Abstract
Background
A new migraine prevention, CGRP monoclonal antibodies (mAbs), is injectable on a monthly or quarterly basis. In clinical practice, some patients reported that drug effectiveness does not last until the upcoming scheduled injection, a so-called “wearing-off” effect. We aimed to evaluate the wearing-off effect of the CGRP mAbs for migraine prevention in patients with different monthly migraine days.
Methods
We conducted a literature search for studies that reported migraine frequency after CGRP monoclonal antibody administration from MEDLINE, SCOPUS, Web of Science, and Cochrane Database from inception through February 2022. A meta-analysis, random-effects model was applied to assess the difference in migraine frequency between early and later weeks after medication to assess the presence of a wearing-off effect. Risk ratio was calculated to report the pooled treatment effect.
Results
Four studies were entered for the analysis, comprising 2409 patients in randomized controlled trials. There was no association between CGRP mAbs and wearing-off effect in patients with galcanezumab with a pooled risk ratio of 1.29 (95% CI 0.73 to 2.28) compared to placebo group. However, there was an association between galcanezumab and wearing-off effect in patients with chronic migraine with a pooled risk ratio of 1.91 (95% CI 1.11 to 3.28) compared to placebo group.
Conclusion
In this meta-analysis, there was a wearing-off efficacy of galcanezumab but only in a small percentage of patients with chronic migraine in randomized controlled trials.
Introduction
Migraine is a severely disabling neurological disease and is associated with significant morbidity and reduced quality of life (1,2). We can categorize migraine into two groups, episodic migraine (EM) and chronic migraine (CM), using the frequency of migraine/headache days per month as less than 15 days, or at least 15 days. (3). EM itself can be divided into three subgroups in accordance with the monthly migraine days (MMDs), including 0–3 MMDs as very low frequency (VLF) EM, 4–7 MMDs as low frequency (LF) EM, and 8–14 MMDs as high frequency (HF) EM (4).
Treatment of migraine consists of two key objectives: to alleviate and to prevent migraine attacks. Migraine acute attack therapies aim to terminate the pain and related symptoms, while migraine preventive attack therapies aim to suppress the frequency, duration, severity and disability of pain episodes; improve acute treatment responsiveness and avoid escalation; enhance function and reduce disability; enhance health-related quality of life; decrease headache-associated suffering and psychological problems (5).
Currently, migraine prevention should be proposed to patients with at least six headache days per month, or at least four headache days per month with some degree of disability, or at least three headache days per month with a severe degree of disability (5).
To date, standard migraine prevention includes oral medications and CGRP-targeted treatments for EM and CM, as well as onabotulinumtoxinA for CM. CGRP-targeted treatments, especially monoclonal antibodies (mAbs), aim to target the CGRP pathway, which is definitely the goal of migraine pathophysiology (6). They are more advantageous than oral migraine prevention due to a monthly or quarterly injection (three monthly), which is more compliant than the oral formulation.
Due to CGRP mAbs, a newer class of migraine prevention, the American Headache Society published an agreement on the utilization of CGRP mAbs for patients with LFEM who have an intolerable or insufficient improvement to two or more oral migraine preventions with at least moderate disability, for patients with HFEM who have an intolerable or insufficient improvement to at least two oral migraine preventions, and for patients with CM who have an intolerable or insufficient improvement to at least two oral migraine preventions or onabotulinumtoxinA (5). The European Headache Federation released a guideline for the use of CGRP mAbs for patients with migraine who were unsuccessful in two or more of the accessible preventive drugs (7).
Currently, there are four products of CGRP mAbs available worldwide, including erenumab, a monthly injection; fremanezumab, a monthly and quarterly injection; galcanezumab, a monthly injection; and eptinezumab, a quarterly infusion. Among those four products, there are differences in pharmacokinetic properties, including half-life (8) and time of maximum observed drug concentration (Tmax) (9). Eptinezumab has the shortest half-life (26 days), fremanezumab has the longest half-life (32 days), and erenumab and galcanezumab are in the middle (28 and 27 days, respectively) (8). The Tmax of the four CGRP mAbs has the same trend as the half-life, including eptinezumab, the shortest (4.8 hours), fremanezumab, the longest period (3–20 days), and erenumab and galcanezumab are in the middle (3–14 days and 7–14 days, respectively) (9).
