Abstract
Background
The Chronic Migraine Epidemiology and Outcomes-International study provides insight into people with migraine in multiple countries.
Methods
This cross-sectional, observational, web-based cohort study was conducted in Canada, France, Germany, Japan, United Kingdom, and United States. An initial Screening Module survey solicited general healthcare information from a representative sample and identified participants with migraine based on modified International Classification of Headache Disorders-3 criteria; those with migraine completed a detailed survey based on validated migraine-specific assessments.
Results
Among 90,613 people who correctly completed the screening surveys, 76,121 respondents did not meet the criteria for migraine, while 14,492 did. Among respondents with migraine, mean age ranged from 40 to 42 years. The median number of monthly headache days ranged from 2.33 to 3.33 across countries, while the proportion of respondents with moderate-to-severe disability (measured by Migraine Disability Assessment) ranged from 30% (Japan) to 52% (Germany). The proportion of respondents with ≥15 monthly headache days ranged from 5.4% (France) to 9.5% (Japan). Fewer than half of respondents with migraine in each country reported having received a migraine diagnosis.
Conclusion
These results demonstrated high rates of migraine-related disability and underdiagnosis of migraine across six countries. This study will characterize country-level burden, treatment patterns, and geographical differences in care.
Introduction
Migraine is the second leading global cause of disability and is estimated to affect 1.04 billion people worldwide, accounting for more than 5% of total years lived with disability (YLDs) (1–3). Clinically, two forms of migraine are defined based primarily on attack frequency: episodic migraine (EM) in people with <15 headache days/month and chronic migraine (CM) in those with ≥15 headache days/month for >3 months and ≥8 migraine days/month (4). However, the specific frequency criteria between the two forms is somewhat arbitrary, and migraine is associated with worsening patient-reported outcomes across a spectrum of migraine frequencies (5–7).
The Chronic Migraine Epidemiology and Outcomes (CaMEO) Study, conducted in the United States (US) from 2012–2013, was a cross-sectional web-based survey study that assessed migraine demographics and disability (8,9), barriers to care (10), the complex relationships between migraine comorbidities, disease severity, and disability (11–14), patterns of migraine medication use (15,16), opioid use among those with migraine (17), and the impacts of migraine on family (18,19) and on relationships, career, and finances (20).
Although data on issues associated with migraine are available from many countries, the studies often differ in methodology and sampling strategy, complicating cross-study comparisons. We conducted an international study in six industrialized countries, focusing on migraine demographics, overall disease burden of migraine, and the impact of migraine on quality of life, social and family life, and work. We describe here the design, methodology, and selected baseline findings from the CaMEO-International (CaMEO-I) study, with an emphasis on respondent demographics, rates of migraine diagnosis, current treatment, and use of medications for people who met criteria for migraine across the six included countries.
Methods
Study design and conduct
CaMEO-I was a prospective, cross-sectional, observational, web-based cohort study conducted in six countries: US, Canada, United Kingdom (UK), Germany, France, and Japan. Data collection occurred from July-November 2021 in the US, September-December 2021 in Canada, November-December 2021 in Germany and France, October-December 2021 in the UK, and October 2021-March 2022 in Japan. The study team included a migraine expert from each country (i.e., a national principal investigator [PI]).
The study comprised two phases: in the first (the Screening Phase), respondents were asked to participate in a ‘health survey’ that included general questions about headache presence and severity, as well as a module designed to assess migraine criteria specified in the modified International Classification of Headache Disorders, 3rd Edition (mICHD-3) using the American Migraine Study (AMS)/American Migraine Prevalence and Prevention Study (AMPP) screening questionnaire (i.e., the AMS/AMPP Diagnostic Module) (4,21,22). The AMS/AMPP diagnostic screening criteria were considered modifications of the ICHD-3 criteria because two criteria were not confirmed: ≥5 lifetime migraine events and duration of attack untreated from 4–72 hours. In the second phase, respondents identified with migraine by the screening survey were transitioned to the Survey Module to complete migraine-specific assessments. The respondents who did not meet mICHD-3 migraine criteria completed the Screening Module and provided data as the non-migraine population but did not advance to the Survey Module.
