Abstract
Background
This narrative review aims to broaden our understanding of the epidemiology, burden and clinical spectrum of cluster headache based on updated findings with a global perspective.
Methods
We conducted a literature search on the following topics: (a) epidemiology; (b) burden: quality of life, disability, economic burden, job-related burden and suicidality; and (c) clinical spectrum: male predominance and its changes, age, pre-cluster and pre-attack symptoms, aura, post-drome, attack characteristics (location, severity, duration and associated symptoms), bout characteristics (attack frequency, bout duration and bout frequency), circadian and seasonal rhythmicity and disease course.
Results
New large-scale population-based reports have suggested a lower prevalence than previous estimations. The impact of cluster headache creates a significant burden in terms of the quality of life, disability, economic and job-related burdens and suicidality. Several studies have reported decreasing male-to-female ratios and a wide age range at disease onset. The non-headache phases of cluster headache, including pre-cluster, pre-attack and postictal symptoms, have recently been revisited. The latest data regarding attack characteristics, bout characteristics, and circadian and seasonal rhythmicity from different countries have shown variability among bouts, attacks, individuals and ethnicities. Studies on the disease course of cluster headache have shown typical characteristics of attacks or bouts that decrease with time.
Conclusions
Cluster headache may be more than a “trigeminal autonomic headache” because it involves complex central nervous system phenomena. The spectrum of attacks and bouts is wider than previously recognised. Cluster headache is a dynamic disorder that evolves or regresses over time.
Keywords
Introduction
Cluster headache (CH) is an extremely debilitating headache disorder presenting with trigeminal autonomic symptoms (1). It has been widely established that CH affects 1 in 1000 people and is typically characterised by strict unilaterality, pain localised to the first division of the trigeminal nerve, autonomic symptoms and symptom clustering during a bout. Recently, reports from previously underrecognised geographical regions, as well as the latest research unveiling the clinical phenotypes and courses of CH, have become available. In this narrative review, we discuss the pre-existing and updated data to broaden the understanding of epidemiology, burden and clinical spectrum, including attack phases, attack phenotype, bout characteristics and clinical course of CH with a global perspective.
Epidemiology
CH is far less common than migraine (2–4), making it challenging to investigate the exact prevalence of CH in the general population. To date, only limited data using rigorous epidemiological methodologies are available (5–18). Most of these studies were conducted in Europe, while others were performed in the USA (6). By contrast, only a limited number of population-based studies have been conducted in Asia, Africa and Latin America (8,10,11,16,17).
Table 1 summarises the results of these epidemiological studies. Fischera et al. (6) conducted a meta-analysis of 16 population-based studies, mostly conducted in European and North American countries. The pooled estimates of lifetime prevalence and 1-year prevalence were 124 per 100,000 [95% confidence interval (CI) = 101–151] and 53 per 100,000 (95% CI = 26–95), respectively. Significant heterogeneity was noted between countries: the lifetime prevalence ranged from 0 (Greece) to 381 (Norway) per 100,000 individuals, and the 1-year prevalence was reported as 0 (Malaysia), 32 (Ethiopia), 119, and 150 (two German studies) per 100,000 individuals (6,9–14).
Prevalence of cluster headache from population-based studies: 2008 meta-analysis and updates.

Following this meta-analysis (6), new data were obtained from Norway, Ethiopia, Georgia and Brazil (15–18). Among the studies included in the aforementioned meta-analysis, the Vågå study conducted in Norway showed the highest lifetime prevalence of CH, with 381 cases per 100,000 individuals (95% Cl = 153–783) (12). However, a recent nationwide epidemiological study conducted in Norway reported a prevalence of 48.6 per 100,000 and an annual incidence of 3.0 per 100,000, which are much lower than the results of the Vågå study (12,18). Torelli et al. (19) conducted a meta-analysis on 10,071 Italian patients and estimated a lifetime prevalence of 279 per 100,000 people (95% CI = 173–427). The prevalence rates were 227 per 100,000 in women and 338 per 100,000 in men. A previous study from Ethiopia reported a 1-year prevalence rate of 32 per 100,000 (95% CI = 10–75) in the 1990s, which was much lower than that reported in European countries (10). In 2013, a study from Ethiopia reported an increased prevalence (3/231), although the sample size was much smaller (16). The first epidemiological data from Georgia and Brazil estimated the lifetime prevalence of CH to be 87 per 100,000 (15) and 41.4 per 100,000 (17), respectively.
Few population-based studies have been conducted on CH in Asia. One study from China, published in 1988, screened 246,812 inhabitants and reported 14 CH cases based on the investigators’ in-house definition (8). Another study from Malaysia evaluated 595 participants and reported no CH cases (11). Hirata et al. (20) analysed data from employees aged 19–74 years who worked for large companies nationwide and registered for health insurance. This study identified 21 (0.098%) CH cases among the 21,480 respondents, similar to the pooled estimates reported by Fischera et al. (6). To address the geographical and ethnic imbalance in current epidemiological studies, an international group of experts is working on the genetic differences in cluster headache among different regions and ethnicities.
