Abstract
Background
Currently, only a few specific blood pressure-lowering medications are recommended for migraine prevention. Whether benefits extend to other classes or drugs is uncertain.
Methods
Embase, MEDLINE, and the Cochrane Central Registry of Controlled Trials were searched for randomized control trials on the effect of blood pressure-lowering medications compared with placebo in participants with episodic migraine. Data were collected on four outcomes – monthly headache or migraine days, and monthly headache or migraine attacks, with a standardised mean difference calculated for overall. Random effect meta-analysis was performed.
Results
In total, 50 trials (70% of which were crossover) were included, comprising 60 comparisons. Overall mean age was 39 years, and 79% were female. Monthly headache days were fewer in all classes compared to placebo, and this was statistically significant for all but one class: alpha-blockers −0.7 (95% CI: −1.2, −0.1), angiotensin-converting enzyme inhibitors −1.3 (95% CI: −2.9, 0.2), angiotensin II receptor blockers −0.9 (−1.6, −0.1), beta-blocker −0.4 (−0.8, −0.0) and calcium channel blockers −1.8 (−3.4, −0.2). Standardised mean difference was significantly reduced for all drug classes and was separately significant for numerous specific drugs: clonidine, candesartan, atenolol, bisoprolol, metoprolol, propranolol, timolol, nicardipine and verapamil.
Conclusion
Among people with episodic migraine, a broader number of blood pressure-lowering medication classes and drugs reduce headache frequency than those currently included in treatment guidelines.
Keywords
Introduction
Among blood pressure (BP)-lowering medications, beta-blockers (BB), including atenolol, bisoprolol, metoprolol, nadolol and propranolol and the angiotensin II receptor blocker (ARB) candesartan, are suggested by guidelines for the prevention of migraine (1,2). In a systematic review and meta-analysis, Jackson et al. (3), found that patients with migraine may benefit from BP-lowering medications and recommended these medications for patients with migraine and concomitant hypertension. Two other meta-analyses found that different classes of BP-lowering medications, including diuretics, angiotensin-converting enzyme inhibitors (ACEi), ARBs and BBs, prevented headache in general compared to placebo (4,5). However, there is little information on whether all classes of BP-lowering medications, or whether combination BP-lowering agents, are effective in the prevention of migraine.
The aim of this systematic review is to evaluate the effect of different classes of antihypertensive medications, including alpha-blockers (AB), ARB, ACEi, BB, and calcium channel blockers (CCB), on the prevention of episodic migraine (EM).
Methods
The current review was performed based on Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) (6) and was registered on the International Prospective Register of Systematic Reviews (PROSPERO) (CRD42017079176).
Literature search
We performed a systematic review of all randomized trials of BP-lowering medications on the prevention of episodic migraine defined as less than 15 headache days per month. We sought to identify potentially relevant studies from searches of Embase, MEDLINE, and the Cochrane Central Registry of Controlled Trials, with each source searched from inception to January 2017 and then updated to December 2019. MEDLINE search terms are provided in Online Supplementary Table 1.
Eligibility criteria
We included randomized controlled trials of adult participants (≥18 years of age) in which BP-lowering medications were compared with placebo for the following drug classes: ACEi, ARB, AB, BB, CCB and combination BP-lowering. Studies in languages other than English and conference abstracts were excluded.
Outcome measures
Headache/migraine frequency was recorded as the evaluated outcome. Four types of frequency outcomes, including headache days per month, headache attacks per month, migraine days per month and migraine attacks per month, are reported in different studies. Therefore, we extracted all of them. If two types of frequency outcomes were reported in a single study, both were extracted.
Based on data availability, we prioritized and used the reported effect measures in the following order: mean difference in means between arms, change from baseline, and post-intervention data. In cases where the frequency was reported for more or less than one month (for example, total number of headache days in three months), we transformed it to one month (e.g., by dividing by three in the mentioned example) (3).
