published in Cephalalgia (29). In addition, a detailed analysis of safety and tolerability of the mAb was performed and published in Cephalalgia (2). The treatments from which we created GRADE tables are recent and all relevant placebo-controlled trials were supported by industry. Academics were, however, involved in the design and conduct of these studies. Industry-independent randomized controlled studies are clearly warranted. Future high-quality RCTs can hopefully provide results with higher certainty of the evidence, providing longer follow-up and more consistent results.
Key findings
Based on a systematic literature search we created GRADE tables for the efficacy of mAb against CGRP or the CGRP-receptor for the prevention of EM and CM. All mAb are superior to placebo for the reduction in monthly migraine days in participants with episodic and CM. There are no major differences between the mAb in terms of efficacy.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231206162 - Supplemental material for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society
Supplemental material, sj-pdf-1-cep-10.1177_03331024231206162 for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society by Katina Aleksovska, Andrew D. Hershey, Marie Deen, Robert de Icco, Mi Ji Lee and Hans-Christoph Diener in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024231206162 - Supplemental material for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society
Supplemental material, sj-pdf-2-cep-10.1177_03331024231206162 for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society by Katina Aleksovska, Andrew D. Hershey, Marie Deen, Robert de Icco, Mi Ji Lee and Hans-Christoph Diener in Cephalalgia
Supplemental Material
sj-pdf-3-cep-10.1177_03331024231206162 - Supplemental material for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society
Supplemental material, sj-pdf-3-cep-10.1177_03331024231206162 for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society by Katina Aleksovska, Andrew D. Hershey, Marie Deen, Robert de Icco, Mi Ji Lee and Hans-Christoph Diener in Cephalalgia
Supplemental Material
sj-pdf-4-cep-10.1177_03331024231206162 - Supplemental material for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society
Supplemental material, sj-pdf-4-cep-10.1177_03331024231206162 for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society by Katina Aleksovska, Andrew D. Hershey, Marie Deen, Robert de Icco, Mi Ji Lee and Hans-Christoph Diener in Cephalalgia
Supplemental Material
sj-pdf-5-cep-10.1177_03331024231206162 - Supplemental material for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society
Supplemental material, sj-pdf-5-cep-10.1177_03331024231206162 for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society by Katina Aleksovska, Andrew D. Hershey, Marie Deen, Robert de Icco, Mi Ji Lee and Hans-Christoph Diener in Cephalalgia
Supplemental Material
sj-pdf-6-cep-10.1177_03331024231206162 - Supplemental material for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society
Supplemental material, sj-pdf-6-cep-10.1177_03331024231206162 for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society by Katina Aleksovska, Andrew D. Hershey, Marie Deen, Robert de Icco, Mi Ji Lee and Hans-Christoph Diener in Cephalalgia
Supplemental Material
sj-pdf-7-cep-10.1177_03331024231206162 - Supplemental material for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society
Supplemental material, sj-pdf-7-cep-10.1177_03331024231206162 for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society by Katina Aleksovska, Andrew D. Hershey, Marie Deen, Robert de Icco, Mi Ji Lee and Hans-Christoph Diener in Cephalalgia
Supplemental Material
sj-pdf-8-cep-10.1177_03331024231206162 - Supplemental material for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society
Supplemental material, sj-pdf-8-cep-10.1177_03331024231206162 for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society by Katina Aleksovska, Andrew D. Hershey, Marie Deen, Robert de Icco, Mi Ji Lee and Hans-Christoph Diener in Cephalalgia
Supplemental Material
sj-pdf-9-cep-10.1177_03331024231206162 - Supplemental material for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society
Supplemental material, sj-pdf-9-cep-10.1177_03331024231206162 for Efficacy and safety of monoclonal antibodies targeting CGRP in migraine prevention. GRADE tables elaborated by the ad hoc working group of the International Headache Society by Katina Aleksovska, Andrew D. Hershey, Marie Deen, Robert de Icco, Mi Ji Lee and Hans-Christoph Diener in Cephalalgia
Footnotes
Author contributions
KA and HCD drafted the manuscript. Two sets of authors performed the literature search for a particular mAb and created data extraction tables. KA calculated the RR and created the figures and GRADE tables. All authors approved the final manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KA received grant from IHS for her work on the analysis. ADH received grants from NIH/NINDS/NICHDS, advisory (paid to institution) to and grants/contracts from Amgen, Allergan, Biohaven/Pfizer, Lilly, Lundbeck, Supernus, Teva, Theranica, and Upsher-Smith, speaker for Theranica, writer for Up-to-date. MD no conflict of interest. RDI received honoraria for scientific presentations from Eli-Lilly, and Teva. MJL has received personal or institutional honoraria for participation in clinical trials, speaker fees, or as consultants from Abbvie, Biohaven, Eli Lilly, Lundbeck, Novartis, Sanofi-Aventis, SK Pharm, Teva (Otsuka), Yuhan Company, and YuYu Pharma, and research support from the National Research Foundation of Korea and Seoul National University. HCD: In the last three years HCD received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Lilly, Novartis, Pfizer, Teva, Weber & Weber and WebMD. The German Research Council (DFG), the German Ministry of Education and Research (BMBF) and the European Union support his headache research. HCD serves on the editorial boards of Cephalalgia, Lancet Neurology and Drugs. HCD is a member of the Clinical Guidelines Committee of the German Society of Neurology and of the Clinical Trials Committee of the IHS.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Scientific Committee of the International Headache Society.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