Burden
CH is characterised by debilitating headache and is often referred to as the “suicide headache” (21). Patients with CH frequently experience headache-related disabilities, decreased quality of life, economic burdens and job-related issues. We reviewed the impact of CH on these issues to highlight the significance of this condition and the need for increased awareness in the society.
Quality of life
CH significantly affects the quality of life. D'Amico et al. (22) reviewed eight studies that utilised quality of life scales and reported that quality of life measures were significantly lower in patients with CH than normative values, controls and patients with migraine. Interestingly, neuromodulation studies of medically refractory CH failed to improve the quality of life, although the attack frequencies reduced. In a recent review by Freeman et al. (23), significantly reduced quality of life was identified in a qualitative analysis of 11 studies conducted in the UK, Sweden, France, Korea, Germany, Denmark, Italy, Spain and Brazil. A significant impact on quality of life was also reported in Asian patients with CH, as reported in Korea (24) and China (25).
The overall impact of CH is not limited to the pain experienced during attacks. Interictal burden, which indicates ongoing symptoms despite pain relief, emotional aspects such as anxiety and worry about future attacks, avoidance of triggers, the need to conceal one’s condition, and the cumulative effect of these factors, all significantly contribute to the overall burden of the disease (26). In surveys examining interictal burden, factors such as fear of pain, self-concealment and difficulties in personal life were found to diminish the quality of life of individuals with CH (27). These burdens, experienced between headache attacks, significantly affect the overall well-being and highlight the challenges of living with this condition. Therefore, CH exerts a profound and dominant influence on individuals’ thoughts and lives, even during pain-free periods.
Disability
CH, particularly when it becomes refractory to available treatment options, is associated with difficult treatment outcomes and imposes a significant burden of disability on patients. D'Amico et al. (22) reviewed 11 studies reporting cross-sectional and longitudinal data on the adverse effects of mainly intractable CH on daily activities in European countries. The average scores on the Headache Impact Test-6 (HIT-6) suggest that CH has a significant impact on patients' lives, with refractory patients having mean scores of >60. The baseline scores in all studies were much higher than 21 on The Migraine Disability Assessment (MIDAS). Longitudinal studies using HIT-6 and MIDAS scores as disability measurements have demonstrated that despite treatments such as sphenopalatine ganglion stimulation, onabotulinum toxin A, ventral tegmental area deep brain stimulation and occipital nerve stimulation that can lead to moderate improvements, patients still experience significant disability, as indicated by high headache disability scores post-treatment (22,28–32).
Jürgens et al. (29) investigated disability caused by CH using the Headache Disability Inventory (HDI) measure. They found that patients with active chronic CH (33) or episodic CH (34) had higher disability scores (HDI total of 62.5 and 59.4, respectively) than patients with episodic cluster headache (ECH) outside active periods and those with migraines (HDI total of 45.3 and 42.0, respectively).
Recent research has demonstrated the burden of CH in Asian countries. Sohn et al. (24) showed that about 85% of Korean patients with CH had severe headache-related impact (HIT-6 score ≥60). They found that anxiety, greater pain intensity and age are significant predictors of severe impact (24). The Taiwanese group also showed a high level of headache-related disability as measured by the MIDAS and HIT-6 in patients with CH (35).
Previous studies have used either generic measures or migraine-related scales (22). However, they are primarily validated for migraine and may not fully capture the unique features of CH, underestimating the burden. For example, the HIT-6 third question assesses motion sensitivity during a headache attack and the 3-month timeframe MIDAS score might not measure the distinctive nature of CH. Factors such as restlessness, agitation and nocturnal attacks are inadequately covered. Specialised CH scales such as Cluster Headache Scales (CHS) (36) for psychosocial impact, Cluster Headache Severity Scale (CHSS) (37) for CH-specific aspects and the 8-item Cluster Headache Impact Questionnaire (CHIQ) assesses disability (38,39). These scales offer insights into CH, though their research usage remains limited, warranting further study. Integrating these tailored scales promises deeper insights into CH.
Economic burden
A high economic burden of CH, particularly chronic cluster headache (CCH), has been reported in Europe and the USA. A clinical study conducted in Germany found that the average direct and indirect costs for a single CH patient over a six-month period were €5963 (40). The costs were significantly higher for individuals with CCH than those with ECH (€10,985 vs. €2583). A Danish study reported the average annual direct costs for CH as €9,158 for CCH and €2,763 for ECH (41). Additionally, the study identified a significant loss of productivity due to missed work, resulting in indirect costs of €11,809 per year per patient for the CCH population and €3,558 per year per patient for the ECH population.