Development of the Screening and Survey Modules in multiple languages (see Online Supplemental Methods) was based on the surveys used in the US CaMEO Study, along with input from clinical experts from each country, people with migraine, and the extant literature.
Study conduct was governed by the Declaration of Helsinki and its amendments, along with applicable national guidelines; where applicable, study documents were approved by a local independent ethics committee. All efforts were made to protect confidentiality consistent with local/national confidentiality and privacy guidelines. Before study recruitment, all study protocols and procedures were reviewed and approved by a central ethics committee and institutional review board that continued to provide oversight across all included countries throughout the study. All study respondents provided informed electronic consent before study enrollment.
Study population
CaMEO-I drew respondents from large panels established in each participating country by Dynata™ (Shelton, CT), a global internet research panel with approximately 4 million members worldwide. Panelists, selected to reflect each country’s demographic and socio-economic makeup, agreed to enroll in surveys on health conditions and other topics. Dynata (previously Research Now) also provided the online panel used in the 2012 US CaMEO study.
Because the study was intended to be broadly inclusive and reflective of national demographics, the only inclusion criteria were age ≥18 years, being an active Dynata Research panelist, ability to read/understand the national language, and willingness/ability to provide informed electronic consent. A demographically representative sample of each country’s panelists was invited via electronic means to participate in a survey about ‘work, health, and lifestyle’ that was also designed to solicit information on the presence and features of headache. Survey completers were provided a small financial incentive.
Screening phase
The Screening Module collected sociodemographic information and information on general health/comorbidities based on a modified Charlson Comorbidity Index (23). All respondents were asked the following question: “how many of your headaches in the past 12 months were caused by head injury, hangover, or an illness such as cold or flu?” with the following response options: “none of them,” “some of them,” “most of them,” “all of them,” and “had no headaches in the past 12 months.” For people who selected any of the response options other than “had no headaches in the past 12 months,” migraine-specific symptom information focusing on the headache phase was collected using the Migraine Symptom Severity Scale (MSSS) (24). The Screening Module also included the Work Productivity and Activity Impairment assessment (25). Although some respondents provided information on race and ethnicity, these data are not reported because not all countries allowed race/ethnicity information to be collected. Similarly, household income information was collected but also is not reported, due to differences in currency between countries.
Respondents who met mICHD-3 criteria for migraine (based on their responses on the AMS/AMPP diagnostic questionnaire [21,22]) were invited to participate in the Survey Module (Online Supplemental Methods). Respondents were defined as meeting mICHD-3 migraine criteria if they indicated having two or more of the four pain symptoms (pain is worse on just one side; pain is pounding, pulsating, or throbbing; pain has moderate or severe intensity; and pain is made worse by routine activities such as walking or climbing stairs) less than half the time or half the time or more and either nausea or both photophobia and phonophobia less than half the time or half the time or more.
Survey Module
The Survey Module comprised multiple patient-reported outcome measures (PROM) addressing migraine clinical aspects, migraine-associated disability, and the impact of migraine on the individual, family, finances, career, and health-related quality of life. Validated PROMs included the 12-item Allodynia Symptom Checklist (ASC-12) (26), the five-item Migraine Disability Assessment (MIDAS) (27), the 14-item Migraine-Specific Quality of Life Questionnaire (MSQ) (28), the six-item Migraine Treatment Optimization Questionnaire (mTOQ) (29), a nine-item subset of the 43-item Subjective Cognitive Impairment Scale for Migraine (Mig-SCog) (30), the six-item Work Productivity and Activity Impairment–General Health (WPAI-GH) assessment (25), the four-item Patient Health Questionnaire-4 (depression and anxiety symptom screener) (PHQ-4) (31), and the 12-item Impact of Migraine on Partners and Adolescent Children (IMPAC) Scale (32). Non-validated assessments included the eight-item MSSS (24), Average Pain Intensity of Most Recent Long-Duration Headache (for individuals with ≥1 headache of 24-hr duration, rated on a 0–10 scale [0 = no pain; 10 = worst pain ever]), and a Barriers to Care assessment.