Study inclusion and data extraction
All stages of the screening (title and abstract and full-text screening) were performed in duplicate-by independent reviewers (FH, JS, PN, AC), and conflicts were resolved by discussion with, or assistance, from a senior researcher (CC). Independent reviewers (FH, JS, PN, AC) extracted data, and verified results using a standard extraction form.
Baseline data for included studies such as first author and year of publication, name of the used drug and its class, drug dose, study duration, number of participants, and the mean age and female percentage were extracted.
Risk of bias assessment
The risk of bias was assessed by two independent reviewers (FH, KV) using the Cochrane risk of bias tool (7) based on the headache frequency outcome. Any conflict was resolved by discussing it, and, if necessary, by seeking advice from a third reviewer. Risk of bias assessment includes the following domains: random sequence generation (selection bias), allocation concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective reporting (reporting bias), and other sources of bias.
Statistical analysis
The outcome of interest in the current study was headache/migraine frequency. We included four types of headache/migraine frequency: headache days per month; headache attacks per month; migraine days per month; and migraine attacks per month. To estimate the effect of the different classes of BP-lowering medications on frequency compared to placebo, analysis was done for each outcome separately using difference in means and 95% confidence intervals (95% CI) by the random-effect model. In order to assess the same outcome that was measured it in a variety of ways, we used standardized difference in means and 95% CI to pool studies regardless of the type of reported frequency outcome. Meta-analysis was performed whenever two or more studies were available. Statistical heterogeneity was assessed reporting I2 and P-value statistics. We used a P-value <0.1 to consider having heterogeneity among the studies. I-squared <40% was considered as no heterogeneity and more than 75% as having a considerable level of heterogeneity among studies (8). Mean standard deviation (SD) of headache frequency was calculated based on the available studies and was used in studies with missing data and in studies with SD < 1. For the final number of participants included in the meta-analysis, the participants were counted two times in cross-over studies. As the number of studies on BBs were adequate for further analysis, we divided these studies into three groups based on the dose of the medication used (less than standard dose, standard dose, and more than standard dose) based on the Law et al. report (9).The standard doses of the antihypertensive medications were derived from the following sources: Mims Online, Defined Daily Dose by the World Health Organization and Martindale: The Complete Drug Reference (10–12). Data were analysed using Comprehensive Meta-Analysis, version 4 (Biostat, Englewood, NJ, USA) and RoB figures were made using the review manager version 5.3.
Data may be obtained from the corresponding author upon reasonable request.
Results
Literature search and included studies
In this systematic review, 50 trials involving 4310 participants and a total of 60 comparisons with placebo were included (Online Supplemental Table 2) (Figure 1). Data from a pilot trial entitled the Headache Prevention Project (HAPPy) performed by the same team (to be submitted separately) were included in this meta-analysis. The HAPPy trial compared a low-dose BP lowering combination medication (telmisartan 20 mg, amlodipine 2.5 mg and indapamide 1.25 mg) with propranolol (160 mg/day) and placebo. Risk of bias assessment using the Cochrane RoB tool is reported in Figure 2 (and in more detail in Online Supplemental Figure 1).