According to a US study involving 6,562 CH patients, those with CH had a higher number of hospital visits and underwent more medical examinations than controls (42). The total cost associated with CH was found to be $16,530 ± 40,068, which was significantly higher than the costs for controls, which were $7197 ± 25,147. In another US study involving 9328 patients with CH, the total annual direct cost was estimated at $3132 per patient (42). The study also identified indirect costs associated with CH, including $4928 per patient per year for absenteeism, $80 for short-term disability and $3374 for both factors combined.
Job-related burden
A US study involving 1134 patients with CH found that nearly 20% of the patients had lost their jobs due to CH, while an additional 8% were either out of work or disabled due to their headache (43). In a Swedish study, it was found that, among 3240 patients with CH, 17% had taken sick leave for 14 days, which was higher than the rate of 9% in the control group (44). Female patients had higher sickness absence rates (25%) than male patients (13%). A comprehensive study conducted in Korea showed that patients with CH were being less employed and tended to work as freelancers or (self-)employers than those with migraine or tension-type headache and headache-free controls. These findings suggest that patients with CH have difficulties in their workplace where they cannot control their work environment and thus change their employment patterns (45). Approximately 85% of patients with CH reported difficulties at work and required sick leave, which was significantly more frequent than that in headache and headache-free controls.
Suicidality
While CH has been referred to as the “suicide headache” (21), to date, the actual incidence rate of suicide has not been reported in a large CH population. Studies have investigated suicidal thoughts, plans and attempts in patients with CH. An internet survey-based US study reported a high prevalence (55%) of suicidal thoughts among patients with CH (43). In a study by Koo et al. (46), patients with CH had high rates of lifetime active suicidal ideation (47%) and suicide risk (38%). A Korean study comprehensively investigated ictal, interictal and between-bouts suicidality (47). Passive suicidal ideation, active suicidal ideation, suicidal planning and suicide attempt were reported in 64%, 36%, 6% and 2% of patients during the attacks, respectively, and these reports markedly dropped to 4%, 4%, 3% and 1% interictally, and further to 0%, 2%, 2% and 0% during between-bouts periods. This study suggests that the suicidality of CH is mostly attributed to headache burden and can be normalised if CH bouts or attacks are properly treated. Furthermore, depressive symptoms were associated with both ictal and interictal suicidality. Therefore, besides proper CH-targeted treatment, careful diagnosis and management of depressive symptoms should be considered to reduce suicide risk.
Personality disorders (48,49), insomnia (50) and substance abuse/addiction (51,52) have showed associations with CH, all of which can potentially influence suicidality. Despite the valuable insights provided by these studies, further research is necessary to fully comprehend whether these factors contribute to the risk of suicidality in CH patients.
Clinical spectrum of CH
Male predominance of CH: secular trends and regional difference
Although CH is considered a male-predominant disorder, a time-related decline in the male-to-female (M:F) ratio has been observed in several countries (35,39,53–59). An Italian study reported a decline in the M:F ratios from 6.2:1 to 2.1:1 according to onset (before 1960 vs. the 1990s) (53,54). A large cohort study in Taiwan also reported a decline in the M:F ratio from 5.2:1 to 4.3:1 according to the time of enrolment (1997–2020 vs. 2011–2021) (35). The M:F ratios reported in studies in Germany decreased from 4.3:1 in 1988 to 2.1:1 in 2021 (39,57,58) and, in France, decreased from 4.7:1 in 2002 to 1.7:1 in 2008 (56,59). A US survey conducted in 2008 showed a female proportion of 28% among 1134 patients with CH (55), whereas a retrospective analysis using a database from 2009 to 2014 in the US showed a female proportion of 42.6%, which is in line with other studies showing a decline in the M:F ratio over time (60). Changes in lifestyle in women (e.g. alcohol consumption or smoking) have been suggested as the cause of changes in the M:F ratios, as a recent genome-wide association study revealed smoking as a causal risk factor of CH (33). However, a Taiwanese study showed that the M:F ratio of current smokers increased from 14.9:1 to 21.6:1 in patients diagnosed before and after 2010, respectively (35). Another explanation is that CH could have been more frequently misdiagnosed in women in the earlier days. Although previous studies did not report a difference in the diagnostic delay of CH between women and men (61–63), Rozen and Fishman (55) claimed that women tended to have a prolonged (>10 years) diagnostic delay compared to men (25% vs. 21%), although statistical significance was not reached. This can be explained by the fact that women with CH experience more vomiting than men (34), and migraine symptoms, such as nausea and vomiting, were found to be associated with diagnostic delay of CH (64). Thus, the recent change in the M:F ratio may, at least in part, may be due to improvements in the identification and awareness of CH among physicians (65).