The Survey Module also assessed the use of over-the-counter (OTC) medications, prescription acute medication for migraine, and prescription preventive medication for migraine. The specific medications available differed across countries (Online Supplemental Table 1). Although certain triptans may be available OTC in pharmacies in some countries, triptans were classified as prescription acute medications in this study.
Statistical methods
Quality checks of the data were performed (Online Supplemental Methods). Samples of approximately 2400 respondents with migraine in each country were targeted to ensure adequate sample sizes from each country for estimating consultation and diagnostic rates. It was estimated that 15% of respondents would meet the mICHD-3 migraine criteria. Invitations were sent to enough panel members to generate ∼93,000 completed Screening Module assessments to achieve a sample of approximately 14,000 respondents with migraine. We continued sampling in each country until the target sample size was achieved. Sampling was reinitiated if the number of eligible individuals in any country fell below the target sample of approximately 2400 individuals with migraine per country.
Response data from the CaMEO-I Survey Module were analyzed on a country-specific basis and also in aggregate, including all respondents. Descriptive statistics were used in this observational study.
Results
Study population
Participant flow
Across all six countries, the e-invitations generated a total of 135,351 views, defined as all unique visitors who reached the first page of the survey site. Of these, approximately 10% did not provide consent or were <18 years of age, leaving 121,436 respondents (90%) who completed all or part of the appropriate survey modules. A total of 104,396 participants completed the appropriate surveys (77% of views), and after quality control evaluation and removal of incomplete or inconsistent responses, a total of 90,613 respondents (67% of views) were included in the study as valid completers. The age and gender distributions of valid completers for each country were similar to those of the overall population (according to national statistics); across the percentages for each age range and each gender in each country, differences between the valid completer and national populations were ≤5% except for the difference in the percentage of Canadian respondents ≥65 years old (8.4%; Online Supplemental Table 2).
Among valid completers, 14,492 respondents (16%) were classified as having migraine based on mICHD-3 criteria gathered via the AMS/AMPP diagnostic questionnaire and were offered the opportunity to enroll in the Survey Module; the remaining 76,121 (84%) did not meet criteria for migraine and were not eligible for the Survey Module (Online Supplemental Results). Each country was represented by at least 2300 people with mICHD-3 migraine. The proportion of respondents classified with migraine was 16%–21% in all countries except Japan (8%). Participant flow, response rates, and sampling dates for each country are summarized in Table 1.
