Abstract
The aim of this study was to assess the efficacy of pharmacological prophylactic treatments of migraine in children. Databases were searched from inception to June 2004 and references were checked. We selected controlled trials on the effects of pharmacological prophylactic treatments in children with migraine. We assessed trial quality using the Delphi list and extracted data. Analyses were carried out according to type of intervention. A total of 20 trials were included. Headache improvement was significantly higher for flunarizine compared with placebo (relative risk 4.00, 95% confidence interval 1.60, 9.97). There is conflicting evidence for the use of propranolol. Nimodipine, clonidine, L-5HTP, trazodone and papaverine showed no effect when compared with placebo. All medications were well tolerated and adverse events showed no significant differences. Flunarizine may be effective as prophylactic treatment for migraine in children. Because of the small number of studies and the methodological shortcomings, conclusions regarding effectiveness have to be drawn with caution.
Introduction
Migraine is the most common cause of chronic recurrent headache in school children. Prevalence of migraine increases with age, ranging from 1.4% to 5% in young children and up to 11.6% in adolescents (15–18 years); the ratio of women to men diverges to become about 2 : 1 after puberty (1–3). The International Headache Society has recently revised its diagnostic criteria and classification system (4). The changes are that migraine may last less than 72 h, may be bifrontal, and that phonophobia and photophobia may not always be present.
Treatment of paediatric migraine has been the subject of debate for many years. Prophylactic pharmacological treatment could be considered when headache frequency exceeds four episodes per month and/or the attacks are so severe or prolonged that they interfere with school or normal activities. Drugs commonly used for migraine are calcium antagonists, β-blockers and pizotifen.
To our knowledge, two systematic reviews including prophylactic treatments have been performed on migraine in children (5, 6). One review included 17 behavioural treatment studies and 24 prophylactic drug studies of migraine in children (5). They concluded that behavioural therapies seemed more effective than prophylactic drug regimens, but they excluded 35% of the behavioural treatment studies and 17% of the drug studies, which they considered to be methodologically inadequate or lacking statistical information. Not all studies analysed had control groups and different study designs were used. The systematic review of Victor and Ryan (6) included 20 studies, of which three were published in a congress book. They identified one single study each of propranolol and flunarizine showing efficacy as prophylactics of paediatric migraine.
The present systematic review distinguishes itself from these reviews by evaluating the literature systematically using up-to-date methodology recommended by the Cochrane Collaboration (7), reporting according to the Quality Of Reporting Of Meta-analyses (QUOROM) statement and by including the most recent literature up to June 2004. In this article, we present a systematic review of controlled trials concerning the efficacy of pharmacological prophylactic treatments of migraine in children. The authors have also reviewed the results of non-pharmacological prophylactic treatments. This is the subject of a separate article (Part 1).
Methods
Search strategy
Medline, Embase, PsycInfo, Web of Science and Cinahl were searched from inception to June 2004 using the terms ‘migraine’, ‘headache’, ‘cephalgia’, ‘cephalalgia’, ‘child∗’, ‘infant’, ‘teenage’, ‘adolescent’ or ‘p(a)ediatric’ together with the search strategy for identifying randomized (RCT) and clinical controlled trials (CCT) described by Robinson and Dickerson (8). The Cochrane Controlled Trials Register, Cochrane Library, issue 2, 2004, was searched using the words ‘migraine’, ‘headache’, ‘cephalgia’, ‘cephalalgia’, ‘child∗’, ‘infant’, ‘teenage’, ‘adolescent’ or ‘p(a)ediatric’. Additional strategies for identifying trials included searching the reference lists of review articles and included studies.