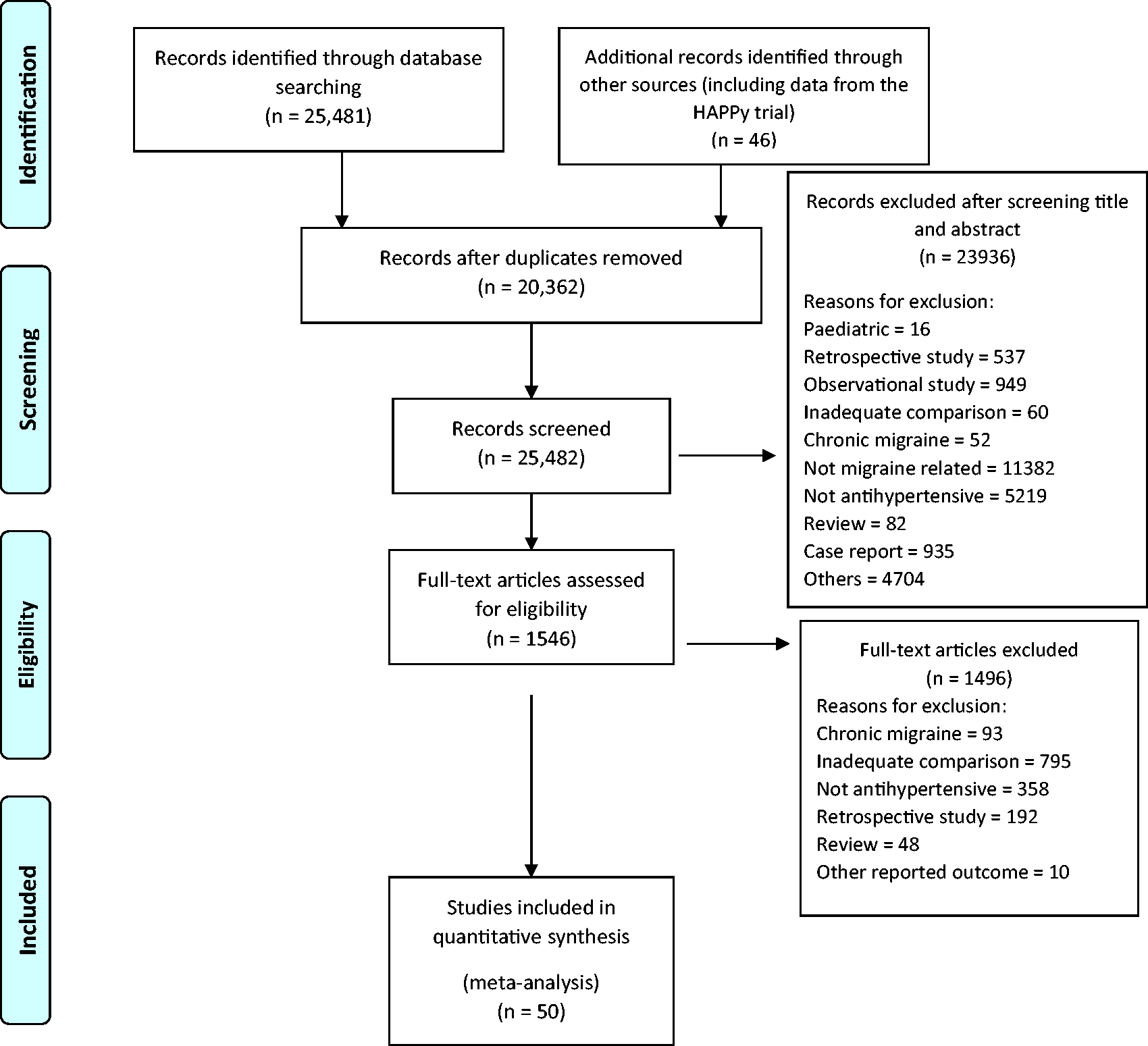
PRISMA flow diagram for the systematic review.