CaMEO-I survey response rates and respondent flow.
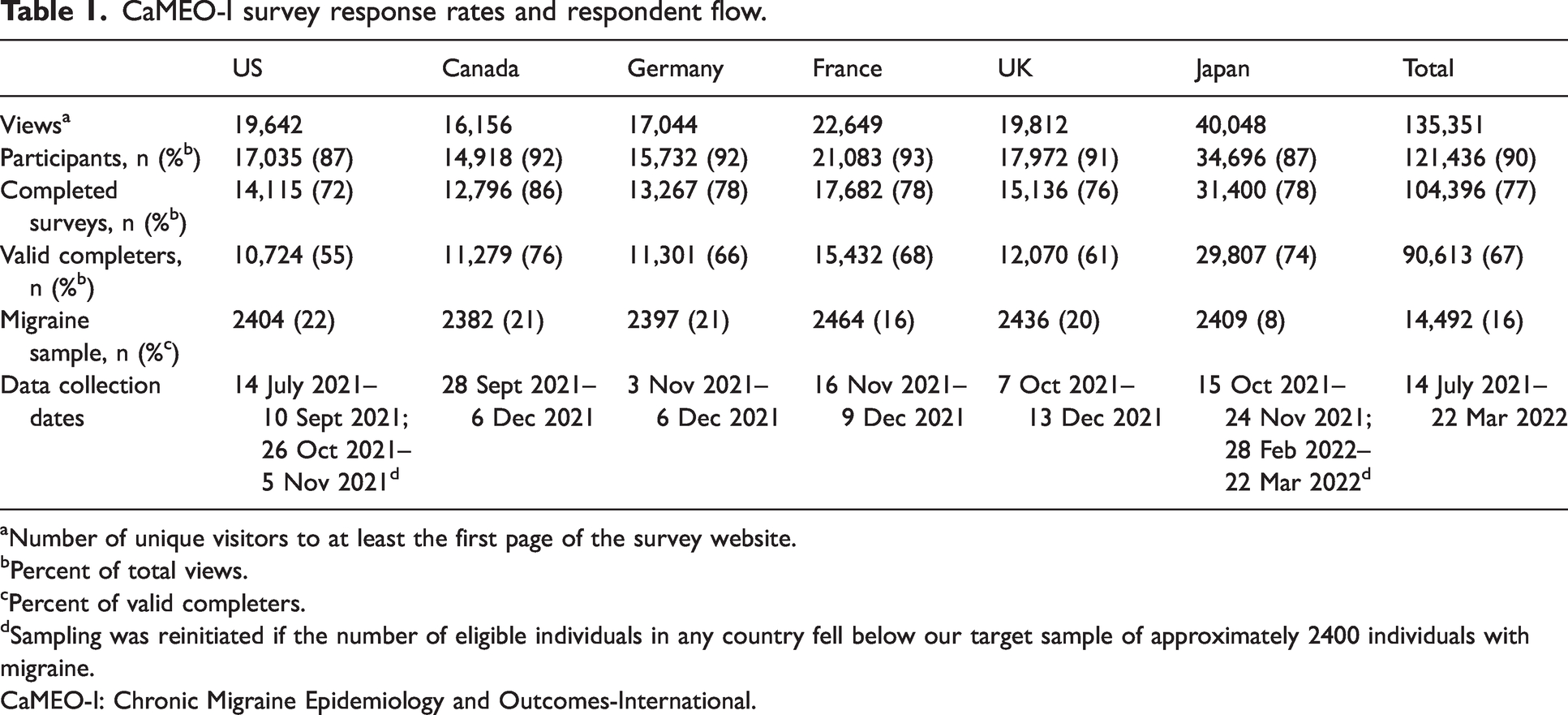
Number of unique visitors to at least the first page of the survey website.
Percent of total views.
Percent of valid completers.
Sampling was reinitiated if the number of eligible individuals in any country fell below our target sample of approximately 2400 individuals with migraine.
CaMEO-I: Chronic Migraine Epidemiology and Outcomes-International.
Population demographics
Key demographic variables for the 14,492 migraine respondents are listed by country in Table 2. Mean age of migraine respondents was similar among countries (range, 40.3–42.3 years); females constituted two-thirds to three-quarters of respondents in all six countries (range, 68.7%–73.8%) and 35%–47% of respondents were married. On average, migraine respondents across all countries had experienced migraine for approximately 19–21 years. Notable between-country differences included lower body mass index (BMI) in Japanese respondents versus other countries, and higher rates of tobacco use in both Germany (45.0%) and the US (40.6%) versus other countries (range, 22.1%–34.3%).
Key demographics for the CaMEO-I migraine population.

Among respondents who provided an answer.
Of respondents in Germany, 44.5% received vocational training or completed professional certification.
Among respondents aged ≥21 years; includes cigarettes, electronic cigarettes, nicotine replacements, smokeless tobacco, heated tobacco, and cigars.
BMI: body mass index; CaMEO-I: Chronic Migraine Epidemiology and Outcomes-International; SD: standard deviation.
Migraine severity: Disability, monthly headache days
The proportions of migraine respondents by MIDAS grade are plotted by country in Figure 1. The rate of moderate-to-severe disability ranged from a low of 30.3% (Japan) to highs of 47.6% in the US and 52.0% in Germany. Notably, median MIDAS scores were lowest in Japan across all MHD categories (Online Supplemental Table 3).

Rates of Migraine Disability Assessment (MIDAS) grades, by country.
Migraine frequency, in terms of monthly headache day (MHD) ranges, is summarized by country in Figure 2. Across all countries, the percentage of respondents with migraine and ≥15 MHDs ranged from 5.4% (France) to 9.5% (Japan). Table 3 summarizes MHD mean and median by country. Mean (SD) MHDs ranged from 4.20 (5.3; UK) to 5.40 (6.3; Japan), and median (IQR) MHDs ranged from 2.33 (3.7 [Canada] and 5.0 [UK]) to 3.33 (5.0 [US, Germany, and Japan]).