Study selection
Only RCTs and CCTs including pharmacological prophylactic interventions used in the treatment or management of migraine conducted among children (age < 18 years), with criteria designed to distinguish migraine from other types of headache, were selected for our review. The use of a specific set of diagnostic criteria (e.g. IHS 1988 (9, 10)) was not required, but migraine diagnoses had to be based on at least some of the distinctive features of migraine, e.g. headache attack lasts 2–48 h, unilateral location, pulsating quality, moderate to severe intensity, aggravation by routine physical activity, nausea and/or vomiting, photophobia and phonophobia. Studies with at least one of the following headache (HA) outcome measures were included: intensity, frequency, duration or improvement. Rescue medication was defined as additional medications different from study medication permitted in non-responders, usually limited to the habitual medications a person uses to treat their migraine headache. No language restriction was applied.
Two authors (L.D., J.B.) independently screened titles and abstracts of studies identified by the literature search for eligibility. Potentially relevant studies were retrieved as full papers and again independently reviewed by two authors (L.D., J.B.). Disagreements regarding the inclusion of trials were resolved through consensus when possible, or by arbitration of a third author (A.P.V.).
Methodological quality and data extraction
Two authors (L.D. and J.B. or A.P.V.) independently assessed the methodological quality of the included trials using the Delphi list (11). The Delphi list is a generic criteria list developed by international consensus and consists of the following nine items: (i) randomization; (ii) adequate allocation concealment; (iii) groups similar at baseline; (iv) specification of eligibility criteria; (v) blinding of outcome assessor; (vi) blinding of care provider; (vii) blinding of patient; (viii) presentation of point estimates and measures of variability; (ix) intention-to-treat-analysis. One extra item was added: (x) withdrawal/drop-out rate (>20% or selective drop-out) unlikely to cause bias because it was found relevant for these studies. All selected methodological criteria were scored as yes (= 1), no (= 0) or don’t know (= 0). The quality score was computed by counting the number of positive scores, with equal weights applied on all items. In case of a disagreement between the two authors, consensus was used to resolve disagreement. When consensus could not be reached, a third author made the final decision (J.B. or A.P.V.).
Extraction of data from the original reports was performed by one author (L.D.) and checked by a second (A.P.V.). Disagreements were resolved by consensus. Extracted information included (if available) demographic data, detailed description of the intervention and control (i.e. dose given, study duration), outcome measures and information on adverse effects.
Data analysis
We calculated standard mean differences (SMD) with 95% confidence interval (CI) for continuous outcomes or relative risks (RR) with 95% CI in case of dichotomous variables. RR > 1 and a SMD > 0 represent a better outcome for the first mentioned intervention group. For all data we include data only on those whose results are known (available case analysis).
In case of cross-over trial ideally we would like to restrict our analysis to first-period data only, or, in case of a sufficient wash-out period and no carry-over effect, data of both periods could be combined. In this review we analysed the cross-over trials as if they were parallel-group trials, because most data did not permit analysis of paired between patient data. If a carry-over effect was found and data were reported by period, then the analysis was restricted to first-period data only.
A qualitative analysis was performed using a rating system with levels of evidence (12). The evidence was judged to be strong when multiple (two or more) high-quality RCTs produced generally consistent findings. Results were considered consistent if 75% or more of the studies reported similar results on the same outcome measure. It was judged to be moderate when one high-quality RCT and/or multiple (two or more) low-quality RCTs and/or CCTs produced generally consistent findings. Evidence was considered to be limited when only one low-quality RCT and/or CCT existed and conflicting if the findings of existing trials were inconsistent. No evidence was considered when no RCTs or CCTs were found or when the authors provide no sufficient data for analysis. We regarded trials with methodological quality scores of ≥6 as of high quality (12).
Results
Search results
A total of 3492 publications were identified by our broad and sensitive search strategy (see Fig. 1). Finally a total of 16 RCTs and four CCTs were included in this review, of which 11 studies used a cross-over design.

Quorum statement flow diagram.
Description of studies
Full details of the included studies are presented in Table 1.