In clinical practice, some patients perceived inconsistent effectiveness over a one- or three-month period of CGRP mAbs before the upcoming scheduled injection. The term “wearing-off” effect has been expressed in some diseases, such as Parkinson's disease, when patients have an early good improvement to levodopa and then the effectiveness is shorter than expected (10). OnabotulinumtoxinA for CM is also observed to have a wearing-off effect, with clinical worsening occurring 8–10 weeks after an early positive improvement (11) and is supported by studies (12–14).
Differences in pharmacokinetic properties of the CGRP mAbs that were addressed earlier, and diversities in MMDs of the patients with migraine, and the number of unresponsiveness CGRP mAbs may be the result of a wearing-off effect. However, data on the presence of wearing-off are also lacking. Only three observational studies were published, with a wide range of wearing-off effects of 9–35% due to different definitions of the wearing-off effect and different single center (15–17). Furthermore, five post-hoc studies reported a difference in migraine days between weeks 1–2 and weeks 3–4 or between weeks 1 and 4, with also differences in the definition of wearing-off effect (18–22) and then high variability of results. Therefore, our objective is to study the wearing-off effect of the CGRP mAbs for prevention in migraine patients with different MMDs in randomized controlled trials.
Methods
Systematic review and search strategy
A systematic search of the literature from MEDLINE (1946 to February 2022), SCOPUS (1988 to February 2022), Web of Science (1900 to February 2022), and the Cochrane Database of Systematic Reviews (database inception to February 2022) was studied to evaluate the differences in the number of weekly migraine/headache days from baseline in randomized controlled trials.
The systematic review of the literature was conducted autonomously by two authors (TA and RC) implementing a search strategy that integrated the words “wearing off”, “monoclonal antibody” and “migraine” combined together (presented in online Supplementary Data 1). A hand-operated manual search for potentially pertinent articles from reference lists of the covered articles and from the proceedings of the annual scientific meetings of the International Headache Society, the European Headache Federation, the American Headache Society, and the American Academy of Neurology, years 2019–2021, was also operated. No language restriction was administered. The study was followed by the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement (23).
Selection criteria
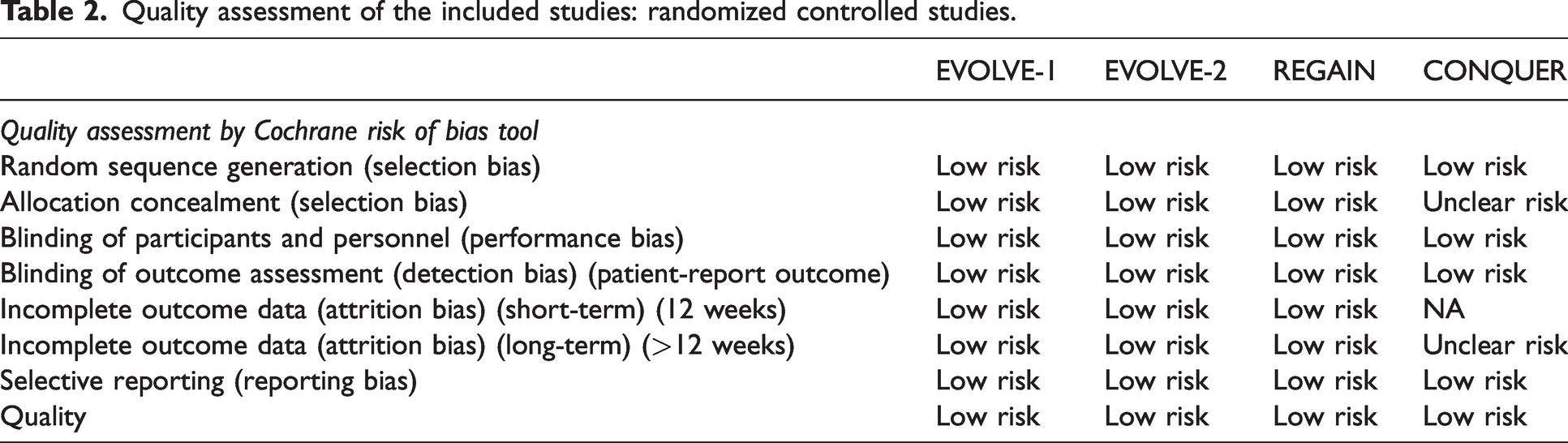
Included studies must be randomized controlled studies. They must present data on clinical features, type of migraine patient, medications and dosage, and the number of migraine days in subsequent periods. Inclusion was not restricted to the number of participants in the studies. The two authors (TA and RC) were blinded and autonomously examined the quality of the retrieved articles. Divergences were debated and a solution was made by a third author (SS). As demonstrated in Table 1 and Table 2, the Cochrane Collaboration's tool was utilized for evaluating the risk of bias in randomized trials.