Proportions of migraine respondents reporting monthly headache day ranges of 0–3, 4–7, 8–14, 15–24, and ≥25, by country.
Monthly headache days, by country, for migraine respondents in CaMEO-I.

CaMEO-I: Chronic Migraine Epidemiology and Outcomes-International; IQR, interquartile range; SD, standard deviation.
Migraine consultation and treatment
The proportion of migraine respondents who had ever consulted any type of healthcare professional about headache ranged from 52.9% (Japan) to 77.2% (France; Figure 3). Although all respondents in the migraine population met the mICHD-3 case definition for migraine, less than half of the people with migraine reported that they had received a diagnosis of migraine. Self-reported healthcare practitioner diagnosis rates for migraine, chronic/transformed migraine, or menstrual migraine ranged from 42.8% (Japan) to 49.3% (US; Figure 4A). Although at least 5.4% of respondents had ≥15 MHDs across all countries, the self-reported migraine diagnosis for chronic/transformed migraine ranged from 0.7% (Japan) to 4.8% (US). The self-reported migraine diagnosis for chronic/transformed migraine ranged from 0.5% (Japan) to 4.0% (US) among respondents who had <15 MHDs (Figure 4B) and from 2.6% (Japan) to 14.2% (US) among respondents who had ≥15 MHDs (Figure 4C).

Proportion of migraine respondents who have ever consulted any type of healthcare professional about headache (error bars represent 95% confidence intervals).

Rates of self-reported medical diagnosis of migraine (any type), of migraine or chronic/transformed migraine, and of chronic/transformed migraine, by country. (a) among all migraine respondents; (b) among migraine respondents with <15 monthly headache days (MHDs); (c) among migraine respondents with ≥15 MHDs; (d) among migraine respondents who have ever consulted a healthcare professional about headache.
Among respondents who had ever consulted any type of healthcare professional about headache, the self-reported migraine diagnosis rates for any type of migraine ranged from 59.3% (Germany) to 66.5% (Japan), and for chronic/transformed migraine ranged from 1.3% (Japan) to 6.8% (US) (Figure 4D). The rate of self-reported migraine diagnosis for any type of migraine (i.e., migraine, chronic/transformed migraine, or menstrual migraine) increased by 10%–20% with consultation with a doctor or other healthcare professional, but diagnosis rates of chronic/transformed migraine remained low.
The percentages of respondents whose migraine was currently being managed or treated by any type of doctor or healthcare professional (e.g., general practitioners; family physicians; internal medicine doctors; nurse practitioners; physician’s assistants; allergists; ear, nose, throat specialists; headache specialists; neurologists; obstetricians/gynecologists; pain specialists; psychiatrists; emergency room doctors; urgent care doctors; ophthalmologists; rheumatologists) were 32.7% (95% CI, 30.8–34.6) in the US, 25.2% (95% CI, 23.5–27.0) in Canada, 29.6% (95% CI, 27.8–31.5) in Germany, 40.0% (95% CI, 38.0–41.9) in France, 23.8% (95% CI, 22.1–25.5) in the UK, and 19.5% (95% CI, 17.9–21.1) in Japan. The most common types of doctor or healthcare professional managing respondents across all countries were general practitioners, family physicians, or internal medicine doctors (45.1% [Japan] to 80.1% [France]).
Patterns of use of OTC and prescription drugs for preventive treatment of migraine attacks were generally similar among countries, but these differed depending on whether respondents received medical consultation; the changes in treatment patterns between respondents who did and did not receive medication consultation were generally similar across countries (Figure 5). Among all migraine respondents, country-specific rates of OTC use for migraine ranged from 71.1% (Japan) to 88.4% (US), rates of acute prescription medication use ranged from 32.7% (Canada) to 49.6% (France), and rates of preventive migraine medication use ranged from 6.4% (Japan) to 16.8% (US) (Figure 5A). Among respondents under current care for migraine by a healthcare professional, rates of OTC use were slightly lower (range: 56.8% [Japan] to 86.5% [US]), but there were higher usage rates for migraine-specific acute prescription medications (range: 70.0% [Canada] to 86.2% [Japan]) and preventive medication (range: 21.3% [France] to 43.1% [UK]) (Figure 5B).