A discrepancy exists between the M:F ratios in Western and Asian countries. A cohort study conducted in the US between 2009 and 2014 reported a low M:F ratio of 1.3:1 (60). Recent studies in Europe showed M:F ratios ranging from 1.35 to 2.8:1 (39,59,60,65–68). However, recent studies conducted in Asian countries still show a higher M:F ratio, ranging from 3.8 to 7:1 (24,25,35,69–72). It is unclear why geographical differences exist in the sex ratios. Potential factors include differences in lifestyle, misdiagnosis or diagnostic delay in women, and genetic influences between Asian and Europe/North American countries. These differences have not yet been proven, and long-term secular trends in sex ratios should be collected and evaluated in more countries.
Gender- or sex-related difference in CH phenotype
In most studies comparing sex or sex-related differences in CH phenotypes, the core clinical manifestations of CH were similar in both women and men (73,74). However, nausea, osmophobia, ptosis, miosis, eyelid oedema, nasal congestion, and comorbid conditions such as thyroid diseases and psychiatric disorders have been reported more frequently in women than in men (34,35,66,75), whereas men seem to experience lacrimation and facial sweating more often than women (55,75). Women tend to present with longer attack duration, greater severity and migraine symptoms (35,66,76). According to Fourier et al. (68), diurnal rhythmicity and family history of CH are reported more often in women than in men. However, these differences have not been universally reported in different cohorts, and require further validation.
Age
The ages at inclusion, onset and diagnosis that have been reported in several cohorts are summarised in Table 2. Most studies included patients aged in their 30s and 40s (with the exception of patients aged in their 50s in Sweden and the Netherlands) (Table 2). Age of onset has been reported as 6–75 years (68,69,77–79), with a mean age of 23.6–33.92 years in several countries (7,24,25,35,43,58,59,65,68,69,77–83). The mean age at diagnosis ranged from 35.7 to 42.5 years (2,9,25,31), suggesting a delay between onset and diagnosis. Although the age of onset was reported most commonly as during the 20s in most studies (25,65,68), one Japanese study showed unique patterns: CH onset was most frequent in men in their 20s and 30s, and in women in their 10s and 60s (69). Taken together, patients with CH are not confined to “typical” young adults, and a greater spectrum of patients, including children, adolescents, middle-aged adults and older adults, can develop CH.
Age at inclusion, age of onset and age at diagnosis.

Data are presented as the mean ± SD or median (IQR) unless otherwise specified.
IQR, interquartile range.
Pre-attack symptoms
Premonitory symptoms of migraine have been well recognised and widely investigated (84–86). Similar to migraine, symptoms preceding bouts or attacks have been recently reported in patients with CH (Table 3). Pre-attack symptoms are symptoms occurring a few minutes before the onset of individual CH attack. Blau et al. (87) conducted a study in 1998 involving 150 patients, where they identified pre-attack symptoms such as sensations in the head and neck, mood and neurological symptoms, alimentary symptoms, and minor sensations in the pain area in 61% of the participants. Two decades later, Danish researchers documented a higher