Characteristics of the included studies: randomized controlled studies.

Abbreviations: CM, chronic migraine (≥15 monthly headache days of which ≥8 are migraine headache days); GMB, galcanezumab; HFEM, high-frequency episodic migraine (8–14 monthly migraine headache days and <15 monthly headache days; LFEM, low-frequency episodic migraine (4–7 monthly migraine headache days); PBO, placebo; EHD, electronic headache diary; EM, episodic migraine; EMR, electronic medical records; CM, chronic migraine; HF, high frequency; LF, low frequency; MHDs, monthly headache days; MMDs, monthly migraine days; OLE, open label extension; RDB, randomized double-blind; WMDs, weekly migraine days.
aMedication categories included propranolol or metoprolol, topiramate, valproate or divalproex, amitriptyline, flunarizine, candesartan, and onabotulinumtoxinA (if taken for CM).
Quality assessment of the included studies: randomized controlled studies.
Data abstraction
A structured form of data collection was applied to extract data from individual studies as follows: title, first author's name, year of the study, publication year, country where the study was carried out, demographic data and characteristics of migraine patients, and number of migraine/headache days before and after mAb administration. If essential data were unavailable, we corresponded with the authors to request missing information. We performed analysis using available data if the authors did not respond within two weeks.
Statistical analysis
The analyses were performed using the STATA software version 17.0. Point values from individual covered studies were adjusted and incorporated by DerSimonian and Laird method (random effect model), which constructed the load of an individual study on the basis of its variance if heterogeneity was revealed and fixed effect model was constructed if heterogeneity not detected (24). To define the heterogeneity between-studies, the I2 statistic was administered. Heterogeneity was defined by the I2 value as follows: 76–100% as high heterogeneity, 51–75% as moderate heterogeneity, 26–50% as low heterogeneity, and 0–25% as insignificant heterogeneity (25). If insignificant heterogeneity was observed, the fixed-effect model was calculated for pooled effect values; otherwise, random-effect model was applied. Publication bias was investigated using both funnel plots and the Egger test (26). If there were any small study effects, Galbraith plots would be applied to identify potential outliers and assess heterogeneity (27) and leave-one-out meta-analysis would be performed by excluding one study that produced exaggerated effect sizes which could distort the overall results at each analysis.
Results
In total 208 records were identified by utilizing our search schemes. After the removal of the duplex records, animal studies, in vitro studies, pediatric patients, correspondences, case reports, review articles, and articles not fulfilling inclusion criteria, six articles remained for complete evaluation (28–33). Two were removed after complete evaluation due to the absence of the outcomes of interest (28,29).