Study characteristics of included studies
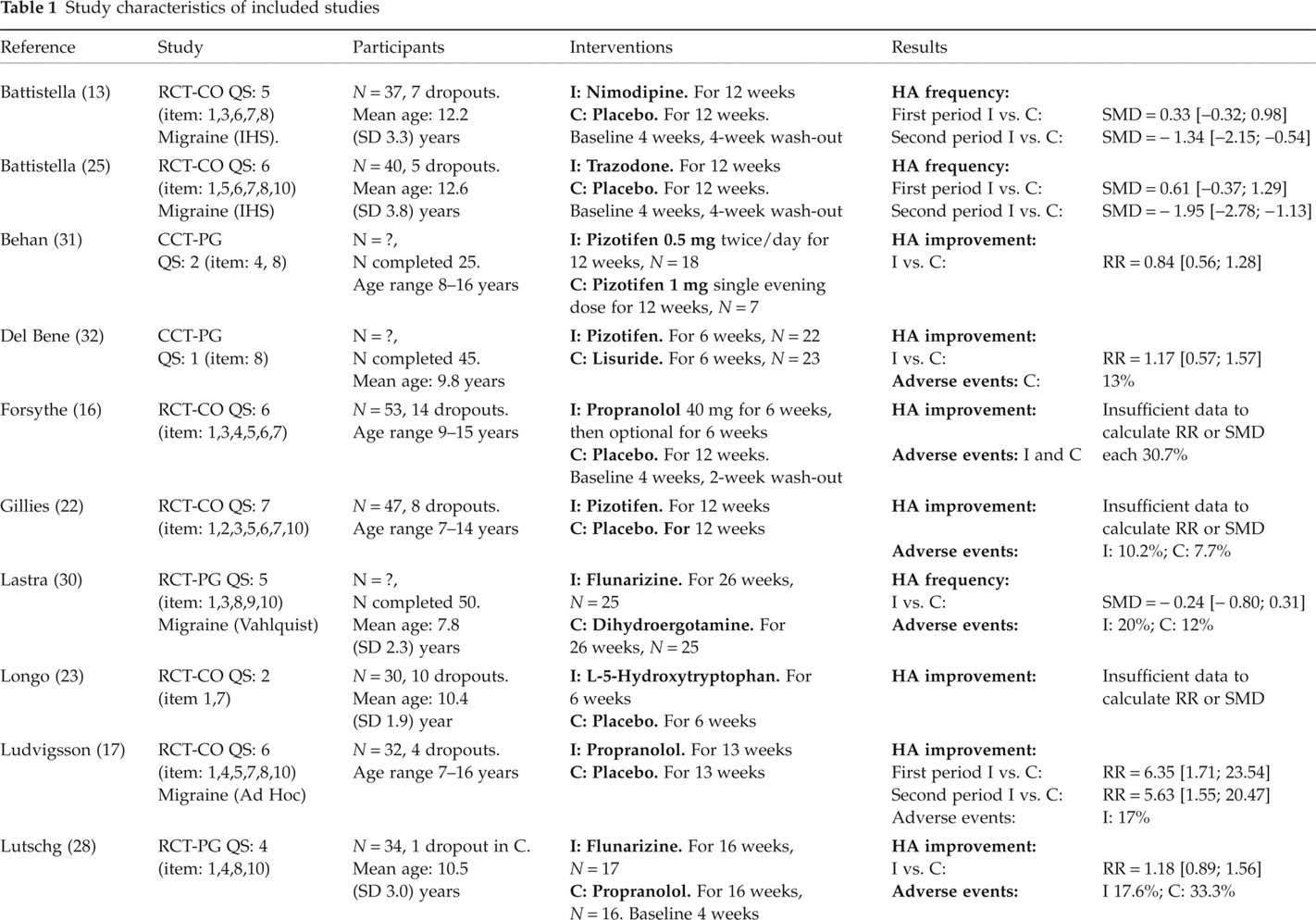

C, control; CCT, controlled clinical trial; CO, cross-over; HA, headache; I, intervention; IHS, International Headache Society 1988; N, number of subjects; QS, quality score on Delphi list with positive items in parentheses; PG, parallel group; RCT, randomized controlled trial; RR, relative risk with 95% confidence interval; SD, standard deviation; SMD, standard mean difference with 95% confidence interval.