prevalence of 86% and categorised the symptoms into three groups: local painful symptoms (shooting pain, dull sensation, ear fullness and neck pain), local painless symptoms (including autonomic symptoms) and general symptoms (difficulty concentrating, restlessness, photophobia, phonophobia, mood changes, frequent urination, etc.) (88). The study findings indicated that local symptoms typically occurred approximately 10 min before the cluster headache attack, while general symptoms preceded the attack by around 20 min (88).
Pre-cluster and pre-attack symptoms.
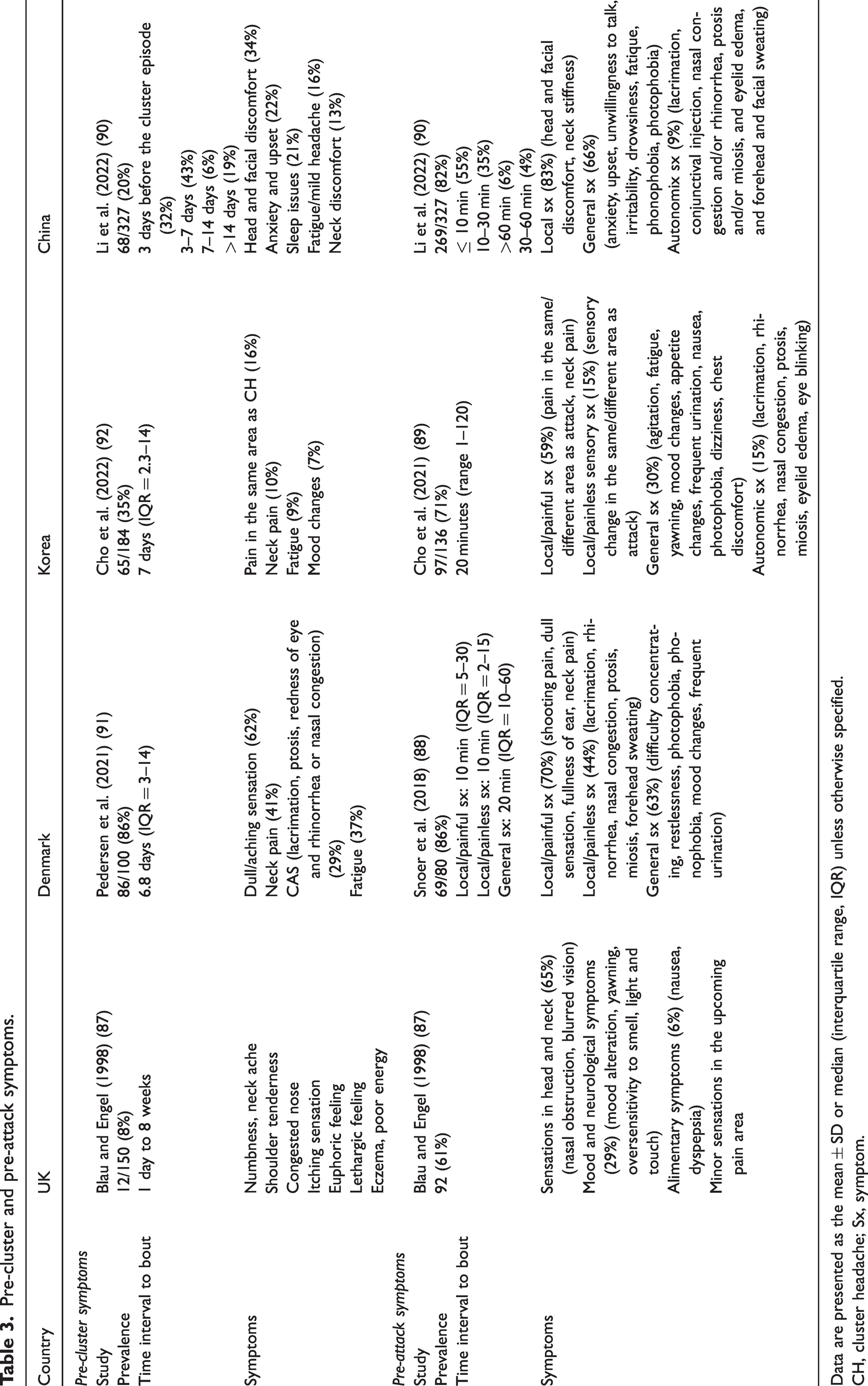
Data are presented as the mean ± SD or median (interquartile range, IQR) unless otherwise specified.
CH, cluster headache; Sx, symptom.
Subsequent replication studies reported a high prevalence of 71% in the Korean study (89) and 82% in the Chinese study (90), with similar time courses and specific symptoms compared to a prior study (88). These consistent findings suggest that pre-attack symptoms are widespread among patients with CH, regardless of their geographic region or ethnicity.
Pre-cluster symptoms
Pre-cluster symptoms indicate symptoms preceding the onset of active bouts by days to a week. Although the prevalence of pre-attack symptoms was similar in subsequent studies, pre-cluster symptoms were much less reported and were not uniform across studies. Blau et al. (87) investigated these symptoms in 150 patients in 1998 and found 8% of patients had pre-cluster symptoms. However, Pedersen et al. (91) reported high prevalence with 86% of Danish patients with ECH experienced pre-cluster symptoms at an average of 6.8 days before the bout, while subsequent studies reported much lower prevalence. Cho et al. (92) reported a 35% prevalence of pre-cluster symptoms in 184 Korean patients, which typically occurred approximately 7 days before the bout. Another recent study of 327 Chinese patients found 21% had pre-cluster symptoms (90). It remains inconclusive whether this variation could be attributed to ethnic distinctions or other contributing factors.
Aura
Aura in CH has not been as well studied as pre-attack symptoms, with the first mention dating back to 1972 in a study by Graham (93). The reported frequencies of aura in CH range from 0% to 35%, with significant variations across different studies and regions (25,35,43,56,67,72,73,81,94–100). Studies conducted in Asia, including China, Taiwan and India, reported a low prevalence of 0–1% (25,35,72,95,98), while studies in European/North American countries reported aura in 3.5–35% of patients with CH (43,56,67,73,81,94,96,97,99,100). However, even within the same country, the reported rates varied widely, as evidenced by German reports, from 3.5% to 23% (96,100). Visual symptoms are the most frequent, while sensory, language/speech, motor and brainstem symptoms have also been reported (35,43,56,67,72,73,81,93,95–97,100). Similar to the migraine aura, the CH aura can occur either before or during an attack (67). The duration of CH aura is usually less than 25 min (43,67,97,100). Considering these similarities with migraine, along with a recent study claiming that CH aura occurs predominantly in patients with migraine with aura, a true CH-attributable aura may be extremely rare (100). A significant difference between Asian and European study results regarding the prevalence of aura in patients with CH may support this idea.