Proportion of migraine respondents reporting use of over-the-counter (OTC) drugs, acute migraine prescription drugs, or preventive migraine prescription drugs, by country. (a) Among all migraine respondents and (b) among migraine respondents under management for headache by a healthcare professional.
Discussion
The CaMEO-I Study, modeled after the US-based 2012 CaMEO Study, compared large samples of migraine respondents from six industrialized countries and revealed generally similar patterns of patient demographics, rates of diagnosis, and medication use. The inclusion criteria allowed age and gender distributions of valid completers for each country to be representative to those of the overall population based on national statistics; however, respondents had a higher level of education than the general population. In this analysis, we assumed that transformed migraine (a term originally used to describe what is known as chronic migraine today [33]) is a proxy for CM so that migraine diagnostic rates would not be underestimated. The data generated in CaMEO-I will serve as the basis of additional analyses to characterize migraine and its impacts more broadly, and ultimately pave the way for improved patient outcomes worldwide. In this analysis, there was also a non-migraine sample of 76,121 respondents.
The US-based 2012 CaMEO study included 16,789 respondents with migraine, corresponding to 28.7% of usable survey returns (9). The demographic and headache characteristics of respondents in the CaMEO study were generally consistent with the CaMEO-I findings in the United States. In CaMEO, the mean ages of respondents were 40.6 years and 41.0 years for EM and CM respondents, respectively; 73.8% and 81.1% of respondents were women, and 34.6% and 41.6% were obese in EM and CM respondents, respectively. The mean (SD) MIDAS scores were 13.1 (22.2) and 60.5 (70.4), and mean (SD) headache frequencies were 3.2 (3.2) and 20.8 (4.9) in the respondents with EM and CM, respectively. These findings from the CaMEO-I study add to previous results from the CaMEO study and provide additional multi-country information.
The present results also add to previous results from the Eurolight project, which was a survey study conducted in 10 European countries from 2008–2009 to evaluate the impact of headache disorders in Europe (34). The Eurolight project reported a lifetime prevalence of any headache of 91.3% and highlighted the burden of headache disorders; men had a mean MHD of 4.7 and women had a mean MHD of 6.3 irrespective of diagnosis (35). Additionally, in people with migraine, mean lost workdays and mean lost housework days were one to two days per month. The substantial prevalence and burden of headache demonstrated in Eurolight highlight the need for additional data, such as from the CaMEO-I study, to better understand migraine.
Although not designed to investigate migraine prevalence, the Screening Module of the two-step survey approach used here identified proportions of respondents with migraine ranging from 16%–22% for five of the six countries included in the study. In Japan, only 8% of Screening Module respondents met criteria for migraine; these results are consistent with those reported in the recent OVERCOME (Japan) study, which found a prevalence of 8.6% (36). Because total response rates were modest in both the CaMEO-I and OVERCOME studies, the prevalence estimates may be biased given the sampling methodology. However, previously conducted Asian community-based surveys similarly reported a migraine prevalence of 8.4% to 12.7% (37). The differences in migraine prevalence between the Asian and Western countries could potentially be due to cultural differences in symptom reporting or pain perception (38). The reasons for prevalence differences among countries merit further exploration.
Most migraine respondents in all countries were in the lowest headache frequency category (0–3 MHDs), consistent with the median MHD values of approximately 3 across countries. However, 30% to 52% of respondents reported moderate-to-severe migraine-related disability based on MIDAS grade. Similarly, headache frequency and migraine-related disability were previously assessed in the 2009 International Burden of Migraine Study (IBMS) conducted in Australia, Canada, France, Germany, Italy, Spain, the UK, Taiwan, and the US (39). The survey study reported that 29.4% of participants had a MIDAS grade of 1, 20.8% had grade 2, 23.1% had grade 3, and 26.7% had grade 4. Depending on the number of headache days respondents had, the distribution of MIDAS grade scores differed. Together, findings from the present CaMEO-I study and the previous IBMS study highlight the substantial burden and disability associated with migraine.