Participants
The number of included participants in each trial ranged from 19 to 118 (mean 44 ± 21 patients), with a total of 887 patients included in this review. Most studies were small; out of 40 study arms 12 included less than 20 subjects, while eight included over 50 subjects. The mean percentage of participants who dropped out was 14.5% (range 0–43.9%). The mean age of participants was 10.7 ± 1.3 years (range 3–18 years). Overall, the percentage of girls was generally the same as boys (mean 48.8%; range 30.8–68.6%). Two trials used the criteria of the International Headache Society (9), two studies used the Ad Hoc criteria to classify migraine, six studies used the Valhquist definition while the remaining studies used varying definitions (see Table 1).
Interventions
The interventions used could be divided into placebo comparisons and drug–drug comparisons. Regarding placebo, comparisons were evaluated for nimodipine (13), flunarizine (14, 15), propranolol (16–18), timolol (19), clonidine (20, 21), pizotifen (22), L-5-hydroxytryptophan (L-5HTP) (23, 24), trazodone (25), magnesium oxide (26), and papaverine (27). Regarding the different drugs, comparisons were made between flunarizine and propranolol (28), flunarizine and acetylsalicylic acid (29), flunarizine and dihydroergotamine (30), pizotifen 0.5 mg and 1.0 mg (31), and pizotifen and lisuride (32).
Outcome measures
All studies used headache diaries to assess outcomes. Using this diary amongst others HA frequency, intensity and duration were scored on a Likert scale. In most studies (11 out of 20) a measure of clinical improvement was calculated. In these studies an improvement was regarded as being clinically relevant when the patients’ headache declines by 50% or more. This score is presented as ‘HA improvement’ in Table 1 and is our primary outcome measure. When this outcome measure was not available, we used mainly headache frequency.
Methodological quality
The quality score (with positive items in parentheses) is presented in the ‘Study quality’ section of Table 1. The median score for methodological quality was 5 with a range of 1–7. Using a cut-off point of six out of 10 criteria, eight studies (40%) were considered to be of high quality. The most prevalent methodological shortcomings were a concealed randomization method (unclear 79%), the intention-to-treat analysis (unclear 28%, negative 72%) and blinding of the care provider (unclear 63%).
Effectiveness of pharmacological prophylactic treatment
1. Placebo comparisons
Calcium antagonists In one cross-over study (13) nimodipine was compared with placebo. During the first treatment period no significant differences were found concerning HA frequency and number of adverse events. None of the patients complained of serious side-effects except for mild abdominal discomfort during the early days of nimodipine treatment (17%), which disappeared spontaneously without altering dosage. Two studies (14, 15) compared flunarizine with placebo, in which one study reported on HA improvement (14). At 3 months we found that HA improvement was significantly higher for flunarizine compared with placebo. The number of adverse events was not significantly different: drowsiness, gastrointestinal complaints, weight gain and fatigue.
There is limited evidence that flunarizine is more effective than placebo.
Anti-hypertensive medications Three high-quality studies compared propranolol with placebo (16–18). Two studies reported on HA improvement, showing inconsistent results (17, 18) and two on adverse events (16, 17). Adverse events mentioned were increased appetite, abdominal pain and worsening of headache, and were not significantly different between groups. One small study compared timolol with placebo, but no data were available to calculate effect estimates (19). One high-quality (20) and one low-quality study (21) compared clonidine with placebo, in which only the low-quality study reported on HA intensity and both studies on adverse events. We found no significant differences in HA intensity or the number of adverse events. Adverse events mentioned for clonidine were fatigue and nausea.
There is conflicting evidence for the use of propanolol, and no evidence is found in favour of the use of timonolol and clonidine.
Serotonergic drugs One high-quality cross-over study (22) compared pizotifen with placebo, but no data were available to calculate effect estimates. Only one patient mentioned excessive weight gain while on pizotifen. Two studies (23, 24) compared L-5HTP with placebo, in which only one reported on HA improvement (24). We found no significant differences in HA improvement or in HA index. In the study of Longo et al. (23) adverse events were not specified per medication. One high-quality cross-over study (25) compared trazodone with placebo and reported no significant differences in HA frequency during the first period. None of the patients complained of any serious side-effects.