Postdrome
There are few reports on the postictal symptoms of CH. Only one study reported that 98% of the patients experienced postictal symptoms in 76% of attacks (101). In this study, local symptoms, such as mild pain and autonomic symptoms, occurred initially after the attack, and was later followed by the general symptoms (101). The most common general symptoms were mood changes, decreased energy levels, and fatigue. Interestingly, prophylactic treatment does not seem to influence the number of postictal symptoms experienced (101).
Attack characteristics: location, severity and duration
CH is defined in the International Classification of Headache Disorders, 3rd edn (ICHD-3) (1) as a severe, unilateral pain that occurs in the orbit, supraorbital, temporal or a combination of these areas and lasts for 15–180 min per episode (1). In the following subsections, we discuss each phenotypic component of a CH attack and describe the syndrome-defining features of each component, as well as a wide range of features reported to date.
Localisation
CH attacks typically involve the orbital, supraorbital and temporal areas as defined in ICHD-3 (1). Orbital pain, including retroorbital location of pain, has been reported in the majority of descriptive studies, with prevalence rates of 68–88% (Table 4). A limited number of studies have examined the prevalence of supraorbital and temporal locations of pain, reported as 50–60% and 52–86%, respectively (Table 4) (25,35,56,67,69,80,102,118). Occasional reports have noted atypical areas including the orofacial, occiput, zygomatic, maxillary, mandibular, nuchal, nose, vertex and shoulder (Table 4) (25,56,58,67,69,118).
Pain location and severity.

Data are presented as the mean ± SD unless otherwise specified.
Although CH is typically characterised by strictly unilateral headache, side shifts have been reported between bouts, within bouts and during attacks (bilateral attacks or spreading patterns). This is discussed in detail below in the section on Disease Course.
Severity
According to ICHD-3 (1), CH is characterised by excruciating headache. Most patients rated the intensity of their pain between 7.5 and 10 on a scale of 0–10 in almost all cross-sectional studies (25,35,58,65,67–69) regardless of subtype (ECH vs. CCH) (Table 4) (39,80). Based on our experience, a reduction in pain severity is clinically noted when a bout is about to remit. Great within-individual variability in attack severity has been reported in a comprehensive case report of a patient with CCH, with mild and moderate severity in 14% and 66% of attacks, respectively (103). Unexpectedly, a decrease in attack severity was noted before remission in approximately 35% of patients with long-term remission (104).
Duration
CH attacks typically last between 15 and 180 min, as defined in ICHD-3 (1). Because the duration criterion should be met to diagnose CH, atypical duration is the most common cause of probable CH (80,104). Other studies have reported that 4–13% of patients have CH attacks lasting over 180 min (25,68,69). Longer attacks have been reported in patients with CCH and women (35,66), although these findings have not been replicated in other studies (34,65,68).
Associated symptoms
ICHD-3 defines CH attacks as those associated with any ipsilateral conjunctival injection, lacrimation, nasal congestion, rhinorrhoea, forehead and facial sweating, miosis, ptosis, eyelid oedema, restlessness or agitation (1).
Numerous studies conducted in various countries, such as Sweden (68), the Netherlands (67), Denmark (65), Germany (58), Norway (81), France (59), Italy (79), China (25), Korea (80), Taiwan (35), Japan (69) and the USA (43) have reported that conjunctival injection or lacrimation is the most prevalent symptom of CH, with a prevalence ranging from 53% to 92% (Table 5). However, in Japan (69), the most common symptom was a feeling of restlessness or agitation, with a prevalence of 70% (Table 5). In one study from Italy (79), the most frequently reported symptom was eyelid oedema, with a prevalence of 75%, while its prevalence was only 13–37% in other countries (Table 5). Other syndrome-defining symptoms include conjunctival injection (30–92%), nasal congestion and/or rhinorrhea (13–37%), forehead and facial sweating (18%–59%), and miosis and/or ptosis (8–67%).
Associated symptoms.

C, conjunctival injection; L, lacrimation; N, nasal congestion or obstruction; R, rhinorrhea; M, miosis; P, ptosis.
Restlessness and/or agitation are another syndrome-defining symptom of CH. Interestingly, restlessness and/or agitation were highly prevalent (70–83%), which is similar to or even more common than conventional autonomic symptoms in Netherland, Norway, Italy, Germany and Japan, whereas it was far less common (38–48%) in Sweden, France and Asian countries such as China, Korea and Taiwan (Table 5).
CH attacks may be accompanied by symptoms not included in the ICHD-3 (Table 5) (1). A few studies using or validating the ICHD-3 beta version (105) reported forehead and facial flushing (14–47%) (81) and ear fullness (9%) (80). Migraine-like symptoms including allodynia, nausea, photophobia and phonophobia were reported up to 36% (67), 60% (25), 57% (35) and 69% (81) of patients with CH, respectively.