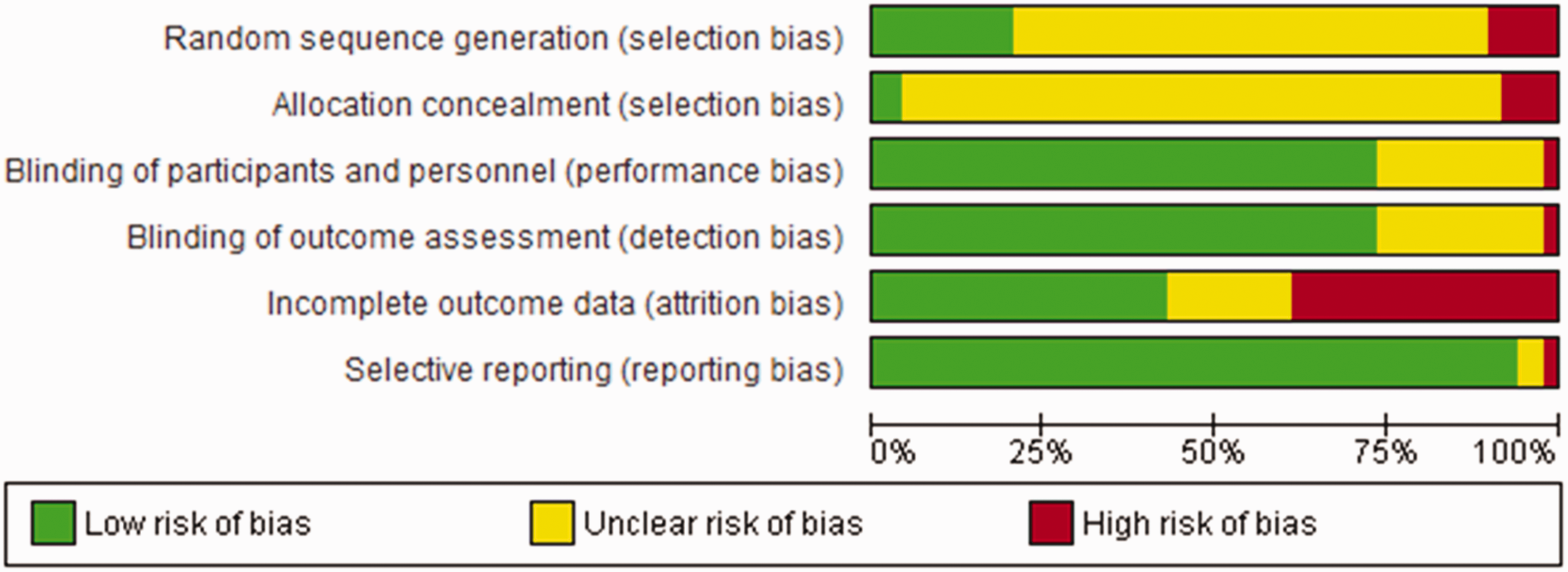
Summary of risk of bias assessment of the studies included in this systematic review.
The majority of the studies (35 out of 50 [70%]) had a cross-over design. Overall mean age of participants was 39 years and 79% were female. A wide variety of BP-lowering drugs were evaluated: ABs (seven comparisons of clonidine (13–19)); ARBs (three comparisons – two candesartan (20,21) and one telmisartan (22)); BBs (38 comparisons −21 propranolol (20,23–39), four timolol (34,38,40,41), four pindolol (42,43), two metoprolol (44,45), two bisoprolol (46), two atenolol (37,47), one alprenolol (48), one acebutolol (49) and one oxprenolol (50)); CCBs (nine comparisons – four nimodipine (51–54), two nifedipine (55,56), two verapamil (57,58) and one nicardipine (59)); ACEi (one enalapril (60) and one lisinopril (61)); and combination therapy (the HAPPy trial)
Effects on headache and migraine outcomes
For each class of BP-lowering drugs, there was a lower number of monthly headache days with treatment compared to placebo, and this was separately statistically significant for each group, with the exception of the two groups (ACEi and combination therapy), for which meta-analysis was not performed (only one study for each drug) (Figure 3). The greatest reduction in monthly headache days was for CCBs with a mean difference of −1.8 (95% CI: −3.4, −0.2). Meta-analysis of the effect of BP-lowering classes on headache attacks per month, migraine attacks per month, and migraine days per month is shown in Figure 3. Generally, there was not a considerable level of heterogeneity among the studies in each group. The overall result of studies with cross-over design was similar to studies with parallel design (Online Supplemental Figure 2).

Meta-analysis of the effect of different classes of BP-lowering drugs on the mean difference of headache/migraine frequency per month in patients with episodic migraine. * trials with more than two eligible arms are counted more than once. Some trials have reported more than one type of frequency outcome. All are reported here. Diamonds represent the meta-analysis of two or more studies.