The final assessment, therefore, covered four randomized controlled trials (30–33). The articles identification, screening, eligibility and inclusion are shown in Figure 1. The characteristics and evaluation of the methodology quality of the covered articles are shown in Table 1 and Table 2.

Flow diagram for meta-analysis of randomized controlled trials.
Wearing-off effect of CGRP mAbs from randomized controlled studies
An analysis of the wearing-off effect from randomized controlled trials was extracted data from the Ailani study (22), which reported the rate of patients who perceived wearing-off in the intervention and control groups from EVOLVE-1 (30), EVOLVE-2 (31), CONQUER (32), and REGAIN (33) studies. EVOLVE-1 and EVOLVE-2 were studied with galcanezumab for EM with three-group comparison including galcanezumab 120 mg, galcanezumab 240 mg, and placebo group, REGAIN for CM with three-group comparison including galcanezumab 120 mg, galcanezumab 240 mg, and placebo group, and CONQUER for both EM and CM with resistance to migraine prevention with two-group comparison including galcanezumab 120 mg and placebo group. Dose of galcanezumab among those studies was either 120 or 240 mg injected monthly. In that study, the definition of wearing-off effect was as a rise of two or more headache days per week compared between week 2 and week 4 in two or more treatment months. The week 2 periods included eight to 14 days after medication, and the week 4 periods was determined as the ultimate seven days prior to the upcoming therapy. Only 2409 patients from those studies had data for analysis of the wearing-off effect. The EVOLVE-1 and EVOLVE-2 for HFEM results were combined together as one study due to unavailable raw data to demonstrate the wearing-off effects from individual studies of EVOLVE-1 and EVOLVE-2. In this study, we used merged data of the EVOLVE-1 and EVOLVE-2 from the Ailani study (22) and demonstrated that there was no significant difference of wearing-off effect between the galcanezumab and the control group in individual study and no significant difference of overall wearing-off effect between the galcanezumab and the control group with a pooled risk ratio (RR) of 1.29 (95% CI 0.73 to 2.28, I2 = 58.18%; Figure 2).

Risk ratio of wearing off effect from randomized controlled studies* among patients with galcanezumab and control.
Galbraith plots and leave-one-out meta-analysis
To examine small studies effects, Galbraith plots were executed to detect potential outliers and assess heterogeneity (Figure 3). The Galbraith plot of three studies, due to unavailable raw data from the original studies of EVOLVE-1 and EVOLVE-2, then merged data from the Ailani study being used, did not demonstrate outliers and heterogeneity. Leave-one-out meta-analysis was also used to show how each individual study affects the overall estimate of the rest of the studies by removing the small sample size studies from the CONQUER study and it still exhibited the same result rate of wearing-off effect in migraine patients between galcanezumab and placebo in randomized controlled trial studies (Figure 4). However, after the removal of the merged data of EVOLVE-1 and EVOLVE-2 for HFEM from the Ailani study due to unavailable raw data from the individual original studies of EVOLVE-1 and EVOLVE-2, the wearing-off effect was revealed to be significantly higher in the galcanezumab group with a pooled RR of 1.78 (95% CI 1.08 to 2.92) (Figure 4).