Bout characteristics: attack frequency, bout duration and bout frequency
Attack frequency
ICHD-3 defines CH attacks as those that occur with a frequency of once every other day up to eight times a day during more than half of the active bouts (1). The frequencies reported in recent studies are summarised in Table 6. In accordance with ICHD criteria (1), the frequency of CH attacks usually ranged from 0.5 to 8 episodes per day during active bout periods. A frequency lower than 0.5 can be seen during the early or late period of the bout, and frequencies of more than 8 per day have been reported in 4% in a Norwegian cohort (81) and up to 20 attacks per day were reported in a German cohort (78) (Table 6).
Attack frequency, bout duration and bout frequency.

Data are presented as the mean ± SD or median (interquartile range) unless otherwise specified.
CCH, chronic cluster headache; ECH, episodic cluster headache.
Several studies have investigated the differences in attack frequency between ECH and CCH. An international survey (7) found that CCH (4.5 ± 2.0) had a higher attack frequency than ECH (3.7 ± 2.0). Similarly, in a Danish survey (106), the frequency of CCH (4.1 ± 2.57) was reported to be higher than that of ECH (3.3 ± 2.1). However, other studies found no significant differences between the two groups (58,75,79,80).
As seen in Table 6, the average attack frequency was generally lower in Asian cohorts (between 1 and 2 attacks per day) than in European or North American cohorts (approximately 3 attacks per day). This was reported in a review by Peng et al. (107), and the latest studies showed a similar trend: more than half of Swedish patients had more than 3 attacks (68) and a mean 1.5 attack per day in a Taiwanese study (35).
Bout duration
Clustering of attacks during an active period (“cluster period” or “bout”) is another characteristic of CH. ICHD-3 classifies CH into ECH and CCH based on the bout duration. In ECH, 2 bouts lasting from 7 days to 1 year are separated by pain-free intervals of at least 3 months (1). If attacks occur without any pain-free intervals or with remission lasting less than 3 months for at least 1 year, a diagnosis of CCH can be made (1). CH bouts usually last for several weeks or months, with 4–8 weeks being the most commonly reported range (Table 6). However, there is variability in the duration of these bouts among individuals.
Bout duration was shorter in the Asian cohort than in the European cohort (Table 6). In Asian countries, CH studies typically report an average bout duration of 4–6 weeks, whereas studies conducted in Europe and North America indicate a longer average bout duration of 8 weeks (107). This may be attributed to true ethnic difference or the proportion of CCH as CCH is much less prevalent in Asian countries.
Bout frequency
The ICHD-3 does not provide specific criteria for bout frequency, and reports on bout frequency have been limited (1). Most cohorts reported a mean frequency of 1–2 bouts per year (Table 6). However, there is considerable variability, with 13% of patients experiencing less than one bout every other year in a Norwegian study (81), while a study from the USA (99) reported that 14% of patients had ≥6 bouts per year.
Circadian and seasonal rhythmicity
Circadian rhythmicity is a characteristic of CH, indicating that CH attacks tend to occur at specific times of the day. Circadian rhythmicity was present in 53–86% of patients (25,35,43,65,68,80,81) (Table 7). Different reports have been published on the most prevalent times of CH attacks. Several studies have reported 2
The presence of circadian and seasonal rhythmicity in cluster headache.

Seasonal rhythmicity in CH refers to the tendency of attacks to occur predominantly during specific seasons of the year. Seasonal rhythmicity ranges from 37% to 73% (25,35,43,65,68,80,81). The highest frequency of CH bouts was reported during both autumn and spring in several studies (43,58,73,108), whereas others reported autumn (65,68,111) and spring (25) individually. By contrast, summer, particularly June, was the season with the lowest number of CH bouts (25,43,65,68,95,108). Environmental factors, such as daylight duration, temperature and rainfall, have been reported to be associated with CH (65,111,112). In a study from Taiwan, mean temperature was found to have the highest correlation with climate-related factors such as absolute temperature, temperature variation, relative humidity, atmospheric pressure, wind speed, sunshine and rainfall. An increase in the frequency of CH attacks has been observed during the seasonal transitions from winter to spring and spring to summer (111).
Disease course
Few long-term follow-up studies have investigated the natural course of CH. In these studies, the follow-up duration was 3–15 years and phone or letter communication was used (104,113–115). The definition of remission varies across studies as a result of the absence of a consensus. Manzoni et al. (114) reported remission rates of 18.5% (>3 years), 5% (>5 years) and 2% (>10 years). Interestingly, discontinuing alcohol consumption led to an immediate remission of active bouts but not a long-term stable remission.