Most evidence was available for BBs which showed on average 0.7 fewer monthly headache attacks (95% CI: −0.1, 1.4), 0.4 fewer monthly headache days (95% CI: 0.0, 0.8), 1.0 fewer monthly migraine attacks (95% CI: 0.7, 1.2) and 1.0 fewer monthly migraine days (95% CI: 0.4, 1.6). On analysis of BBs studies divided into three groups based on the standard dose of this medication, there was no clear trend to increased efficacy with increased dose (Online Supplemental Figure 3).
After pooling different outcomes using standardized difference in means, specific BP-lowering medications that demonstrated benefits were: clonidine with the seven studies (standardized mean difference = −0.2, 95% CI: −0.3, 0.0), candesartan with the two studies (standardized mean difference = −0.4, 95% CI: −0.7, −0.2), atenolol with the two studies (standardized mean difference = −0.6, 95% CI: −1.0, −0.1), bisoprolol with the two studies (standardized mean difference = −0.5, 95% CI: −0.7, −0.2), propranolol with the 21 studies (standardized mean difference = −0.3, 95% CI: −0.4, −0.2), timolol with the four studies (standardized mean difference = −0.3, 95% CI: −0.5, −0.2), nicardipine with the one study (standardized mean difference = −0.9, 95% CI: −1.4, −0.4) and verapamil with the two studies (standardized mean difference = −0.7, 95% CI: −1.2, −0.1) were effective in reducing monthly frequency in people with episodic migraine (Figure 4). All classes of BP-lowering medications showed efficacy in preventing episodic migraine.

Meta-analysis of the effect of different BP-lowering medications on the standardized difference in means of monthly headache/migraine frequency in patients with episodic migraine. All are reported here. Each white diamond represents the pooled meta-analysis of all studies on the respective drug. Black diamonds are the pooled meta-analysis of all studies on the respective class. Meta-analysis was performed if ≥2 studies were available. Otherwise, the result of a single study is reported (no diamond). (* trials with more than two eligible arms are counted more than once).
Overall, only six trials reported the BP difference between groups, with an average 9.3 mmHg systolic blood pressure (SBP) and 3.0 mmHg diastolic blood pressure (DBP) reduction compared to placebo (21,22,32,45,46,61). Therefore, there was very limited ability to assess whether the amount of BP reduction per se was a determinant of the degree of headache/migraine frequency reduction.
Discussion
Our systematic review and meta-analysis demonstrated that different BP-lowering classes reduced the frequency of headache days per month compared to placebo in EM. This was statistically significant for all classes and was separately statistically significant for numerous specific drugs within the classes: clonidine, candesartan, atenolol, bisoprolol, propranolol, timolol, nicardipine, and verapamil.
This systematic review is an inclusive study of BP-lowering medications used in the management of EM with analysis of class effect. It has, however, some important limitations. The main ones are the small sample size and the limited number of studies for all classes except for BBs. In addition, many studies are decades old and do not adhere to current accepted guidelines; definitions of outcomes are often not well explained, and the endpoints reported differ between studies (e.g., based on change from baseline to last month, last week or during the study period). However, we would note that these factors are not expected to result in a bias or an overestimation of treatment effect – it is well reported that use of trial outcomes that are non-optimal with regard to specificity do in fact have a bias toward the null hypothesis (62). Furthermore, it is unclear in many studies if the participants had only migraine or if other headache disorders were present. After evaluating the risk of bias, judgment for most studies was unclear for selection bias. Finally, while the trials used BP-lowering medications, reports on SBP and DBP changes were scarce. Due to these limitations, the results should be interpreted cautiously but they might, at least, serve to generate hypotheses about whether BP lowering per se might be one mechanism whereby these various drugs with different modes of action might have a role in migraine prevention, at least in EM patients.
A systematic review and meta-analysis (3) in 2015 compared different types of migraine prevention medications but did not include an assessment by BP-lowering drug class. There were three or more trials per medication showing the preventive effect of metoprolol and propranolol and less than three per medication reporting the significant effect of enalapril, lisinopril, captopril, candesartan, telmisartan, atenolol, bisoprolol and timolol compared to placebo. Studies on the effect of BP-lowering medications on headache (not just migraine) showed that all classes reduced headache compared to placebo (4,5). The authors suggested that BP reduction (4) or a reduction in BP variability (5) may be the reason for this effect.