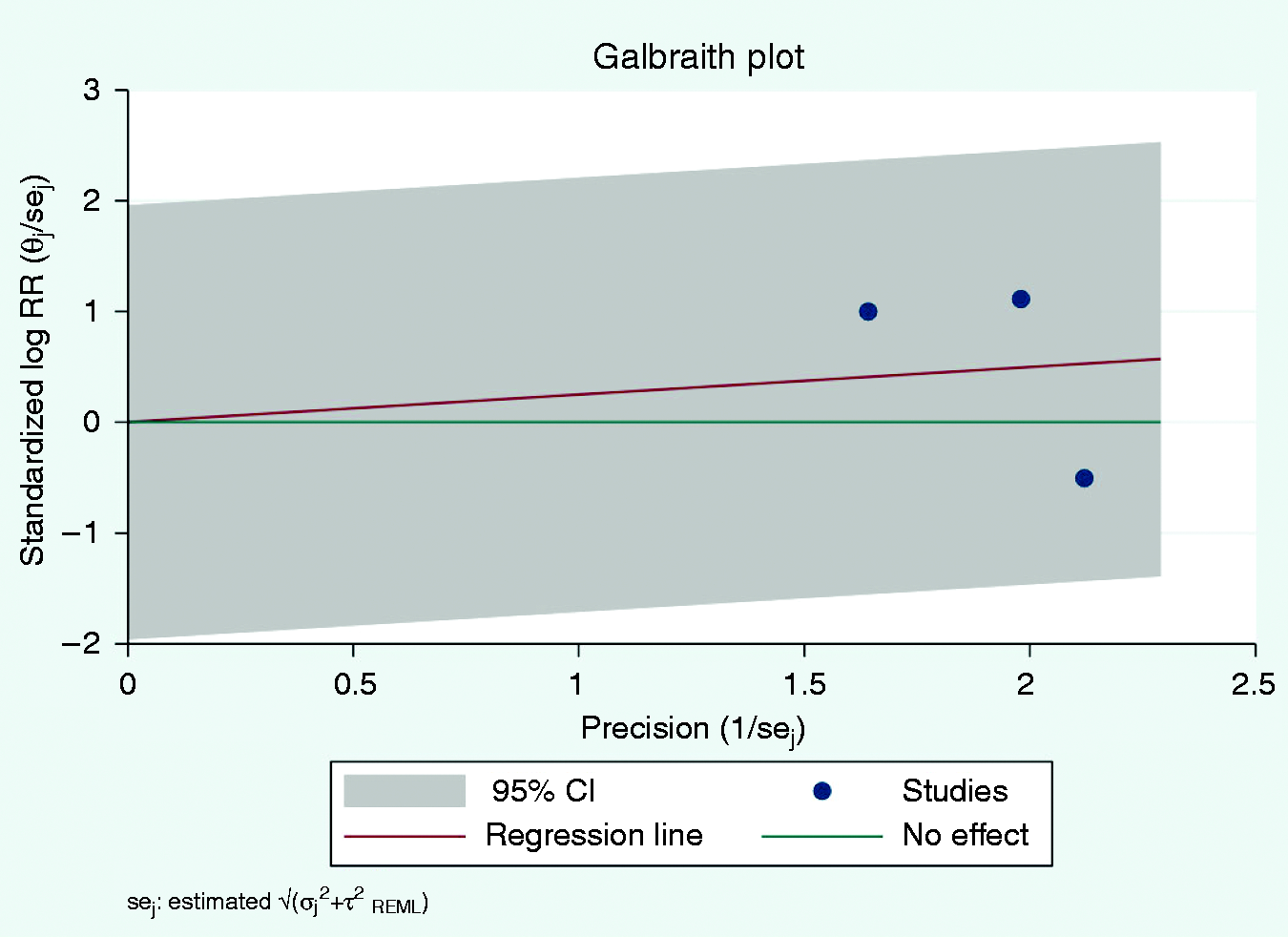
Galbraith plot.*

Leave-one-out meta-analysis.*
Meta-regression and subgroup analysis
Due to heterogeneity of the overall wearing-off effect between the galcanezumab and control groups (Figure 2), meta-regression was, therefore, performed to identify which variables caused variability of the results including migraine subtypes, HFEM or CM and galcanezumab dose, 120 or 240 mg. it revealed that CM had a significantly higher wearing-off effect than those with HFEM (Figure 5) but no significant difference between doses of 120 and 240 mg.

Meta-regression.
Subgroup analysis of chronic and episodic migraine also revealed that in CM group, there was a significantly higher rate of wearing-off effect in galcanezumab compared to placebo group with a pooled RR of 1.91 (95%CI 1.11 to 3.28; I2 0.0%) but no significant difference in HFEM group with a pooled RR of 0.83 (95%CI 0.53 to 1.30; I2 0.0%) (Figure 6). However, subgroup analysis of galcanezumab dose 120 or 240 mg demonstrated no significant difference in wearing-off effect between galcanezumab and placebo (Figure 7).