In a study by Pearce et al. (115), 12% of patients achieved remission (>4 years). Lee defined remission as lasting longer than twice the longest between-bouts period and >5 years and found that 33% of lost-to-follow-up CH patients had remission. Predictive factors for remission were not identified. Patients in remission showed only some trends toward fewer lifetime bouts, shorter disease duration and reduced seasonal/circadian rhythmicity (104). Thus, it can be speculated that higher disease activity is associated with more frequent bouts, longer disease duration and pronounced seasonal/circadian rhythmicity.
The typical characteristics of CH attacks or bouts appear to decrease over time. Strict unilaterality is considered a characteristic of CH; however, side shift occurs in 2–17% of patients (114,115). Even bilaterality can be found in approximately 3%–6% of patients when followed up for a long period (104,114). In addition, there can be changes in the seasonal and circadian rhythmicity. Lee et al. (104) reported that 40% of patients showed changes in seasonal and circadian rhythmicity (26% lost rhythmicity and 13% shifted seasonal predilection) and 35% showed changes in circadian rhythmicity (22% lost rhythmicity and 13% shifted circadian predilection) during the course of the disease. Changes in bout duration were observed in two studies: increased, unchanged and decreased (48%, 44% and 6%, respectively) in patients with active disease (114) and unchanged, decreased and increased (60%, 23% and 17%, respectively) in a more recent study including both remitted and active patients (104). Similarly, in one study, changes in bout frequency along the disease course were reported, with bout intervals increasing over time: the first between-bouts interval and the last interval was 1.4 and 1.9 years respectively, during a 5–6 year follow-up of both remitted and active patients (104).
The transition from episodic to chronic subtypes of CH within an individual's disease course holds substantial significance. For example, the study conducted by Manzoni et al. (114) in 1991 demonstrated that patients experienced a progression towards a chronic form (13%), or alternatively towards a combined form (6%) which suggests dynamic or evolving nature of CH progression. Clinic-based case series endeavored to assess the risk associated with transitioning from an episodic onset to a chronic manifestation, with estimated probabilities spanning from 2% to 19% (116). Moreover, factors comprising personal habits such as smoking and alcohol consumption, head injuries, higher frequency of clusters and shorter intervals of remission periods are suggested as potential predictors of the shift towards a chronic state (117).
Conclusions
A wide range of reported prevalence of CH has been noted in previous and recent studies, even within the same country, suggesting the need for a more rigorous methodology for population-based surveys of CH. CH-related burdens, including quality of life, disability, economic and job-related burdens and suicidality, are significant and consistent worldwide. CH constitutes more than a “trigeminal autonomic headache” because it involves complex central nervous system phenomena as evidenced by pre-cluster and pre-attack symptoms, side shifting, and circadian and seasonal rhythmicity. Although the current diagnostic criteria apply well globally, the spectrum of attack and bout characteristics reported from different countries and ethnicities is wider than that previously recognised. Despite the limited literature on the disease course, CH appears to be a dynamic disorder that evolves or regresses over time.
Clinical implications
Recent population-based studies indicate a lower prevalence of CH globally than previously estimated. CH significantly impacts patients' quality of life, leading to disability, economic burdens, job-related issues and increased risk of suicidality. The male-to-female ratio in CH is decreasing, and CH characteristics, including attack patterns and rhythmicity, vary among countries and ethnicities. The spectrum of attacks and bouts is wider than previously recognised. Non-headache phases of CH and its involvement of complex central nervous system phenomena have gained renewed attention.
Footnotes
Declaration of conflicting interests
MJL has received personal or institutional honoraria for participation in clinical trials, speaker fees, or as a consultant from Abbvie, Biohaven, Eli Lilly, Lundbeck, Novartis, Otsuka, Sanofi-Aventis, SK Pharm, Teva, Yuhan Company and YuYu Pharma. PPR has received honoraria as a consultant and speaker in the last 3 years, from AbbVie, Amgen, Biohaven, Chiesi, Eli Lilly, Lundbeck, Medscape, Novartis, Pfizer and Teva. Her research group has received research grants from AbbVie, Novartis and Teva and also has received funding for clinical trials from Alder, AbbVie, Amgen, Biohaven, Electrocore, Eli Lilly, Lundbeck, Novartis and Teva. She is the Honorary Secretary of the International Headache Society. She is on the editorial board of Revista de Neurologia and associate editor for Cephalalgia, Headache, Neurologia, Frontiers of Neurology. She is a member of the Clinical Trials Guidelines Committee and Scientific Committee of the International Headache Society. She has also edited the Guidelines for the Diagnosis and Treatment of Headache of the Spanish Neurological Society. She is the founder of ![]() . Pozo‐Rosich does not own stocks from any pharmaceutical company. The other authors report having nothing to declare.
. Pozo‐Rosich does not own stocks from any pharmaceutical company. The other authors report having nothing to declare.
Funding
This study was supported by the New Faculty Start-up Fund of Seoul National University and National Research Foundation of Korea (NRF) grant funded by the Korean Government (MSIP; No. 2020R1A2B5B01001826).