We found no evidence for or against the use of pizotifen, L-5HTP or trazodone.
Other medications One high-quality study compared magnesium oxide with placebo, but no data were available to calculate effect estimates (26). Adverse events were not significantly different between magnesium oxide and placebo: diarrhoea or soft stools. One small study compared papaverin with placebo and found no significant differences for HA improvement (27).
2. Drug–drug comparisons
All drug–drug comparisons were made in single low-quality studies, of which three were rather small and reported no significant differences between both drugs on HA improvement, HA frequency or adverse events. Adverse events were all minor and included fatigue, sleepiness, wait gain or abdominal pain.
Discussion
Based on the available literature, we found limited evidence that flunarizine is more effective than placebo. There is conflicting evidence from two studies for the use of propranolol. Nimodipine, clonidine, L-5HTP, trazodone and papaverine showed no HA improvement or efficacy in HA reduction of frequency attacks compared with placebo. Available studies on drugs like timolol, pizotifen and magnesium oxide compared with placebo reported insufficient data to calculate effect estimates.
Although systematic reviews offer the least biased method of summarizing research literature, our results must be interpreted with consideration of the quality of evidence from which they were obtained. First, we decided not to contact the authors for additional information, because 19 of the 20 trials included in this review were published before 1994 and most authors would be difficult to find. Second, our inclusion criteria greatly reduced the number of studies selected. Positive results for the use of prophylactic treatments in children and adolescents have frequently emerged from open-label or uncontrolled studies (33–39). Third, the methodological shortcomings of many of the currently available studies limit conclusions about the effectiveness of pharmacological prophylactic treatments. These shortcomings include an unconcealed randomization method, inadequate statistical analysis (intention-to-treat analysis), and most studies suffered from the lack of (reported) credible blinding of the care provider. Finally, most treatments have been evaluated in only one or two studies with a small number of patients, which limits the generalizability of the findings.
Commonly described drugs such as pizotifen and anti-epileptic drugs have not been adequately studied in controlled studies. Calcium antagonists and antihypertensive drugs have been well studied, but larger and better trials are still required in children. Therefore we strongly recommend performing large high-quality RCTs evaluating most frequently offered pharmacological prophylactic treatments, because at the moment no firm conclusions can be drawn based on the available literature.
One of the reasons most studies show no significant differences is that it is difficult for any prophylactic treatment to show additional benefit taking the favourable natural course of childhood migraine into account. Furthermore, it may also be due to the small sample sizes of most studies or the outcome measure ‘HA improvement’, which was a main outcome measure in most studies. It indicated that only people with over 50% improvement are considered clinically improved, which is a large improvement. The Philadelphia panel advises cut-off scores for clinically relevant differences in muscular skeletal diseases of 15% improvement (40). Headache improvement and adverse events are two outcome measures frequently used in the included trials. Although most studies described the adverse events as mild and safe, the simple description of the kind and number of adverse events often gives insufficient insight into the severity and appreciation of the adverse events for a child. Therefore, we suggest that other outcome measures like quality of life, satisfaction of child and/or parents, and repeated administration should also be used as an outcome measure in studies involving treatment of migraine in children.
In conclusion, this review shows that there is a clear need for high-quality research evaluating pharmacological prophylactic treatment of children with migraine. Favourably high quality studies should be performed and reported according to the Consolidated Standards of Reporting of Trials (CONSORT) statement to improve the quality of trials reports. Headache clinical improvement should be used as the primary outcome measure, but lower cut-off points for recovered and not recovered are recommended. Quality of life and satisfaction of child and/or parents should also be used as an outcome measure in studies involving pharmacological prophylactic treatment of children with migraine.
Footnotes
Acknowledgements
The authors thank the Netherlands Organization for Health Research and Development (ZONMw) for funding this research.
Conflict of interest
None declared.