The size of the benefits seen with BP-lowering drugs here – on average, one fewer headache day per month – is clinically relevant. In addition, BP-lowering medication classes differ in their adverse effects profile – for example, ARBs typically have no measurable side effects compared to placebo (63,64). There are several other drug classes used to prevent migraine attacks with on average approximately similar size of benefits as BP-lowering medications, including: antiepileptics such as topiramate, and sodium valproate; antidepressants such as amitriptyline and venlafaxine; BTX-A; and anti-CGRP monoclonal antibodies (3,65–68). However, important factors such as cost, health insurance coverage, concomitant disorders, potential side effects, patients’ preference, and availability of the treatment need to be considered (65,68). Furthermore, there are very few direct randomized comparisons between different modalities, and little is known about whether there may be any synergistic benefit from combined treatment with BP-lowering medications.
There may be other antihypertensive agents and classes that have benefits for migraine prevention, beyond those currently recommended. This would have clinical relevance in terms of therapeutic options if a currently used medication has unacceptable side effects. It also suggests that BP lowering per se may be a relevant mechanism.
Conclusion
Different classes of BP-lowering medications reduced headache/migraine frequency in people with episodic migraine. These data suggest that new trials using BP-lowering medications and conforming to present guidelines for migraine prevention might be useful. In addition, further research is required to determine whether the degree of BP-lowering matters, and on the efficacy and safety of combining BP-lowering drugs with other established effective therapies, such as CGRP monoclonal antibodies and CGRP antagonists.
Article highlights
All classes of BP-lowering medications studied, including alpha-blockers, angiotensin II receptor blockers, angiotensin-converting enzyme inhibitors, beta-blockers and calcium channel blockers reduced headache days per month more than placebo. Rigorous trials adhering to the most up to date guidelines for clinical trials in headache are required to study a wider range of BP-lowering agents for migraine prevention than the relatively few which are included in current guidelines. The present results, despite their significant limitations, may at least, serve to generate hypotheses about whether BP lowering per se might be one mechanism whereby these various drugs with different modes of action might have a role in migraine prevention, at least in EM patients.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231183166 - Supplemental material for The effect of blood pressure lowering medications on the prevention of episodic migraine: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-cep-10.1177_03331024231183166 for The effect of blood pressure lowering medications on the prevention of episodic migraine: A systematic review and meta-analysis by Cheryl Carcel, Faraidoon Haghdoost, Joanne Shen, Puneet Nanda, Yu Bai, Emily Atkins, Takako Torii-Yoshimura, Alexander J. Clough, Leo Davies, Dennis Cordato, Lyn R. Griffiths, Grace Balicki, Xia Wang, Kota Vidyasagar, Alejandra Malavera, Craig S. Anderson, Alessandro S. Zagami, Candice Delcourt and Anthony Rodgers in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LRG reports migraine research funding from the Australian National Health and Medical Research Council (NHMRC-APP1122387), US Migraine Research Foundation, US Dept of Defence and Teva. ASZ is an associate editor for Cephalalgia. George Health Enterprises, the social enterprise arm of The George Institute for Global Health, has received investment to develop fixed-dose combination products containing aspirin, statin and blood pressure lowering drugs; and has submitted patents for low-dose blood pressure combinations, on which AR is listed as one of the inventors. AR is seconded part-time to George Medicines Pty Ltd (GM) which is developing a low-dose blood pressure lowering combination for FDA approval. The George Institute has an institutional interest to declare with respect to George Health Enterprises. None of the authors have a financial interest in these investments.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CC acknowledges the support of the National Heart Foundation of Australia (Postdoctoral fellowship 102741) and National Health and Medical Research Council (NHMRC) investigator grant (APP2009726). CSA holds a NHMRC Senior Principal Investigator Fellowship.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