Findings from the present study showed a low rate of migraine diagnosis; less than 50% of respondents who met the study criteria for migraine had a self-reported medical diagnosis of migraine across all six countries. Although self-reported migraine diagnosis rates were not surprisingly higher for respondents who had received medical consultation for headache, 30%–40% of migraine patients did not report receiving a migraine diagnosis. These results highlight that underdiagnosis of migraine is a concern in each country studied and point to the need to increase the rates of consultation and diagnosis.
In addition, among the migraine respondents overall, OTC medications were much more commonly used than either prescription acute or preventive treatments for migraine. Current medical consultation was associated with increased use of prescription migraine medications; in some countries, use of current acute prescription medications was higher than that of OTC drugs. The rate of using preventive medications for migraine was also increased in this respondent subset compared with other epidemiologic studies, reaching >40% in the US and the UK.
This study is limited by self-reported data and the potential for selection bias resulting from use of an online platform and modest participation rates. These survey results were not confirmed by medical record review. Additionally, as these data were collected during the COVID-19 pandemic, medical consultation patterns could have been affected. Findings might not be generalizable to the overall population of people with migraine. Notably, the percentages of respondents in each country with university education or higher in this analysis were higher than national statistics for each country (40), suggesting that people with lower socio-economic status may have been underrepresented in this study. Additionally, the study was conducted in three English-speaking countries and in three countries with other languages. Although we engaged in translation and backtranslation of questionnaires, performed cognitive debriefing of translated measures, and consulted with clinical experts from each country, there may be some differences in survey performance among the countries and in the different languages. Finally, we used a modified version of the ICHD-3 migraine criteria, the AMS/AMPP Diagnostic Module (4,21,22), to identify participants with migraine. Although our results may be less comparable to those of studies in which the full ICHD-3 criteria were used, the AMS/AMPP criteria have been used in a number of other US epidemiological studies, including CaMEO, Migraine in America Symptoms and Treatment (MAST), and OVERCOME (9,41,42) In addition, the large sample size, rigorous prevention of duplicate/multiple entries, and quality checks on survey response validity help to ensure this study’s overall validity and meaning.
Conclusion
This initial report of baseline results from the CaMEO-I Study highlights the extent of migraine-related disability, with one-third to one-half of respondents reporting moderate-to-severe disability across countries. Despite minor country-to-country differences in rates of self-reported medical diagnosis of migraine and CM, underdiagnosis of migraine was a concern in each country studied. Additionally, results from this study suggest a potential underuse of prescription migraine-specific medications and overuse of OTC drugs, especially among those not under current medical care for migraine. These findings provide insights to aid in improving the management of people with migraine.
Article highlights
The CaMEO-I Study demonstrated generally consistent demographics and disease burden among migraine respondents from six countries. Although all migraine respondents met diagnostic criteria for migraine, the rate of self-reported medical diagnosis of migraine was <50% (42.8%–49.3%) across all six countries. Individuals with migraine, especially those not currently managed by a healthcare professional, potentially underuse prescription acute and preventive treatments.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231180611 - Supplemental material for Chronic Migraine Epidemiology and Outcomes – International (CaMEO-I) Study: Methods and multi-country baseline findings for diagnosis rates and care
Supplemental material, sj-pdf-1-cep-10.1177_03331024231180611 for Chronic Migraine Epidemiology and Outcomes – International (CaMEO-I) Study: Methods and multi-country baseline findings for diagnosis rates and care by Aubrey Manack Adams, Dawn C. Buse, Elizabeth Leroux, Michel Lanteri-Minet, Fumihiko Sakai, Manjit S. Matharu, Zaza Katsarava, Michael L. Reed, Kristina Fanning, Katherine Sommer and Richard B. Lipton in Cephalalgia
Footnotes
Acknowledgments
Medical writing and editorial assistance was provided to the authors by Anny Wu, PharmD, of Peloton Advantage, LLC, an OPEN Health company, Parsippany, NJ, and was funded by AbbVie. The authors wish to acknowledge the contributions of Ryan Bostic of MIST Research, Wilmington, DE, for data analyses, and Valerie Marske of Vedanta Research, Chapel Hill, NC, for survey development. The authors would also like to thank the study participants, site collaborators, and survey translators.