Subgroup analysis of chronic and episodic migraine.

Subgroup analysis of galcanezumab dose.
Evaluation for publication bias
Funnel plots (online Supplementary Figure S1) and Egger's regression asymmetry tests were operated to assess for publication bias in analyses investigating the wearing-off effect in CGRP mAbs-treated migraine patients. There was no significant publication bias. For those that covered randomized controlled studies, we find that −0.30 is the intercept of the regression model without a significant level and a beta value greater than zero (beta = 3.75, p = 0.23), and confirms the asymmetry of the funnel plot. On the whole, this confirms our early discovery of small-study effects. Yet, to restate, it is ambiguous whether bias in publication caused this figure.
Retrospective statistical power analysis
Due to no a priori estimation of sample size and participant size per study, we calculate retrospective statistical power for this meta-analysis. The power for meta-analysis according to the number of included studies of 3, I2 of 58.18%, and delta (RR) of 1.29 was approximately 20% (34).
Discussion
The aim of migraine prevention is to cover effectiveness throughout the therapy period. This meta-analysis examines whether the wearing-off effect of CGRP mAbs at the final therapy period do exist, employing data from four RCTs. However, due to unavailable raw data from the individual studies of EVOLVE-1 and EVOLVE-2, merged data of those two studies from the Ailany study (22) was used for analysis. To our knowledge, this is the first study of meta-analysis to examine the wearing-off effect of CGRP mAbs. We report no association between CGRP mAbs and a wearing-off effect with a pooled RR of 1.29 (95% CI 0.73 to 2.28, I2 = 58.18%) but an association in chronic migraine with a pooled RR of 1.91 (95% CI 1.11 to 3.28) between those with galcanezumab compared to placebo group.
Before this study, five post-hoc analyses of fremanezumab and galcanezumab clinical trials (18–22) consistently confirmed the absence of evidence of wearing-off for patients with either EM or CM who received therapy every month or every three months. Contrastingly, our findings indicated that a wearing-off effect occurred in a small percentage of chronic migraine patients. Four previous post-hoc analyses of randomized studies (18–21) looked at the wearing-off effect of CGRP mAbs by examining differences in headache days comparing between the second week after the prior injection and the final week of the course of therapy. Given that wearing-off may develop only in a small group of migraine patients, these surged headache days from a small group of patients will simply be normalized in the analysis by a large number of suppressed headache days from patients who did not perceive the wearing-off phenomenon.
Another post-hoc study by Ailani et al. (22) tries to explore this effect with a different approach. They first specified wearing-off at the level of each patient and then compared the proportion of patients who perceived wearing-off among galcanezumab doses of 120 mg, 240 mg, and placebo at the final course of therapy by applying their prespecified threshold. However, that study showed an absence of evidence of reduced efficacy at the final course of therapy both for total patients or the subgroup of those with chronic or episodic migraine. It is notable that they used two-way analysis of variance (ANOVA) with treatment group to examine the wearing off effect. The use of two-way ANOVA to compare multiple treatment groups from multiple RCTs may underestimate the treatment effect than the use of meta-analysis in this situation.
In our study, we abstracted data from the Ailani study (22) for a random-effect meta-analysis. We report no association between CGRP mAbs and a wearing-off effect with a pooled RR of 1.29 (95% CI 0.73 to 2.28, I2 = 58.18%) but an association in chronic migraine with a pooled RR of 1.91 (95% CI 1.11 to 3.28) between those with galcanezumab compared to placebo group. Due to no a priori estimation of sample size and participant size per study, we calculated retrospective statistical power for this meta-analysis. The power for meta-analysis according to the number of included studies of 3, I2 of 58.18%, and delta (RR) of 1.29 was approximately 20% (34). In conclusion, that a wearing-off effect does exist but only in a small group of patients with chronic migraine.