Data availability statement
AbbVie is committed to responsible data sharing regarding the clinical trials we sponsor. This includes access to anonymized, individual, and trial-level data (analysis data sets), as well as other information (e.g., protocols, clinical study reports, or analysis plans), as long as the trials are not part of an ongoing or planned regulatory submission. This includes requests for clinical trial data for unlicensed products and indications.
These clinical trial data can be requested by any qualified researchers who engage in rigorous, independent, scientific research, and will be provided following review and approval of a research proposal, Statistical Analysis Plan (SAP), and execution of a Data Sharing Agreement (DSA). Data requests can be submitted at any time after approval in the US and Europe and after acceptance of this manuscript for publication. The data will be accessible for 12 months, with possible extensions considered. For more information on the process or to submit a request, visit the following link: ![]()
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AMA and KS are employees of AbbVie and may hold AbbVie stock. DCB has received grant support and honoraria from AbbVie, Amgen, Biohaven, Collegium, Eli Lilly and Company, Lundbeck, and Teva and for work on the editorial board of Current Pain and Headache Reports. EL has received speaker fees and consulting fees from Allergan, Eli Lilly, and Teva Neuroscience; consulting fees from Aralez Pharmaceuticals, McKesson Canada, and Medscape; speaking fees, consulting fees, and reimbursement for travel from Novartis; and reimbursement for travel from IHS-GPAC. ML-M reports personal fees for advisory boards, speaker panels or investigation studies from Allergan, Amgen, Astellas, ATI, BioElectronics, BMS, Boehringer, Boston Scientific, CoLucid, Convergence, GlaxoSmithKline, Grunenthal, IPSEN, Lundbeck, Lilly, Medtronic, Menarini, MSD, Novartis, Pfizer, Reckitt Benckiser, Saint-Jude, Salvia, Sanofi-Aventis, Teva, UCB, UPSA, and Zambon. FS is a consultant, speaker, or scientific advisor for Amgen, Otsuka, Eli Lilly, Eisai, and Teva. MSM serves on the advisory board for Abbott, AbbVie, Eli Lilly, Medtronic, Salvia, and Teva, and has received payment for the development of educational presentations from AbbVie, electroCore, Eli Lilly, Novartis, and Teva. ZK has been a speaker and/or consultant, and/or received research support from Allergan, Amgen/Novartis, Eli Lilly, Merck, and Teva. MLR is Managing Director of Vedanta Research, which has received research funding from AbbVie, Allay Lamp, Dr. Reddy’s Laboratories, Eli Lilly, and GlaxoSmithKline via grants to the National Headache Foundation. Vedanta Research has received funding directly from AbbVie for work on the CaMEO and CaMEO-I Studies. KF is Managing Director of MIST Research which, in collaboration with Vedanta Research, has received research funding from AbbVie, Allay Lamp, Dr. Reddy’s Laboratories, Eli Lilly, and GlaxoSmithKline via grants to the National Headache Foundation. MIST Research has received funding directly from AbbVie for work on the CaMEO and CaMEO-I Studies. RBL has received research support from the National Institutes of Health, the FDA, and the National Headache Foundation. He serves as consultant, advisory board member, or has received honoraria or research support from AbbVie/Allergan, Amgen, Biohaven, Dr. Reddy’s Laboratories (Promius), electroCore, Eli Lilly, GlaxoSmithKline, Lundbeck, Merck, Novartis, Pfizer, Teva, Vector, and Vedanta Research. He receives royalties from Wolff’s Headache, 8th edition (Oxford University Press, 2009), and Informa. He holds stock/options in Biohaven and Manistee.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by study funding from Allergan (prior to its acquisition by AbbVie), which contributed to the study design, the collection, analysis, and interpretation of data, and the review and approval of the final version for publication. All authors had access to relevant data and participated in the drafting, review, and approval of this publication. No honoraria or payments were made for authorship.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