Several important limitations of our study should be considered. First, there was statistical heterogeneity (I2 = 58.18%) between studies in meta-analysis examining association between CGRP mAbs and wearing off effect.
The between-study heterogeneity variance was estimated at τ2 = 0.14 with an I2 value of 58.18% (95%CI: 98–99%). The Q test showed an exaggerated number compared to what we expected based on the degree of freedom at 2. These implied an existence of between-study heterogeneity. The possible source of this heterogeneity includes the difference in population with either episodic or chronic migraine, dose of Galcanezumab and small study effects. To examine the heterogeneity in our study, Galbraith plots were executed to detect potential outliers and assess heterogeneity (Figure 3). The Galbraith plot did not demonstrate outliers and heterogeneity. In addition, Leave-one-out meta-analysis was also used to show how each individual study affects the overall estimate of the rest of the studies by removing the small sample size studies from the CONQUER study and it still exhibited the same result of the rate of the wearing-off effect in migraine patients between galcanezumab and placebo in randomized controlled trial studies (Figure 4). However, after the removal of the EVOLVE-1 and EVOLVE-2 for HFEM, the wearing-off effect was revealed significantly higher in the galcanezumab group with a pooled RR of 1.78 (95% CI 1.08 to 2.92) (Figure 4). Next, we performed meta-regression to identify which variables caused variability of the results including migraine subtypes, HFEM or CM and galcanezumab dose, 120 or 240 mg. This revealed that CM had a significantly higher wearing-off effect than those with HFEM (beta = 0.82, p = 0.008 [Figure 5]) but no significant difference between doses of 120 and 240 mg. From meta-regression, 100% of the variability in our data can be attributed to the type of migraine, either chronic or episodic. Nevertheless, we executed a random-effect model to incorporate this heterogeneity into the primary analyses in our study. Subgroup analysis of chronic and episodic migraine also performed to confirm that in CM group there was a significantly higher rate of wearing-off effect in galcanezumab compared to placebo group with a pooled RR of 1.91 (95%CI 1.11 to 3.28; I2 0.0%) but no significant difference in HFEM group with a pooled RR of 0.83 (95%CI 0.53 to 1.30; I2 0.0%) (Figure 6).
Moreover, our meta-analysis is based on published data from the Ailani study (22). Without access to individual patient data and data from RCTs of other CGRP mAbs such as fremanezumab, erenumab and eptinezumab, we were unable to perform additional analyses to examine whether wearing off phenomenon is exclusive to galcanezumab or not. Our finding has limited generalizability to other CGRP mAbs.
Key findings
We report no association between CGRP mAbs and a wearing-off effect with a pooled RR of 1.29 (95% CI 0.73 to 2.28, I2 = 58.18%) but an association in chronic migraine with a pooled RR of 1.91 (95% CI 1.11 to 3.28) between those with galcanezumab compared to placebo group. Our findings suggested that a wearing-off effect does exist but only in a small group of patients with chronic migraine.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231161261 - Supplemental material for “Wearing-off” efficacy of CGRP monoclonal antibodies for migraine prevention: A meta-analysis of randomized controlled trials
Supplemental material, sj-pdf-1-cep-10.1177_03331024231161261 for “Wearing-off” efficacy of CGRP monoclonal antibodies for migraine prevention: A meta-analysis of randomized controlled trials by Thanin Asawavichienjinda, Seelwan Sathitratanacheewin and Ronpichai Chokesuwattanaskul in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024231161261 - Supplemental material for “Wearing-off” efficacy of CGRP monoclonal antibodies for migraine prevention: A meta-analysis of randomized controlled trials
Supplemental material, sj-pdf-2-cep-10.1177_03331024231161261 for “Wearing-off” efficacy of CGRP monoclonal antibodies for migraine prevention: A meta-analysis of randomized controlled trials by Thanin Asawavichienjinda, Seelwan Sathitratanacheewin and Ronpichai Chokesuwattanaskul in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
